Cryoballoon-Induced Circumferential Pulmonary Vein Fibrosis, Assessed by Late Gadolinium-Enhancement Cardiac Magnetic Resonance Imaging, and Its Correlation with Clinical Atrial Fibrillation Recurrence
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.1.1. Study Inclusion Criteria
- −
- Age 18–75;
- −
- Symptomatic AF, refractory to at least one anti-arrhythmic medication.
2.1.2. Study Exclusion Criteria
- −
- Significant structural heart disease as defined above, except of hypertension (HTN);
- −
- Prior heart surgery or prior ablation involving the left atrium;
- −
- Pregnant women;
- −
- Inability to undergo cardiac MRI with gadolinium injection either due to claustrophobia or significant renal disease (GFR < 30 mL/min).
2.1.3. Study Endpoints
2.2. Cryoballoon AF Ablation Procedure
2.3. Cardiac MRI Protocol
2.4. MRI Definitions and Categorization of Ablation-Induced PV Ostial Fibrosis
- −
- Complete—circumferential PV ostial LGE without gaps;
- −
- Sub-complete—an almost circumferential LGE around PV ostium with the presence of a minor LGE gap only;
- −
- Partial—incomplete circumferential LGE around PV ostium due to the presence of a major LGE gap;
- −
- Absent PV fibrosis—no LGE around PV ostium.
2.5. Statistics
3. Results
| AF Recurrence (N = 5) | No AF Recurrence (N = 14) | Total | |
|---|---|---|---|
| Patients with major PV gap in one or more PVs | 5 (True Positive) | 4 (False Positive) | 9 |
| Patients without major PV gap in any PV | 0 (False Negative) | 10 (True Negative) | 10 |
| Sensitivity = 100% Specificity = 71.4% Positive predictive Value (PPV) = 55.6% Negative Predictive Value (NPV) = 100% | Sensitivity = TP/(Tp + FN) Specificity = TN/(TN + FP) PPV = TP/(Tp + FP) NPV = Tn/(FN + TN) |
4. Discussion
- In many prior MRI studies, both high LGE intensities (corresponding to dense scar) and intermediate intensities (corresponding to interstitial fibrosis) were considered as scar for the sake of gap analysis to avoid gap over-estimation, suggesting that MRI has limited ability to differentiate dense scar from interstitial fibrosis and has a tendency for over-estimation of PV gaps [7,15,16,19];
- Changing gap definition from 3 to 5 mm on post-ablation MRI did not change AF recurrence results [19];
