Predictors of Post-Intensive Care Syndrome in ICU Survivors After Discharge: An Observational Study
Abstract
1. Introduction
Aim
2. Materials and Methods
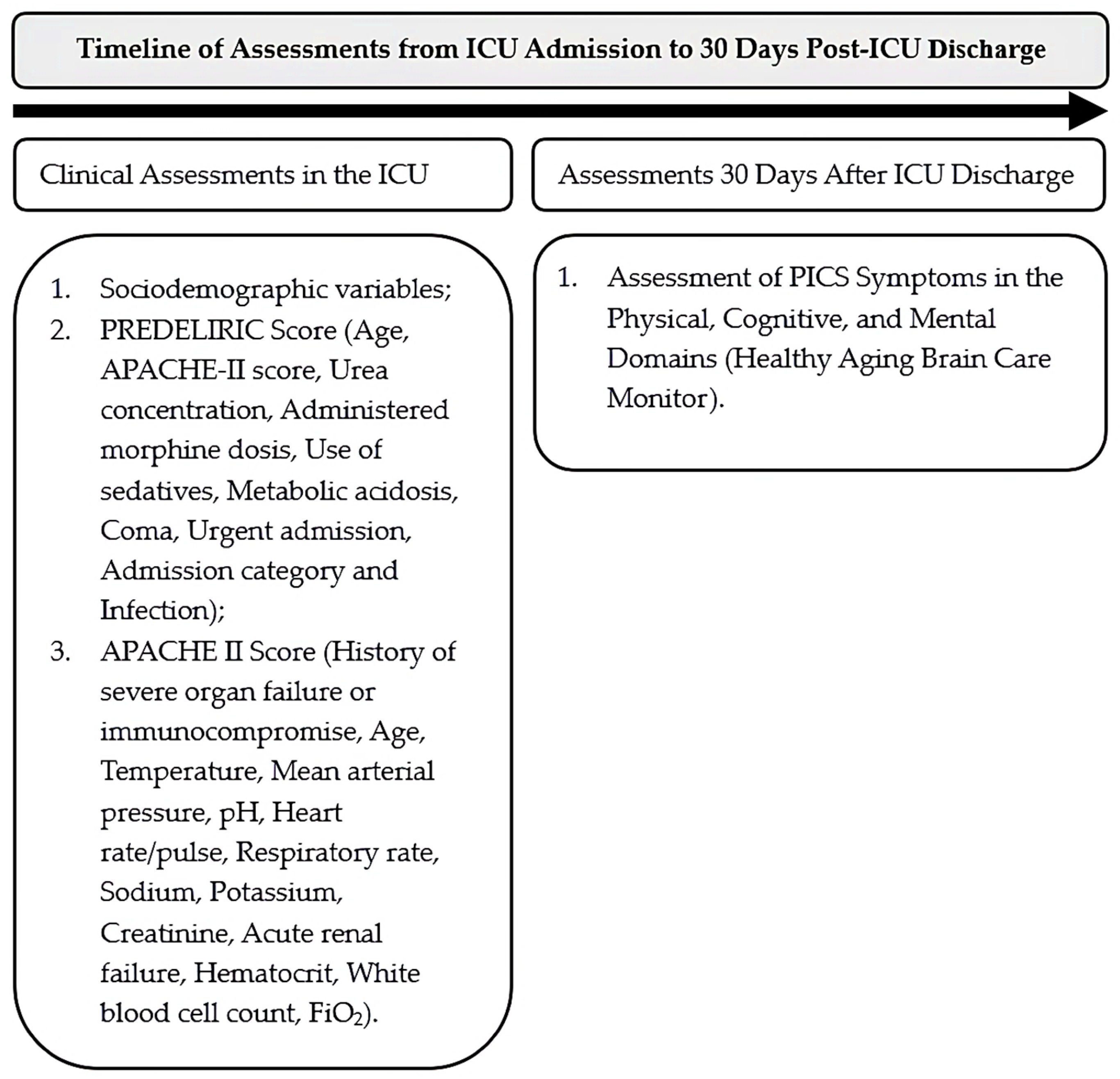
2.1. Study Design and Research Questions
2.2. Data Collection and Participants
2.3. Variables and Predictors
2.4. Outcome
2.5. Instruments
2.6. Statistical Analysis
2.7. Ethical Considerations
3. Results
3.1. Sociodemographic Characteristics of the Sample and PICS
3.2. Multivariate Regression Models
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johnson, C. Design, Organization and Staffing of the Intensive Care Unit. Surgery 2018, 36, 159–165. [Google Scholar] [CrossRef]
- Doherty, Z.; Kippen, R.; Bevan, D.; Duke, G.; Williams, S.; Wilson, A.; Pilcher, D. Long-Term Outcomes of Hospital Survivors Following an ICU Stay: A Multi-Centre Retrospective Cohort Study. PLoS ONE 2022, 17, e0266038. [Google Scholar] [CrossRef] [PubMed]
- Gravante, F.; Trotta, F.; Latina, S.; Simeone, S.; Alvaro, R.; Vellone, E.; Pucciarelli, G. Quality of Life in ICU Survivors and Their Relatives with Post-intensive Care Syndrome: A Systematic Review. Nurs. Crit. Care 2024, 29, 807–823. [Google Scholar] [CrossRef] [PubMed]
- Needham, D.M.; Davidson, J.; Cohen, H.; Hopkins, R.O.; Weinert, C.; Wunsch, H.; Zawistowski, C.; Bemis-Dougherty, A.; Berney, S.C.; Bienvenu, O.J.; et al. Improving Long-Term Outcomes after Discharge from Intensive Care Unit: Report from a Stakeholders’ Conference. Crit. Care Med. 2012, 40, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Schwitzer, E.; Jensen, K.S.; Brinkman, L.; DeFrancia, L.; VanVleet, J.; Baqi, E.; Aysola, R.; Qadir, N. Survival ≠ Recovery. Chest Crit. Care 2023, 1, 100003. [Google Scholar] [CrossRef]
- Yuan, C.; Timmins, F.; Thompson, D.R. Post-Intensive Care Syndrome: A Concept Analysis. Int. J. Nurs. Stud. 2021, 114, 103814. [Google Scholar] [CrossRef]
- Bottom-Tanzer, S.F.; Poyant, J.O.; Louzada, M.T.; Ahmed, S.E.; Boudouvas, A.; Poon, E.; Hojman, H.M.; Bugaev, N.; Johnson, B.P.; Van Kirk, A.L.; et al. High Occurrence of Postintensive Care Syndrome Identified in Surgical ICU Survivors after Implementation of a Multidisciplinary Clinic. J. Trauma Acute Care Surg. 2021, 91, 406–412. [Google Scholar] [CrossRef]
- Kawakami, D.; Fujitani, S.; Morimoto, T.; Dote, H.; Takita, M.; Takaba, A.; Hino, M.; Nakamura, M.; Irie, H.; Adachi, T.; et al. Prevalence of Post-Intensive Care Syndrome among Japanese Intensive Care Unit Patients: A Prospective, Multicenter, Observational J-PICS Study. Crit. Care 2021, 25, 69. [Google Scholar] [CrossRef]
