Beyond Seizures: A Comprehensive Review of Giant Somatosensory Evoked Potentials
Abstract
1. Introduction
2. Neurophysiological Basis of gSEPs
2.1. Preclinical Animal Model of Giant Somatosensory Evoked Potentials
2.2. Human Studies and Pathophysiology of Giant Somatosensory Evoked Potentials
3. Giant Somatosensory Evoked Potentials in Epileptic Conditions
4. Giant Somatosensory Evoked Potentials in Non-Epileptic Disorders
4.1. Neurodegenerative Diseases
4.2. Focal Structural Lesions
4.3. Metabolic and Similar Conditions
4.4. Functional and Other Non-Epileptic Disorders
5. Clinical Utility and Diagnostic Applications
5.1. Applications in Critical Care and Coma Prognostication
5.2. Early Detection and Longitudinal Monitoring
5.3. Neurorehabilitation and Cortical Plasticity
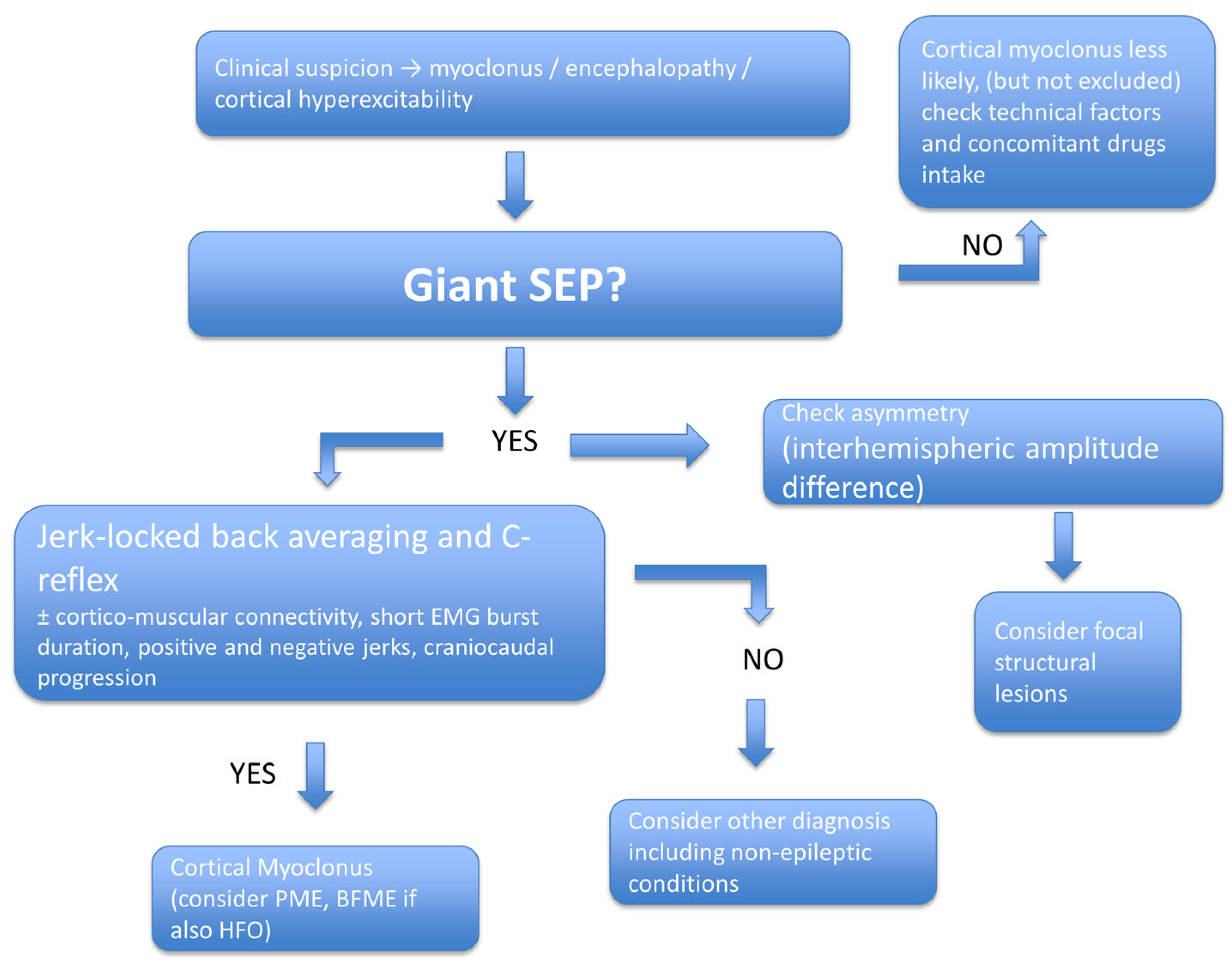
6. Diagnosis and Interpretation
7. Future Directions and Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cruccu, G.; Aminoff, M.J.; Curio, G.; Guerit, J.M.; Kakigi, R.; Mauguiere, F.; Rossini, P.M.; Treede, R.D.; Garcia-Larrea, L. Recommendations for the clinical use of somatosensory-evoked potentials. Clin. Neurophysiol. 2008, 119, 1705–1719. [Google Scholar] [CrossRef]
- Muzyka, I.M.; Estephan, B. Chapter 35—Somatosensory evoked potentials. In Handbook of Clinical Neurology; Levin, K.H., Chauvel, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 160, pp. 523–540. [Google Scholar]
- Dawson, G.D. Cerebral Responses to Electrical Stimulation of Peripheral Nerve in Man. J. Neurol. Neurosurg. Psychiatry 1947, 10, 134–140. [Google Scholar] [CrossRef]
- Shibasaki, H.; Yamashita, Y.; Neshige, R.; Tobimatsu, S.; Fukui, R. Pathogenesis of giant somatosensory evoked potentials in progressive myoclonic epilepsy. Brain 1985, 108 Pt 1, 225–240. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, H.; Yamashita, Y.; Tsuji, S. Somatosensory evoked potentials: Diagnostic criteria and abnormalities in cerebral lesions. J. Neurol. Sci. 1977, 34, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Canafoglia, L.; Ragona, F.; Panzica, F.; Piazza, E.; Freri, E.; Binelli, S.; Scaioli, V.; Avanzini, G.; Granata, T.; Franceschetti, S. Movement-activated cortical myoclonus in Dravet syndrome. Epilepsy Res. 2017, 130, 47–52. [Google Scholar] [CrossRef]
- Latorre, A.; Belvisi, D.; Rothwell, J.C.; Bhatia, K.P.; Rocchi, L. Rethinking the neurophysiological concept of cortical myoclonus. Clin. Neurophysiol. 2023, 156, 125–139. [Google Scholar] [CrossRef]
- So, N.; Berkovic, S.; Andermann, F.; Kuzniecky, R.; Gendron, D.; Quesney, L.F. Myoclonus Epilepsy and Ragged-Red Fibres (Merrf): 2Electrophysiological Studies and Comparison with Other Progressive Myoclonus Epilepsies. Brain 1989, 112, 1261–1276. [Google Scholar] [CrossRef]
