The Effect of Aerobic Exercise Training on Patients with Type III Spinal Muscular Atrophy
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Ethical Approval
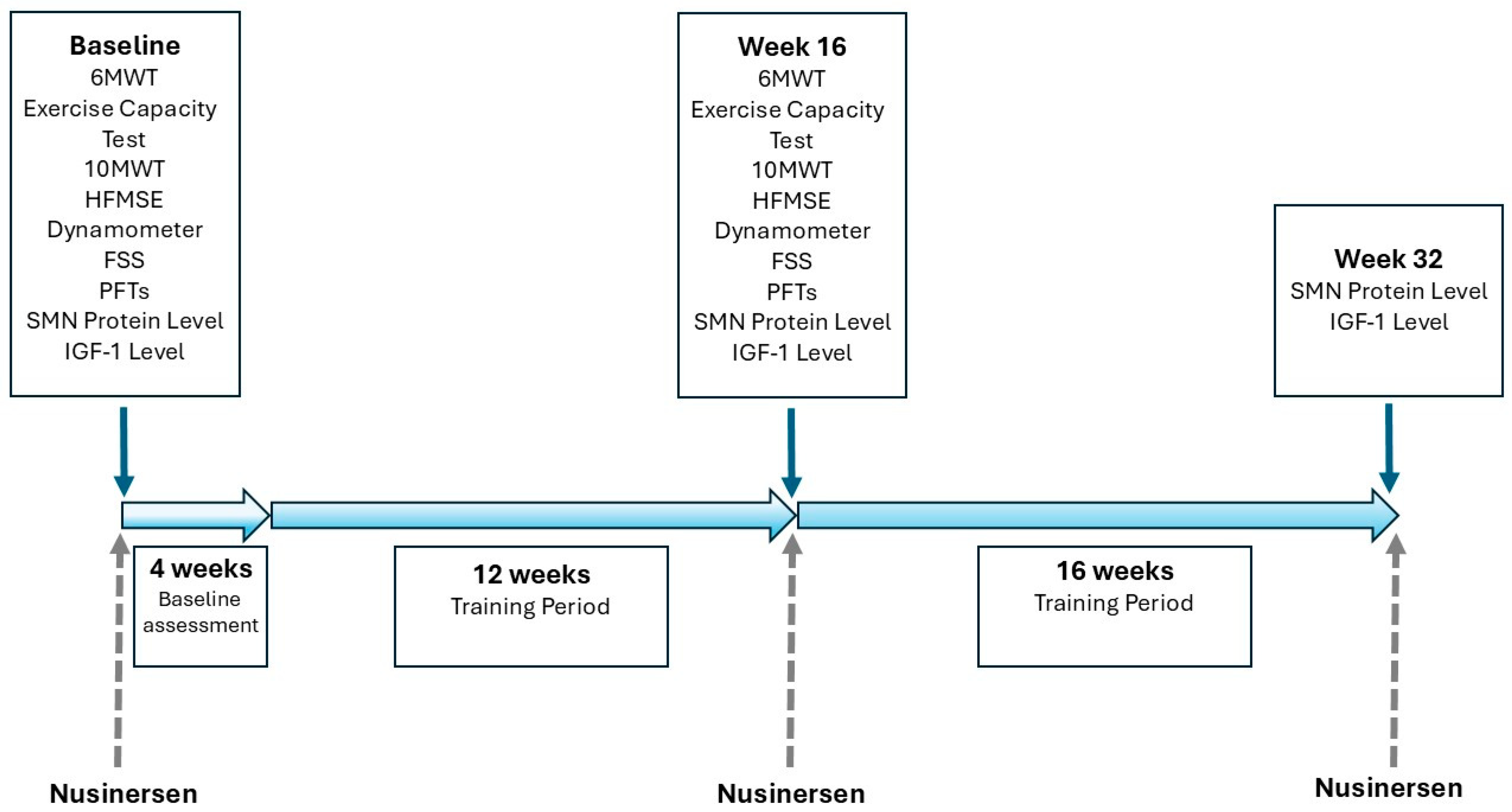
2.2. Study Design
2.3. Aerobic Exercise Training Protocol
2.4. Outcome Measures
2.5. Primary Outcomes
2.6. Secondary Outcomes
2.7. Analysis of IGF-1 in Serum Samples
2.8. Western Blot Testing
2.9. Statistical Analysis
3. Results
3.1. Demographic and Clinical Patient Characteristics
3.2. The Effect of 12 Weeks of Aerobic Exercise Training on Physical Function
3.3. The Effect of Aerobic Exercise Training on Blood SMN Protein and IGF-1 Levels
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lefebvre, S.; Burglen, L.; Reboullet, S.; Clermont, O.; Burlet, P.; Viollet, L.; Benichou, B.; Cruaud, C.; Millasseau, P.; Zeviani, M.; et al. Identification and characterization of a spinal muscular atrophy-determining gene. Cell 1995, 80, 155–165. [Google Scholar] [CrossRef]
- Kolb, S.J.; Kissel, J.T. Spinal muscular atrophy: A timely review. Arch. Neurol. 2011, 68, 979–984. [Google Scholar] [CrossRef]
- Darras, B.T. Spinal muscular atrophies. Pediatr. Clin. N. Am. 2015, 62, 743–766. [Google Scholar] [CrossRef]
- Chaytow, H.; Huang, Y.T.; Gillingwater, T.H.; Faller, K.M.E. The role of survival motor neuron protein (SMN) in protein homeostasis. Cell Mol. Life Sci. 2018, 75, 3877–3894. [Google Scholar] [CrossRef]
- Markowitz, J.A.; Singh, P.; Darras, B.T. Spinal muscular atrophy: A clinical and research update. Pediatr. Neurol. 2012, 46, 1–12. [Google Scholar] [CrossRef]
- Mercuri, E.; Bertini, E.; Iannaccone, S.T. Childhood spinal muscular atrophy: Controversies and challenges. Lancet Neurol. 2012, 11, 443–452. [Google Scholar] [CrossRef]
- Scoto, M.; Finkel, R.S.; Mercuri, E.; Muntoni, F. Therapeutic approaches for spinal muscular atrophy (SMA). Gene Ther. 2017, 24, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Lorson, C.L.; Rindt, H.; Shababi, M. Spinal muscular atrophy: Mechanisms and therapeutic strategies. Hum. Mol. Genet. 2010, 19, R111–R118. [Google Scholar] [CrossRef] [PubMed]
- Chiriboga, C.A.; Swoboda, K.J.; Darras, B.T.; Iannaccone, S.T.; Montes, J.; De Vivo, D.C.; Norris, D.A.; Bennett, C.F.; Bishop, K.M. Results from a phase 1 study of nusinersen (ISIS-SMN(Rx)) in children with spinal muscular atrophy. Neurology 2016, 86, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Trenkle, J.; Brugman, J.; Peterson, A.; Roback, K.; Krosschell, K.J. Filling the gaps in knowledge translation: Physical therapy recommendations for individuals with spinal muscular atrophy compared to standard of care guidelines. Neuromuscul. Disord. 2021, 31, 397–408. [Google Scholar] [CrossRef]
