Management of Statin Intolerant Patients in the Era of Novel Lipid Lowering Therapies: A Critical Approach in Clinical Practice
Abstract
1. Introduction
2. Methodology for Literature Searching
3. Statin Intolerance
3.1. Definition and Clinical Manifestations
3.2. Statin Intolerance and Cardiovascular Events
4. Management of Statin Intolerance
- -
- Switch
- -
- Lower dose
- -
- Alternate dosage regimen
- -
- Polypharmacy
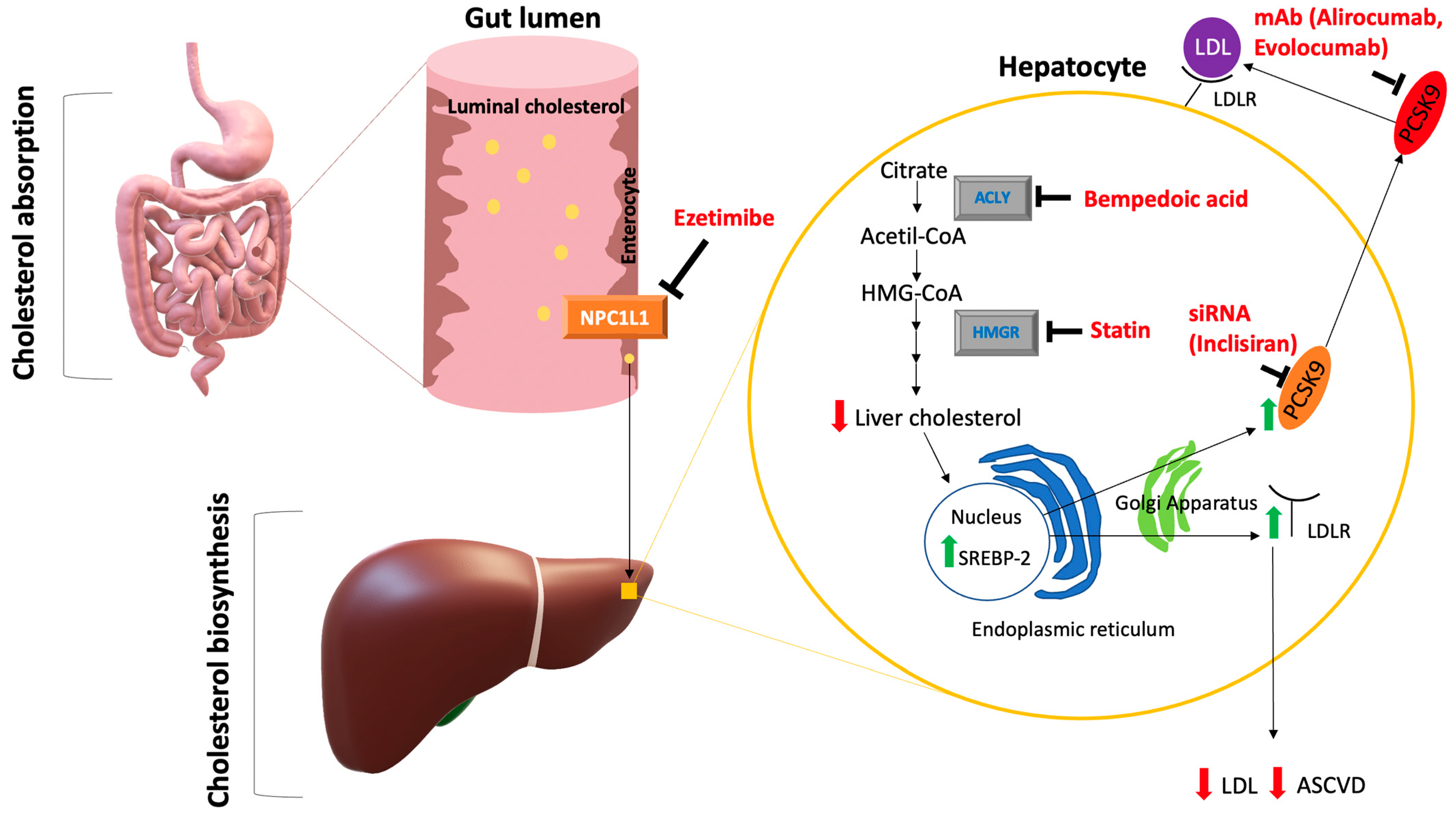
5. Non-Statin Lipid Lowering Treatments
5.1. Ezetimibe
5.2. PCSK9 Inhibitors
5.3. Bempedoic Acid
5.4. Nutraceuticals
5.5. Other Therapeutic Strategies for LDL-C Reduction
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Borén, J.; Williams, K.J. The Central Role of Arterial Retention of Cholesterolrich Apolipoprotein-B-Containing Lipoproteins in the Pathogenesis of Atherosclerosis: A Triumph of Simplicity. Curr. Opin. Lipidol. 2016, 27, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-Density Lipoproteins Cause Atherosclerotic Cardiovascular Disease. 1. Evidence from Genetic, Epidemiologic, and Clinical Studies. A Consensus Statement Fromthe European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2017, 38, 2459–2472. [Google Scholar] [CrossRef] [PubMed]
- Scicali, R.; Di Pino, A.; Ferrara, V.; Urbano, F.; Piro, S.; Rabuazzo, A.M.; Purrello, F. New Treatment Options for Lipid-Lowering Therapy in Subjects with Type 2 Diabetes. Acta Diabetol. 2018, 55, 209–218. [Google Scholar] [CrossRef]
- Stone, N.J.; Robinson, J.G.; Lichtenstein, A.H.; Bairey Merz, C.N.; Blum, C.B.; Eckel, R.H.; Goldberg, A.C.; Gordon, D.; Levy, D.; Lloyd-Jones, D.M.; et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 2889–2934. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Hong, S.J.; Lee, Y.J. A RACING Vizsgálat Jelentôsége. Lege Artis Med. 2022, 32, 381–383. [Google Scholar]
- Saxon, D.R.; Eckel, R.H. Statin Intolerance: A Literature Review and Management Strategies. Prog. Cardiovasc. Dis. 2016, 59, 153–164. [Google Scholar] [CrossRef]
- Banach, M.; Cannon, C.P.; Paneni, F.; Penson, P.E. Individualized Therapy in Statin Intolerance: The Key to Success. Eur. Heart J. 2023, 44, 544–546. [Google Scholar] [CrossRef]
- Jia, X.; Al Rifai, M.; Saeed, A.; Ballantyne, C.M.; Virani, S.S. PCSK9 Inhibitors in the Management of Cardiovascular Risk: A Practical Guidance. Vasc. Health Risk Manag. 2022, 18, 555–566. [Google Scholar] [CrossRef]
- Cannon, C.P.; Blazing, M.A.; Giugliano, R.P.; McCagg, A.; White, J.A.; Theroux, P.; Darius, H.; Lewis, B.S.; Ophuis, T.O.; Jukema, J.W.; et al. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes. N. Engl. J. Med. 2015, 372, 2387–2397. [Google Scholar] [CrossRef]