- Oi, K.; Neshige, S.; Hitomi, T.; Kobayashi, K.; Tojima, M.; Matsuhashi, M.; Shimotake, A.; Fujii, D.; Matsumoto, R.; Kasama, S.; et al. Low-dose perampanel improves refractory cortical myoclonus by the dispersed and suppressed paroxysmal depolarization shifts in the sensorimotor cortex. Clin. Neurophysiol. 2019, 130, 1804–1812. [Google Scholar] [CrossRef]
- Rothwell, J.C.; Obeso, J.A.; Marsden, C.D. On the significance of giant somatosensory evoked potentials in cortical myoclonus. J. Neurol. Neurosurg. Psychiatry 1984, 47, 33–42. [Google Scholar] [CrossRef]
- Caviness, J.N.; Adler, C.H.; Beach, T.G.; Wetjen, K.L.; Caselli, R.J. Small-amplitude cortical myoclonus in Parkinson’s disease: Physiology and clinical observations. Mov. Disord. 2002, 17, 657–662. [Google Scholar] [CrossRef]
- Hitomi, T.; Ikeda, A.; Kondo, T.; Imamura, H.; Inouchi, M.; Matsumoto, R.; Terada, K.; Kanda, M.; Matsuhashi, M.; Nagamine, T.; et al. Increased cortical hyperexcitability and exaggerated myoclonus with aging in benign adult familial myoclonus epilepsy. Mov. Disord. 2011, 26, 1509–1514. [Google Scholar] [CrossRef]
- Ikeda, A.; Shibasaki, H.; Nagamine, T.; Xu, X.; Terada, K.; Mima, T.; Kaji, R.; Kawai, I.; Tatsuoka, Y.; Kimura, J. Peri-rolandic and fronto-parietal components of scalp-recorded giant SEPs in cortical myoclonus. Electroencephalogr. Clin. Neurophysiol. 1995, 96, 300–309. [Google Scholar] [CrossRef]
- Terada, K.; Ikeda, A.; Mima, T.; Kimura, M.; Nagahama, Y.; Kamioka, Y.; Murone, I.; Kimura, J.; Shibasaki, H. Familial cortical myoclonic tremor as a unique form of cortical reflex myoclonus. Mov. Disord. 1997, 12, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Visani, E.; Canafoglia, L.; Rossi Sebastiano, D.; Agazzi, P.; Panzica, F.; Scaioli, V.; Ciano, C.; Franceschetti, S. Giant SEPs and SEP-recovery function in Unverricht–Lundborg disease. Clin. Neurophysiol. 2013, 124, 1013–1018. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.E.; Artieda, J.; Zubieta, J.L.; Obeso, J.A. Reflex myoclonus in olivopontocerebellar atrophy. J. Neurol. Neurosurg. Psychiatry 1994, 57, 316–319. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thompson, P.D.; Day, B.L.; Rothwell, J.C.; Brown, P.; Britton, T.C.; Marsden, C.D. The myoclonus in corticobasal degeneration: Evidence for two forms of cortical reflex myoclonus. Brain 1994, 117, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.; Day, B.L.; Rothwell, J.C.; Thompson, P.D.; Marsden, C.D. Intrahemispheric and Interhemispheric Spread of Cerebral Cortical Myoclonic Activity and Its Relevance to Epilepsy. Brain 1991, 114, 2333–2351. [Google Scholar] [CrossRef]
- Lu, C.S.; Ikeda, A.; Terada, K.; Mima, T.; Nagamine, T.; Fukuyama, H.; Kohara, N.; Kojima, Y.; Yonekura, Y.; Chen, R.S.; et al. Electrophysiological studies of early stage corticobasal degeneration. Mov. Disord. 1998, 13, 140–146. [Google Scholar] [CrossRef]
- Okuma, Y.; Fujishima, K.; Miwa, H.; Mori, H.; Mizuno, Y. Myoclonic tremulous movements in multiple system atrophy are a form of cortical myoclonus. Mov. Disord. 2005, 20, 451–456. [Google Scholar] [CrossRef]