- Wang, C.H.; Finkel, R.S.; Bertini, E.S.; Schroth, M.; Simonds, A.; Wong, B.; Aloysius, A.; Morrison, L.; Main, M.; Crawford, T.O.; et al. Consensus statement for standard of care in spinal muscular atrophy. J. Child. Neurol. 2007, 22, 1027–1049. [Google Scholar] [CrossRef]
- Grondard, C.; Biondi, O.; Armand, A.S.; Lecolle, S.; Della Gaspera, B.; Pariset, C.; Li, H.; Gallien, C.L.; Vidal, P.P.; Chanoine, C.; et al. Regular exercise prolongs survival in a type 2 spinal muscular atrophy model mouse. J. Neurosci. 2005, 25, 7615–7622. [Google Scholar] [CrossRef]
- Biondi, O.; Branchu, J.; Ben Salah, A.; Houdebine, L.; Bertin, L.; Chali, F.; Desseille, C.; Weill, L.; Sanchez, G.; Lancelin, C.; et al. IGF-1R Reduction Triggers Neuroprotective Signaling Pathways in Spinal Muscular Atrophy Mice. J. Neurosci. 2015, 35, 12063–12079. [Google Scholar] [CrossRef]
- Bosch-Marce, M.; Wee, C.D.; Martinez, T.L.; Lipkes, C.E.; Choe, D.W.; Kong, L.; Van Meerbeke, J.P.; Musaro, A.; Sumner, C.J. Increased IGF-1 in muscle modulates the phenotype of severe SMA mice. Hum. Mol. Genet. 2011, 20, 1844–1853. [Google Scholar] [CrossRef] [PubMed]
- Lewelt, A.; Krosschell, K.J.; Stoddard, G.J.; Weng, C.; Xue, M.; Marcus, R.L.; Gappmaier, E.; Viollet, L.; Johnson, B.A.; White, A.T.; et al. Resistance strength training exercise in children with spinal muscular atrophy. Muscle Nerve 2015, 52, 559–567. [Google Scholar] [CrossRef]
- Montes, J.; Garber, C.E.; Kramer, S.S.; Montgomery, M.J.; Dunaway, S.; Kamil-Rosenberg, S.; Carr, B.; Cruz, R.; Strauss, N.E.; Sproule, D.; et al. Single-Blind, Randomized, Controlled Clinical Trial of Exercise in Ambulatory Spinal Muscular Atrophy: Why are the Results Negative? J. Neuromuscul. Dis. 2015, 2, 463–470. [Google Scholar] [CrossRef]
- Madsen, K.L.; Hansen, R.S.; Preisler, N.; Thogersen, F.; Berthelsen, M.P.; Vissing, J. Training improves oxidative capacity, but not function, in spinal muscular atrophy type III. Muscle Nerve 2015, 52, 240–244. [Google Scholar] [CrossRef]
