Only EBUS-Guided Mediastinal Lymph Node Cryobiopsy Enabled Immunotherapy in a Patient with Non-Small Cell Lung Cancer
Abstract
1. Introduction
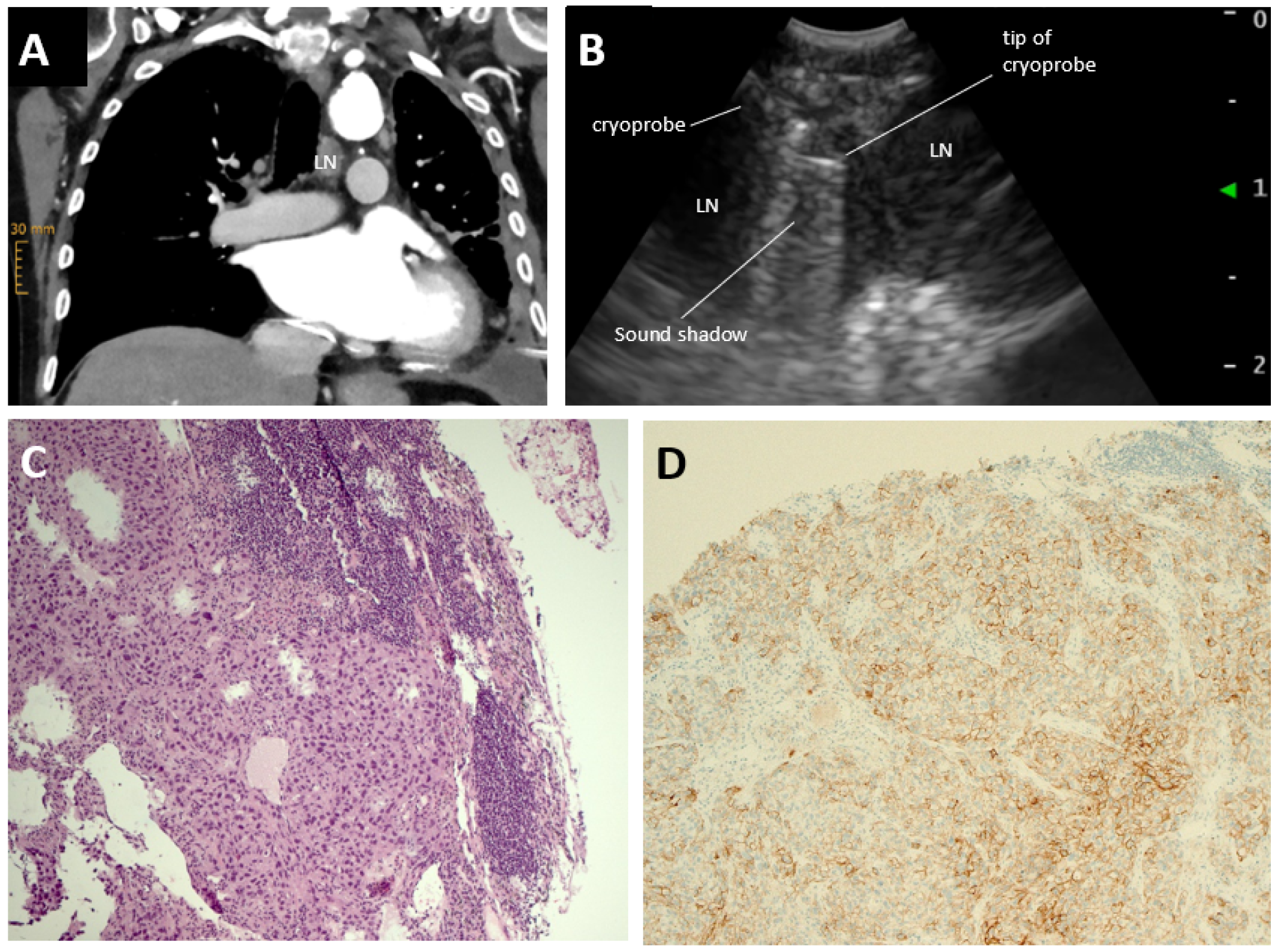
2. Case Report
3. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhou, C.; Wu, Y.L.; Chen, G.; Feng, J.; Liu, X.-Q.; Wang, C.; Zhang, S.; Wang, J.; Zhou, S.; Ren, S.; et al. Final overall survival results from a randomised, phase III study of erlotinib versus chemotherapy as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer (OPTIMAL, CTONG-0802). Ann. Oncol. 2015, 26, 1877–1883. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.C.H.; Schuler, M.; Sebastian, M.; Popat, S.; Yamamoto, N.; Zhou, C.; Hu, C.P.; O’Byrne, K.; Feng, J.; Lu, S.; et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): Analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol. 2015, 16, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, S.S.; Vansteenkiste, J.; Planchard, D.; Cho, B.C.; Gray, J.E.; Ohe, Y.; Zhou, C.; Reungwetwattana, T.; Cheng, Y.; Chewaskulyong, B.; et al. Overall Survival with Osimertinib in Untreated, EGFR-Mutated Advanced NSCLC. N. Engl. J. Med. 2020, 382, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Mazzaschi, G.; Barbieri, F.; Passiglia, F.; Mazzoni, F.; Berardi, R.; Proto, C.; Cecere, F.L.; Pilotto, S.; Scotti, V.; et al. First-line pembrolizumab in advanced non–small cell lung cancer patients with poor performance status. Eur. J. Cancer 2020, 130, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Nosaki, K.; Saka, H.; Hosomi, Y.; Baas, P.; de Castro, G.; Reck, M.; Wu, Y.-L.; Brahmer, J.R.; Felip, E.; Sawada, T.; et al. Safety and efficacy of pembrolizumab monotherapy in elderly patients with PD-L1–positive advanced non–small-cell lung cancer: Pooled analysis from the KEYNOTE-010, KEYNOTE-024, and KEYNOTE-042 studies. Lung Cancer 2019, 135, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Elias, R.; Giobbie-Hurder, A.; McCleary, N.J.; Ott, P.; Hodi, F.S.; Rahma, O. Efficacy of PD-1 & PD-L1 inhibitors in older adults: A meta-analysis. J. Immunother. Cancer 2018, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Griesinger, F.; Absenger, F.; Bleckmann, A.; Eberhardt, W.; Eichhorn, M.; Früh, M.; Gautschi, O.; Hilbe, W.; Hoffmann, H.; Huber, R.M.; et al. Lungenkarzinom, Nicht-Kleinzellig (NSCLC). 2022. Available online: https://www.onkopedia.com/de/onkopedia/guidelines/lungenkarzinom-nicht-kleinzellig-nsclc/@@guideline/html/index.html#ID0EGLAC (accessed on 10 January 2022).
