Transthyretin Amyloid Cardiomyopathy Treatment: An Updated Review
Abstract
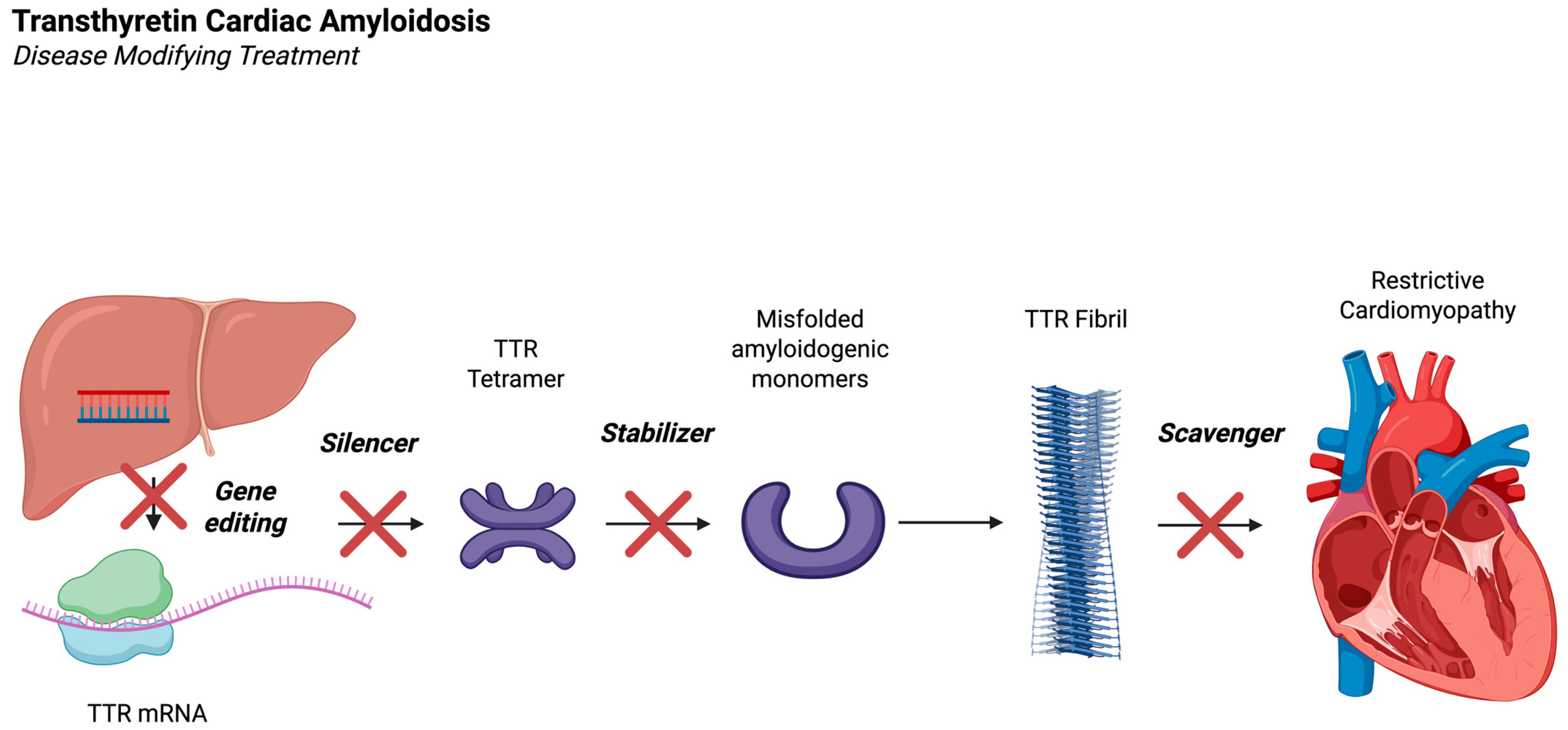
1. Introduction
2. Clinical Manifestations and Diagnosis
3. Current Disease-Directed Therapies
3.1. Transthyretin Stabilizers
3.1.1. Tafamidis
3.1.2. Diflunisal
3.1.3. Acoramidis
3.2. TTR Gene Silencing Therapies
3.2.1. Patisiran
3.2.2. Vutrisiran
3.2.3. Eplontersen
3.3. Transthyretin Gene Editing
4. TTR Scavenging Therapy
5. Management of Cardiovascular Complications
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Prapunpoj, P.; Leelawatwattana, L. Evolutionary changes to transthyretin: Structure-function relationships. FEBS J. 2009, 276, 5330–5341. [Google Scholar] [CrossRef]
- Maurer, M.S.; Schwartz, J.H.; Gundapaneni, B.; Elliott, P.M.; Merlini, G.; Waddington-Cruz, M.; Kristen, A.V.; Grogan, M.; Witteles, R.; Damy, T.; et al. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2018, 379, 1007–1016. [Google Scholar] [CrossRef]
- Ruberg, F.L.; Grogan, M.; Hanna, M.; Kelly, J.W.; Maurer, M.S. Transthyretin Amyloid Cardiomyopathy: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 2872–2891. [Google Scholar] [CrossRef]
- Mohammed, S.F.; Mirzoyev, S.A.; Edwards, W.D.; Dogan, A.; Grogan, D.R.; Dunlay, S.M.; Roger, V.L.; Gertz, M.A.; Dispenzieri, A.; Zeldenrust, S.R.; et al. Left ventricular amyloid deposition in patients with heart failure and preserved ejection fraction. JACC Heart Fail 2014, 2, 113–122. [Google Scholar] [CrossRef] [PubMed]
- González-López, E.; Gallego-Delgado, M.; Guzzo-Merello, G.; de Haro-Del Moral, F.J.; Cobo-Marcos, M.; Robles, C.; Bornstein, B.; Salas, C.; Lara-Pezzi, E.; Alonso-Pulpon, L.; et al. Wild-type transthyretin amyloidosis as a cause of heart failure with preserved ejection fraction. Eur. Heart J. 2015, 36, 2585–2594. [Google Scholar] [CrossRef]
- González-López, E.; Gagliardi, C.; Dominguez, F.; Quarta, C.C.; de Haro-Del Moral, F.J.; Milandri, A.; Salas, C.; Cinelli, M.; Cobo-Marcos, M.; Lorenzini, M.; et al. Clinical characteristics of wild-type transthyretin cardiac amyloidosis: Disproving myths. Eur. Heart J. 2017, 38, 1895–1904. [Google Scholar] [CrossRef]
