Splenic Arterial Pulsatility Index to Predict Hepatic Fibrosis in Hemodialysis Patients with Chronic Hepatitis C Virus Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
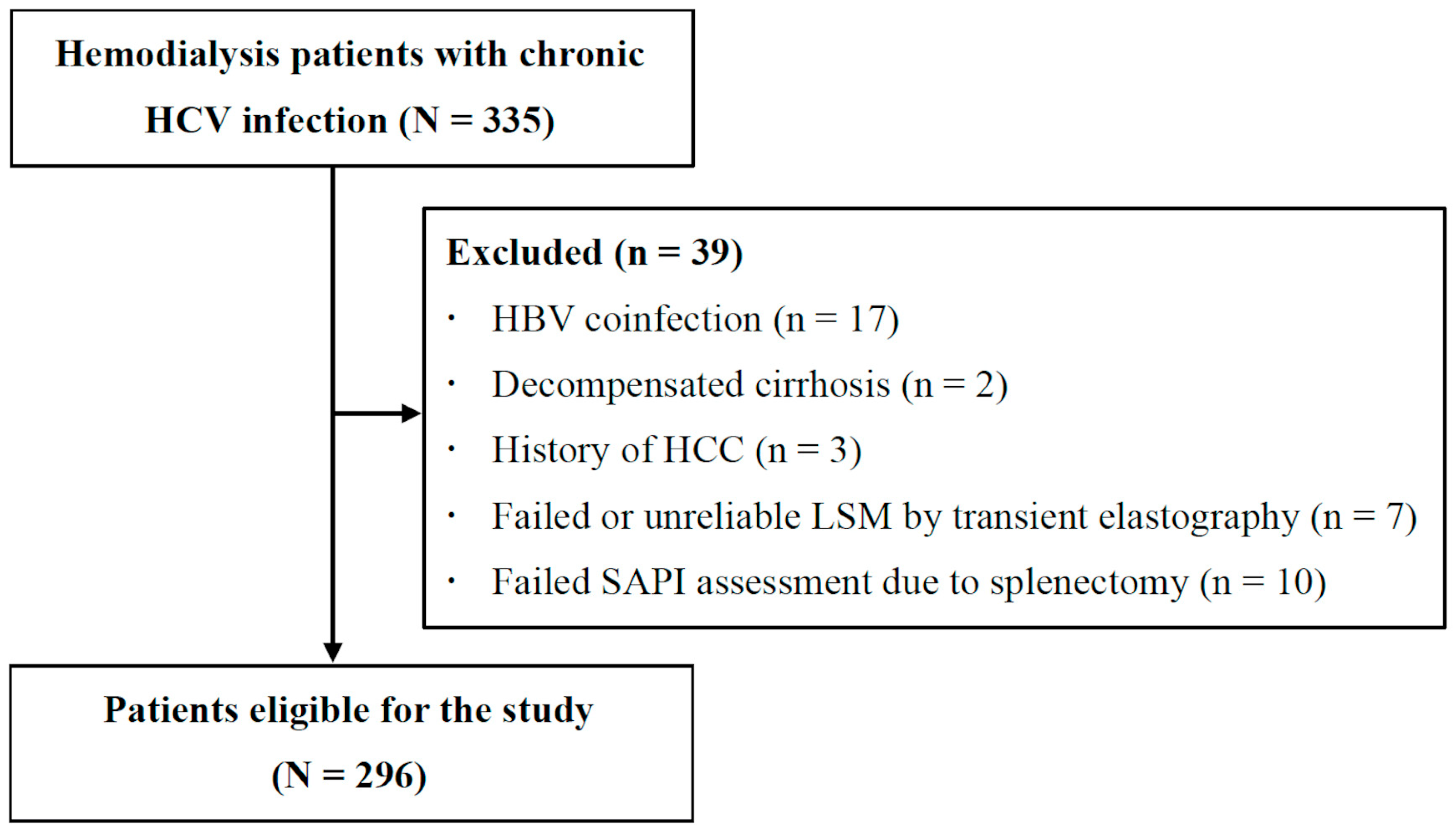
2.2. Study Design
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
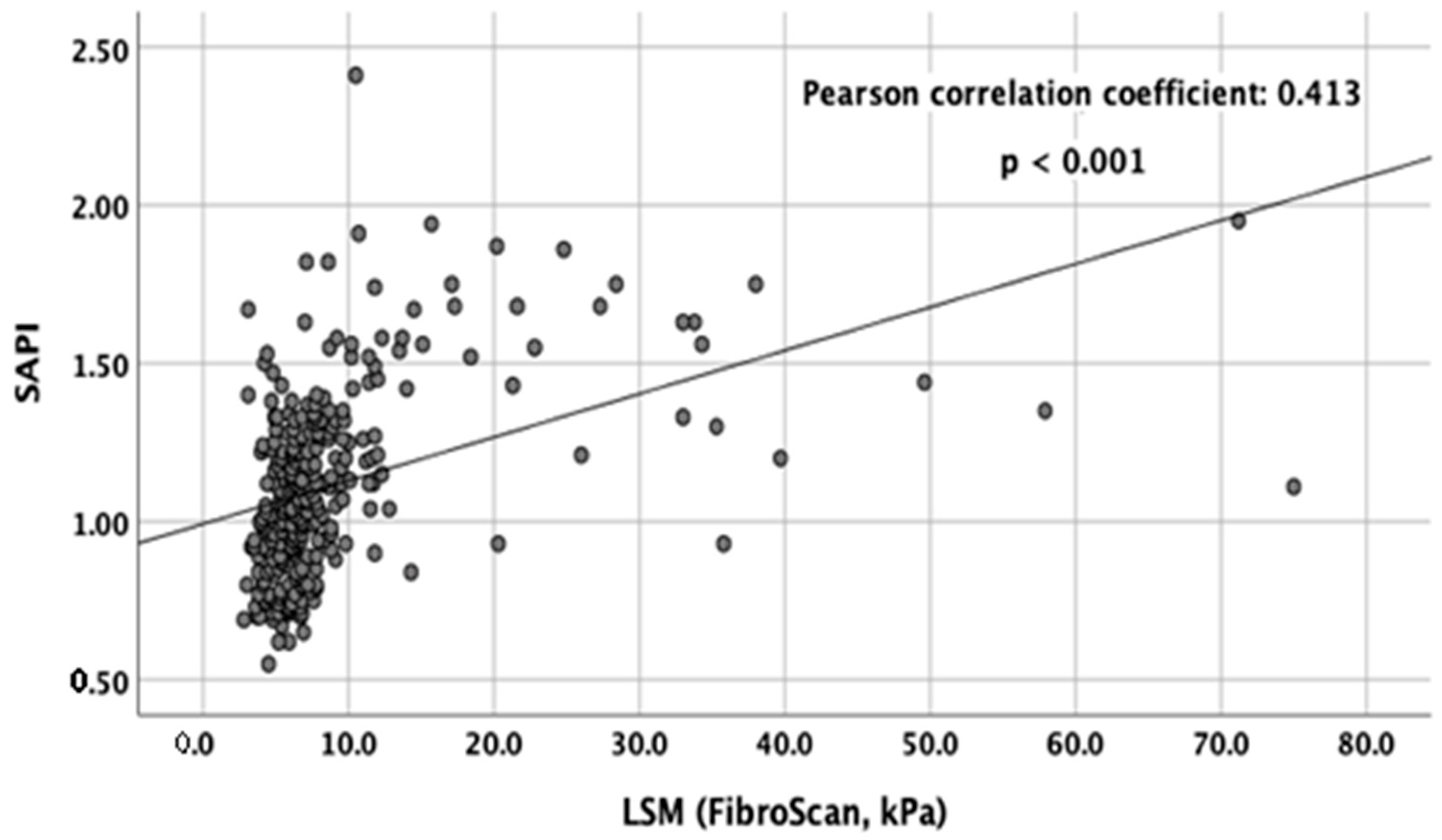
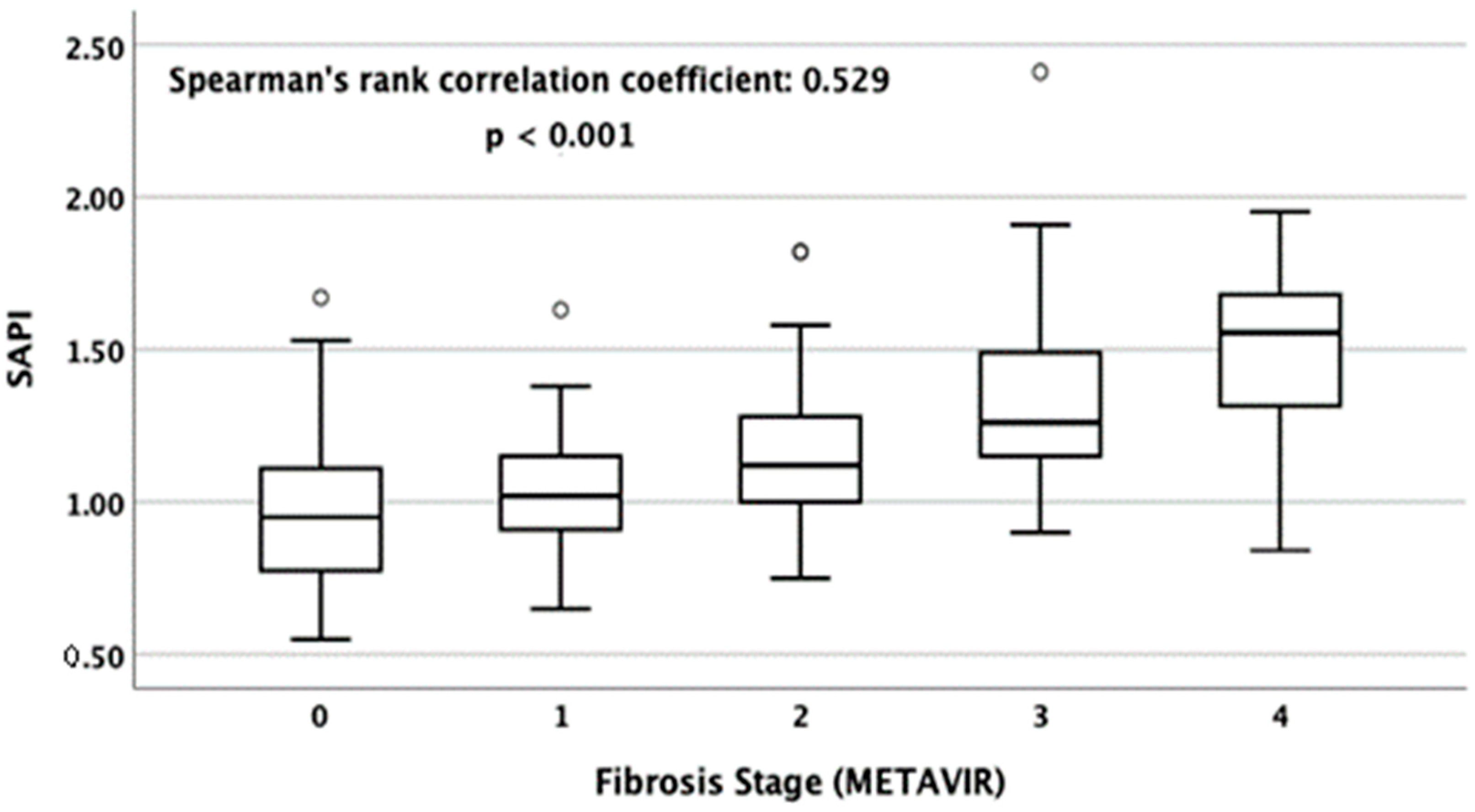
3.2. Correlation between SAPI and LSM and Stage of Hepatic Fibrosis
3.3. AUROCs of SAPI to Predict the Severity of Hepatic Fibrosis
3.4. Selective Cutoff Values for SAPI to Predict the Severity of Hepatic Fibrosis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jadoul, M.; Bieber, B.A.; Martin, P.; Akiba, T.; Nwankwo, C.; Arduino, J.M.; Goodkin, D.A.; Pisoni, R.L. Prevalence, incidence, and risk factors for hepatitis C virus infection in hemodialysis patients. Kidney Int. 2019, 95, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Polaris Observatory HCV Collaborators. Global change in hepatitis C virus prevalence and cascade of care between 2015 and 2020: A modelling study. Lancet Gastroenterol. Hepatol. 2022, 7, 396–415. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Kao, J.-H. Pan-genotypic direct-acting antivirals for patients with hepatitis C virus infection and chronic kidney disease stage 4 or 5. Hepatol. Int. 2022, 16, 1001–1019. [Google Scholar] [CrossRef]
- Okuda, K.; Hayashi, H.; Kobayashi, S.; Irie, Y. Mode of hepatitis C infection not associated with blood transfusion among chronic hemodialysis patients. J. Hepatol. 1995, 23, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Kanda, T.; Yokosuka, O.; Hayashi, H.; Yokozeki, K.; Ohtake, Y.; Irie, Y. GB virus-C infection among chronic haemodialysis patients: Clinical implications. J. Gastroenterol. Hepatol. 1997, 12, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Yokosuka, O. Natural history of chronic hepatitis C in patients on hemodialysis: Case control study with 4–23 years of follow-up. World J. Gastroenterol. 2004, 10, 2209–2212. [Google Scholar] [CrossRef]