- Bora, G.; Subasi-Yildiz, S.; Yesbek-Kaymaz, A.; Bulut, N.; Alemdaroglu, I.; Tunca-Yilmaz, O.; Topaloglu, H.; Karaduman, A.A.; Erdem-Yurter, H. Effects of Arm Cycling Exercise in Spinal Muscular Atrophy Type II Patients: A Pilot Study. J. Child. Neurol. 2018, 33, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Salem, Y.; Gropack, S.J. Aquatic therapy for a child with type III spinal muscular atrophy: A case report. Phys. Occup. Ther. Pediatr. 2010, 30, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Bulut, N.; Yardimci, B.N.; Ayvat, E.; Aran, O.T.; Yilmaz, O.; Karaduman, A. The effect of two different aerobic training modalities in a child with spinal muscular atrophy type II: A case report. J. Exerc. Rehabil. 2019, 15, 322–326. [Google Scholar] [CrossRef]
- Dobrowolny, G.; Giacinti, C.; Pelosi, L.; Nicoletti, C.; Winn, N.; Barberi, L.; Molinaro, M.; Rosenthal, N.; Musaro, A. Muscle expression of a local Igf-1 isoform protects motor neurons in an ALS mouse model. J. Cell Biol. 2005, 168, 193–199. [Google Scholar] [CrossRef]
- Simone, C.; Ramirez, A.; Bucchia, M.; Rinchetti, P.; Rideout, H.; Papadimitriou, D.; Re, D.B.; Corti, S. Is spinal muscular atrophy a disease of the motor neurons only: Pathogenesis and therapeutic implications? Cell Mol. Life Sci. 2016, 73, 1003–1020. [Google Scholar] [CrossRef]
- Mercuri, E.; Darras, B.T.; Chiriboga, C.A.; Day, J.W.; Campbell, C.; Connolly, A.M.; Iannaccone, S.T.; Kirschner, J.; Kuntz, N.L.; Saito, K.; et al. Nusinersen versus Sham Control in Later-Onset Spinal Muscular Atrophy. N. Engl. J. Med. 2018, 378, 625–635. [Google Scholar] [CrossRef]
- Karvonen, J.; Vuorimaa, T. Heart rate and exercise intensity during sports activities. Practical application. Sports Med. 1988, 5, 303–311. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Voorn, E.L.; Gerrits, K.H.; Koopman, F.S.; Nollet, F.; Beelen, A. Determining the anaerobic threshold in postpolio syndrome: Comparison with current guidelines for training intensity prescription. Arch. Phys. Med. Rehabil. 2014, 95, 935–940. [Google Scholar] [CrossRef]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Dunaway Young, S.; Montes, J.; Kramer, S.S.; Marra, J.; Salazar, R.; Cruz, R.; Chiriboga, C.A.; Garber, C.E.; De Vivo, D.C. Six-minute walk test is reliable and valid in spinal muscular atrophy. Muscle Nerve 2016, 54, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Coratti, G.; Bovis, F.; Pera, M.C.; Scoto, M.; Montes, J.; Pasternak, A.; Mayhew, A.; Muni-Lofra, R.; Duong, T.; Rohwer, A.; et al. Determining minimal clinically important differences in the Hammersmith Functional Motor Scale Expanded for untreated spinal muscular atrophy patients: An international study. Eur. J. Neurol. 