- Yamamoto, H.; Yokochi, T. Transthyretin cardiac amyloidosis: An update on diagnosis and treatment. ESC Heart Fail. 2019, 6, 1128–1139. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee; Kittleson, M.M.; Ruberg, F.L.; Ambardekar, A.V.; Brannagan, T.H.; Cheng, R.K.; Clarke, J.O.; Dember, L.M.; Frantz, J.G.; Hershberger, R.E.; et al. 2023 ACC Expert Consensus Decision Pathway on Comprehensive Multidisciplinary Care for the Patient With Cardiac Amyloidosis: A Report of the American College of Cardiology Solution Set Oversight Committee. J. Am. Coll. Cardiol. 2023, 81, 1076–1126. [Google Scholar]
- Tschöpe, C.; Elsanhoury, A. Treatment of Transthyretin Amyloid Cardiomyopathy: The Current Options, the Future, and the Challenges. J. Clin. Med. 2022, 11, 2148. [Google Scholar] [CrossRef]
- Bulawa, C.E.; Connelly, S.; Devit, M.; Wang, L.; Weigel, C.; Fleming, J.A.; Packman, J.; Powers, E.T.; Wiseman, R.L.; Foss, T.R.; et al. Tafamidis, a potent and selective transthyretin kinetic stabilizer that inhibits the amyloid cascade. Proc. Natl. Acad. Sci. USA 2012, 109, 9629–9634. [Google Scholar] [CrossRef]
- Coelho, T.; Maia, L.F.; Martins da Silva, A.; Waddington Cruz, M.; Planté-Bordeneuve, V.; Lozeron, P.; Suhr, O.B.; Campistol, J.M.; Conceição, I.M.; Schmidt, H.H.; et al. Tafamidis for transthyretin familial amyloid polyneuropathy: A randomized, controlled trial. Neurology 2012, 79, 785–792. [Google Scholar] [CrossRef]
- Lohrmann, G.; Pipilas, A.; Mussinelli, R.; Gopal, D.M.; Berk, J.L.; Connors, L.H.; Vellanki, N.; Hellawell, J.; Siddiqi, O.K.; Fox, J.; et al. Stabilization of Cardiac Function With Diflunisal in Transthyretin (ATTR) Cardiac Amyloidosis. J. Card. Fail. 2022, 26, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Castaño, A.; Helmke, S.; Alvarez, J.; Delisle, S.; Maurer, M.S. Diflunisal for ATTR cardiac amyloidosis. Congest. Heart Fail. 2012, 18, 315–319. [Google Scholar] [CrossRef]
- Siddiqi, O.K.; Mints, Y.Y.; Berk, J.L.; Connors, L.; Doros, G.; Gopal, D.M.; Kataria, S.; Lohrmann, G.; Pipilas, A.R.; Ruberg, F.L. Diflunisal treatment is associated with improved survival for patients with early stage wild-type transthyretin (ATTR) amyloid cardiomyopathy: The Boston University Amyloidosis Center experience. Amyloid Int. J. Exp. Clin. Investig. Off. J. Int. Soc. Amyloidosis 2022, 29, 71–78. [Google Scholar] [CrossRef]
