Digital Health Technologies for Post-Discharge Care after Heart Failure Hospitalisation to Relieve Symptoms and Improve Clinical Outcomes
Abstract
1. Introduction
2. Telecare and Remote Disease Management
3. Invasive Remote Monitoring
4. Non-Invasive Monitoring of Volume Status
5. Virtual Wards
6. Telerehabilitation
7. Prognostic Digital Algorithms
8. Patients’ and Healthcare Providers’ Perspectives
9. Recommendations, Knowledge Gaps, and Challenges
10. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. ESC Scientific Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Butt, J.H.; Fosbøl, E.L.; Gerds, T.A.; Andersson, C.; McMurray, J.J.V.; Petrie, M.C.; Gustafsson, F.; Madelaire, C.; Kristensen, S.L.; Gislason, G.H.; et al. Readmission and death in patients admitted with new-onset versus worsening of chronic heart failure: Insights from a nationwide cohort. Eur. J. Heart Fail. 2020, 22, 1777–1785. [Google Scholar] [CrossRef]
- Fraiche, A.M.; Eapen, Z.J.; McClellan, M.B. Moving Beyond the Walls of the Clinic: Opportunities and Challenges to the Future of Telehealth in Heart Failure. JACC Heart Fail. 2017, 5, 297–304. [Google Scholar] [CrossRef]
- Hill, L. Producing an effective care plan in advanced heart failure. Eur. Heart J. Suppl. 2019, 21 (Suppl. M), M61–M63. [Google Scholar] [CrossRef]
- Drews, T.E.I.; Laukkanen, J.; Nieminen, T. Non-invasive home telemonitoring in patients with decompensated heart failure: A systematic review and meta-analysis. ESC Heart Fail. 2021, 8, 3696–3708. [Google Scholar] [CrossRef]
- Romero-Brufau, S.; Wyatt, K.D.; Boyum, P.; Mickelson, M.; Moore, M.; Cognetta-Rieke, C. Implementation of Artificial Intelligence-Based Clinical Decision Support to Reduce Hospital Readmissions at a Regional Hospital. Appl. Clin. Inform. 2020, 11, 570–577. [Google Scholar] [CrossRef]
- Chaudhry, S.I.; Mattera, J.A.; Curtis, J.P.; Spertus, J.A.; Herrin, J.; Lin, Z.; Phillips, C.O.; Hodshon, B.V.; Cooper, L.S.; Krumholz, H.M. Telemonitoring in patients with heart failure. N. Engl. J. Med. 2010, 363, 2301–2309. [Google Scholar] [CrossRef] [PubMed]
- Lyngå, P.; Persson, H.; Hägg-Martinell, A.; Hägglund, E.; Hagerman, I.; Langius-Eklöf, A.; Rosenqvist, M. Weight monitoring in patients with severe heart failure (WISH). A randomized controlled trial. Eur. J. Heart Fail. 2012, 14, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Angermann, C.E.; Störk, S.; Gelbrich, G.; Faller, H.; Jahns, R.; Frantz, S.; Loeffler, M.; Ertl, G.; Competence Network Heart Failure. Mode of action and effects of standardized collaborative disease management on mortality and morbidity in patients with systolic heart failure: The Interdisciplinary Network for Heart Failure (INH) study. Circ. Heart Fail. 2012, 5, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.G.; Louis, A.A.; Rigby, A.S.; Janssens, U.; Balk, A.H.; TEN-HMS Investigators. Noninvasive home telemonitoring for patients with heart failure at high risk of recurrent admission and death: The Trans-European Network-Home-Care Management System (TEN-HMS) study. J. Am. Coll. Cardiol. 2005, 45, 1654–1664. [Google Scholar] [CrossRef]