- Lee, M.; Kang, J.; Jeong, Y.J. Risk Factors for Post–Intensive Care Syndrome: A Systematic Review and Meta-Analysis. Aust. Crit. Care 2020, 33, 287–294. [Google Scholar] [CrossRef]
- Gupta, L.; Subair, M.N.; Munjal, J.; Singh, B.; Bansal, V.; Gupta, V.; Jain, R. Beyond Survival: Understanding Post-Intensive Care Syndrome. Acute Crit. Care 2024, 39, 226–233. [Google Scholar] [CrossRef]
- Inoue, S.; Nakanishi, N.; Sugiyama, J.; Moriyama, N.; Miyazaki, Y.; Sugimoto, T.; Fujinami, Y.; Ono, Y.; Kotani, J. Prevalence and Long-Term Prognosis of Post-Intensive Care Syndrome after Sepsis: A Single-Center Prospective Observational Study. J. Clin. Med. 2022, 11, 5257. [Google Scholar] [CrossRef]
- Van Beusekom, I.; Bakhshi-Raiez, F.; De Keizer, N.F.; Van Der Schaaf, M.; Busschers, W.B.; Dongelmans, D.A. Healthcare Costs of ICU Survivors Are Higher before and after ICU Admission Compared to a Population Based Control Group: A Descriptive Study Combining Healthcare Insurance Data and Data from a Dutch National Quality Registry. J. Crit. Care 2018, 44, 345–351. [Google Scholar] [CrossRef]
- Gravante, F.; Giannarelli, D.; Pucci, A.; Pisani, L.; Latina, R. Calibration of the PREdiction of DELIRium in ICu Patients (PRE-DELIRIC) Score in a Cohort of Critically Ill Patients: A Retrospective Cohort Study. Dimens. Crit. Care Nurs. 2023, 42, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Villa, M.; Villa, S.; Vimercati, S.; Andreossi, M.; Mauri, F.; Ferlicca, D.; Rona, R.; Foti, G.; Lucchini, A. Implementation of a Follow-Up Program for Intensive Care Unit Survivors. Int. J. Environ. Res. Public Health 2021, 18, 10122. [Google Scholar] [CrossRef] [PubMed]
- Renner, C.; Jeitziner, M.-M.; Albert, M.; Brinkmann, S.; Diserens, K.; Dzialowski, I.; Heidler, M.-D.; Lück, M.; Nusser-Müller-Busch, R.; Sandor, P.S.; et al. Guideline on Multimodal Rehabilitation for Patients with Post-Intensive Care Syndrome. Crit. Care 2023, 27, 301. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, A.-F.; Prescott, H.C.; Brett, S.J.; Weiss, B.; Azoulay, E.; Creteur, J.; Latronico, N.; Hough, C.L.; Weber-Carstens, S.; Vincent, J.-L.; et al. Long-Term Outcomes after Critical Illness: Recent Insights. Crit. Care 2021, 25, 108. [Google Scholar] [CrossRef]
- Gao, S.; Liang, X.; Pan, Z.; Zhang, X.; Zhang, L. Effect Size Estimates of Risk Factors for Post-Intensive Care Syndrome: A Systematic Review and Meta-Analysis. Intensive Crit. Care Nurs. 2025, 87, 103888. [Google Scholar] [CrossRef]