- Fabrizi, F.; Takkouche, B.; Lunghi, G.; Dixit, V.; Messa, P.; Martin, P. The impact of hepatitis C virus infection on survival in dialysis patients: Meta-analysis of observational studies. J. Viral Hepat. 2007, 14, 697–703. [Google Scholar] [CrossRef]
- Fabrizi, F.; Dixit, V.; Messa, P. Impact of hepatitis C on survival in dialysis patients: A link with cardiovascular mortality? J. Viral Hepat. 2012, 19, 601–607. [Google Scholar] [CrossRef]
- Goodkin, D.A.; Bieber, B.; Jadoul, M.; Martin, P.; Kanda, E.; Pisoni, R.L. Mortality, hospitalization, and quality of life among patients with hepatitis C infection on hemodialysis. Clin. J. Am. Soc. Nephrol. 2017, 12, 287–297. [Google Scholar] [CrossRef]
- Söderholm, J.; Millbourn, C.; Büsch, K.; Kövamees, J.; Schvarcz, R.; Lindahl, K.; Bruchfeld, A. Higher risk of renal disease in chronic hepatitis C patients: Antiviral therapy survival benefit in patients on hemodialysis. J. Hepatol. 2018, 68, 904–911. [Google Scholar] [CrossRef]
- Goodkin, D.A.; Bieber, B.; Gillespie, B.; Robinson, B.M.; Jadoul, M. Hepatitis C infection is very rarely treated among hemodialysis patients. Am. J. Nephrol. 2013, 38, 405–412. [Google Scholar] [CrossRef]
- Liu, C.-H.; Huang, C.-F.; Liu, C.-J.; Dai, C.-Y.; Liang, C.-C.; Huang, J.-F.; Hung, P.-H.; Tsai, H.-B.; Tsai, M.-K.; Chen, S.-I.; et al. Pegylated interferon-α2a with or without low-dose ribavirin for treatment-naive patients with hepatitis C virus genotype 1 receiving hemodialysis: A randomized trial. Ann. Intern. Med. 2013, 159, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Liu, C.-J.; Huang, C.-F.; Lin, J.-W.; Dai, C.-Y.; Liang, C.-C.; Huang, J.-F.; Hung, P.-H.; Tsai, H.-B.; Tsai, M.-K.; et al. Peginterferon alfa-2a with or without low-dose ribavirin for treatment-naive patients with hepatitis C virus genotype 2 receiving haemodialysis: A randomised trial. Gut 2015, 64, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Pockros, P.J.; Reddy, K.R.; Mantry, P.S.; Cohen, E.; Bennett, M.; Sulkowski, M.S.; Bernstein, D.E.; Cohen, D.E.; Shulman, N.S.; Wang, D.; et al. Efficacy of direct-acting antiviral combination for patients with hepatitis C virus genotype 1 infection and severe renal impairment or end-stage renal disease. Gastroenterology 2016, 150, 1590–1598. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.; Nelson, D.R.; Bruchfeld, A.; Liapakis, A.; Silva, M.; Monsour, H., Jr.; Martin, P.; Pol, S.; Londoño, M.-C.; Hassanein, T.; et al. Grazoprevir plus elbasvir in treatment-naive and treatment-experienced patients with hepatitis C virus genotype 1 infection and stage 4–5 chronic kidney disease (the C-SURFER study): A combination phase 3 study. Lancet 2015, 386, 1537–1545. [Google Scholar] [CrossRef]
- Gane, E.; Lawitz, E.; Pugatch, D.; Papatheodoridis, G.; Bräu, N.; Brown, A.; Pol, S.; Leroy, V.; Persico, M.; Moreno, C.; et al. Glecaprevir and pibrentasvir in patients with HCV and severe renal impairment. N. Engl. J. Med. 2017, 377, 1448–1455. [Google Scholar] [CrossRef]
- Lawitz, E.; Flisiak, R.; Abunimeh, M.; Sise, M.E.; Park, J.Y.; Kaskas, M.; Bruchfeld, A.; Wörns, M.-A.; Aglitti, A.; Zamor, P.J.; et al. Efficacy and safety of glecaprevir/pibrentasvir in renally impaired patients with chronic HCV infection. Liver Int. 2020, 40, 1032–1041. [Google Scholar] [CrossRef]
- Liu, C.-H.; Yang, S.-S.; Peng, C.-Y.; Lin, W.-T.; Liu, C.-J.; Su, T.-H.; Tseng, T.-C.; Chen, P.-J.; Chen, D.-S.; Kao, J.-H. Glecaprevir/pibrentasvir for patients with chronic hepatitis C virus infection and severe renal impairment. J. Viral Hepat. 2020, 27, 568–575. [Google Scholar] [CrossRef]
- Borgia, S.M.; Dearden, J.; Yoshida, E.M.; Shafran, S.D.; Brown, A.; Ben-Ari, Z.; Cramp, M.E.; Cooper, C.; Foxton, M.; Rodriguez, C.F.; et al. Sofosbuvir/velpatasvir for 12 weeks in hepatitis C virus-infected patients with end-stage renal disease undergoing dialysis. J. Hepatol. 2019, 71, 660–665. [Google Scholar] [CrossRef]
- Liu, C.-H.; Chen, C.-Y.; Su, W.-W.; Tseng, K.-C.; Lo, C.-C.; Liu, C.-J.; Chen, J.-J.; Peng, C.-Y.; Shih, Y.-L.; Yang, S.-S.; et al. Sofosbuvir/velpatasvir with or without low-dose ribavirin for patients with chronic hepatitis C virus infection and severe renal impairment. Gut 2022, 71, 176–184. [Google Scholar] [CrossRef]