- Ong, M.K.; Romano, P.S.; Edgington, S.; Aronow, H.U.; Auerbach, A.D.; Black, J.T.; De Marco, T.; Escarce, J.J.; Evangelista, L.S.; Hanna, B.; et al. Better Effectiveness After Transition–Heart Failure (BEAT-HF) Research Group. Effectiveness of Remote Patient Monitoring After Discharge of Hospitalized Patients With Heart Failure: The Better Effectiveness After Transition—Heart Failure (BEAT-HF) Randomized Clinical Trial. JAMA Intern. Med. 2016, 176, 310–318. [Google Scholar]
- Koehler, F.; Winkler, S.; Schieber, M.; Sechtem, U.; Stangl, K.; Böhm, M.; Boll, H.; Baumann, G.; Honold, M.; Koehler, K.; et al. Telemedical Interventional Monitoring in Heart Failure Investigators. Impact of remote telemedical management on mortality and hospitalizations in ambulatory patients with chronic heart failure: The telemedical interventional monitoring in heart failure study. Circulation 2011, 123, 1873–1880. [Google Scholar] [PubMed]
- Galinier, M.; Roubille, F.; Berdague, P.; Brierre, G.; Cantie, P.; Dary, P.; Ferradou, J.M.; Fondard, O.; Labarre, J.P.; Mansourati, J.; et al. OSICAT Investigators. Telemonitoring versus standard care in heart failure: A randomised multicentre trial. Eur. J. Heart Fail. 2020, 22, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Haynes, S.C.; Tancredi, D.J.; Tong, K.; Hoch, J.S.; Ong, M.K.; Ganiats, T.G.; Evangelista, L.S.; Black, J.T.; Auerbach, A.; Romano, P.S. Better Effectiveness After Transition–Heart Failure (BEAT-HF) Research Group. Association of Adherence to Weight Telemonitoring With Health Care Use and Death: A Secondary Analysis of a Randomized Clinical Trial. JAMA Netw. Open. 2020, 3, e2010174. [Google Scholar] [CrossRef] [PubMed]
- Koehler, F.; Koehler, K.; Deckwart, O.; Prescher, S.; Wegscheider, K.; Kirwan, B.A.; Winkler, S.; Vettorazzi, E.; Bruch, L.; Oeff, M.; et al. Efficacy of telemedical interventional management in patients with heart failure (TIM-HF2): A randomised, controlled, parallel-group, unmasked trial. Lancet 2018, 392, 1047–1057. [Google Scholar] [CrossRef]
- Nouryan, C.N.; Morahan, S.; Pecinka, K.; Akerman, M.; Lesser, M.; Chaikin, D.; Castillo, S.; Zhang, M.; Pekmezaris, R. Home Telemonitoring of Community-Dwelling Heart Failure Patients After Home Care Discharge. Telemed. J. e-Health 2019, 25, 447–454. [Google Scholar] [CrossRef]
- Dawson, N.L.; Hull, B.P.; Vijapura, P.; Dumitrascu, A.G.; Ball, C.T.; Thiemann, K.M.; Maniaci, M.J.; Burton, M.C. Home Telemonitoring to Reduce Readmission of High-Risk Patients: A Modified Intention-to-Treat Randomized Clinical Trial. J. Gen. Intern. Med. 2021, 36, 3395–3401. [Google Scholar] [CrossRef]