- Animal studies showing conduction block persisting in the presence of post-ablation MRI gaps of up to 4 mm [22].
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Oakes, R.S.; Badger, T.J.; Kholmovski, E.G.; Akoum, N.; Burgon, N.S.; Fish, E.N.; Blauer, J.J.E.; Rao, S.N.; DiBella, E.V.R.; Segerson, N.M.; et al. Detection and quantification of left atrial structural remodeling with delayed-enhancement magnetic resonance imaging in patients with atrial fibrillation. Circulation 2009, 119, 1758–1767. [Google Scholar] [CrossRef] [PubMed]
- Akoum, N.; Daccarett, M.; McGann, C.; Segerson, N.; Vergara, G.; Kuppahally, S.; Badger, T.; Burgon, N.; Haslam, T.; Kholmovski, E.; et al. Atrial fibrosis helps select the appropriate patient and strategy in catheter ablation of atrial fibrillation: A DE-MRI guided approach. J. Cardiovasc. Electrophysiol. 2011, 22, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Marrouche, N.F.; Wilber, D.; Hindricks, G.; Jais, P.; Akoum, N.; Marchlinski, F.; Kholmovski, E.; Burgon, N.; Hu, N.; Mont, L.; et al. Association of atrial tissue fibrosis identified by delayed enhancement MRI and atrial fibrillation catheter ablation: The DECAAF study. JAMA 2014, 311, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498. [Google Scholar]
- Quinto, L.; Cozzari, J.; Benito, E.; Alarcón, F.; Bisbal, F.; Trotta, O.; Caixal, G.; Antonio, R.S.; Garre, P.; Prat-Gonzalez, S.; et al. Magnetic resonance guided re-ablation for atrial fibrillation is associated with a lower recurrence rate: A case-control study. Europace 2020, 22, 1805–1811. [Google Scholar] [CrossRef]
- Fochler, F.; Yamaguchi, T.; Kheirkahan, M.; Kholmovski, E.G.; Morris, A.K.; Marrouche, N.F. Late gadolinium enhancement magnetic resonance imaging guided treatment of post-atrial fibrillation ablation recurrent arrhythmia. Circ. Arrhythm. Electrophysiol. 2019, 12, e007174. [Google Scholar] [CrossRef]
- Bisbal, F.; Guiu, E.; Cabanas-Grandío, P.; Berruezo, A.; Prat-Gonzalez, S.; Vidal, B.; Garrido, C.; Andreu, D.; Fernandez-Armenta, J.; Tolosana, J.M.; et al. CMR-guided approach to localize and ablate gaps in repeat AF ablation procedure. JACC Cardiovasc. Imaging 2014, 7, 653–663. [Google Scholar] [CrossRef]