- Kim, N.J.; Vutien, P.; Cleveland, E.; Cravero, A.; Ioannou, G.N. Fibrosis stage-specific incidence of hepatocellular cancer after hepatitis C cure with direct-acting antivirals: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2022. ahead of print. [Google Scholar] [CrossRef]
- Bravo, A.A.; Sheth, S.G.; Chopra, S. Liver biopsy. N. Engl. J. Med. 2001, 344, 495–500. [Google Scholar] [CrossRef]
- Pawa, S.; Ehrinpreis, M.; Mutchnick, M.; Janisse, J.; Dhar, R.; Siddiqui, F.A. Percutaneous liver biopsy is safe in chronic hepatitis C patients with end-stage renal disease. Clin. Gastroenterol. Hepatol. 2007, 5, 1316–1320. [Google Scholar] [CrossRef]
- Cotler, S.J.; Diaz, G.; Gundlapalli, S.; Jakate, S.; Chawla, A.; Mital, D.; Jensik, S.; Jensen, D.M. Characteristics of hepatitis C in renal transplant candidates. J. Clin. Gastroenterol. 2002, 35, 191–195. [Google Scholar] [CrossRef]
- McGill, D.B.; Rakela, J.; Zinsmeister, A.R.; Ott, B.J. A 21-year experience with major hemorrhage after percutaneous liver biopsy. Gastroenterology 1990, 99, 1396–1400. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, B.; Leary, W.P.; Naran, A.D.; Maharaj, R.J.; Cooppan, R.M.; Pirie, D.; Pudifin, D.J. Sampling variability and its influence on the diagnostic yield of percutaneous needle biopsy of the liver. Lancet 1986, 1, 523–525. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Kao, J.-H. Noninvasive diagnosis of hepatic fibrosis in hemodialysis patients with hepatitis C virus infection. Diagnostics 2022, 12, 2282. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, M.; Sacerdoti, D.; Merkel, C.; Gerunda, G.; Maffei-Faccioli, A.; Angeli, P.; Jemmolo, R.M.; Bombonato, G.; Gatta, A. Splenic Doppler impedance indices: Influence of different portal hemodynamic conditions. Hepatology 1996, 23, 1035–1040. [Google Scholar] [CrossRef]
- Liu, C.-H.; Lin, J.-W.; Tsai, F.-C.; Yang, P.-M.; Lai, M.-Y.; Chen, J.-H.; Kao, J.-H.; Chen, D.-S. Noninvasive tests for the prediction of significant hepatic fibrosis in hepatitis C virus carriers with persistently normal alanine aminotransferases. Liver Int. 2006, 26, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Ullah, H.; Rehman, A.-U.; Aslam, H. Diagnostic accuracy of splenic arterial pulsatility index in predicting fibrosis associated with chronic hepatitis C. Pak. Armed Forces Med. J. 2018, 68, 1630–1635. [Google Scholar]