- Krzesiński, P.; Jankowska, E.A.; Siebert, J.; Galas, A.; Piotrowicz, K.; Stańczyk, A.; Siwołowski, P.; Gutknecht, P.; Chrom, P.; Murawski, P.; et al. Effects of an outpatient intervention comprising nurse-led non-invasive assessments, telemedicine support and remote cardiologists’ decisions in patients with heart failure (AMULET Study): A randomised controlled trial. Eur. J. Heart Fail. 2022, 24, 565–577. [Google Scholar] [CrossRef]
- Inglis, S.C.; Clark, R.A.; Dierckx, R.; Prieto-Merino, D.; Cleland, J.G. Structured telephone support or non-invasive telemonitoring for patients with heart failure. Cochrane Database Syst. Rev. 2015, 2015, CD007228. [Google Scholar] [CrossRef]
- Lin, M.H.; Yuan, W.L.; Huang, T.C.; Zhang, H.F.; Mai, J.T.; Wang, J.F. Clinical effectiveness of telemedicine for chronic heart failure: A systematic review and meta-analysis. J. Investig. Med. 2017, 65, 899–911. [Google Scholar] [CrossRef]
- Zhu, Y.; Gu, X.; Xu, C. Effectiveness of telemedicine systems for adults with heart failure: A meta-analysis of randomized controlled trials. Heart Fail. Rev. 2020, 25, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Hafkamp, F.J.; Tio, R.A.; Otterspoor, L.C.; de Greef, T.; van Steenbergen, G.J.; van de Ven, A.R.T.; Smits, G.; Post, H.; van Veghel, D. Optimal effectiveness of heart failure management—An umbrella review of meta-analyses examining the effectiveness of interventions to reduce (re)hospitalizations in heart failure. Heart Fail. Rev. 2022, 27, 1683–1748. [Google Scholar] [CrossRef]
- Berry, R.; Keeling, P. Compliance with Telemonitoring in Heart Failure. Are Study Findings Representative of Reality?: A Narrative Literature Review. Telemed. J. e-Health. 2022, 28, 467–480. [Google Scholar] [CrossRef]
- Aimo, A.; Vergaro, G.; Giannoni, A.; Emdin, M. Wet is bad: Residual congestion predicts worse prognosis in acute heart failure. Int. J. Cardiol. 2018, 258, 201–202. [Google Scholar] [CrossRef]
- Abraham, W.T.; Adamson, P.B.; Bourge, R.C.; Aaron, M.F.; Costanzo, M.R.; Stevenson, L.W.; Strickland, W.; Neelagaru, S.; Raval, N.; Krueger, S.; et al. CHAMPION Trial Study Group. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: A randomised controlled trial. Lancet 2011, 377, 658–666. [Google Scholar] [CrossRef]
- Givertz, M.M.; Stevenson, L.W.; Costanzo, M.R.; Bourge, R.C.; Bauman, J.G.; Ginn, G.; Abraham, W.T.; CHAMPION Trial Investigators. Pulmonary Artery Pressure-Guided Management of Patients With Heart Failure and Reduced Ejection Fraction. J. Am. Coll. Cardiol. 2017, 70, 1875–1886. [Google Scholar] [CrossRef] [PubMed]
- Angermann, C.E.; Assmus, B.; Anker, S.D.; Asselbergs, F.W.; Brachmann, J.; Brett, M.E.; Brugts, J.J.; Ertl, G.; Ginn, G.; Hilker, L.; et al. MEMS-HF Investigators. Pulmonary artery pressure-guided therapy in ambulatory patients with symptomatic heart failure: The CardioMEMS European Monitoring Study for Heart Failure (MEMS-HF). Eur. J. Heart Fail. 2020, 22, 1891–1901. [Google Scholar] [CrossRef]