2024, 31, e16309. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.L.; Steenbruggen, I.; Miller, M.R.; Barjaktarevic, I.Z.; Cooper, B.G.; Hall, G.L.; Hallstrand, T.S.; Kaminsky, D.A.; McCarthy, K.; McCormack, M.C.; et al. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am. J. Respir. Crit. Care Med. 2019, 200, e70–e88. [Google Scholar] [CrossRef] [PubMed]
- Merlini, L.; Mazzone, E.S.; Solari, A.; Morandi, L. Reliability of hand-held dynamometry in spinal muscular atrophy. Muscle Nerve 2002, 26, 64–70. [Google Scholar] [CrossRef]
- Krosschell, K.J.; Townsend, E.L.; Kiefer, M.; Simeone, S.D.; Zumpf, K.; Welty, L.; Swoboda, K.J. Natural history of 10-m walk/run test performance in spinal muscular atrophy: A longitudinal analysis. Neuromuscul. Disord. 2022, 32, 125–134. [Google Scholar] [CrossRef]
- Werlauff, U.; Hojberg, A.; Firla-Holme, R.; Steffensen, B.F.; Vissing, J. Fatigue in patients with spinal muscular atrophy type II and congenital myopathies: Evaluation of the fatigue severity scale. Qual. Life Res. 2014, 23, 1479–1488. [Google Scholar] [CrossRef]
- Orngreen, M.C.; Olsen, D.B.; Vissing, J. Aerobic training in patients with myotonic dystrophy type 1. Ann. Neurol. 2005, 57, 754–757. [Google Scholar] [CrossRef]
- Bartels, B.; Montes, J.; van der Pol, W.L.; de Groot, J.F. Physical exercise training for type 3 spinal muscular atrophy. Cochrane Database Syst. Rev. 2019, 3, CD012120. [Google Scholar] [CrossRef]
- Perera, S.; Mody, S.H.; Woodman, R.C.; Studenski, S.A. Meaningful change and responsiveness in common physical performance measures in older adults. J. Am. Geriatr. Soc. 2006, 54, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Kirn, D.R.; Reid, K.F.; Hau, C.; Phillips, E.M.; Fielding, R.A. What is a Clinically Meaningful Improvement in Leg-Extensor Power for Mobility-limited Older Adults? J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Sumner, C.J.; Kolb, S.J.; Harmison, G.G.; Jeffries, N.O.; Schadt, K.; Finkel, R.S.; Dreyfuss, G.; Fischbeck, K.H. SMN mRNA and protein levels in peripheral blood: Biomarkers for SMA clinical trials. Neurology 2006, 66, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, B.K.; Frost, L.M.; Christian, L.; Umapathi, P.; Gage, F.H. Synergy of insulin-like growth factor-1 and exercise in amyotrophic lateral sclerosis. Ann. Neurol. 2005, 57, 649–655. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-Induced Neuroplasticity: A Mechanistic Model and Prospects for Promoting Plasticity. Neuroscientist 2019, 25, 65–85. [Google Scholar] [CrossRef]