- Planchard, D.; Popat, S.; Kerr, K.; Novello, S.; Smit, E.F.; Faivre-Finn, C.; Mok, T.S.; Reck, M.; Van Schil, P.E.; Hellmann, M.D.; et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29 (Suppl. S4), iv192–iv237. [Google Scholar] [CrossRef] [PubMed]
- Cicek, T.; Ozturk, A.; Yılmaz, A.; Aktas, Z.; Demirag, F.; Akyurek, N. Adequacy of EBUS-TBNA specimen for mutation analysis of lung cancer. Clin. Respir. J. 2019, 13, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, K.; Inoue, Y.; Karayama, M.; Tsuchiya, K.; Mori, K.; Suzuki, Y.; Iwashita, Y.; Kahyo, T.; Kawase, A.; Tanahashi, M.; et al. Heterogeneity analysis of PD-L1 expression and copy number status in EBUS-TBNA biopsy specimens of non-small cell lung cancer: Comparative assessment of primary and metastatic sites. Lung Cancer 2019, 134, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Haentschel, M.; Boeckeler, M.; Ehab, A.; Wagner, R.; Spengler, W.; Steger, V.; Boesmueller, H.; Horger, M.; Lewis, R.A.; Fend, F.; et al. Cryobiopsy increases the EGFR detection rate in non-small cell lung cancer. Lung Cancer 2020, 141, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Gershman, E.; Ikan, A.; Pertzov, B.; Rosengarten, D.; Kramer, M.R. Mediastinal “deep freeze”—Transcarinal lymph node cryobiopsy. Thorac. Cancer 2022, 13, 1592–1596. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, J.-R.; Huang, Z.-S.; Fu, W.-L.; Wu, X.-L.; Wu, N.; Kuebler, W.M.; Herth, F.J.; Fan, Y. Transbronchial mediastinal cryobiopsy in the diagnosis of mediastinal lesions: A randomised trial. Eur. Respir. J. 2021, 58, 2100055. [Google Scholar] [CrossRef] [PubMed]
- Genova, C.; Tagliabue, E.; Mora, M.; Aloè, T.; Dono, M.; Salvi, S.; Zullo, L.; Barisione, E. Potential application of cryobiopsy for histo-molecular characterization of mediastinal lymph nodes in patients with thoracic malignancies: A case presentation series and implications for future developments. BMC Pulm. Med. 2022, 22, 5. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus Chemotherapy for PD-L1–Positive Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Five-Year Outcomes with Pembrolizumab Versus Chemotherapy for Metastatic Non–Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score ≥ 50%. J. Clin. Oncol. 2021, 39, 2339–2349. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Li, M.; Wang, Z.; An, D.; Li, B. Adverse events of immunotherapy in non-small cell lung cancer: A systematic review and network meta-analysis. Int. Immunopharmacol. 2021, 102, 108353. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hetzel, J.; Mauti, L.A.; Winkler, J.; Cardoso Almeida, S.; Jermann, P.; Pless, M.; Bubendorf, L.; Bode, P.K.; Häntschel, M. Only EBUS-Guided Mediastinal Lymph Node Cryobiopsy Enabled Immunotherapy in a Patient with Non-Small Cell Lung Cancer. J. Clin. Med. 2023, 12, 2355. https://doi.org/10.3390/jcm12062355
Hetzel J, Mauti LA, Winkler J, Cardoso Almeida S, Jermann P, Pless M, Bubendorf L, Bode PK, Häntschel M. Only EBUS-Guided Mediastinal Lymph Node Cryobiopsy Enabled Immunotherapy in a Patient with Non-Small Cell Lung Cancer. Journal of Clinical Medicine. 2023; 12(6):2355. https://doi.org/10.3390/jcm12062355
Chicago/Turabian StyleHetzel, Jürgen, Laetitia A. Mauti, Jonas Winkler, Sabine Cardoso Almeida, Philip Jermann, Miklos Pless, Lukas Bubendorf, Peter Karl Bode, and Maik Häntschel. 2023. "Only EBUS-Guided Mediastinal Lymph Node Cryobiopsy Enabled Immunotherapy in a Patient with Non-Small Cell Lung Cancer" Journal of Clinical Medicine 12, no. 6: 2355. https://doi.org/10.3390/jcm12062355
APA StyleHetzel, J., Mauti, L. A., Winkler, J., Cardoso Almeida, S., Jermann, P., Pless, M., Bubendorf, L., Bode, P. K., & Häntschel, M. (2023). Only EBUS-Guided Mediastinal Lymph Node Cryobiopsy Enabled Immunotherapy in a Patient with Non-Small Cell Lung Cancer. Journal of Clinical Medicine, 12(6), 2355. https://doi.org/10.3390/jcm12062355