- Storti, S.F.; Del Felice, A.; Canafoglia, L.; Formaggio, E.; Brigo, F.; Alessandrini, F.; Bongiovanni, L.G.; Menegaz, G.; Manganotti, P. Neurophysiological and BOLD signal uncoupling of giant somatosensory evoked potentials in progressive myoclonic epilepsy: A case-series study. Sci. Rep. 2017, 7, 44664. [Google Scholar] [CrossRef]
- van Egmond, M.E.; Verschuuren-Bemelmans, C.C.; Nibbeling, E.A.; Elting, J.W.J.; Sival, D.A.; Brouwer, O.F.; de Vries, J.J.; Kremer, H.P.; Sinke, R.J.; Tijssen, M.A.; et al. Ramsay hunt syndrome: Clinical characterization of progressive myoclonus ataxia caused by GOSR2 mutation. Mov. Disord. 2014, 29, 139–143. [Google Scholar] [CrossRef]
- Rossi Sebastiano, D.; Cazzato, D.; Visani, E.; Dalla Bella, E.; Brambilla, L.; Devigili, G.; Caroppo, P.; Maggi, L.; Nanetti, L.; Salsano, E.; et al. Significance and clinical suggestions for the somatosensory evoked potentials increased in amplitude revealed by a large sample of neurological patients. Neurol. Sci. 2022, 43, 5553–5562. [Google Scholar] [CrossRef] [PubMed]
- Martín-Palomeque, G.; Castro-Ortiz, A.; Pamplona-Valenzuela, P.; Saiz-Sepúlveda, M.; Cabañes-Martínez, L.; López, J.R. Large Amplitude Cortical Evoked Potentials in Nonepileptic Patients. Reviving an Old Neurophysiologic Tool to Help Detect CNS Pathology. J. Clin. Neurophysiol. 2017, 34, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Nuwer, M.R.; Aminoff, M.; Desmedt, J.; Eisen, A.A.; Goodin, D.; Matsuoka, S.; Mauguière, F.; Shibasaki, H.; Sutherling, W.; Vibert, J.F. IFCN recommended standards for short latency somatosensory evoked potentials. Report of an IFCN committee. International Federation of Clinical Neurophysiology. Electroencephalogr. Clin. Neurophysiol. 1994, 91, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Hoareau, G.L.; Peters, A.; Hilgart, D.; Iversen, M.; Clark, G.; Zabriskie, M.; Rieke, V.; Floyd, C.; Shah, L. Feasibility of non-invasive recording of somatosensory evoked potential in pigs. Lab. Anim. Res. 2022, 38, 9. [Google Scholar] [CrossRef]
- Hilgart, D.R.; Iversen, M.M.; Peters, A.Y.; Zabriskie, M.S.; Hoareau, G.L.; Vapniarsky, N.; Clark, G.A.; Shah, L.M.; Rieke, V. Non-invasive central nervous system assessment of a porcine model of neuropathic pain demonstrates increased latency of somatosensory-evoked potentials. J. Neurosci. Methods 2023, 396, 109934. [Google Scholar] [CrossRef]
- Cui, H.Y.; Wu, Y.X.; Li, R.; Li, G.S.; Hu, Y. A translational study of somatosensory evoked potential time-frequency components in rats, goats, and humans. Neural. Regen. Res. 2021, 16, 2269–2275. [Google Scholar] [CrossRef]
- Fesenko, Z.; Ptukha, M.; da Silva, M.M.; de Carvalho, R.S.M.; Tsytsarev, V.; Gainetdinov, R.R.; Faber, J.; Volnova, A.B. Electrophysiological and Behavioral Markers of Hyperdopaminergia in DAT-KO Rats. Biomedicines 2024, 12, 2114. [Google Scholar] [CrossRef]