- Liu, C.-H.; Hsu, S.-J.; Lin, J.-W.; Hwang, J.-J.; Liu, C.-J.; Yang, P.-M.; Lai, M.-Y.; Chen, P.-J.; Chen, J.-H.; Kao, J.-H.; et al. Noninvasive diagnosis of hepatic fibrosis in patients with chronic hepatitis C by splenic Doppler impedance index. Clin. Gastroenterol. Hepatol. 2007, 5, 1199–1206.e1. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Iijima, H.; Nishikawa, H.; Kondo, R.; Yano, H.; Kage, M.; Aoki, T.; Nakano, C.; Yuri, Y.; Ishii, N.; et al. Liver fibrosis markers as assessed by ultrasound elastography and serum samples: A large comparative study in hepatitis virus B and C liver diseases. Hepatol. Res. 2019, 49, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Liu, C.-H.; Lee, M.-H.; Lin, J.-W.; Liu, C.-J.; Su, T.-H.; Tseng, T.-C.; Chen, P.-J.; Chen, D.-S.; Kao, J.-H. Evolution of eGFR in chronic HCV patients receiving sofosbuvir-based or sofosbuvir-free direct-acting antivirals. J. Hepatol. 2020, 72, 839–846. [Google Scholar] [CrossRef]
- Liu, C.-H.; Lin, J.-W.; Liu, C.-J.; Su, T.-H.; Wu, J.-H.; Tseng, T.-C.; Chen, P.-J.; Kao, J.-H. Long-term evolution of estimated glomerular filtration rate in patients with antiviral treatment for hepatitis C virus infection. Clin. Gastroenterol. Hepatol. 2023, 21, 424–434.e5. [Google Scholar] [CrossRef]
- Liu, C.-H.; Liang, C.-C.; Liu, C.-J.; Lin, C.-L.; Su, T.-H.; Yang, H.-C.; Chen, P.-J.; Chen, D.-S.; Kao, J.-H. Comparison of Abbott RealTime HCV Genotype II with Versant line probe assay 2.0 for hepatitis C virus genotyping. J. Clin. Microbiol. 2015, 53, 1754–1757. [Google Scholar] [CrossRef] [PubMed]
- Prati, D.; Taioli, E.; Zanella, A.; Della Torre, E.; Butelli, S.; Del Vecchio, E.; Vianello, L.; Zanuso, F.; Mozzi, F.; Milani, S.; et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann. Intern. Med. 2002, 137, 1–10. [Google Scholar] [CrossRef]