- Chu, Y.; Thompson, D.R.; Eustace-Cook, J.; Timmins, F. Instruments to Measure Post-intensive Care Syndrome: A Scoping Review. Nurs. Crit. Care 2023, 28, 484–498. [Google Scholar] [CrossRef]
- Wang, F.; Li, J.; Fan, Y.; Qi, X. Construction of a Risk Prediction Model for Detecting Postintensive Care Syndrome—Mental Disorders. Nurs. Crit. Care 2024, 29, 646–660. [Google Scholar] [CrossRef]
- Wang, S.; Allen, D.; Perkins, A.; Monahan, P.; Khan, S.; Lasiter, S.; Boustani, M.; Khan, B. Validation of a New Clinical Tool for Post–Intensive Care Syndrome. Am. J. Crit. Care 2019, 28, 10–18. [Google Scholar] [CrossRef]
- Boogaard, M.V.D.; Pickkers, P.; Slooter, A.J.C.; Kuiper, M.A.; Spronk, P.E.; Voort, P.H.J.V.D.; Hoeven, J.G.V.D.; Donders, R.; Achterberg, T.V.; Schoonhoven, L. Development and Validation of PRE-DELIRIC (PREdiction of DELIRium in ICu Patients) Delirium Prediction Model for Intensive Care Patients: Observational Multicentre Study. BMJ 2012, 344, e420. [Google Scholar] [CrossRef]
- Knaus, W.A.; Draper, E.A.; Wagner, D.P.; Zimmerman, J.E. APACHE II: A Severity of Disease Classification System. Crit. Care Med. 1985, 13, 818–829. [Google Scholar] [CrossRef]
- Weidman, K.; LaFond, E.; Hoffman, K.L.; Goyal, P.; Parkhurst, C.N.; Derry-Vick, H.; Schenck, E.; Lief, L. Post–Intensive Care Unit Syndrome in a Cohort of COVID-19 Survivors in New York City. Ann. Am. Thorac. Soc. 2022, 19, 1158–1168. [Google Scholar] [CrossRef]
- Lwanga, S.K.; Lemeshow, S.; World Health Organization. Dťermination de la Taille d’ un Čhantillon Dans les Ťudes Sanomťriques: Manuel Pratique; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- Chadda, K.R.; Blakey, E.E.; Davies, T.W.; Puthucheary, Z. Risk Factors, Biomarkers, and Mechanisms for Persistent Inflammation, Immunosuppression, and Catabolism Syndrome (PICS): A Systematic Review and Meta-Analysis. Br. J. Anaesth. 2024, 133, 538–549. [Google Scholar] [CrossRef]
- Palakshappa, J.A.; Krall, J.T.W.; Belfield, L.T.; Files, D.C. Long-Term Outcomes in Acute Respiratory Distress Syndrome: Epidemiology, Mechanisms, and Patient Evaluation. Crit. Care Clin. 2021, 37, 895–911. [Google Scholar] [CrossRef]
- Pang, H.; Kumar, S.; Ely, E.W.; Gezalian, M.M.; Lahiri, S. Acute Kidney Injury-Associated Delirium: A Review of Clinical and Pathophysiological Mechanisms. Crit. Care 2022, 26, 258. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.; Sigala, I.; Roussos, C. Mechanical Ventilation Clinical Applications and Pathophysiology—CHAPTER 11—Muscle Function; Saunders: Philadelphia, PA, USA, 2008. [Google Scholar]