- Gotoh, M.; Nagasaka, K.; Nakata, M.; Takashima, I.; Yamamoto, S. Brain Temperature Alters Contributions of Excitatory and Inhibitory Inputs to Evoked Field Potentials in the Rat Frontal Cortex. Front. Cell. Neurosci. 2020, 14, 593027. [Google Scholar] [CrossRef]
- Hitomi, T.; Ikeda, A.; Matsumoto, R.; Kinoshita, M.; Taki, J.; Usui, K.; Mikuni, N.; Nagamine, T.; Hashimoto, N.; Shibasaki, H.; et al. Generators and temporal succession of giant somatosensory evoked potentials in cortical reflex myoclonus: Epicortical recording from sensorimotor cortex. Clin. Neurophysiol. 2006, 117, 1481–1486. [Google Scholar] [CrossRef]
- Latorre, A.; Rocchi, L.; Magrinelli, F.; Mulroy, E.; Berardelli, A.; Rothwell, J.C.; Bhatia, K.P. Unravelling the enigma of cortical tremor and other forms of cortical myoclonus. Brain 2020, 143, 2653–2663. [Google Scholar] [CrossRef]
- Ikeda, A.; Kakigi, R.; Funai, N.; Neshige, R.; Kuroda, Y.; Shibasaki, H. Cortical tremor: A variant of cortical reflex myoclonus. Neurology 1990, 40, 1561–1565. [Google Scholar] [CrossRef]
- Alegre, M.; Urriza, J.; Valencia, M.; Muruzábal, J.; Iriarte, J.; Artieda, J. High-frequency oscillations in the somatosensory evoked potentials of patients with cortical myoclonus: Pathophysiologic implications. J. Clin. Neurophysiol. 2006, 23, 265–272. [Google Scholar] [CrossRef]
- Assenza, G.; Lanzone, J.; Dubbioso, R.; Coppola, A.; Boscarino, M.; Ricci, L.; Insola, A.; Bilo, L.; Tombini, M.; Di Lazzaro, V. Thalamic and cortical hyperexcitability in juvenile myoclonic epilepsy. Clin. Neurophysiol. 2020, 131, 2041–2046. [Google Scholar] [CrossRef]
- Dubbioso, R.; Striano, P.; Tomasevic, L.; Bilo, L.; Esposito, M.; Manganelli, F.; Coppola, A. Abnormal sensorimotor cortex and thalamo-cortical networks in familial adult myoclonic epilepsy type 2: Pathophysiology and diagnostic implications. Brain Commun. 2022, 4, fcac037. [Google Scholar] [CrossRef] [PubMed]
- Hanajima, R.; Okabe, S.; Terao, Y.; Furubayashi, T.; Arai, N.; Inomata-Terada, S.; Hamada, M.; Yugeta, A.; Ugawa, Y. Difference in intracortical inhibition of the motor cortex between cortical myoclonus and focal hand dystonia. Clin. Neurophysiol. 2008, 119, 1400–1407. [Google Scholar] [CrossRef] [PubMed]
- Anzellotti, F.; Onofrj, M.; Bonanni, L.; Saracino, A.; Franciotti, R. Giant early components of somatosensory evoked potentials to tibial nerve stimulation in cortical myoclonus. NeuroImage Clin. 2016, 12, 212–218. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kalina, A.; Jezdik, P.; Fabera, P.; Marusic, P.; Hammer, J. Electrical Source Imaging of Somatosensory Evoked Potentials from Intracranial EEG Signals. Brain Topogr. 2023, 36, 835–853. [Google Scholar] [CrossRef]
- Autti, S.; Hirvi, P.; Keitaanniemi, M.; Mustaniemi, H.; Kotilahti, K.; Renvall, H.; Nissilä, I. Simultaneously Acquired Magnetoencephalography and Diffuse Optical Tomography Data Reveals Correlated Somatosensory Activity. Hum. Brain Mapp. 2025, 46, e70293. [Google Scholar] [CrossRef]