- Lindenfeld, J.; Zile, M.R.; Desai, A.S.; Bhatt, K.; Ducharme, A.; Horstmanshof, D.; Krim, S.R.; Maisel, A.; Mehra, M.R.; Paul, S.; et al. Haemodynamic-guided management of heart failure (GUIDE-HF): A randomised controlled trial. Lancet 2021, 398, 991–1001. [Google Scholar] [CrossRef]
- Conraads, V.M.; Tavazzi, L.; Santini, M.; Oliva, F.; Gerritse, B.; Yu, C.M.; Cowie, M.R. Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: The SENSE-HF trial. Eur. Heart J. 2011, 32, 2266–2273. [Google Scholar] [CrossRef] [PubMed]
- Domenichini, G.; Rahneva, T.; Diab, I.G.; Dhillon, O.S.; Campbell, N.G.; Finlay, M.C.; Baker, V.; Hunter, R.J.; Earley, M.J.; Schilling, R.J. The lung impedance monitoring in treatment of chronic heart failure (the LIMIT-CHF study). Europace 2016, 18, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Heist, E.K.; Herre, J.M.; Binkley, P.F.; Van Bakel, A.B.; Porterfield, J.G.; Porterfield, L.M.; Qu, F.; Turkel, M.; Pavri, B.B.; DEFEAT-PE Study Investigators. Analysis of different device-based intrathoracic impedance vectors for detection of heart failure events (from the Detect Fluid Early from Intrathoracic Impedance Monitoring study). Am. J. Cardiol. 2014, 114, 1249–1256. [Google Scholar] [CrossRef]
- Whellan, D.J.; Ousdigian, K.T.; Al-Khatib, S.M.; Pu, W.; Sarkar, S.; Porter, C.B.; Pavri, B.B.; O’Connor, C.M.; PARTNERS Study Investigators. Combined heart failure device diagnostics identify patients at higher risk of subsequent heart failure hospitalizations: Results from PARTNERS HF (Program to Access and Review Trending Information and Evaluate Correlation to Symptoms in Patients With Heart Failure) study. J. Am. Coll. Cardiol. 2010, 55, 1803–1810. [Google Scholar]
- Zile, M.R.; Koehler, J.; Sarkar, S.; Butler, J. Prediction of worsening heart failure events and all-cause mortality using an individualized risk stratification strategy. ESC Heart Fail. 2020, 7, 4277–4289. [Google Scholar] [CrossRef]
- Santini, L.; D’Onofrio, A.; Dello Russo, A.; Calò, L.; Pecora, D.; Favale, S.; Petracci, B.; Molon, G.; Bianchi, V.; De Ruvo, E.; et al. Prospective evaluation of the multisensor HeartLogic algorithm for heart failure monitoring. Clin. Cardiol. 2020, 43, 691–697. [Google Scholar] [CrossRef]
- Hindricks, G.; Taborsky, M.; Glikson, M.; Heinrich, U.; Schumacher, B.; Katz, A.; Brachmann, J.; Lewalter, T.; Goette, A.; Block, M.; et al. IN-TIME study group*. Implant-based multiparameter telemonitoring of patients with heart failure (IN-TIME): A randomised controlled trial. Lancet 2014, 384, 583–590. [Google Scholar] [CrossRef] [PubMed]
- De Simone, A.; Leoni, L.; Luzi, M.; Amellone, C.; Stabile, G.; La Rocca, V.; Capucci, A.; D’onofrio, A.; Ammendola, E.; Accardi, F.; et al. Remote monitoring improves outcome after ICD implantation: The clinical efficacy in the management of heart failure (EFFECT) study. Europace 2015, 17, 1267–1275. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. ESC Scientific Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [PubMed]