- Henney, N.C.; Banach, M.; Penson, P.E. RNA Silencing in the Management of Dyslipidemias. Curr. Atheroscler. Rep. 2021, 23, 69. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Bays, H.; Catapano, A.L.; Goldberg, A.; Ray, K.K.; Saseen, J.J. Role of Bempedoic Acid in Clinical Practice. Cardiovasc. Drugs Ther. 2021, 35, 853–864. [Google Scholar] [CrossRef]
- Cheeley, M.K.; Saseen, J.J.; Agarwala, A.; Ravilla, S.; Ciffone, N.; Jacobson, T.A.; Dixon, D.L.; Maki, K.C. NLA Scientific Statement on Statin Intolerance: A New Definition and Key Considerations for ASCVD Risk Reduction in the Statin Intolerant Patient. J. Clin. Lipidol. 2022, 16, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef] [PubMed]
- Stroes, E.S.; Thompson, P.D.; Corsini, A.; Vladutiu, G.D.; Raal, F.J.; Ray, K.K.; Roden, M.; Stein, E.; Tokgözoğlu, L.; Nordestgaard, B.G.; et al. Statin-Associated Muscle Symptoms: Impact on Statin Therapy—European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur. Heart J. 2015, 36, 1012–1022. [Google Scholar] [CrossRef] [PubMed]
- Newman, C.B.; Preiss, D.; Tobert, J.A.; Jacobson, T.A.; Page, R.L.; Goldstein, L.B.; Chin, C.; Tannock, L.R.; Miller, M.; Raghuveer, G.; et al. Statin Safety and Associated Adverse Events A Scientific Statement from the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 2019, 39, E38–E81. [Google Scholar] [CrossRef]
- Casula, M.; Gazzotti, M.; Bonaiti, F.; OImastroni, E.; Arca, M.; Averna, M.; Zambon, A.; Catapano, A.L.; Montali, A.; Averna, M.; et al. Reported Muscle Symptoms during Statin Treatment amongst Italian Dyslipidaemic Patients in the Real-Life Setting: The PROSISA Study. J. Intern. Med. 2021, 290, 116–128. [Google Scholar] [CrossRef]
- Fiorentino, R.; Chiarelli, F. Statins in Children, an Update. Int. J. Mol. Sci. 2023, 24, 1366. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Baker, S.K.; Jacobson, T.A.; Kopecky, S.L.; Parker, B.A. An Assessment by the Statin Muscle Safety Task Force: 2014 Update. J. Clin. Lipidol. 2014, 8, S58–S71. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Miller, K.; Bayliss, M.; Sanchez, R.J.; Baccara-Dinet, M.T.; Chibedi-De-Roche, D.; Taylor, B.; Khan, I.; Manvelian, G.; White, M.; et al. The Statin-Associated Muscle Symptom Clinical Index (SAMS-CI): Revision for Clinical Use, Content Validation, and Inter-Rater Reliability. Cardiovasc. Drugs Ther. 2017, 31, 179–186. [Google Scholar] [CrossRef]
- Gulizia, M.M.; Colivicchi, F.; Arca, M.; Abrignani, M.G.; Perna, G.P.; Mureddu, G.F.; Nardi, F.; Riccio, C. ANMCO Position Paper: Diagnostic–Therapeutic Pathway in Patients with Hypercholesterolaemia and Statin Intolerance. Eur. Heart J. Suppl. 2017, 19, D55–D63. [Google Scholar] [CrossRef]
- Gheorghe, G.; Toth, P.P.; Bungau, S.; Behl, T.; Ilie, M.; Pantea Stoian, A.; Bratu, O.G.; Bacalbasa, N.; Rus, M.; Diaconu, C.C. Cardiovascular Risk and Statin Therapy Considerations in Women. Diagnostics 2020, 10, 483. [Google Scholar] [CrossRef]