- Schreiner, L.; Jordan, M.; Sieghartsleitner, S.; Kapeller, C.; Pretl, H.; Kamada, K.; Asman, P.; Ince, N.F.; Miller, K.J.; Guger, C. Mapping of the central sulcus using non-invasive ultra-high-density brain recordings. Sci. Rep. 2024, 14, 6527. [Google Scholar] [CrossRef]
- Ishibashi, H.; Kobayashi, K.; Yamanaka, H.; Tojima, M.; Oi, K.; Neshige, S.; Hitomi, T.; Matsuhashi, M.; Maruyama, H.; Takahashi, R.; et al. Redefined giant somatosensory evoked potentials: Evoked epileptic complexes of excitatory and inhibitory components. Clin. Neurophysiol. 2024, 164, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P.; Schmidt, R.F. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp. Brain Res. 1999, 129, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Latorre, A.; Rocchi, L.; Paparella, G.; Manzo, N.; Bhatia, K.P.; Rothwell, J.C. Changes in cerebellar output abnormally modulate cortical myoclonus sensorimotor hyperexcitability. Brain 2024, 147, 1412–1422. [Google Scholar] [CrossRef] [PubMed]
- van van Hugte, E.J.H.; Schubert, D.; Nadif Kasri, N. Excitatory/inhibitory balance in epilepsies and neurodevelopmental disorders: Depolarizing γ-aminobutyric acid as a common mechanism. Epilepsia 2023, 64, 1975–1990. [Google Scholar] [CrossRef]
- Trevelyan, A.J.; Schevon, C.A. How inhibition influences seizure propagation. Neuropharmacology 2013, 69, 45–54. [Google Scholar] [CrossRef]
- Tojima, M.; Hitomi, T.; Matsuhashi, M.; Neshige, S.; Usami, K.; Oi, K.; Kobayashi, K.; Takeyama, H.; Shimotake, A.; Takahashi, R.; et al. A Biomarker for Benign Adult Familial Myoclonus Epilepsy: High-Frequency Activities in Giant Somatosensory Evoked Potentials. Mov. Disord. 2021, 36, 2335–2345. [Google Scholar] [CrossRef]
- Hitomi, T.; Kobayashi, K.; Murai, T.; Sakurai, T.; Matsumoto, R.; Takahashi, R.; Ikeda, A. Positive rate of giant somatosensory evoked potential (giant SEP) and c reflex in benign adult familial myoclonus epilepsy (BAFME). J. Neurol. Sci. 2017, 381, 343. [Google Scholar] [CrossRef]
- Jacobs, J.; Kobayashi, K.; Gotman, J. High-frequency changes during interictal spikes detected by time-frequency analysis. Clin. Neurophysiol. 2011, 122, 32–42. [Google Scholar] [CrossRef]
- Houlden, D.A.; Boulos, M.I.; Murray, B.J.; Norton, L.; Young, G.B. Giant Somatosensory Evoked Potentials Coincident With Epileptiform Discharges in Acutely Comatose Patients. Can. J. Neurol. Sci. 2015, 42, 317–323. [Google Scholar] [CrossRef][Green Version]
- Canafoglia, L.; Ciano, C.; Panzica, F.; Scaioli, V.; Zucca, C.; Agazzi, P.; Visani, E.; Avanzini, G.; Franceschetti, S. Sensorimotor cortex excitability in Unverricht-Lundborg disease and Lafora body disease. Neurology 2004, 63, 2309–2315. [Google Scholar] [CrossRef]