- Tsai, L.K.; Chen, C.L.; Ting, C.H.; Lin-Chao, S.; Hwu, W.L.; Dodge, J.C.; Passini, M.A.; Cheng, S.H. Systemic administration of a recombinant AAV1 vector encoding IGF-1 improves disease manifestations in SMA mice. Mol. Ther. 2014, 22, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- Shababi, M.; Glascock, J.; Lorson, C.L. Combination of SMN trans-splicing and a neurotrophic factor increases the life span and body mass in a severe model of spinal muscular atrophy. Hum. Gene Ther. 2011, 22, 135–144. [Google Scholar] [CrossRef] [PubMed]
| Training Group | Home-Exercise Group | p-Value | |
|---|---|---|---|
| (n = 12) | (n = 11) | ||
| Age (year) | 28.50 (10.00) | 30.00 (21.00) | 0.487 |
| Height (cm) | 169.50 (17.00) | 167.00 (12.00) | 0.928 |
| Weight (kg) | 62.50 (25.00) | 56.00 (24.00) | 0.449 |
| BMI (kg/m2) | 22.65 (4.30) | 19.80 (6.30) | 0.487 |
| HFMSE Total | 58.50 (6.00) | 55.00 (13.00) | 0.449 |
| Age at symptom onset (month) | 84.00 (123.00) | 84.00 (96.00) | 0.928 |
| Gender | 0.292 | ||
| Male (n) | 7 (58.30%) | 4 (36.40%) | |
| Female (n) | 5 (41.70%) | 7 (63.60%) | |
| HFMSE Total | 55.00 (12.00) | 58.50 (6.00) | 0.379 |
| 6MWT Distance (m) | 307.00 (215.00) | 382.50 (170.25) | 0.413 |
| Exercise Capacity (mL/kg/min) | 9.62 (4.40) | 14.65 (10.12) | 0.374 |
| FVC (% predicted) | 86.00 (23.00) | 90.00 (18.00) | 0.740 |
| FEV1 (% predicted) | 92.00 (22.00) | 95.50 (23.00) | 0.976 |
| PEF (% predicted) | 87.00 (31.00) | 81.00 (23.75) | 0.449 |
| FVC (L) | 3.53 (1.42) | 3.84 (1.79) | 0.566 |
| FEV1 (L) | 3.08 (1.02) | 3.51 (1.81) | 0.566 |
| PEF (L) | 6.01 (3.79) | 5.96 (2.21) | 0.928 |
| HHD Quadriceps right force (kg) | 3.17 (2.00) | 3.63 (1.87) | 0.608 |
| HHD Quadriceps left force (kg) | 3.13 (2.04) | 3.27 (1.98) | 0.976 |
| HHD Gastrocnemius right force (kg) | 7.30 (1.86) | 8.07 (1.19) | 0.044 |
| HHD Gastrocnemius left force (kg) | 7.12 (1.32) | 7.26 (1.21) | 0.525 |
| 10MWT (s) | 8.34 (4.20) | 7.29 (1.85) | 0.487 |
| FSS | 4.40 (1.20) | 4.10 (1.48) | 0.651 |
| SNM Protein | 0.70 (0.64) | 0.65 (0.69) | 0.674 |
| IGF-1 (ng/mL) | 3.40 (2.05) | 4.09 (4.95) | 0.190 |
| Training Group (n = 12) | Home-Exercise Groups (n = 11) | p Value | ||