- Cannon, C.P. Statin Intolerance: How Common Is It and How Do We Work with Patients to Overcome It? Eur. Heart J. 2022, 43, 3224–3226. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Budhani, S.B.; Masineni, S.S.; Abuchaibe, C.; Khan, N.; Wang, P.; Goldenberg, N. Vitamin D Deficiency, Myositis–Myalgia, and Reversible Statin Intolerance. Curr. Med. Res. Opin. 2011, 27, 1683–1690. [Google Scholar] [CrossRef]
- Ward, N.C.; Watts, G.F.; Eckel, R.H. Statin Toxicity: Mechanistic Insights and Clinical Implications. Circ. Res. 2019, 124, 328–350. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.V.; Stelzle, D.; Lee, K.K.; Beck, E.J.; Alam, S.; Clifford, S.; Longenecker, C.T.; Strachan, F.; Bagchi, S.; Whiteley, W.; et al. Global Burden of Atherosclerotic Cardiovascular Disease in People Living with HIV. Circulation 2018, 138, 1100–1112. [Google Scholar] [CrossRef] [PubMed]
- Banach, M.; Rizzo, M.; Toth, P.P.; Farnier, M.; Davidson, M.H.; Al-Rasadi, K.; Aronow, W.S.; Athyros, V.; Djuric, D.M.; Ezhov, M.V.; et al. Statin Intolerance—An Attempt at a Unified Definition. Position Paper from an International Lipid Expert Panel. Expert Opin. Drug Saf. 2015, 14, 935–955. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Plutzky, J.; Skentzos, S.; Morrison, F.; Mar, P.; Shubina, M.; Turchin, A. Discontinuation of Statins in Routine Care Settings, A Cohort Study. Ann. Intern. Med. 2013, 158, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Gomez Sandoval, Y.-H.; Braganza, M.V.; Daskalopoulou, S.S. Statin Discontinuation in High-Risk Patients: A Systematic Review of the Evidence. Curr. Pharm. Des. 2011, 17, 3669–3689. [Google Scholar] [CrossRef]
- Jones, P.H.; Davidson, M.H.; Stein, E.A.; Bays, H.E.; McKenney, J.M.; Miller, E.; Cain, V.A.; Blasetto, J.W. Comparison of the Efficacy and Safety of Rosuvastatin versus Atorvastatin, Simvastatin, and Pravastatin across Doses (STELLAR**STELLAR = Statin Therapies for Elevated Lipid Levels Compared Across Doses to Rosuvastatin. Trial). Am. J. Cardiol. 2003, 92, 152–160. [Google Scholar] [CrossRef]
- Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and Safety of LDL-Lowering Therapy among Men and Women: Meta-Analysis of Individual Data from 174 000 Participants in 27 Randomised Trials. Lancet 2015, 385, 1397–1405. [Google Scholar] [CrossRef]
- Serban, M.-C.; Colantonio, L.D.; Manthripragada, A.D.; Monda, K.L.; Bittner, V.A.; Banach, M.; Chen, L.; Huang, L.; Dent, R.; Kent, S.T.; et al. Statin Intolerance and Risk of Coronary Heart Events and All-Cause Mortality Following Myocardial Infarction. J. Am. Coll. Cardiol. 2017, 69, 1386–1395. [Google Scholar] [CrossRef] [PubMed]