- Misuri, G.; Lanini, B.; Gigliotti, F.; Iandelli, I.; Pizzi, A.; Bertolini, M.G.; Scano, G. Mechanism of CO2 Retention in Patients with Neuromuscular Disease. Chest 2000, 117, 447–453. [Google Scholar] [CrossRef]
- Wei, Z.; Zhao, L.; Yan, J.; Wang, X.; Li, Q.; Ji, Y.; Liu, J.; Cui, Y.; Xie, K. Dynamic Monitoring of Neutrophil/Lymphocyte Ratio, APACHE II Score, and SOFA Score Predict Prognosis and Drug Resistance in Patients with Acinetobacter Baumannii-Calcoaceticus Complex Bloodstream Infection: A Single-Center Retrospective Study. Front. Microbiol. 2024, 15, 1296059. [Google Scholar] [CrossRef]
- Stewart, J.; Bradley, J.; Smith, S.; McPeake, J.; Walsh, T.; Haines, K.; Leggett, N.; Hart, N.; McAuley, D. Do Critical Illness Survivors with Multimorbidity Need a Different Model of Care? Crit. Care 2023, 27, 485. [Google Scholar] [CrossRef]
- Bowles, K.H.; Murtaugh, C.M.; Jordan, L.; Barrón, Y.; Mikkelsen, M.E.; Whitehouse, C.R.; Chase, J.-A.D.; Ryvicker, M.; Feldman, P.H. Sepsis Survivors Transitioned to Home Health Care: Characteristics and Early Readmission Risk Factors. J. Am. Med. Dir. Assoc. 2020, 21, 84–90.e2. [Google Scholar] [CrossRef]
- Stenholt, P.O.O.; Abdullah, S.M.O.B.; Sørensen, R.H.; Nielsen, F.E. Independent Predictors for 90-Day Readmission of Emergency Department Patients Admitted with Sepsis: A Prospective Cohort Study. BMC Infect. Dis. 2021, 21, 315. [Google Scholar] [CrossRef]
- Prescott, H.C.; Langa, K.M.; Iwashyna, T.J. Readmission Diagnoses After Hospitalization for Severe Sepsis and Other Acute Medical Conditions. JAMA 2015, 313, 1055. [Google Scholar] [CrossRef]
- Boelens, Y.F.N.; Melchers, M.; Van Zanten, A.R.H. Poor Physical Recovery after Critical Illness: Incidence, Features, Risk Factors, Pathophysiology, and Evidence-Based Therapies. Curr. Opin. Crit. Care 2022, 28, 409–416. [Google Scholar] [CrossRef]
- Bourke, S.C.; Piraino, T.; Pisani, L.; Brochard, L.; Elliott, M.W. Beyond the Guidelines for Non-Invasive Ventilation in Acute Respiratory Failure: Implications for Practice. Lancet Respir. Med. 2018, 6, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Markousis-Mavrogenis, G.; Bacopoulou, F.; Kolovou, G.; Pons, M.-R.; Giannakopoulou, A.; Papavasiliou, A.; Kitas, G.; Chrousos, G.; Mavrogeni, S. Pathophysiology of Cognitive Dysfunction and the Role of Combined Brain/Heart Magnetic Resonance Imaging (Review). Exp. Ther. Med. 2022, 24, 569. [Google Scholar] [CrossRef]