- Spragg, D.D.; Khurram, I.; Zimmerman, S.L.; Yarmohammadi, H.; Barcelon, B.; Needleman, M.; Edwards, D.; Marine, J.E.; Calkins, H.; Nazarian, S. Initial experience with magnetic resonance imaging of atrial scar and co-registration with electroanatomic voltage mapping during atrial fibrillation: Success and limitations. Heart Rhythm. 2012, 9, 2003–2009. [Google Scholar] [CrossRef]
- Akoum, N.; Wilber, D.; Hindricks, G.; Jais, P.; Cates, J.; Marchlinski, F.; Kholmovski, E.; Burgon, N.; Hu, N.; Mont, L.; et al. MRI assessment of ablation-induced scarring in atrial fibrillation: Analysis for the DECAAF study. J. Cardiovasc. Electrophysiol. 2015, 26, 473–480. [Google Scholar] [CrossRef]
- Peters, D.C.; Wylie, J.V.; Hauser, T.H.; Nezafat, R.; Han, Y.; Woo, J.J.; Taclas, J.; Kissinger, K.V.; Goddu, B.; Josephson, M.E.; et al. Recurrence of atrial fibrillation correlates with the extent of post-procedural late gadolinium enhancement: A pilot study. J. Am. Coll. Cardiol. Imaging 2009, 2, 308–316. [Google Scholar] [CrossRef]
- Harrison, J.L.; Sohns, C.; Linton, N.W.; Karim, R.; Williams, S.E.; Rhode, K.S.; Gill, J.; Cooklin, M.; Rinaldi, C.A.; Wright, M.; et al. Repeat left atrial catheter ablation: Cardiac magnetic resonance prediction of endocardial voltage and gaps in ablation lesion sets. Circ. Arrhythm. Electrophysiol. 2015, 8, 270–278. [Google Scholar] [CrossRef]
- Chen, S.; Schmidt, B.; Bordignon, S.; Bologna, F.; Perrotta, L.; Nagase, T.; Chun, K.R.J. Atrial fibrillation ablation using cryoballoon technology: Recent advances and practical techniques. J. Cardiovasc. Electrophysiol. 2018, 29, 932–943. [Google Scholar] [CrossRef]
- Sarabanda, A.V.; Bunch, T.J.; Johnson, S.B.; Mahapatra, S.; Milton, M.A.; Leite, L.R.; Bruce, G.K.; Packer, D.L. Efficacy and safety of circumferential pulmonary vein isolation using a novel cryothermal balloon ablation system. J. Am. Coll. Cardiol. 2005, 46, 1902–1912. [Google Scholar] [CrossRef]
- Aviltall, B.; Uroboniene, D.; Rozmus, G.; Lafontaine, D.; Helms, R.; Urbonas, A. New cryotechnology for electrical isolation of pulmonary veins. J. Cardiovasc. Electrophysiol. 2003, 14, 281–286. [Google Scholar] [CrossRef]