- Banach, M.; Mikhailidis, D.P. Statin Intolerance. Cardiol. Clin. 2018, 36, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Mancini, G.B.J.; Baker, S.; Bergeron, J.; Fitchett, D.; Frohlich, J.; Genest, J.; Gupta, M.; Hegele, R.A.; Ng, D.; Pearson, G.J.; et al. Diagnosis, Prevention, and Management of Statin Adverse Effects and Intolerance: Canadian Consensus Working Group Update. Can. J. Cardiol. 2016, 32, S35–S65. [Google Scholar] [CrossRef] [PubMed]
- Penson, P.E.; Bruckert, E.; Marais, D.; Reiner, Ž.; Pirro, M.; Sahebkar, A.; Bajraktari, G.; Mirrakhimov, E.; Rizzo, M.; Mikhailidis, D.P.; et al. Step-by-step Diagnosis and Management of the Nocebo/Drucebo Effect in Statin-associated Muscle Symptoms Patients: A Position Paper from the International Lipid Expert Panel (ILEP). J. Cachexia Sarcopenia Muscle 2022, 13, 1596–1622. [Google Scholar] [CrossRef] [PubMed]
- Awad, K.; Mikhailidis, D.P.; Toth, P.P.; Jones, S.R.; Moriarty, P.; Lip, G.Y.H.; Muntner, P.; Catapano, A.L.; Pencina, M.J.; Rosenson, R.S.; et al. Efficacy and Safety of Alternate-Day Versus Daily Dosing of Statins: A Systematic Review and Meta-Analysis. Cardiovasc. Drugs Ther. 2017, 31, 419–431. [Google Scholar] [CrossRef]
- Ray, K.K.; Molemans, B.; Schoonen, W.M.; Giovas, P.; Bray, S.; Kiru, G.; Murphy, J.; Banach, M.; De Servi, S.; Gaita, D.; et al. EU-Wide Cross-Sectional Observational Study of Lipid-Modifying Therapy Use in Secondary and Primary Care: The DA VINCI Study. Eur. J. Prev. Cardiol. 2021, 28, 1279–1289. [Google Scholar] [CrossRef]
- Ray, K.K.; Haq, I.; Bilitou, A.; Aguiar, C.; Arca, M.; Connolly, D.L.; Eriksson, M.; Ferrières, J.; Hildebrandt, P.; Laufs, U.; et al. Evaluation of Contemporary Treatment of High- and Very High-Risk Patients for the Prevention of Cardiovascular Events in Europe—Methodology and Rationale for the Multinational Observational SANTORINI Study. Atheroscler. Plus 2021, 43, 24–30. [Google Scholar] [CrossRef]
- Pandor, A.; Ara, R.M.; Tumur, I.; Wilkinson, A.J.; Paisley, S.; Duenas, A.; Durrington, P.N.; Chilcott, J. Ezetimibe Monotherapy for Cholesterol Lowering in 2722 People: Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Intern. Med. 2009, 265, 568–580. [Google Scholar] [CrossRef]
- Mandraffino, G.; Scicali, R.; Rodríguez-Carrio, J.; Savarino, F.; Mamone, F.; Scuruchi, M.; Cinquegrani, M.; Imbalzano, E.; Di Pino, A.; Piro, S.; et al. Arterial Stiffness Improvement after Adding on PCSK9 Inhibitors or Ezetimibe to High-Intensity Statins in Patients with Familial Hypercholesterolemia: A Two–Lipid Center Real-World Experience. J. Clin. Lipidol. 2020, 14, 231–240. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the Management of Dyslipidaemias: Lipid Modification to Reduce Cardiovascular Risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- McDonagh, M.; Peterson, K.; Holzhammer, B.; Fazio, S. A Systematic Review of PCSK9 Inhibitors Alirocumab and Evolocumab. J. Manag. Care Spec. Pharm. 2016, 22, 641–653q. [Google Scholar] [CrossRef] [PubMed]