- Iaconelli, A.; Pellicori, P.; Caiazzo, E.; Rezig, A.O.M.; Bruzzese, D.; Maffia, P.; Cleland, J.G.F. Implanted haemodynamic telemonitoring devices to guide management of heart failure: A review and meta-analysis of randomised trials. Clin. Res. Cardiol. 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Shochat, M.; Shotan, A.; Blondheim, D.S.; Kazatsker, M.; Dahan, I.; Asif, A.; Shochat, I.; Frimerman, A.; Rozenman, Y.; Meisel, S.R. Derivation of baseline lung impedance in chronic heart failure patients: Use for monitoring pulmonary congestion and predicting admissions for decompensation. J. Clin. Monit. Comput. 2015, 29, 341–349. [Google Scholar] [CrossRef]
- Shochat, M.K.; Shotan, A.; Blondheim, D.S.; Kazatsker, M.; Dahan, I.; Asif, A.; Rozenman, Y.; Kleiner, I.; Weinstein, J.M.; Frimerman, A.; et al. Non-Invasive Lung IMPEDANCE-Guided Preemptive Treatment in Chronic Heart Failure Patients: A Randomized Controlled Trial (IMPEDANCE-HF Trial). J. Card. Fail. 2016, 22, 713–722. [Google Scholar] [CrossRef]
- Shochat, M.K.; Fudim, M.; Shotan, A.; Blondheim, D.S.; Kazatsker, M.; Dahan, I.; Asif, A.; Rozenman, Y.; Kleiner, I.; Weinstein, J.M.; et al. Prediction of readmissions and mortality in patients with heart failure: Lessons from the IMPEDANCE-HF extended trial. ESC Heart Fail. 2018, 5, 788–799. [Google Scholar] [CrossRef]
- Amir, O.; Rappaport, D.; Zafrir, B.; Abraham, W.T. A novel approach to monitoring pulmonary congestion in heart failure: Initial animal and clinical experiences using remote dielectric sensing technology. Congest. Heart Fail. 2013, 19, 149–155. [Google Scholar] [CrossRef]
- Abraham, W.T.; Anker, A.; Burkhoff, D.; Cleland, J.; Gorodeski, E.; Jaarsma, T.; Small, R.; Lindenfeld, J.; Miller, A.; Ogenstad, S.; et al. Primary Results of the Sensible Medical Innovations Lung Fluid Status Monitor Allows Reducing Readmission Rate of Heart Failure Patients (smile) Trial. J. Card. Fail. 2019, 25, 938. [Google Scholar] [CrossRef]
- Bensimhon, D.; Alali, S.A.; Curran, L.; Gelbart, E.; Garman, D.W.V.; Taylor, R.; Chase, P.; Peacock, W.F. The use of the reds noninvasive lung fluid monitoring system to assess readiness for discharge in patients hospitalized with acute heart failure: A pilot study. Heart Lung. 2021, 50, 59–64. [Google Scholar] [CrossRef]
- Dovancescu, S.; Saczynski, J.S.; Darling, C.E.; Riistama, J.; Sert Kuniyoshi, F.; Meyer, T.; Goldberg, R.; McManus, D.D. Detecting Heart Failure Decompensation by Measuring Transthoracic Bioimpedance in the Outpatient Setting: Rationale and Design of the SENTINEL-HF Study. JMIR Res. Protoc. 2015, 4, e121. [Google Scholar] [CrossRef]