- Hsueh, S.-J.; Lin, C.-H.; Lee, N.-C.; Chang, T.-M.; Fan, S.-P.; Huang, W.-D.; Lin, Y.-H.; Tsai, L.-K.; Chien, Y.-H.; Lee, M.-J.; et al. Unique clinical and electrophysiological features in the peripheral nerve system in patients with sialidosis—A case series study. Orphanet J. Rare Dis. 2024, 19, 217. [Google Scholar] [CrossRef]
- Thompson, P.D.; Hammans, S.R.; Harding, A.E. Cortical reflex myoclonus in patients with the mitochondrial DNA transfer RNA(Lys)(8344) (MERRF) mutation. J. Neurol. 1994, 241, 335–340. [Google Scholar] [CrossRef]
- Shiga, Y.; Seki, H.; Onuma, A.; Shimizu, H.; Itoyama, Y. Decrement of N20 amplitude of the median nerve somatosensory evoked potential in Creutzfeldt-Jakob disease patients. J. Clin. Neurophysiol. 2001, 18, 576–582. [Google Scholar] [CrossRef]
- Binelli, S.; Agazzi, P.; Canafoglia, L.; Scaioli, V.; Panzica, F.; Visani, E.; Di Fede, G.; Giaccone, G.; Bizzi, A.; Bugiani, O.; et al. Myoclonus in Creutzfeldt-Jakob disease: Polygraphic and video-electroencephalography assessment of 109 patients. Mov. Disord. 2010, 25, 2818–2827. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, K.; Uozumi, T.; Akamatsu, N.; Nagashio, Y.; Qingrui, L.; Hashimoto, T.; Tsuji, S. Negative myoclonus in Creutzfeldt-Jakob disease. Clin. Neurophysiol. 2000, 111, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Calleja, J.; Carpizo, R.; Berciano, J.; Quintial, C.; Polo, J. Serial waking-sleep EEGs and evolution of somatosensory potentials in Creutzfeldt-Jakob disease. Electroencephalogr. Clin. Neurophysiol. 1985, 60, 504–508. [Google Scholar] [CrossRef] [PubMed]
- Miwa, H.; Mizuno, Y. Enlargements of somatosensory-evoked potentials in progressive supranuclear palsy. Acta Neurol. Scand. 2002, 106, 209–212. [Google Scholar] [CrossRef]
- Kofler, M.; Müller, J.; Reggiani, L.; Wenning, G.K. Somatosensory evoked potentials in progressive supranuclear palsy. J. Neurol. Sci. 2000, 179, 85–91. [Google Scholar] [CrossRef]
- Alsallom, F.; Simon, M.V. Giant Somatosensory Evoked Potentials in Focal Epilepsy Secondary to Glioblastoma Multiforme. Neurohospitalist 2023, 13, 202–203. [Google Scholar] [CrossRef]
- Lee, M.S.; Kim, W.J.; Lyoo, C.H.; Kim, S.J.; Suh, G. Giant somatosensory evoked potential in a patient with shaking TIA. Mov. Disord. 2009, 24, 2301–2303. [Google Scholar] [CrossRef]
- Ueno, Y.; Fujishima, K.; Kobayashi, H.; Mizuno, Y.; Okuma, Y. Cortical myoclonus due to hypocalcemia 12 years after thyroidectomy. Clin. Neurol. Neurosurg. 2006, 108, 400–403. [Google Scholar] [CrossRef] [PubMed]
- Mailis-Gagnon, A.; Giannoylis, I.; Downar, J.; Kwan, C.L.; Mikulis, D.J.; Crawley, A.P.; Nicholson, K.; Davis, K.D. Altered central somatosensory processing in chronic pain patients with “hysterical” anesthesia. Neurology 2003, 60, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Squintani, G.; Geroin, C.; Pasquali, A.; Cavazzana, E.; Segatti, A.; Lippolis, M.; Bonetto, C.; Antelmi, E.; Tinazzi, M. Abnormalities of the Descending Inhibitory Nociceptive Pathway in Functional Motor Disorders. Mov. Disord. 2024, 39, 905–910. [Google Scholar] [CrossRef]