- Alarcón, F.; Cabanelas, N.; Izquierdo, M.; Benito, E.; Ventura, R.F.I.; Guasch, E.; Prat-Gonzalez, S.; Perea, R.J.; Borràs, R.; Trotta, O.; et al. Cryoballoon vs. radiofrequency lesions as detected by late-enhancement cardiac magnetic resonance after ablation of paroxysmal atrial fibrillation: A case-control study. Europace 2020, 22, 382–387. [Google Scholar] [CrossRef]
- Trotta, O.; Alarcon, F.; Guasch, E.; Benito, E.M.; San Antonio, R.; Perea, R.J.; Prat-Gonzalez, S.; Apolo, J.; Sitges, M.; Tolosana, J.M.; et al. Impact of cryoballoon applications on lesion gaps detected by magnetic resonance imaging after pulmonary vein isolation. JCE 2020, 31, 638–646. [Google Scholar]
- Kurose, J.; Kiuchi, K.; Fukuzawa, K.; Takami, M.; Mori, S.; Suehiro, H.; Nagamatsu, Y.; Akita, T.; Takemoto, M.; Yatomi, A.; et al. Lesion characteristics between cryoballoon ablation and radiofrequency ablation with a contact force-sensing catheter: Late-gadolinium enhancement magnetic resonance imaging assessment. J. Cardiovasc. Electrophysiol. 2020, 31, 2572–2581. [Google Scholar] [CrossRef]
- Khurram, I.M.; Catanzaro, J.N.; Zimmerman, S.; Zipunnikov, V.; Berger, R.D.; Cheng, A.; Sinha, S.; Dewire, J.; Marine, J.; Spragg, D.; et al. MRI Evaluation of Radiofrequency, Cryothermal, and Laser Left Atrial Lesion Formation in Patients with Atrial Fibrillation. Pacing Clin. Electrophysiol. 2015, 38, 1317–1324. [Google Scholar] [CrossRef]
- Linhart, M.; Alarcon, F.; Borràs, R.; Benito, E.M.; Chipa, F.; Cozzari, J.; Caixal, G.; Enomoto, N.; Carlosena, A.; Guasch, E.; et al. Delayed Gadolinium Enhancement Magnetic Resonance Imaging Detected Anatomic Gap Length in Wide Circumferential Pulmonary Vein Ablation Lesions Is Associated with Recurrence of Atrial Fibrillation. Circ. Arrhythm. Electrophysiol. 2018, 11, e006659. [Google Scholar] [CrossRef]
- Benito, E.M.; Carlosena-Remirez, A.; Guasch, E.; Prat-González, S.; Perea, R.J.; Figueras, R.; Borràs, R.; Andreu, D.; Arbelo, E.; Tolosana, J.M.; et al. Left atrial fibrosis quantification by late gadolinium-enhanced magnetic resonance: A new method to standardize the thresholds for reproducibility. Europace 2017, 19, 1272–1279. [Google Scholar] [CrossRef]