- Wai, C.-T.; Greenson, J.K.; Fontana, R.J.; Kalbfleisch, J.D.; Marrero, J.A.; Conjeevaram, H.S.; Lok, A.S.-F. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology 2003, 38, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Liu, C.-H.; Liang, C.-C.; Huang, K.-W.; Liu, C.-J.; Chen, S.-I.; Lin, J.-W.; Hung, P.-H.; Tsai, H.-B.; Lai, M.-Y.; Chen, P.-J.; et al. Transient elastography to assess hepatic fibrosis in hemodialysis chronic hepatitis C patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 1057–1065. [Google Scholar] [CrossRef]
- Hanley, J.A.; McNeil, B.J. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology 1983, 148, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Trevizoli, J.E.; de Paula Menezes, R.; Ribeiro Velasco, L.F.; Amorim, R.; de Carvalho, M.B.; Mendes, L.S.; Neto, C.J.; de Deus Macedo, J.R.; de Assis, F.; Neves, R. Hepatitis C is less aggressive in hemodialysis patients than in nonuremic patients. Clin. J. Am. Soc. Nephrol. 2008, 3, 1385–1390. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Simcox, T.; Twaddell, W.; Drachenberg, C.; Flasar, M. Dialysis reduces portal pressure in patients with chronic hepatitis C. Artif. Organs 2010, 34, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Ergun, T.; Lakadamyali, H. Doppler ultrasound evaluation of morphological and hemodynamical changes of hepatic and mesenteric structures in end-stage renal disease patients on regular hemodialysis. Int. Urol. Nephrol. 2010, 42, 205–210. [Google Scholar] [CrossRef]
- Schiavon, L.L.; Schiavon, J.L.N.; Filho, R.J.C.; Sampaio, J.P.; Lanzoni, V.P.; Silva, A.E.B.; Ferraz, M.L.G. Simple blood tests as noninvasive markers of liver fibrosis in hemodialysis patients with chronic hepatitis C virus infection. Hepatology 2007, 46, 307–314. [Google Scholar] [CrossRef]
- Liu, C.-H.; Liang, C.-C.; Liu, C.-J.; Hsu, S.-J.; Lin, J.-W.; Chen, S.-I.; Hung, P.-H.; Tsai, H.-B.; Lai, M.-Y.; Chen, P.-J.; et al. The ratio of aminotransferase to platelets is a useful index for predicting hepatic fibrosis in hemodialysis patients with chronic hepatitis C. Kidney Int. 2010, 78, 103–109. [Google Scholar] [CrossRef]