- Kamble, N.L.; Pal, P.K. Electrophysiological evaluation of psychogenic movement disorders. Park. Relat. Disord. 2016, 22 (Suppl. S1), S153–S158. [Google Scholar] [CrossRef] [PubMed]
- Popa, I.; Jung, J.; Jeannin-Mayer, S.; Boulogne, S.; Andre-Obadia, N. Giant somatosensory evoked potentials are uncommon in patients with post-anoxic coma and paroxysmal electroencephalographic activity. Neurophysiol. Clin. 2018, 48, 137–138. [Google Scholar] [CrossRef]
- Schorl, M. Giant somatosensory evoked potentials as indicator of nonconvulsive status epilepticus. Clin. Neurophysiol. 2008, 119, 726–728. [Google Scholar] [CrossRef]
- Endisch, C.; Storm, C.; Ploner, C.J.; Leithner, C. Amplitudes of SSEP and outcome in cardiac arrest survivors. Neurology 2015, 85, 1752–1760. [Google Scholar] [CrossRef]
- Glimmerveen, A.B.; Keijzer, H.M.; Ruijter, B.J.; Tjepkema-Cloostermans, M.C.; van Putten, M.; Hofmeijer, J. Relevance of Somatosensory Evoked Potential Amplitude After Cardiac Arrest. Front. Neurol. 2020, 11, 335. [Google Scholar] [CrossRef]
- Kobayashi, K.; Hitomi, T.; Matsumoto, R.; Kondo, T.; Kawamata, J.; Matsuhashi, M.; Hashimoto, S.; Ikeda, H.; Koide, Y.; Inoue, Y.; et al. Long-term follow-up of cortical hyperexcitability in Japanese Unverricht-Lundborg disease. Seizure 2014, 23, 746–750. [Google Scholar] [CrossRef]
- Acharya, J.N.; Satishchandra, P.; Shankar, S.K. Familial progressive myoclonus epilepsy: Clinical and electrophysiologic observations. Epilepsia 1995, 36, 429–434. [Google Scholar] [CrossRef]
- Giaquinto, S. Evoked potentials in rehabilitation. A review. Funct. Neurol. 2004, 19, 219–225. [Google Scholar]
- Yang, S.S.; Chu, N.H.; Wu, C.H. Subcortical somatosensory evoked potentials in patients with hepatic encephalopathy caused by severe acute hepatitis. J. Gastroenterol. Hepatol. 1993, 8, 545–549. [Google Scholar] [CrossRef]
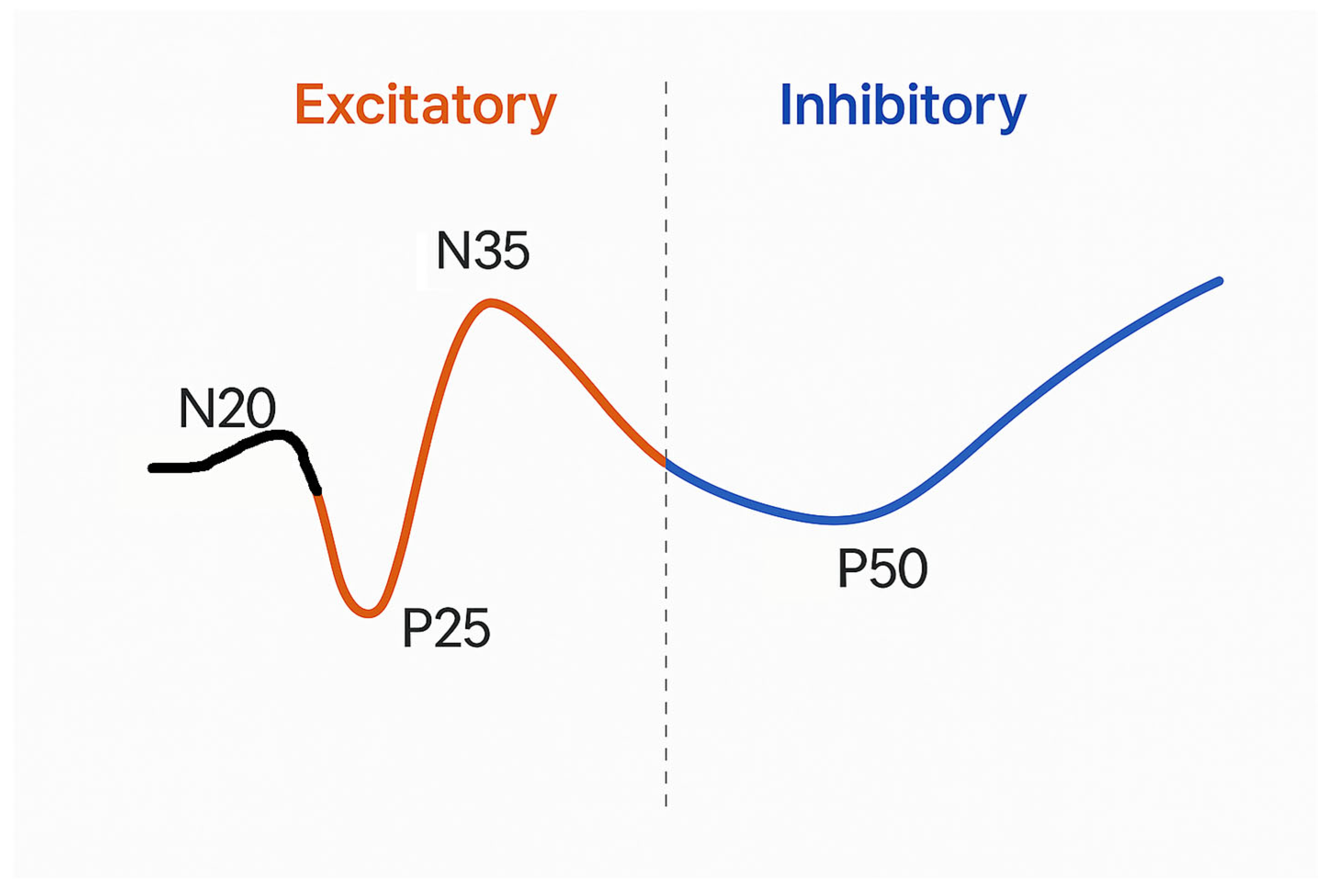
| Authors and References | Definition of Giant SEPs (gSEPs) |
|---|---|
| Shibasaki et al., 1977 [5] | Giant SEP defined as P25 amplitude > 8.6 μV or N33 amplitude > 8.4 μV |
| Canafoglia et al., 2017; Caviness et al., 2002; Hitomi et al., 2011; Ikeda et al., 1995; So et al., 1989; Terada et al., 1997; Visani et al., 2013 [6,8,11,12,13,14,15] | SEP amplitude greater than the mean plus 2–3 standard deviations of healthy control subjects. |
| Rodriguez et al., 1994; Thompson et al., 1994c [16,17] | Threshold N20-P25 ≥ 10 μV. |
| Brown et al., 1991; Lu et al., 1998; Okuma et al., 2005; Storti et al., 2017; van Egmond et al., 2014 [18,19,20,21,22] | No clearly defined amplitude cut-off provided. |
| General variations | Some authors require increased amplitudes of both N20–P25 and P25–N33 components, while others consider enlargement of only one component sufficient. |
| Syndrome | gSEP Frequency (%) | SEP Components (gSEP) | Typical Amplitude (μV) | SEP–Myoclonus Correlation | References |