- Althoff, T.F.; Garre, P.; Caixal, G.; Perea, R.; Prat, S.; Tolosana, J.M.; Guasch, E.; Roca-Luque, I.; Arbelo, E.; Sitges, M.; et al. Late gadolinium enhancement-MRI determines definite lesion formation most accurately at three months post-ablation compared to later time points. Pacing Clin. Electrophysiol. 2022, 45, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, R.; Kato, R.; Zviman, M.M.; Dickfeld, T.M.; Roguin, A.; Berger, R.D.; Tomaselli, G.F.; Halperin, H.R. Gaps in the ablation line as a potential cause of recovery from electrical isolation and their visualization using MRI. Circ. Arrhythm. Electrophysiol. 2011, 4, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.P.; Galand, V.; Behar, N.; Daubert, J.-C.; Mabo, P.; Leclercq, C.; Pavin, D. Localization of Residual Conduction Gaps After Wide Antral Circumferential Ablation of Pulmonary Veins. JACC Clin. Electrophysiol. 2019, 5, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.-H.; Po, S.S.; Tung, R.; Liu, Q.; Sheng, X.; Zhang, Z.-W.; Sun, Y.-X.; Yu, L.; Zhang, P.; Fu, G.-S.; et al. Incidence of pulmonary vein conduction recovery in patients without clinical recurrence after ablation of paroxysmal atrial fibrillation: Mechanistic implications. Heart Rhythm. 2014, 11, 969–976. [Google Scholar] [CrossRef]
- Kuck, K.H.; Hoffmann, B.A.; Ernst, S.; Wegscheider, K.; Treszl, A.; Metzner, A.; Eckardt, L.; Lewalter, T.; Breithardt, G.; Willems, S.; et al. Gap-AF–AFNET 1 Investigators. Impact of Complete Versus Incomplete Circumferential Lines Around the Pulmonary Veins During Catheter Ablation of Paroxysmal Atrial Fibrillation: Results From the Gap-Atrial Fibrillation-German Atrial Fibrillation Competence Network 1 Trial. Circ. Arrhythm. Electrophysiol. 2016, 9, e003337. [Google Scholar]
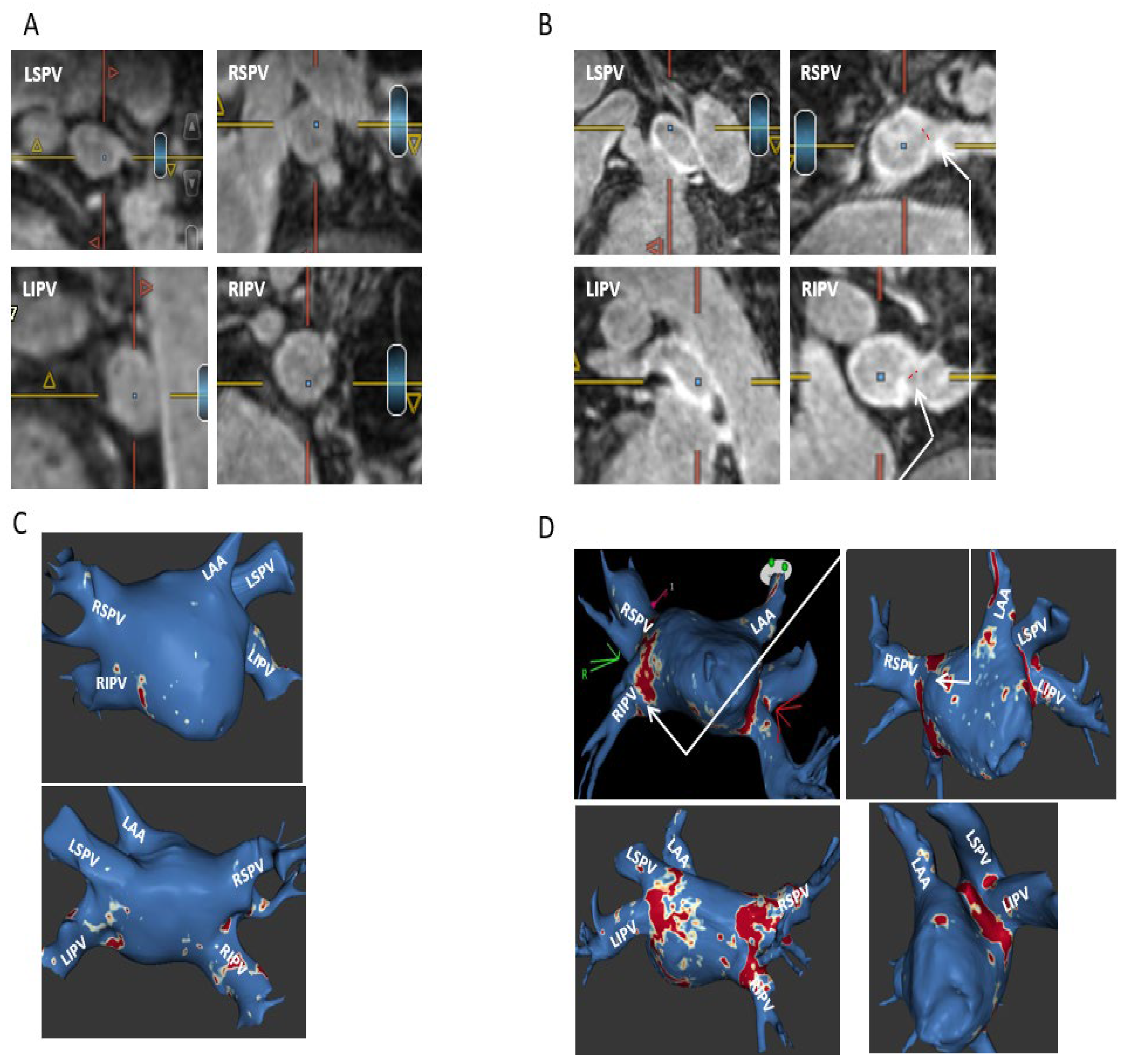
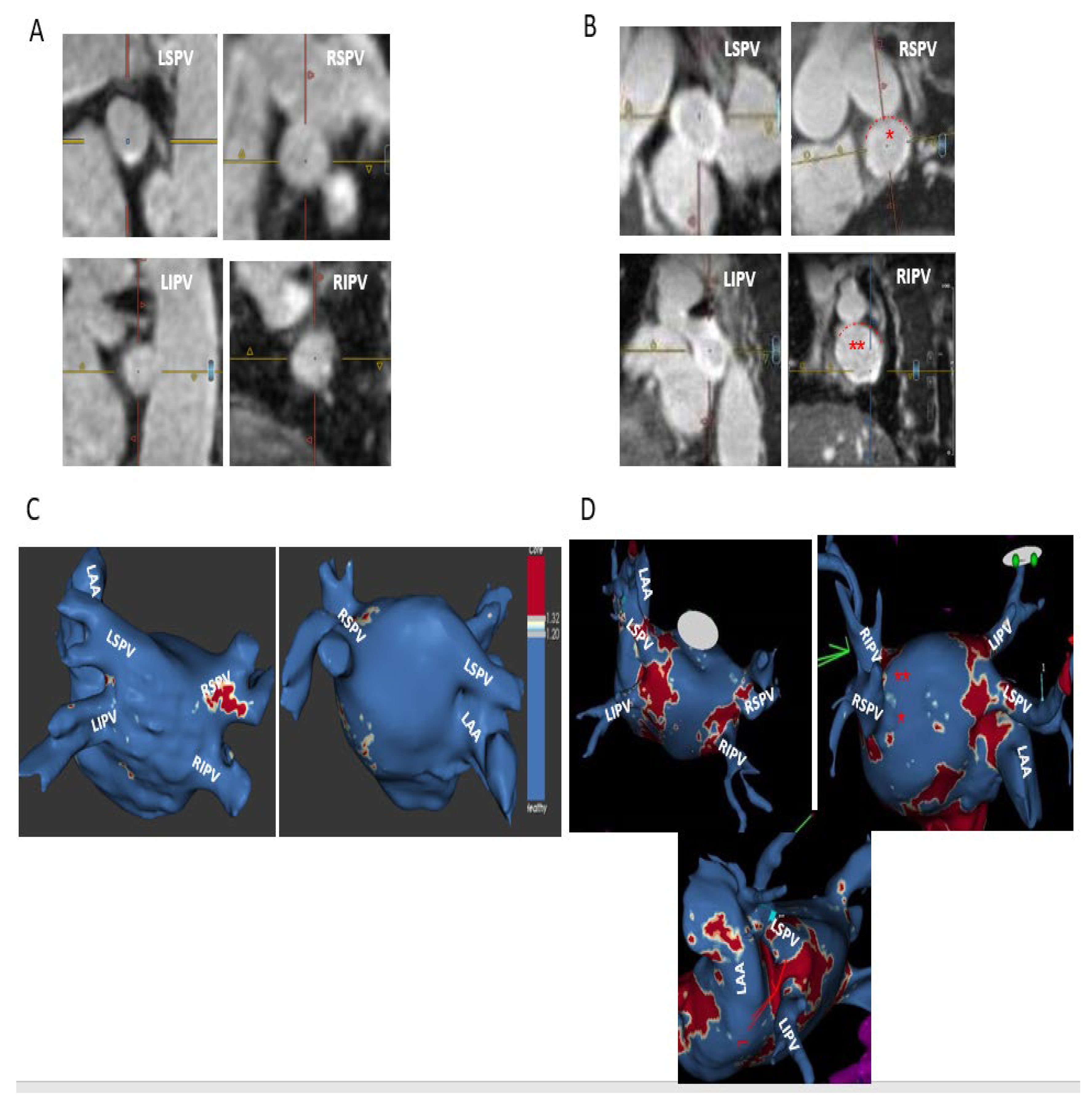
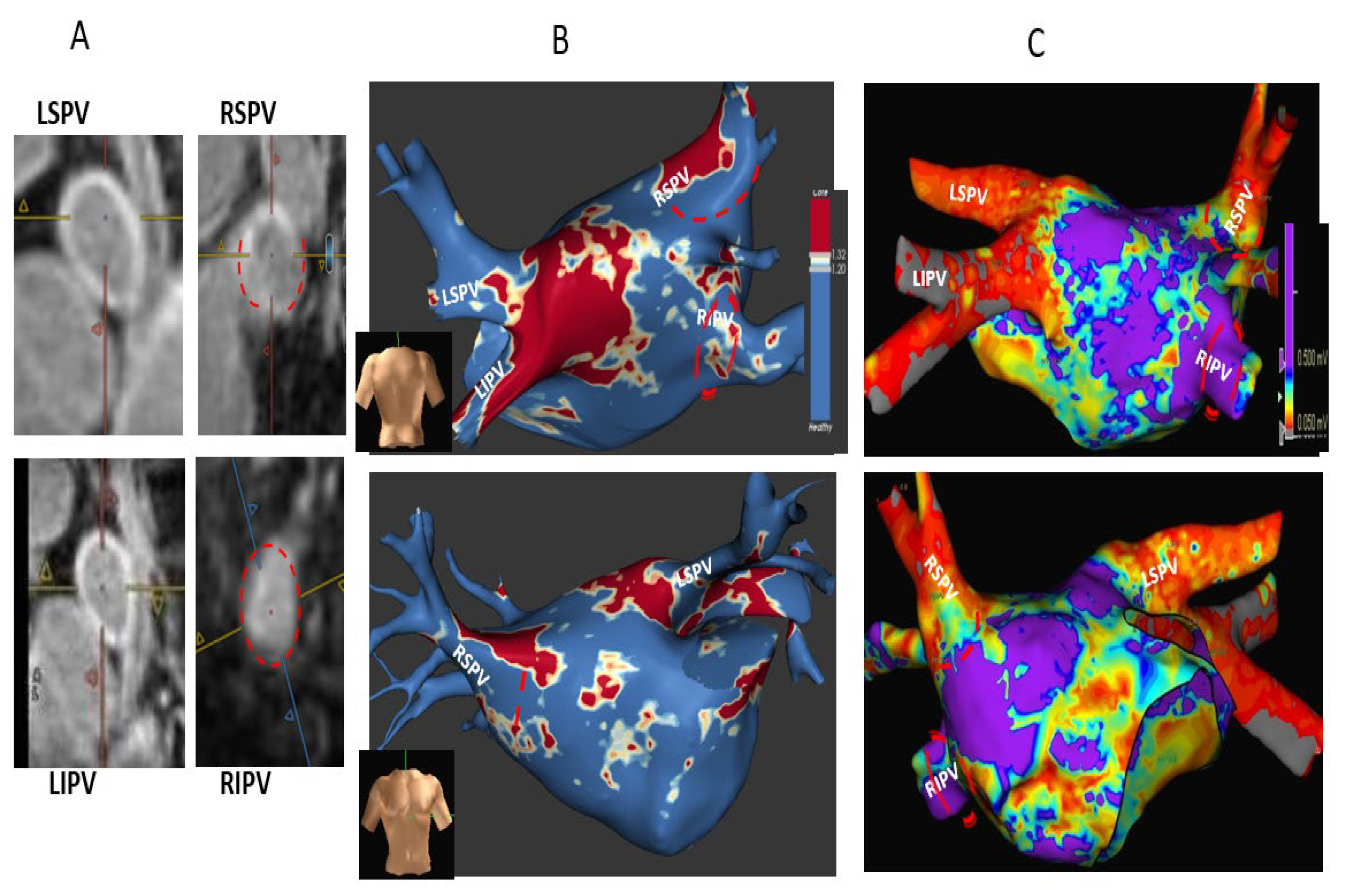
 ) and thus are defined as partial incomplete circumferential PV ostial fibrosis. The right PV gaps are marked (
) and thus are defined as partial incomplete circumferential PV ostial fibrosis. The right PV gaps are marked ( ) both on the orthogonal plane images and the cross-sectional view.