- Pestana, N.F.; Equi, C.M.A.; Gomes, C.P.; Cardoso, A.C.; Zumack, J.P.; Villela-Nogueira, C.A.; Perez, R.M. Aminotransferase-to-platelet ratio index and Fibrosis-4 index score predict hepatic fibrosis evaluated by transient hepatic elastography in hepatitis C virus-infected hemodialysis patients. Eur. J. Gastroenterol. Hepatol. 2021, 33, e260–e265. [Google Scholar] [CrossRef]
- Lee, J.-J.; Wei, Y.-J.; Lin, M.-Y.; Niu, S.-W.; Hsu, P.-Y.; Huang, J.-C.; Jang, T.-Y.; Yeh, M.-L.; Huang, C.-I.; Liang, P.-C.; et al. The applicability of non-invasive methods for assessing liver fibrosis in hemodialysis patients with chronic hepatitis C. PLoS ONE 2020, 15, e0242601. [Google Scholar] [CrossRef]
- Nakano, C.; Nishimura, T.; Tada, T.; Yoshida, M.; Takashima, T.; Aizawa, N.; Ikeda, N.; Nishikawa, H.; Enomoto, H.; Hatano, E.; et al. Severity of liver fibrosis using shear wave elastography is influenced by hepatic necroinflammation in chronic hepatitis patients, but not in cirrhotic patients. Hepatol. Res. 2021, 51, 436–444. [Google Scholar] [CrossRef]

| Characteristics a | Patient (N = 296) |
|---|---|
| Age, year | 55 (23–81) |
| Male, n (%) | 186 (62.8) |
| HCV RNA, log10, IU/mL | 5.80 (1.83–8.00) |
| HCV genotype, (%) | |
| 1a | 17 (5.7) |
| 1b | 189 (63.9) |
| 2 | 74 (25.0) |
| 6 | 7 (2.4) |
| Mixed | 8 (2.7) |
| Indeterminate | 1 (0.3) |
| LSM, kPa b | 6.7 (2.8–75.0) |
| Fibrosis stage (METAVIR), n (%) c | |
| F0 | 108 (36.5) |
| F1 | 61 (20.6) |
| F2 | 65 (22.0) |
| F3 | 30 (10.1) |
| F4 | 32 (10.8) |
| SAPI | 1.09 (0.55–2.41) |
| BMI, kg/m2 | 22.7 (14.9–37.2) |
| Hemoglobin, g/dL | 11.7 (7.9–17.2) |
| White blood cell count, 109 cells/L | 5.7 (1.2–11.6) |
| Platelet count, 109/L | 180 (39–432) |
| Albumin, g/dL | 4.3 (2.2–5.4) |
| Total bilirubin, mg/dL | 0.5 (0.1–1.6) |
| AST, ULN d | 1.2 (0.3–13.7) |
| ALT, ULN d | 1.4 (0.1–24.7) |
| APRI | 0.69 (0.14–8.45) |
| FIB-4 e | 1.67 (0.32–10.74) |
| eGFR, mg/dL/1.73 m2 e | 6 (2–14) |
| Stage of Hepatic Fibrosis | SAPI | APRI | FIB-4 | |||
|---|---|---|---|---|---|---|
| AUROC | 95% CI | AUROC | 95% CI | AUROC | 95% CI | |
| ≥F1 | 0.730 | 0.671–0.789 | 0.674 | 0.611–0.738 | 0.735 | 0.678–0.793 |
| ≥F2 | 0.782 | 0.730–0.834 | 0.680 | 0.619–0.741 | 0.768 | 0.714–0.822 |