- Moriarty, P.M.; Thompson, P.D.; Cannon, C.P.; Guyton, J.R.; Bergeron, J.; Zieve, F.J.; Bruckert, E.; Jacobson, T.A.; Kopecky, S.L.; Baccara-Dinet, M.T.; et al. Efficacy and Safety of Alirocumab vs Ezetimibe in Statin-Intolerant Patients, with a Statin Rechallenge Arm: The ODYSSEY ALTERNATIVE Randomized Trial. J. Clin. Lipidol. 2015, 9, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.E.; Stroes, E.; Dent-Acosta, R.E.; Rosenson, R.S.; Lehman, S.J.; Sattar, N.; Preiss, D.; Bruckert, E.; Ceška, R.; Lepor, N.; et al. Efficacy and Tolerability of Evolocumab vs Ezetimibe in Patients with Muscle-Related Statin Intolerance. JAMA 2016, 315, 1580. [Google Scholar] [CrossRef]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Steg, P.G.; Szarek, M.; Bhatt, D.L.; Bittner, V.A.; Diaz, R.; Edelberg, J.M.; Goodman, S.G.; Hanotin, C.; Harrington, R.A.; et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N. Engl. J. Med. 2018, 379, 2097–2107. [Google Scholar] [CrossRef]
- Martirossian, A.N.; Goldberg, A.C. Management of Patients with Statin Intolerance. Best Pract. Res. Clin. Endocrinol. Metab. 2022, 101714. [Google Scholar] [CrossRef]
- Ray, K.K.; Raal, F.J.; Kallend, D.G.; Jaros, M.J.; Koenig, W.; Leiter, L.A.; Landmesser, U.; Schwartz, G.G.; Lawrence, D.; Friedman, A.; et al. Inclisiran and Cardiovascular Events: A Patient-Level Analysis of Phase III Trials. Eur. Heart J. 2023, 44, 129–138. [Google Scholar] [CrossRef]
- Wright, R.S.; Kallend, D.; Raal, F.J.; Stoekenbroek, R.; Koenig, W.; Leiter, L.A.; Landmesser, U.; Schwartz, G.; Wijngaard, P.L.J.; Kastelein, J.J.P.; et al. Pooled Safety and Efficacy of Inclisiran in Patients with Statin Intolerance (ORION-10 and ORION-11). Eur. Heart J. 2020, 41, ehaa946.3009. [Google Scholar] [CrossRef]
- Merćep, I.; Friščić, N.; Strikić, D.; Reiner, Ž. Advantages and Disadvantages of Inclisiran: A Small Interfering Ribonucleic Acid Molecule Targeting PCSK9—A Narrative Review. Cardiovasc. Ther. 2022, 2022, 8129513. [Google Scholar] [CrossRef]
- Laufs, U.; Banach, M.; Mancini, G.B.J.; Gaudet, D.; Bloedon, L.T.; Sterling, L.R.; Kelly, S.; Stroes, E.S.G. Efficacy and Safety of Bempedoic Acid in Patients with Hypercholesterolemia and Statin Intolerance. J. Am. Heart Assoc. 2019, 8, e011662. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Laufs, U.; Ray, K.K.; Leiter, L.A.; Bays, H.E.; Goldberg, A.C.; Stroes, E.S.; MacDougall, D.; Zhao, X.; Catapano, A.L. Bempedoic Acid plus Ezetimibe Fixed-Dose Combination in Patients with Hypercholesterolemia and High CVD Risk Treated with Maximally Tolerated Statin Therapy. Eur. J. Prev. Cardiol. 2020, 27, 593–603. [Google Scholar] [CrossRef] [PubMed]