- Smeets, C.J.P.; Lee, S.; Groenendaal, W.; Squillace, G.; Vranken, J.; De Cannière, H.; Van Hoof, C.; Grieten, L.; Mullens, W.; Nijst, P.; et al. The Added Value of In-Hospital Tracking of the Efficacy of Decongestion Therapy and Prognostic Value of a Wearable Thoracic Impedance Sensor in Acutely Decompensated Heart Failure With Volume Overload: Prospective Cohort Study. JMIR Cardio 2020, 4, e12141. [Google Scholar] [CrossRef]
- Dhalla, I.A.; O’Brien, T.; Morra, D.; Thorpe, K.E.; Wong, B.M.; Mehta, R.; Frost, D.W.; Abrams, H.; Ko, F.; Van Rooyen, P.; et al. Effect of a postdischarge virtual ward on readmission or death for high-risk patients: A randomized clinical trial. JAMA 2014, 312, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Low, L.L.; Tan, S.Y.; Ng, M.J.; Tay, W.Y.; Ng, L.B.; Balasubramaniam, K.; Towle, R.M.; Lee, K.H. Applying the Integrated Practice Unit Concept to a Modified Virtual Ward Model of Care for Patients at Highest Risk of Readmission: A Randomized Controlled Trial. PLoS ONE 2017, 12, e0168757. [Google Scholar] [CrossRef]
- Uminski, K.; Komenda, P.; Whitlock, R.; Ferguson, T.; Nadurak, S.; Hochheim, L.; Tangri, N.; Rigatto, C. Effect of post-discharge virtual wards on improving outcomes in heart failure and non-heart failure populations: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0196114. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.M.; Goldberg, J.M.; Molinsky, R.L.; Hanson, M.A.; Castaño, A.; Raza, S.S.; Janas, N.; Celano, P.; Kapoor, K.; Telaraja, J.; et al. A Virtual Cardiovascular Care Program for Prevention of Heart Failure Readmissions in a Skilled Nursing Facility Population: Retrospective Analysis. JMIR Cardio 2021, 5, e29101. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, U.; McAlister, F.A. Comparison of Mortality and Hospital Readmissions Among Patients Receiving Virtual Ward Transitional Care vs Usual Postdischarge Care: A Systematic Review and Meta-analysis. JAMA Netw. Open 2022, 5, e2219113. [Google Scholar] [CrossRef]
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Bäck, M.; Benetos, A.; Biffi, A.; Boavida, J.M.; Capodanno, D.; et al. ESC Scientific Document Group. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2021, 42, 3227–3337. [Google Scholar] [CrossRef] [PubMed]
- Passantino, A.; Dalla Vecchia, L.A.; Corrà, U.; Scalvini, S.; Pistono, M.; Bussotti, M.; Gambarin, F.I.; Scrutinio, D.; La Rovere, M.T. The Future of Exercise-Based Cardiac Rehabilitation for Patients With Heart Failure. Front. Cardiovasc. Med. 2021, 8, 709898. [Google Scholar] [CrossRef] [PubMed]
- Isernia, S.; Pagliari, C.; Morici, N.; Toccafondi, A.; Banfi, P.I.; Rossetto, F.; Borgnis, F.; Tavanelli, M.; Brambilla, L.; Baglio, F.; et al. Telerehabilitation Approaches for People with Chronic Heart Failure: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 12, 64. [Google Scholar] [CrossRef]