| ≥F3 | 0.838 | 0.781–0.894 | 0.751 | 0.682–0.820 | 0.836 | 0.781–0.890 |
| F4 | 0.851 | 0.771–0.931 | 0.766 | 0.674–0.857 | 0.822 | 0.745–0.898 |
| SAPI a | Fibrosis Stage | All Patients (N = 296) | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | ||
|---|---|---|---|---|---|---|---|---|---|
| Tested Positive, n (%) | Actual Positive, n (%) | Actual Negative, n (%) | |||||||
| 1.04 | ≥F1 | 166 (56.1) | 188 (63.5) | 108 (36.5) | 70.2 | 68.5 | 79.5 | 56.9 | 69.6 |
| 1.06 | ≥F2 | 148 (50.0) | 127 (42.9) | 169 (57.1) | 78.0 | 65.1 | 62.7 | 79.8 | 67.2 |
| 1.19 | ≥F3 | 108 (36.5) | 62 (20.9) | 234 (79.1) | 77.4 | 74.4 | 44.4 | 92.6 | 75.0 |
| 1.30 | F4 | 68 (23.0) | 32 (10.8) | 264 (89.2) | 78.1 | 83.7 | 36.7 | 96.9 | 83.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.-H.; Fang, Y.-J.; Liu, C.-J.; Su, T.-H.; Huang, S.-C.; Tseng, T.-C.; Wu, J.-H.; Chen, P.-J.; Kao, J.-H. Splenic Arterial Pulsatility Index to Predict Hepatic Fibrosis in Hemodialysis Patients with Chronic Hepatitis C Virus Infection. J. Clin. Med. 2023, 12, 2020. https://doi.org/10.3390/jcm12052020
Liu C-H, Fang Y-J, Liu C-J, Su T-H, Huang S-C, Tseng T-C, Wu J-H, Chen P-J, Kao J-H. Splenic Arterial Pulsatility Index to Predict Hepatic Fibrosis in Hemodialysis Patients with Chronic Hepatitis C Virus Infection. Journal of Clinical Medicine. 2023; 12(5):2020. https://doi.org/10.3390/jcm12052020
Chicago/Turabian StyleLiu, Chen-Hua, Yu-Jen Fang, Chun-Jen Liu, Tung-Hung Su, Shang-Chin Huang, Tai-Chung Tseng, Jo-Hsuan Wu, Pei-Jer Chen, and Jia-Horng Kao. 2023. "Splenic Arterial Pulsatility Index to Predict Hepatic Fibrosis in Hemodialysis Patients with Chronic Hepatitis C Virus Infection" Journal of Clinical Medicine 12, no. 5: 2020. https://doi.org/10.3390/jcm12052020
APA StyleLiu, C.-H., Fang, Y.-J., Liu, C.-J., Su, T.-H., Huang, S.-C., Tseng, T.-C., Wu, J.-H., Chen, P.-J., & Kao, J.-H. (2023). Splenic Arterial Pulsatility Index to Predict Hepatic Fibrosis in Hemodialysis Patients with Chronic Hepatitis C Virus Infection. Journal of Clinical Medicine, 12(5), 2020. https://doi.org/10.3390/jcm12052020