- Rubino, J.; MacDougall, D.E.; Sterling, L.R.; Hanselman, J.C.; Nicholls, S.J. Combination of Bempedoic Acid, Ezetimibe, and Atorvastatin in Patients with Hypercholesterolemia: A Randomized Clinical Trial. Atherosclerosis 2021, 320, 122–128. [Google Scholar] [CrossRef]
- Rubino, J.; MacDougall, D.E.; Sterling, L.R.; Kelly, S.E.; McKenney, J.M.; Lalwani, N.D. Lipid Lowering with Bempedoic Acid Added to a Proprotein Convertase Subtilisin/Kexin Type 9 Inhibitor Therapy: A Randomized, Controlled Trial. J. Clin. Lipidol. 2021, 15, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Pirro, M.; Vetrani, C.; Bianchi, C.; Mannarino, M.R.; Bernini, F.; Rivellese, A.A. Joint Position Statement on “Nutraceuticals for the Treatment of Hypercholesterolemia” of the Italian Society of Diabetology (SID) and of the Italian Society for the Study of Arteriosclerosis (SISA). Nutr. Metab. Cardiovasc. Dis. 2017, 27, 2–17. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Kennedy, C.; Knežević, T.; Bove, M.; Georges, C.M.G.; Šatrauskienė, A.; Toth, P.P.; Fogacci, F. Efficacy and Safety of Armolipid Plus®: An Updated PRISMA Compliant Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. Nutrients 2021, 13, 638. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Stoian, A.P.; Vrablik, M.; Al Rasadi, K.; Banach, M.; Toth, P.P.; Rizzo, M. Nutraceuticals in the Management of Dyslipidemia: Which, When, and for Whom? Could Nutraceuticals Help Low-Risk Individuals with Non-Optimal Lipid Levels? Curr. Atheroscler. Rep. 2021, 23, 57. [Google Scholar] [CrossRef] [PubMed]
- Vesa, C.M.; Bungau, S.G. Novel Molecules in Diabetes Mellitus, Dyslipidemia and Cardiovascular Disease. Int. J. Mol. Sci. 2023, 24, 4029. [Google Scholar] [CrossRef]
- Chen, R.; Lin, S.; Chen, X. The Promising Novel Therapies for Familial Hypercholesterolemia. J. Clin. Lab. Anal. 2022, 36, e24552. [Google Scholar] [CrossRef]
- Behl, T.; Chadha, S.; Sachdeva, M.; Sehgal, A.; Kumar, A.; Dhruv; Venkatachalam, T.; Hafeez, A.; Aleya, L.; Arora, S.; et al. Understanding the Possible Role of Endocannabinoid System in Obesity. Prostaglandins Other Lipid Mediat. 2021, 152, 106520. [Google Scholar] [CrossRef]


| Statin-Associated Side Effects (SASE) | Frequency |
|---|---|
| Statin-Associated Muscle Symptom (SAMS) | |
| Infrequent (1–5%) in RCTs; frequent (5–10%) in observational studies and in clinical practice |
| Rare |
| Rare |
| Rare |
| New onset diabetes mellitus | It depends on population. Frequency increases in the presence of diabetes mellitus risk factors, such as BMI ≥ 30, fasting blood glucose ≥ 100, metabolic syndrome, HbA1c ≥ 6% |
| Hepatic injury | |
| Infrequent |
| Rare |
| Central nervous system | |