- Hammarström, P.; Jiang, X.; Hurshman, A.R.; Powers, E.T.; Kelly, J.W. Sequence-dependent denaturation energetics: A major determinant in amyloid disease diversity. Proc. Natl. Acad. Sci. USA 2022, 99 (Suppl. 4), 16427–16432. [Google Scholar] [CrossRef]
- Gillmore, J.D.; Judge, D.P.; Cappelli, F.; Fontana, M.; Garcia-Pavia, P.; Gibbs, S.; Grogan, M.; Hanna, M.; Hoffman, J.; Masri, A.; et al. Efficacy and Safety of Acoramidis in Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2024, 390, 132–142. [Google Scholar] [CrossRef]
- Miller, M.; Pal, A.; Albusairi, W.; Joo, H.; Pappas, B.; Haque Tuhin, M.T.; Liang, D.; Jampala, R.; Liu, F.; Khan, J.; et al. Enthalpy-Driven Stabilization of Transthyretin by AG10 Mimics a Naturally Occurring Genetic Variant That Protects from Transthyretin Amyloidosis. J. Med. Chem. 2018, 61, 7862–7876. [Google Scholar] [CrossRef] [PubMed]
- Yonezawa, S.; Koide, H.; Asai, T. Recent advances in siRNA delivery mediated by lipid-based nanoparticles. Adv. Drug Deliv. Rev. 2020, 154–155, 64–78. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.S.; Kale, P.; Fontana, M.; Berk, J.L.; Grogan, M.; Gustafsson, F.; Hung, R.R.; Gottlieb, R.L.; Damy, T.; González-Duarte, A.; et al. Patisiran Treatment in Patients with Transthyretin Cardiac Amyloidosis. N. Engl. J. Med. 2023, 389, 1553–1565. [Google Scholar] [CrossRef]
- Fontana, M.; Berk, J.L.; Gillmore, J.D.; Witteles, R.M.; Grogan, M.; Drachman, B.; Damy, T.; Garcia-Pavia, P.; Taubel, J.; Solomon, S.D.; et al. Vutrisiran in Patients with Transthyretin Amyloidosis with Cardiomyopathy. N. Engl. J. Med. 2024, 392, 33–44. [Google Scholar] [CrossRef]
- Nie, T. Eplontersen: First Approval. Drugs 2024, 84, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Ionis Pharmaceuticals, Inc. A Phase 3 Global, Double-Blind, Randomized, Placebo-Controlled Study to Evaluate the Efficacy and Safety of ION-682884 in Patients with Transthyretin-Mediated Amyloid Cardiomyopathy (ATTR CM) [Internet]. ClinicalTrials.gov Identifier NCT04136171; First posted 23 Oct 2019. Available online: https://clinicaltrials.gov/ct2/show/NCT04136171 (accessed on 10 February 2025).