|---|---|---|---|---|
| HFMSE Total | Pre-I | 58.50 (5.75) | 55.00 (12.00) | 0.695 * |
| Post-I | 60.00 (6.75) | 55.00 (13.00) | ||
| Δ | 0.00 (1.50) | 0.00 (0.00) | ||
| p | 0.102 # | 0.180 # | ||
| 6MWT Distance (m) | Pre-I | 382.50 (170.25) | 307.00 (215.00) | 0.169 * |
| Post-I | 409.00 (186.00) | 346.00 (230.00) | ||
| Δ | 30.50 (16.75) | 0.00 (49.00) | ||
| p | 0.003 # | 0.213 # | ||
| Exercise Capacity (mL/kg/min) | Pre-I | 14.65 (10.13) | 9.60 (4.40) | <0.001 * |
| Post-I | 19.63 (12.59) | 11.00 (6.60) | ||
| Δ | 5.44 (4.48) | 0.51 (1.47) | ||
| p | 0.002 # | 0.028 # | ||
| HHD Quadriceps right force (kg) | Pre-I | 3.63 (1.87) | 3.17 (2.00) | 0.004 * |
| Post-I | 4.78 (1.26) | 3.10 (1.41) | ||
| Δ | 1.00 (1.46) | 0.04 (0.27) | ||
| p | 0.005 # | 0.561 # | ||
| HHD Quadriceps left force (kg) | Pre-I | 3.27 (1.98) | 3.13 (2.04) | 0.069 * |
| Post-I | 4.60 (2.08) | 3.22 (1.77) | ||
| Δ | 0.52 (1.73) | 0.04 (0.54) | ||
| p | 0.031 # | 0.859 # | ||
| HHD Gastrocnemius right force (kg) | Pre-I | 8.07 (1.19) | 7.30 (1.86) | 0.104 * |
| Post-I | 9.18 (1.86) | 7.93 (2.10) | ||
| Δ | 0.95 (1.92) | 0.00 (0.59) | ||
| p | 0.034 # | 0.373 # | ||
| HHD Gastrocnemius left force (kg) | Pre-I | 7.26 (1.21) | 7.12 (1.32) | 0.169 * |
| Post-I | 8.35 (1.29) | 7.12 (1.35) | ||
| Δ | 1.00 (2.29) | −0.18 (1.09) | ||
| p | 0.068 # | 0.306 # | ||
| 10MWT (seconds) | Pre-I | 7.29 (1.85) | 8.34 (4.20) | 0.079 * |
| Post-I | 6.24 (3.21) | 8.47 (3.30) | ||
| Δ | −1.15 (1.57) | −0.03 (0.97) | ||
| p | 0.019 # | 0.722 # | ||
| FSS | Pre-I | 4.10 (1.48) | 4.40 (1.20) | 0.037 * |
| Post-I | 3.20 (1.05) | 4.20 (0.50) | ||
| Δ | −0.90 (1.25) | −0.20 (0.90) | ||
| p | 0.005 # | 0.476 # |
| Training Group (n = 12) | Home-Exercise Groups (n = 11) | p Value | ||
|---|---|---|---|---|
| FVC (% predicted) | Pre-I | 90.00 (18.00) | 86.00 (23.00) | 0.091 * |
| Post-I | 92.50 (17.25) | 88.00 (23.00) | ||
| Δ | 3.50 (4.00) | −2.00 (8.00) | ||
| p value | 0.016 # | 0.475 # | ||
| FEV1 (% predicted) | Pre-I | 95.50 (23.00) | 92.00 (22.00) | 0.190 * |
| Post-I | 95.00 (21.00) | 90.00 (22.00) | ||
| Δ | 0.50 (2.75) | −4.00 (8.00) | ||
| p value | 0.535 # | 0.247 # | ||
| PEF (% predicted) | Pre-I | 81.00 (23.75) | 87.00 (31.00) | 0.880 * |
| Post-I | 74.50 (26.75) | 84.00 (16.00) | ||
| Δ | −2.00 (12.25) | 0.00 (19.00) | ||
| p value | 0.783 # | 0.838 # | ||
| FVC (L) | Pre-I | 3.84 (1.79) | 3.50 (1.40) | 0.169 * |