| Rare |
| Cancer | No clear association |
| Kidney disease | No clear association |
| Cataract | No clear association |
| Tendon rupture | No clear association |
| Haemorrhagic stroke | No clear association |
| Interstitial lung disease | No clear association |
| Hypogonadism | No clear association |
| Endogenous Risk Factors | Exogenous Risk Factor |
|---|---|
| Elderly | Intensive physical activity |
| Female sex | High intensity statin therapy |
| Asian ethnicity | Alcohol abuse |
| Positive history of muscle and/or joint pain | Drug abuse |
Inflammatory or metabolic neuromuscular disease
| Drug interactions
|
| Positive history of increased CK (especially if CK > 10 ULN) | Grapefruit or blueberry juice consumption (>1 L/day) |
| Positive family history of myopathy | Unregulated supplements (e.g., red yeast rice, pleurotus mushrooms, etc.). |
| Induced myopathy by statins or other hypolipidemic drugs | Surgical procedures |
| Low body mass index | |
| Severe kidney failure (III-IV stage KDOQI) | |
| Acute or decompensated hepatopathy | |
| Hypertension/heart failure (secondary to kidney disease) | |
| Untreated or undertreated hypothyroidism | |
| Diabetes mellitus | |
| Acute infection | |
| Biliary obstruction | |
| Major trauma with increased metabolic demand | |
| Vitamin D deficiency | |
Genetic polymorphisms
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bosco, G.; Di Giacomo Barbagallo, F.; Spampinato, S.; Lanzafame, L.; Di Pino, A.; Piro, S.; Purrello, F.; Scicali, R. Management of Statin Intolerant Patients in the Era of Novel Lipid Lowering Therapies: A Critical Approach in Clinical Practice. J. Clin. Med. 2023, 12, 2444. https://doi.org/10.3390/jcm12062444
Bosco G, Di Giacomo Barbagallo F, Spampinato S, Lanzafame L, Di Pino A, Piro S, Purrello F, Scicali R. Management of Statin Intolerant Patients in the Era of Novel Lipid Lowering Therapies: A Critical Approach in Clinical Practice. Journal of Clinical Medicine. 2023; 12(6):2444. https://doi.org/10.3390/jcm12062444
Chicago/Turabian StyleBosco, Giosiana, Francesco Di Giacomo Barbagallo, Salvatore Spampinato, Lorena Lanzafame, Antonino Di Pino, Salvatore Piro, Francesco Purrello, and Roberto Scicali. 2023. "Management of Statin Intolerant Patients in the Era of Novel Lipid Lowering Therapies: A Critical Approach in Clinical Practice" Journal of Clinical Medicine 12, no. 6: 2444. https://doi.org/10.3390/jcm12062444
APA StyleBosco, G., Di Giacomo Barbagallo, F., Spampinato, S., Lanzafame, L., Di Pino, A., Piro, S., Purrello, F., & Scicali, R. (2023). Management of Statin Intolerant Patients in the Era of Novel Lipid Lowering Therapies: A Critical Approach in Clinical Practice. Journal of Clinical Medicine, 12(6), 2444. https://doi.org/10.3390/jcm12062444