) both on the orthogonal plane images and the cross-sectional view.
 ) and thus are defined as partial incomplete circumferential PV ostial fibrosis. The right PV gaps are marked (
) and thus are defined as partial incomplete circumferential PV ostial fibrosis. The right PV gaps are marked ( ) both on the orthogonal plane images and the cross-sectional view.
) both on the orthogonal plane images and the cross-sectional view.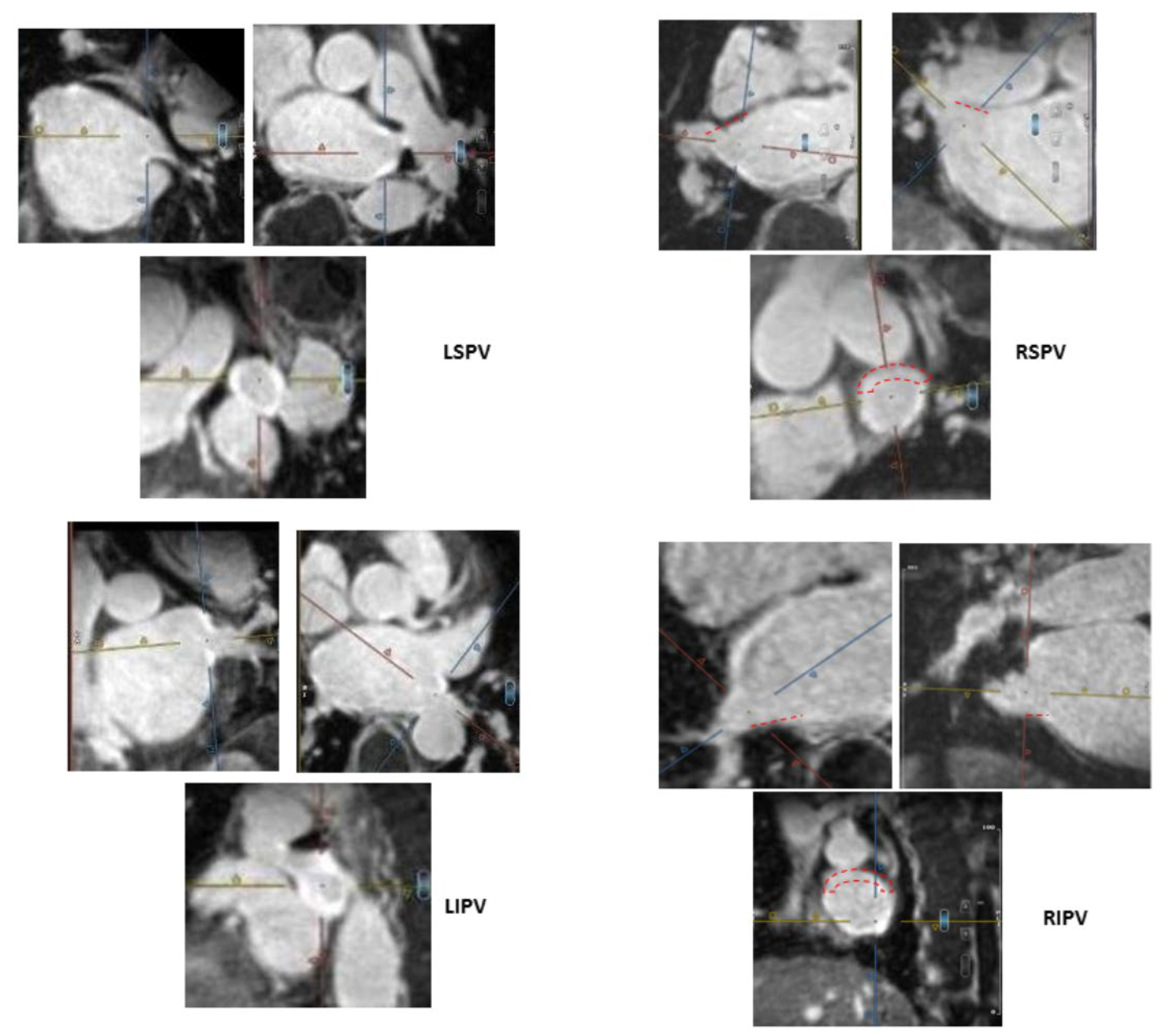

| Total N = 19 | No AF Recurrence N = 14 | AF Recurrence N = 5 | p | |
|---|---|---|---|---|
| Age | 55.2 ± 12 | 54 ± 1 | 59 ± 9 | 0.6 |
| Female | 10/19 (52.6%) | 7/14 (50%) | 3/5 (60%) | 0.9 |
| BMI | 26 ± 2.8 | 25.9 ± 3 | 26 ± 1.8 | 0.9 |
| HTN | 11/19 (57.9%) | 7/14 (50%) | 4/5 (80%) | 0.3 |
| DM | 3/19 (15.8%) | 2/14 (14.3%) | 1/5 (20%) | 0.9 |
| Chronic renal failure | 1/19 (5.2%) | 1/14 (7.2%) | 0/5 (0%) | 0.8 |
| Heart failure | 1/19 (5.2%) | 1/14 (7.2%) | 0/5 (0%) | 0.8 |
| Ischemic heart disease | 0/19 (0%) | 0/14 (0%) | 0/5 (0%) | 1 |
| Obstructive sleep apnea | 3/19 (15.8%) | 2/14 (14.3%) | 1/5 (20%) | 0.9 |
| Persistent AF | 2/19 (10.5%) | 2/14 (14.3%) | 0/5 (0%) | 0.5 |