| Post-I | 4.02 (2.03) | 3.70 (0.70) | ||
| Δ | 0.09 (0.18) | −0.11 (0.38) | ||
| p value | 0.116 # | 0.398 # | ||
| FEV1 (L) | Pre-I | 3.51 (1.81) | 3.08 (1.02) | 0.169 * |
| Post-I | 3.58 (1.60) | 3.06 (1.04) | ||
| Δ | 0.02 (0.35) | −0.10 (0.26) | ||
| p value | 0.505 # | 0.213 # | ||
| PEF (L) | Pre-I | 5.96 (2.21) | 6.01 (3.79) | 0.833 * |
| Post-I | 6.56 (2.54) | 6.01 (1.74) | ||
| Δ | −0.10 (1.06) | −0.06 (1.40) | ||
| p value | 0.814 # | 1.000 # |
| Training Group (n = 12) | Home-Exercise Groups (n = 11) | p Value * | ||
|---|---|---|---|---|
| SMN Protein | Pre-I | 0.65 (0.70) | 0.70 (0.64) | 0.298 a |
| Post-I | 1.18 (1.07) | 0.94 (0.91) | ||
| Post-II. | 1.42 (2.20) | 1.11 (0.83) | ||
| p value | 0.027 c | 0.651 c | ||
| Pre-I.–Post-I | 0.539 b | |||
| Pre-I–Post-II. | 0.022 b | |||
| Post-I–Post-II | 0.539 b | |||
| IGF-1 (ng/mL) | Pre-I | 4.09 (4.95) | 3.40 (2.05) | 0.900 a |
| Post-I | 4.04 (3.98) | 3.69 (1.21) | ||
| Post-II | 8.41 (14.24) | 4.12 (1.57) | ||
| p value | 0.014 c | 0.169 c | ||
| Pre-I–Post-I | 1.000 b | |||
| Pre-I–Post-II | 0.016 b | |||
| Post-I–Post-II | 0.172 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mergen Kilic, S.; Mutluay, F.; Cakar, A.; Giris, M.; Durmus, H.; Bingul, I.; Gedikbasi, A.; Kucukgergin, C.; Uyguner, Z.O.; Parman, Y. The Effect of Aerobic Exercise Training on Patients with Type III Spinal Muscular Atrophy. J. Clin. Med. 2025, 14, 6087. https://doi.org/10.3390/jcm14176087
Mergen Kilic S, Mutluay F, Cakar A, Giris M, Durmus H, Bingul I, Gedikbasi A, Kucukgergin C, Uyguner ZO, Parman Y. The Effect of Aerobic Exercise Training on Patients with Type III Spinal Muscular Atrophy. Journal of Clinical Medicine. 2025; 14(17):6087. https://doi.org/10.3390/jcm14176087
Chicago/Turabian StyleMergen Kilic, Sezan, Fatma Mutluay, Arman Cakar, Murat Giris, Hacer Durmus, Ilknur Bingul, Asuman Gedikbasi, Canan Kucukgergin, Zehra Oya Uyguner, and Yesim Parman. 2025. "The Effect of Aerobic Exercise Training on Patients with Type III Spinal Muscular Atrophy" Journal of Clinical Medicine 14, no. 17: 6087. https://doi.org/10.3390/jcm14176087
APA StyleMergen Kilic, S., Mutluay, F., Cakar, A., Giris, M., Durmus, H., Bingul, I., Gedikbasi, A., Kucukgergin, C., Uyguner, Z. O., & Parman, Y. (2025). The Effect of Aerobic Exercise Training on Patients with Type III Spinal Muscular Atrophy. Journal of Clinical Medicine, 14(17), 6087. https://doi.org/10.3390/jcm14176087






