The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population and Clinical Characteristics
2.2. Statistical Analysis
2.3. Institutional Ethics
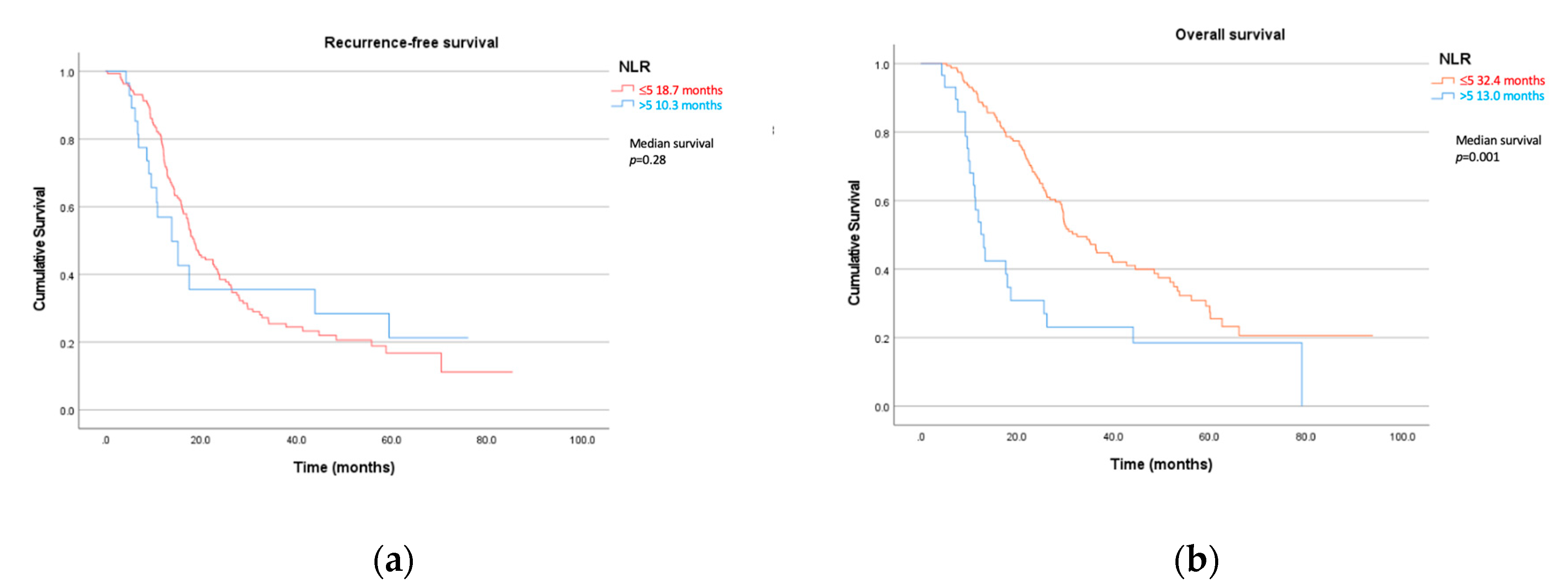
3. Results
3.1. Predictive Biomarkers
3.2. Prognostic Biomarkers
3.2.1. Recurrence-Free Survival
| Recurrence-Free Survival | ||||
|---|---|---|---|---|
| Univariate | Multivariate | |||
| p- Value | Hazard Ratio (95%CI)_ | p- Value | Hazard Ratio (95% CI) | |
| ECOG | 0.228 | 1.27 (0.86–1.89) | 0.825 | 1.1 (0.52–2.25) |
| CCI | 0.981 | 1.00 (0.91–1.10) | 0.566 | 0.95 (0.79–1.14) |
| Weight loss | 0.346 | 1.20 (0.82–1.75) | 0.935 | 1.03 (0.54–1.95) |
| Back pain | 0.581 | 1.22 (0.60–2.51) | 0.014 * | 4.45 (1.34–14.26) |
| Surgical stage | 0.001 * | 1.36 (1.15–1.62) | 0.003 * | 1.66 (1.18–2.34) |
| NLR > 5 | 0.283 | 1.34 (0.79–2.27) | 0.109 | 2.41 (0.82–7.04) |
| PLR > 150 | 0.671 | 1.08 (0.76–1.54) | 0.975 | 1.00 (0.47–2.07) |
| LMR > 3 | 0.275 | 1.21 (0.86–1.71) | 0.019 * | 2.32 (1.15–4.68) |
| Bilirubin > 20 µmol/L | 0.079 | 1.40 (0.96–2.04) | 0.138 | 1.66 (0.85–3.26) |
| mGPS | 0.238 | 0.82 (0.59–1.14)) | 0.459 | 0.84 (0.52–1.34) |
| CA 19–9 > 1000 kU/L | 0.114 | 1.47 (0.91–2.38) | 0.396 | 1.38 (0.65–2.92) |
3.2.2. Overall Survival
| Overall Survival | ||||
|---|---|---|---|---|
| Univariate | Multivariate | |||
| p- Value | Hazard Ratio (95%CI)_ | p- Value | Hazard Ratio (95% CI) | |
| ECOG | 0.144 | 1.30 (0.92–1.84) | 0.785 | 1.10 (0.55–2.2) |
| CCI | 0.383 | 1.04 (0.95–1.15) | 0.583 | 1.05 (0.88–1.27) |
| Weight loss | 0.147 | 1.33 (0.91–1.94) | 0.964 | 0.99 (0.52–1.88) |
| Back pain | 0.484 | 1.30 (0.63–2.66) | 0.010 * | 4.21 (1.4–12.63) |
| Surgical stage | 0.021 * | 1.23 (1.03–1.46) | 0.092 | 1.33 (0.95–1.87) |
| NLR > 5 | 0.001 * | 2.43 (1.54–3.85) | 0.019 * | 3.04 (1.20–7.71) |
| PLR > 150 | 0.163 | 1.30 (0.90–1.90) | 0.504 | 1.33 (0.58–3.08) |
| LMR > 3 | 0.966 | 1.01 (0.70–1.45) | 0.427 | 1.346 (0.65–2.80) |
| Bilirubin > 20 µmol/L | 0.037 * | 1.52 (1.03–2.26) | 0.051 | 2.07 (1.00–4.27) |
| mGPS | 0.234 | 0.81 (0.57–1.15) | 0.028 * | 0.58 (0.36–0.94) |
| CA 19–9 > 1000 kU/L | 0.270 | 1.30 (0.81–2.16) | 0.953 | 1.02 (0.46–2.30) |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Australian Institute of Welfare. Cancer in Australia. AIHW. 2019. Available online: https://www.aihw.gov.au/reports/cancer/cancer-in-australia-2019/contents/summary (accessed on 1 June 2019).