- Piotrowicz, E.; Pencina, M.J.; Opolski, G.; Zareba, W.; Banach, M.; Kowalik, I.; Orzechowski, P.; Szalewska, D.; Pluta, S.; Glówczynska, R.; et al. Effects of a 9-Week Hybrid Comprehensive Telerehabilitation Program on Long-term Outcomes in Patients With Heart Failure: The Telerehabilitation in Heart Failure Patients (TELEREH-HF) Randomized Clinical Trial. JAMA Cardiol. 2020, 5, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, A.H.; Silva Cardoso, J.; Rocha, A.; Moreira, E.; Azevedo, L.F. Effectiveness of Tele-rehabilitation Programs in Heart Failure: A Systematic Review and Meta-analysis. Health Serv. Insights 2021, 14, 11786329211021668. [Google Scholar] [CrossRef]
- Allain, F.; Loizeau, V.; Chaufourier, L.; Hallouche, M.; Herrou, L.; Hodzic, A.; Blanchart, K.; Belin, A.; Manrique, A.; Milliez, P.; et al. Usefulness of a personalized algorithm-based discharge checklist in patients hospitalized for acute heart failure. ESC Heart Fail. 2020, 7, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Fahimi, F.; Guo, Y.; Tong, S.C.; Ng, A.; Bing, S.O.Y.; Choo, B.; Weiliang, H.; Lee, S.; Ramasamy, S.; Chow, W.L.; et al. Vital Signs Telemonitoring Programme Improves the Dynamic Prediction of Readmission Risk in Patients with Heart Failure. AMIA Annu. Symp. Proc. 2021, 2020, 432–441. [Google Scholar] [PubMed]
- Stehlik, J.; Schmalfuss, C.; Bozkurt, B.; Nativi-Nicolau, J.; Wohlfahrt, P.; Wegerich, S.; Rose, K.; Ray, R.; Schofield, R.; Deswal, A.; et al. Continuous Wearable Monitoring Analytics Predict Heart Failure Hospitalization: The LINK-HF Multicenter Study. Circ. Heart Fail. 2020, 13, e006513. [Google Scholar] [CrossRef]
- Croon, P.M.; Selder, J.L.; Allaart, C.P.; Bleijendaal, H.; Chamuleau, S.A.J.; Hofstra, L.; Išgum, I.; Ziesemer, K.A.; Winter, M.M. Current state of artificial intelligence-based algorithms for hospital admission prediction in patients with heart failure: A scoping review. Eur. Heart J. Digit. Health 2022, 3, 415–425. [Google Scholar] [CrossRef]
- Seto, E.; Ross, H.; Tibbles, A.; Wong, S.; Ware, P.; Etchells, E.; Kobulnik, J.; Chibber, T.; Poon, S. A Mobile Phone-Based Telemonitoring Program for Heart Failure Patients After an Incidence of Acute Decompensation (Medly-AID): Protocol for a Randomized Controlled Trial. JMIR Res. Protoc. 2020, 9, e15753. [Google Scholar] [CrossRef]
- Tersalvi, G.; Winterton, D.; Cioffi, G.M.; Ghidini, S.; Roberto, M.; Biasco, L.; Pedrazzini, G.; Dauw, J.; Ameri, P.; Vicenzi, M. Telemedicine in Heart Failure During COVID-19: A Step Into the Future. Front. Cardiovasc. Med. 2020, 7, 612818. [Google Scholar] [CrossRef]
- Cowie, M.R.; Lam, C.S.P. Remote monitoring and digital health tools in CVD management. Nat. Rev. Cardiol. 2021, 18, 457–458. [Google Scholar] [CrossRef]
- Viitanen, J.; Valkonen, P.; Savolainen, K.; Karisalmi, N.; Hölsä, S.; Kujala, S. Patient Experience from an eHealth Perspective: A Scoping Review of Approaches and Recent Trends. Yearb. Med. Inform. 2022, 31, 136–145. [Google Scholar] [CrossRef]
- Meyer, M.A. COVID-19 Pandemic Accelerates Need to Improve Online Patient Engagement Practices to Enhance Patient Experience. J. Patient Exp. 2020, 7, 657–664. [Google Scholar] [CrossRef]