| AF duration (months) * | 37 ± 31 | 41 ± 33 | 36 ± 30 | 0.7 |
| AAD use post index ablation ** | 11/19 (57.9%) | 7/14 (50%) | 4/5 (80%) | 0.3 |
| LV dysfunction | 0/19 (0%) | 0/19 | 0/5 | 1 |
| Mitral regurgitation mild/mild-moderate | 14/19 (73.7%) | 4/5 (80%) | 11/14 (78.5%) | 0.9 |
| Tricuspid regurgitation mild/mild-moderate | 13/19 (68.5%) | 3/5 (60%) | 10/14 (71.5%) | 0.6 |
| Tricuspid gradient (mmHg) | 21.5 ± 7.5 | 23 ± 4 | 21 ± 9 | 0.5 |
| LA diameter (mm) *** | 39.5 ± 4.3 | 38 ± 3.8 | 42 ± 5.5 | 0.1 |
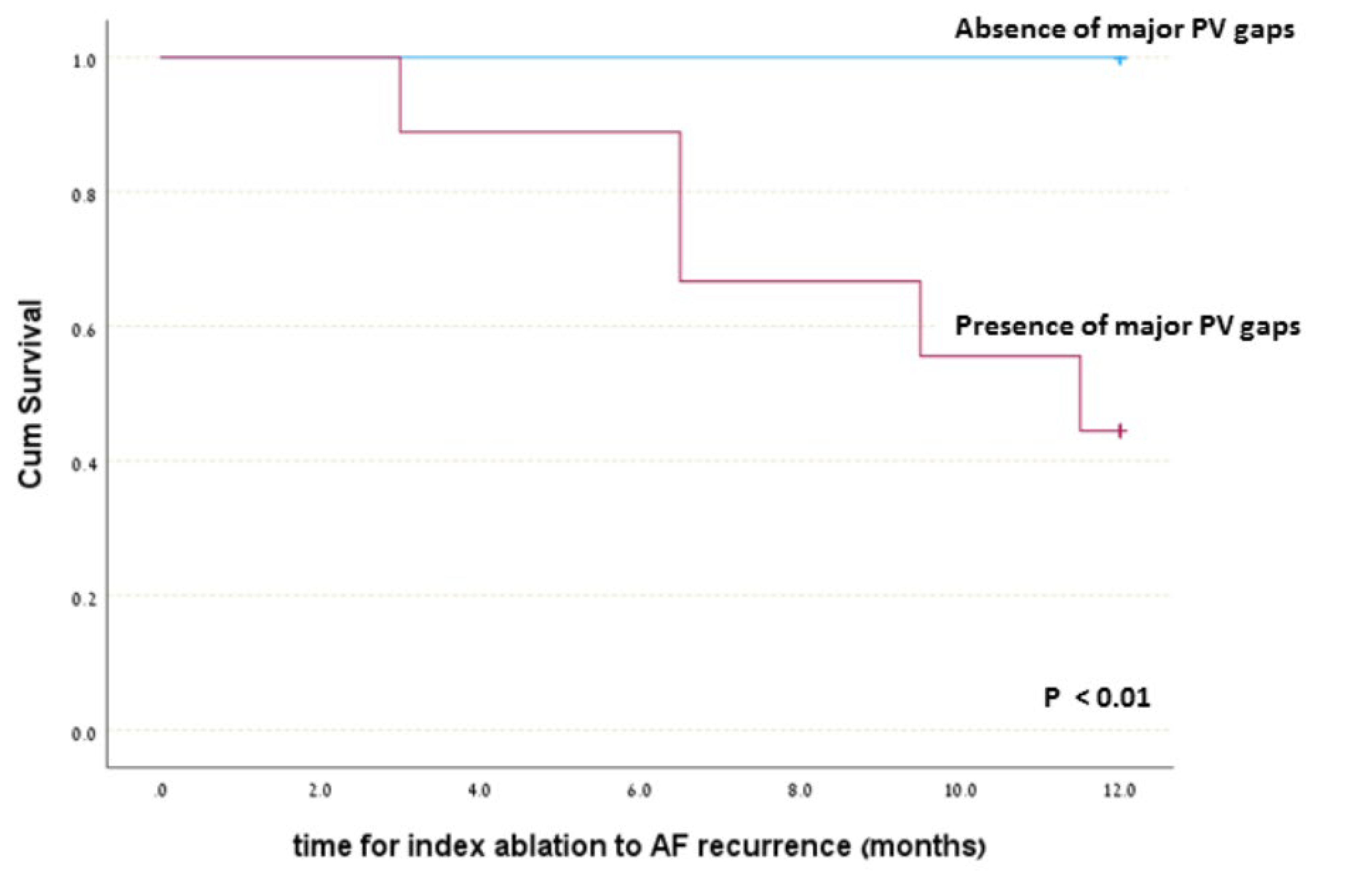
| Partial PV fibrosis (major gap) on post-ablation MRI | 9/19 (47.4%) | 4/14 (28.5%) | 5/5 (100%) | 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rav Acha, M.; Tovia-Brodie, O.; Michowitz, Y.; Bayya, F.; Shaheen, F.F.; Abuhatzera, S.; Medina, A.; Glikson, M.; Wolak, A. Cryoballoon-Induced Circumferential Pulmonary Vein Fibrosis, Assessed by Late Gadolinium-Enhancement Cardiac Magnetic Resonance Imaging, and Its Correlation with Clinical Atrial Fibrillation Recurrence. J. Clin. Med. 2023, 12, 2442. https://doi.org/10.3390/jcm12062442