- Oettle, H.; Neuhaus, P.; Hochhaus, A.; Hartmann, J.T.; Gellert, K.; Ridwelski, K.; Niedergethmann, M.; Zülke, C.; Fahlke, J.; Arning, M.B.; et al. Adjuvant Chemotherapy With Gemcitabine and Long-term Outcomes Among Patients with Resected Pancreatic Cancer: The CONKO-001 Randomized Trial. JAMA 2013, 310, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Chua, W.; Charles, K.A.; Baracos, V.E.; Clarke, S.J. Neutrophil/lymphocyte ratio predicts chemotherapy outcomes in patients with advanced colorectal cancer. Br. J. Cancer 2011, 104, 1288–1295. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Guo, M.; Liu, Z.; Xiao, Z.; Jin, K.; Long, J.; Liu, L.; Liu, C.; Xu, J.; Ni, Q.; et al. Blood neutrophil-lymphocyte ratio predicts survival in patients with advanced pancreatic cancer treated with chemotherapy. Ann. Surg. Oncol. 2015, 22, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.J.; Hu, Z.G.; Shi, W.X.; Deng, T.; He, S.Q.; Yuan, S.G. Prognostic significance of neutrophil to lymphocyte ratio in pancreatic cancer: A meta-analysis. World J. Gastroenterol. 2015, 21, 2807–2815. [Google Scholar] [CrossRef]
- Szuster-Ciesielska, A.; Hryciuk-Umer, E.; Stepulak, A.; Kupisz, K.; Kandefer-Szerszeń, M. Reactive Oxygen Species Production by Blood Neutrophils of Patients With Laryngeal Carcinoma and Antioxidative Enzyme Activity in Their Blood. Acta Oncol. 2004, 43, 252–258. [Google Scholar] [CrossRef]
- Qiao, B.; Luo, W.; Liu, Y.; Wang, J.; Liu, C.; Liu, Z.; Chen, S.; Gu, J.; Qi, X.; Wu, T. The prognostic value of CXC chemokine receptor 2 (CXCR2) in cancers: A meta-analysis. Oncotarget 2018, 9, 15068–15076. [Google Scholar] [CrossRef]
- Jin, L.; Kim, H.S.; Shi, J. Neutrophil in the Pancreatic Tumor Microenvironment. Biomolecules 2021, 11, 1170. [Google Scholar] [CrossRef]
- Pointer, D.T.; Roife, D.; Powers, B.D.; Murimwa, G.; Elessawy, S.; Thompson, Z.J.; Schell, M.J.; Hodul, P.J.; Pimiento, J.M.; Fleming, J.B.; et al. Neutrophil to lymphocyte ratio, not platelet to lymphocyte or lymphocyte to monocyte ratio, is predictive of patient survival after resection of early-stage pancreatic ductal adenocarcinoma. BMC Cancer 2020, 20, 750. [Google Scholar] [CrossRef]