- Gillmore, J.D.; Gane, E.; Taubel, J.; Kao, J.; Fontana, M.; Maitland, M.L.; Seitzer, J.; O’Connell, D.; Walsh, K.R.; Wood, K.; et al. CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. N. Engl. J. Med. 2021, 385, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Fontana, M.; Solomon, S.D.; Kachadourian, J.; Walsh, L.; Rocha, R.; Lebwohl, D.; Smith, D.; Täubel, J.; Gane, E.J.; Pilebro, B.; et al. CRISPR-Cas9 Gene Editing with Nexiguran Ziclumeran for ATTR Cardiomyopathy. N. Engl. J. Med. 2024, 391, 2231–2241. [Google Scholar] [CrossRef] [PubMed]
- Fontana, M.; Gilbertson, J.; Verona, G.; Riefolo, M.; Slamova, I.; Leone, O.; Rowczenio, D.; Botcher, N.; Ioannou, A.; Patel, R.K.; et al. Antibody-Associated Reversal of ATTR Amyloidosis-Related Cardiomyopathy. N. Engl. J. Med. 2023, 388, 2199–2201. [Google Scholar] [CrossRef]
- Higaki, J.N.; Chakrabartty, A.; Galant, N.J.; Hadley, K.C.; Hammerson, B.; Nijjar, T.; Torres, R.; Tapia, J.R.; Salmans, J.; Barbour, R.; et al. Novel conformation-specific monoclonal antibodies against amyloidogenic forms of transthyretin. Amyloid Int. J. Exp. Clin. Investig. Off. J. Int. Soc. Amyloidosis 2016, 23, 86–97. [Google Scholar] [CrossRef]
- Michalon, A.; Hagenbuch, A.; Huy, C.; Varela, E.; Combaluzier, B.; Damy, T.; Suhr, O.B.; Saraiva, M.J.; Hock, C.; Nitsch, R.M.; et al. A human antibody selective for transthyretin amyloid removes cardiac amyloid through phagocytic immune cells. Nat. Commun. 2021, 12, 3142. [Google Scholar] [CrossRef]
- Garcia-Pavia, P.; Aus dem Siepen, F.; Donal, E.; Lairez, O.; van der Meer, P.; Kristen, A.V.; Mercuri, M.F.; Michalon, A.; Frost, R.J.A.; Grimm, J.; et al. Phase 1 Trial of Antibody NI006 for Depletion of Cardiac Transthyretin Amyloid. N. Engl. J. Med. 2023, 389, 239–250. [Google Scholar] [CrossRef]
- Suhr, O.B.; Grogan, M.; Silva, A.M.D.; Karam, C.; Garcia-Pavia, P.; Drachman, B.; Zago, W.; Tripuraneni, R.; Kinney, G.G. PRX004 in variant amyloid transthyretin (ATTRv) amyloidosis: Results of a phase 1, open-label, dose-escalation study. Amyloid 2024, 32, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Ternacle, J.; Krapf, L.; Mohty, D.; Magne, J.; Nguyen, A.; Galat, A.; Gallet, R.; Teiger, E.; Côté, N.; Clavel, M.A.; et al. Aortic Stenosis and Cardiac Amyloidosis: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 74, 2638–2651. [Google Scholar] [CrossRef] [PubMed]

| Trial Name | Drug Studied | Mechanism of Action | Baseline Characteristics (Treatment Group) | Key Results | Survival at 30 Months |
|---|---|---|---|---|---|
| ATTR-ACT NEJM, 2018 | Tafamidis | TTR stabilizer | 91.3% Men 79.9% White Median age: 75 years Median NT-proBNP (pg/mL): 2995.9 NYHA II symptoms: 61.4% NYHA III symptoms: 29.5% | 441 patients enrolled 267 → Tafamidis 177 → Placebo 30% reduction in all-cause mortality (29.5% in tafamidis group vs. 42.9% in placebo group, HR 0.70; 95% CI 0.51 to 0.96) 32% reduction in cardiovascular-related hospitalizations (0.48 per year vs. 0.70 per year; RR 0.68, 95% CI, 0.56 to 0.81) | Pooled Tafamidis: 70.5% Placebo: 57.1% RRR: 32% |