Rav Acha M, Tovia-Brodie O, Michowitz Y, Bayya F, Shaheen FF, Abuhatzera S, Medina A, Glikson M, Wolak A. Cryoballoon-Induced Circumferential Pulmonary Vein Fibrosis, Assessed by Late Gadolinium-Enhancement Cardiac Magnetic Resonance Imaging, and Its Correlation with Clinical Atrial Fibrillation Recurrence. Journal of Clinical Medicine. 2023; 12(6):2442. https://doi.org/10.3390/jcm12062442
Chicago/Turabian StyleRav Acha, Moshe, Oholi Tovia-Brodie, Yoav Michowitz, Feras Bayya, Fauzi F. Shaheen, Shalom Abuhatzera, Aharon Medina, Michael Glikson, and Arik Wolak. 2023. "Cryoballoon-Induced Circumferential Pulmonary Vein Fibrosis, Assessed by Late Gadolinium-Enhancement Cardiac Magnetic Resonance Imaging, and Its Correlation with Clinical Atrial Fibrillation Recurrence" Journal of Clinical Medicine 12, no. 6: 2442. https://doi.org/10.3390/jcm12062442
APA StyleRav Acha, M., Tovia-Brodie, O., Michowitz, Y., Bayya, F., Shaheen, F. F., Abuhatzera, S., Medina, A., Glikson, M., & Wolak, A. (2023). Cryoballoon-Induced Circumferential Pulmonary Vein Fibrosis, Assessed by Late Gadolinium-Enhancement Cardiac Magnetic Resonance Imaging, and Its Correlation with Clinical Atrial Fibrillation Recurrence. Journal of Clinical Medicine, 12(6), 2442. https://doi.org/10.3390/jcm12062442