- Pandharipande, P.; Hughes, C.G.; Grane, M. Sedation in the Intensive Care Setting. Clin. Pharmacol. Adv. Appl. 2012, 4, 53–63. [Google Scholar] [CrossRef]
- Sanz, L.R.D.; Laureys, S.; Gosseries, O. Towards Modern Post-Coma Care Based on Neuroscientific Evidence. Int. J. Clin. Health Psychol. 2023, 23, 100370. [Google Scholar] [CrossRef]
- Inoue, S.; Hatakeyama, J.; Kondo, Y.; Hifumi, T.; Sakuramoto, H.; Kawasaki, T.; Taito, S.; Nakamura, K.; Unoki, T.; Kawai, Y.; et al. Post-intensive Care Syndrome: Its Pathophysiology, Prevention, and Future Directions. Acute Med. Surg. 2019, 6, 233–246. [Google Scholar] [CrossRef]
- Fuke, R.; Hifumi, T.; Kondo, Y.; Hatakeyama, J.; Takei, T.; Yamakawa, K.; Inoue, S.; Nishida, O. Early Rehabilitation to Prevent Postintensive Care Syndrome in Patients with Critical Illness: A Systematic Review and Meta-Analysis. BMJ Open 2018, 8, E019998. [Google Scholar] [CrossRef]
- Hong, J.; Kang, J. Cutoff Values for Screening Post-Intensive Care Syndrome Using the Post-Intensive Care Syndrome Questionnaire. J. Clin. Med. 2025, 14, 3897. [Google Scholar] [CrossRef]
- Bornemann-Cimenti, H.; Lang, J.; Hammer, S.; Lang-Illievich, K.; Labenbacher, S.; Neuwersch-Sommeregger, S.; Klivinyi, C. Consensus-Based Recommendations for Assessing Post-Intensive Care Syndrome: A Systematic Review. J. Clin. Med. 2025, 14, 3595. [Google Scholar] [CrossRef]

| Sociodemographic and Clinical Characteristics | N (%) or m (SD) |
|---|---|
| Gender (female) | 37 (41.1) |
| Age (years) | 63.3 (16.03) |
| Ethnicity (Italian) | 90 (100) |
| Formal education (>8 yrs) | 33 (36.7) |
| Smoking habit | |
| Use in the past | 49 (54.4) |
| Actual use | 5 (5.6) |
| Alcohol habit (no) | |
| Never | 78 (86.7) |
| Use in the past | 12 (13.3) |
| Coma (yes) | 52 (57.8) |
| Sedation (yes) | 52 (57.8) |
| APACHE II score | 15.33 (9.25) |
| Predeliric score | |
| low risk | 30 (33.3) |
| moderate risk | 37 (41.1) |
| high risk | 16 (17.8) |
| very high risk | 7 (7.8) |
| Length of stay (days) | 16.51 (14.06) |
| Mechanical ventilation (days) | 5.61 (6.02) |
| HABCM | |
| Cognitive score (0–18) | 5.16 (4.47) |
| Functional score (0–33) | 9.24 (10.12) |
| Behavioural score (0–30) | 5.14 (4.04) |
| Total score (0–81) | 19.54 (16.08) |
| Model 1 | Model 2 | Model 3 | Model 4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total Score | Cognitive Score | Functional Score | Behavioral Score | |||||||||