|---|---|---|---|---|---|
| Unverricht–Lundborg (ULD) | ~52% | N20–P25; P25-N33 | N20–P25 22.0 ± 8.5 μV; | Mostly no | Visani [15] |
| P25–N33 29.0 ± 17.9 μV | |||||
| BAFME (FAME) | Variable ~100% | N20–P25 markedly increased; P25-HFOs superimposed | P25 17.8 ± 7.5 μV; | Time locked | Dubbioso [36] |
| N35 30.1 ± 18.0 μV | |||||
| Lafora Disease | ~75% | Both N20–P25 and P25–N33 markedly elevated | N20–P25 14 ± 10.1 μV; | No identifiable time-locked correlate with myoclonus | Canafoglia [51] |
| P25–N60 33.5 ± 20.5 μV | |||||
| Sialidosis | ~100% | N20–P25, mid-latency | NA | Usually present | Hsueh [52] |
| MERRF | ~100% | Enlarged early cortical SEPs | P1–N2 13–30 μV | Not always | Thompson [53] |
| Disorder/Etiology | Possible Interpretation | Myoclonus/EEG Correlation |
|---|---|---|
| Creutzfeldt–Jakob disease (CJD) | Cortical disinhibition followed by degeneration | Variable correlation with gSEP may vary with disease progression |
| Progressive Supranuclear Palsy (PSP) | Subcortical disinhibition | Rare or absent |
| CNS (central nervous system) Tumors | Focal cortical disinhibition | Rarely reported |
| Vascular causes (Stroke, etc) | Contralateral, possibly maladaptive or compensatory mechanism | Absent |
| Multiple Sclerosis (MS) | Maladaptive plasticity | Rare |
| Cervical myelopathy | Altered afferent conduction/cortical reorganization | Absent |
| Hypocalcemia (post-thyroidectomy) | Reversible cortical disinhibition | No reported |
| Vitamin B12 deficiency | Metabolic encephalopathy with hyperexcitability | Not specified |
| Hydroxychloroquine toxicity | Drug-induced cortical dysfunction | Not specified |
| Functional Neurological Disorders (FND) | Maladaptive cortical sensitization | Absent |
| Motor Neuron Disease (ALS) | Widespread cortical dysfunction | Absent |
| Neuropathic pain/central sensitization | Plasticity in the somatosensory cortex and afferents | Absent |
| Peripheral Neuropathies (e.g., anti-MAG, diabetic) | Sensory deafferentation and compensatory plasticity | Absent |
| Spinal cord tumors/intramedullary lesions | Cortical response to altered spinal afferents | Very Rare |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magro, G. Beyond Seizures: A Comprehensive Review of Giant Somatosensory Evoked Potentials. J. Clin. Med. 2025, 14, 5755. https://doi.org/10.3390/jcm14165755
Magro G. Beyond Seizures: A Comprehensive Review of Giant Somatosensory Evoked Potentials. Journal of Clinical Medicine. 2025; 14(16):5755. https://doi.org/10.3390/jcm14165755
Chicago/Turabian StyleMagro, Giuseppe. 2025. "Beyond Seizures: A Comprehensive Review of Giant Somatosensory Evoked Potentials" Journal of Clinical Medicine 14, no. 16: 5755. https://doi.org/10.3390/jcm14165755
APA StyleMagro, G. (2025). Beyond Seizures: A Comprehensive Review of Giant Somatosensory Evoked Potentials. Journal of Clinical Medicine, 14(16), 5755. https://doi.org/10.3390/jcm14165755