| ATTRibute-CM NEJM, 2024 | Acoramidis | TTR stabilizer | 91.2% Men 87.4% White Median age: 77 years Median NT-pro BNP (pg/mL): 2326 NYHA II symptoms: 69.6% NYHA III symptoms: 18.3% | 632 patients enrolled 421 → Acoramidis 211 → Placebo Four-step primary hierarchical analysis (includes death from any cause, CV-related hospitalization, change from baseline in NT-proBNP level, and change from baseline in 6 MWD) yielded a win ratio of 1.8 (95% CI, 1.4 to 2.2) in acoramidis group vs. placebo | Acoramidis: 80.7% Placebo: 74.3% RRR: 25% |
| APOLLO-B NEJM, 2023 | Patisiran | RNA interference (RNAi) therapeutic | 89% Men 77% White Median age: 76 years Median NT-pro BNP (pg/mL): 2008 NYHA II symptoms: 86% NYHA III symptoms: 8% | 360 patients enrolled 181 → Patisiran 179 → Placebo At 12 months, magnitude of decline in 6 MWD was significantly lower in the treatment group (median change from baseline of −8.15 m (95% confidence interval [CI], −16.42 to 1.50) in the patisiran group and −21.35 m (95% CI, −34.05 to −7.52) in the placebo group | 30-month data N/A 12-month follow-up period did not provide sufficient power to assess the effects of treatment on end points related to mortality and hospitalization |
| HELIOS-B NEJM, 2024 | Vutrisiran | RNAi therapeutic | 92% Men 85% White Median age: 77 years Median NT-pro BNP (pg/mL): 2021 NYHA II symptoms: 77% NYHA III symptoms: 8% | 655 patients enrolled 326 → Vutrisiran 329 → Placebo Approximately 60% of both groups not on tafamidis (monotherapy group) Vutrisiran showed a significant reduction in mortality, with a HR of 0.72; 95% CI, 0.56 to 0.93; p = 0.01, and a HR of 0.67 in the monotherapy population, 95% CI, 0.49 to 0.93; p = 0.02 | Vutrisiran: 84% Placebo: 79% RRR: 24% |
| CARDIO-TTRansform | Eplontersen | Antisense oligonucleotide | Inclusion Criteria: Confirmed diagnosis of ATTR-CM End-diastolic interventricular septum thickness of >12 mm (mm) New York Heart Association (NYHA) class I-III | Trial is ongoing; results are pending. | N/A |
| DepleTTR-CM | ALXN2220 | Monoclonal antibody against misfolded TTR | Inclusion criteria: Confirmed diagnosis of ATTR-CM End-diastolic septal wall thickness > 11 mm for women or >12 mm for men NT-proBNP > 2000 pg/mL Treatment with loop diuretic 30 days prior to screening NYHA class II-IV Life expectancy of >6 months | Trial is ongoing: results are pending | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wanniarachchige, D.; Khan, S.; Pan, S. Transthyretin Amyloid Cardiomyopathy Treatment: An Updated Review. J. Clin. Med. 2025, 14, 6089. https://doi.org/10.3390/jcm14176089
Wanniarachchige D, Khan S, Pan S. Transthyretin Amyloid Cardiomyopathy Treatment: An Updated Review. Journal of Clinical Medicine. 2025; 14(17):6089. https://doi.org/10.3390/jcm14176089
Chicago/Turabian StyleWanniarachchige, Dinusha, Shazli Khan, and Stephen Pan. 2025. "Transthyretin Amyloid Cardiomyopathy Treatment: An Updated Review" Journal of Clinical Medicine 14, no. 17: 6089. https://doi.org/10.3390/jcm14176089
APA StyleWanniarachchige, D., Khan, S., & Pan, S. (2025). Transthyretin Amyloid Cardiomyopathy Treatment: An Updated Review. Journal of Clinical Medicine, 14(17), 6089. https://doi.org/10.3390/jcm14176089