- Nanda, M.; Sharma, R. A Review of Patient Satisfaction and Experience with Telemedicine: A Virtual Solution During and Beyond COVID-19 Pandemic. Telemed. J. e-Health 2021, 27, 1325–1331. [Google Scholar] [CrossRef]
- Khader, H.; Alsayed, A.; Hasoun, L.Z.; Alnatour, D.; Awajan, D.; Alhosanie, T.N.; Samara, A. Pharmaceutical care and telemedicine during COVID-19: A cross-sectional study based on pharmacy students, pharmacists, and physicians in Jordan. Pharmacia 2022, 69, 891–901. [Google Scholar] [CrossRef]
- Alsayed, A.R.; Halloush, S.; Hasoun, L.; Alnatour, D.; Al-Dulaimi, A.; Alnajjar, M.S.; Blaibleh, A.; Al-Imam, A.; Alshammari, F.; Khader, H.A. Perspectives of the community in the developing countries toward telemedicine and pharmaceutical care during the COVID-19 pandemic. Pharm. Pract. 2022, 20, 2618. [Google Scholar] [CrossRef]
- Ndayishimiye, C.; Lopes, H.; Middleton, J. A systematic scoping review of digital health technologies during COVID-19: A new normal in primary health care delivery. Health Technol. 2023, 13, 273–284. [Google Scholar] [CrossRef]
- Wrzosek, N.; Zimmermann, A.; Balwicki, Ł. A Survey of Patients’ Opinions and Preferences on the Use of E-Prescriptions in Poland. Int. J. Environ. Res. Public Health 2021, 18, 9769. [Google Scholar] [CrossRef]
- Addotey-Delove, M.; Scott, R.E.; Mars, M. Healthcare Workers’ Perspectives of mHealth Adoption Factors in the Developing World: Scoping Review. Int. J. Environ. Res. Public Health 2023, 20, 1244. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee Members; ACC/AHA Joint Committee Members. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J. Card. Fail. 2022, 28, e1–e167. [Google Scholar] [CrossRef] [PubMed]
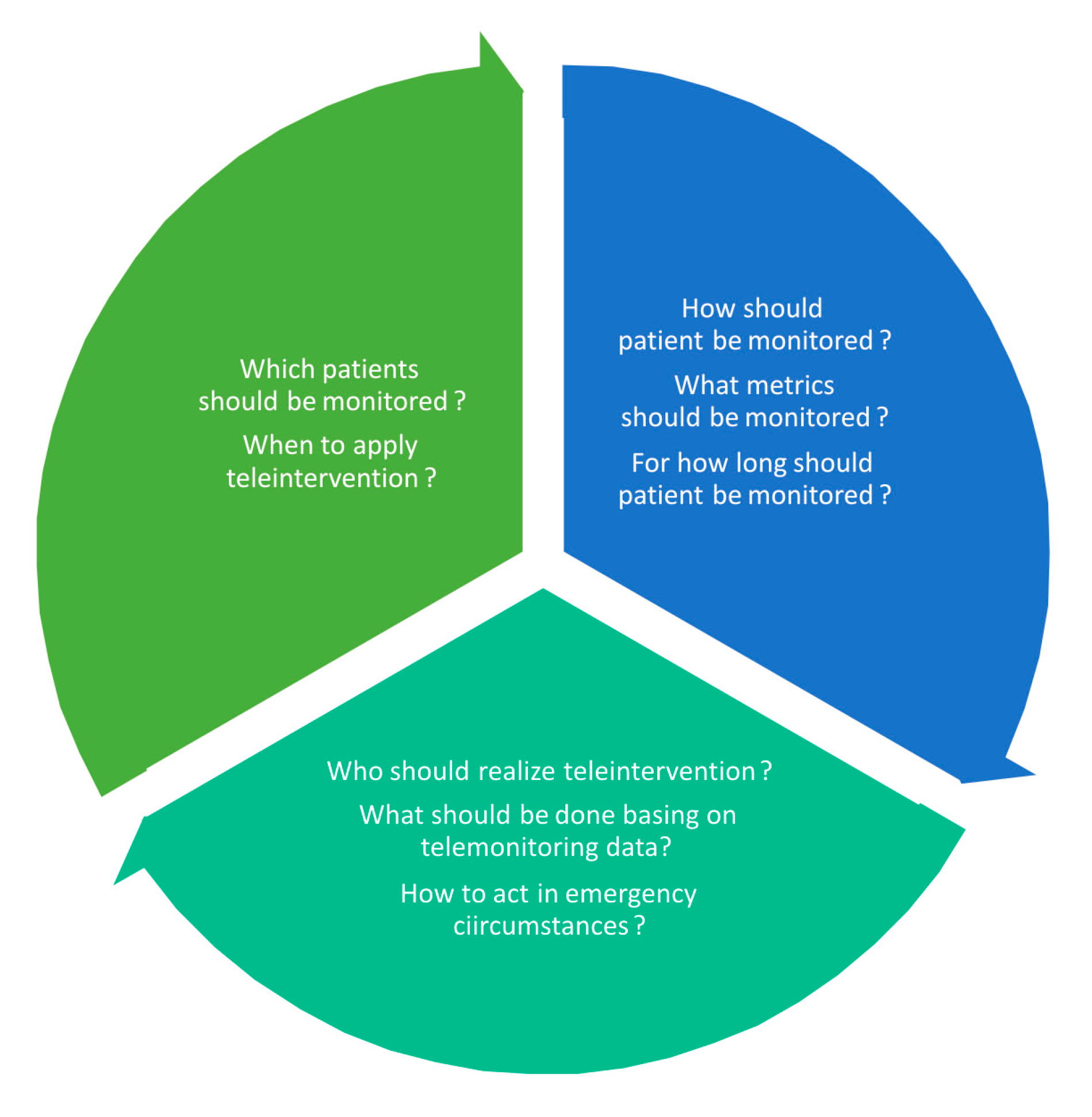
| Telecare and remote disease management |
|
| Telemonitoring |
|
| Supporting digital tools |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krzesiński, P. Digital Health Technologies for Post-Discharge Care after Heart Failure Hospitalisation to Relieve Symptoms and Improve Clinical Outcomes. J. Clin. Med. 2023, 12, 2373. https://doi.org/10.3390/jcm12062373
Krzesiński P. Digital Health Technologies for Post-Discharge Care after Heart Failure Hospitalisation to Relieve Symptoms and Improve Clinical Outcomes. Journal of Clinical Medicine. 2023; 12(6):2373. https://doi.org/10.3390/jcm12062373
Chicago/Turabian StyleKrzesiński, Paweł. 2023. "Digital Health Technologies for Post-Discharge Care after Heart Failure Hospitalisation to Relieve Symptoms and Improve Clinical Outcomes" Journal of Clinical Medicine 12, no. 6: 2373. https://doi.org/10.3390/jcm12062373
APA StyleKrzesiński, P. (2023). Digital Health Technologies for Post-Discharge Care after Heart Failure Hospitalisation to Relieve Symptoms and Improve Clinical Outcomes. Journal of Clinical Medicine, 12(6), 2373. https://doi.org/10.3390/jcm12062373