| B | SE | p | B | SE | p | B | SE | p | B | SE | p | |
| Gender (female) | −0.41 | 3.05 | 0.893 | −0.48 | 0.81 | 0.556 | 0.50 | 1.92 | 0.794 | −0.43 | 0.85 | 0.609 |
| Age (one year-unit) | −0.09 | 0.11 | 0.389 | −0.02 | 0.03 | 0.548 | −0.08 | 0.07 | 0.224 | 0.01 | 0.03 | 0.811 |
| Coma (yes) | −9.26 | 10.53 | 0.380 | −0.99 | 2.80 | 0.724 | −1.53 | 6.61 | 0.530 | −6.74 | 2.93 | 0.023 |
| Sedation (yes) | 12.71 | 10.53 | 0.231 | 1.07 | 2.81 | 0.703 | 4.18 | 6.63 | 0.530 | 7.46 | 2.94 | 0.013 |
| APACHE II score | 0.60 | 0.18 | 0.002 | 0.17 | 0.05 | 0.001 | 0.36 | 0.11 | 0.002 | 0.07 | 0.05 | 0.158 |
| PREDELIRIC score * | ||||||||||||
| 1 | −0.20 | 3.67 | 0.957 | −1.57 | 0.98 | 0.112 | −0.87 | 2.31 | 0.707 | 2.24 | 1.02 | 0.031 |
| 2 | 12.45 | 4.21 | 0.004 | 3.11 | 1.12 | 0.007 | 7.12 | 2.65 | 0.009 | 2.22 | 1.17 | 0.061 |
| Length of stay (days) | −0.13 | 0.18 | 0.463 | −0.03 | 0.05 | 0.554 | −0.16 | 0.11 | 0.156 | 0.06 | 0.05 | 0.249 |
| Mechanical ventilation (days) | 0.38 | 0.40 | 0.351 | 0.12 | 0.11 | 0.271 | 0.36 | 0.25 | 0.154 | −0.11 | 0.11 | 0.344 |
| Intercept | 11.36 | 8.37 | 0.179 | 3.49 | 2.23 | 0.122 | 6.37 | 5.27 | 0.231 | 1.51 | 2.33 | 0.519 |
| R2 | 0.33 | 0.37 | 0.33 | 0.18 | ||||||||
| Adjusted R2 | 0.26 | 0.31 | 0.25 | 0.09 | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gravante, F.; Iovino, P.; Trotta, F.; Meucci, B.; Abagnale, M.; Bambi, S.; Pucciarelli, G. Predictors of Post-Intensive Care Syndrome in ICU Survivors After Discharge: An Observational Study. J. Clin. Med. 2025, 14, 6043. https://doi.org/10.3390/jcm14176043
Gravante F, Iovino P, Trotta F, Meucci B, Abagnale M, Bambi S, Pucciarelli G. Predictors of Post-Intensive Care Syndrome in ICU Survivors After Discharge: An Observational Study. Journal of Clinical Medicine. 2025; 14(17):6043. https://doi.org/10.3390/jcm14176043
Chicago/Turabian StyleGravante, Francesco, Paolo Iovino, Francesca Trotta, Beatrice Meucci, Marco Abagnale, Stefano Bambi, and Gianluca Pucciarelli. 2025. "Predictors of Post-Intensive Care Syndrome in ICU Survivors After Discharge: An Observational Study" Journal of Clinical Medicine 14, no. 17: 6043. https://doi.org/10.3390/jcm14176043
APA StyleGravante, F., Iovino, P., Trotta, F., Meucci, B., Abagnale, M., Bambi, S., & Pucciarelli, G. (2025). Predictors of Post-Intensive Care Syndrome in ICU Survivors After Discharge: An Observational Study. Journal of Clinical Medicine, 14(17), 6043. https://doi.org/10.3390/jcm14176043