- Stevens, L.; Pathak, S.; Nunes, Q.M.; Pandanaboyana, S.; Macutkiewicz, C.; Smart, N.; Smith, A.M. Prognostic significance of pre-operative C-reactive protein and the neutrophil-lymphocyte ratio in resectable pancreatic cancer: A systematic review. HPB 2015, 17, 285–291. [Google Scholar] [CrossRef]
- Bermudez, E.A.; Rifai, N.; Buring, J.; Manson, J.E.; Ridker, P.M. Interrelationships Among Circulating Interleukin-6, C-Reactive Protein, and Traditional Cardiovascular Risk Factors in Women. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1668–1673. [Google Scholar] [CrossRef]
- Soeters, P.B.; Wolfe, R.R.; Shenkin, A. Hypoalbuminemia: Pathogenesis and Clinical Significance. J. Parenter. Enteral. Nutr. 2019, 43, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Proctor, M.J.; Morrison, D.S.; Talwar, D.; Balmer, S.M.; O’Reilly, D.S.J.; Foulis, A.K.; Horgan, P.G.; McMillan, D.C. An inflammation-based prognostic score (mGPS) predicts cancer survival independent of tumour site: A Glasgow Inflammation Outcome Study. Br. J. Cancer 2011, 104, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Townend, P.; de Reuver, P.R.; Chua, T.C.; Mittal, A.; Clark, S.J.; Pavlakis, N.; Gill, A.J.; Samra, J.S. Histopathological tumour viability after neoadjuvant chemotherapy influences survival in resected pancreatic cancer: Analysis of early outcome data. ANZ J. Surg. 2018, 88, E167–E172. [Google Scholar] [CrossRef] [PubMed]
- Chou, A.; Ahadi, M.; Arena, J.; Sioson, L.; Sheen, A.; Fuchs, T.L.; Pavlakis, N.; Clarke, S.; Kneebone, A.; Hruby, G.; et al. A Critical Assessment of Postneoadjuvant Therapy Pancreatic Cancer Regression Grading Schemes With a Proposal for a Novel Approach. Am. J. Surg. Pathol. 2021, 45, 394–404. [Google Scholar] [CrossRef]
- Insilla, A.C.; Vivaldi, C.; Giordano, M.; Vasile, E.; Cappelli, C.; Kauffmann, E.; Napoli, N.; Falcone, A.; Boggi, U.; Campani, D. Tumor Regression Grading Assessment in Locally Advanced Pancreatic Cancer After Neoadjuvant FOLFIRINOX: Interobserver Agreement and Prognostic Implications. Front. Oncol. 2020, 10, 64. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. Pancreatic Adenocarcinoma. 2019. Available online: https://www.nccn.org/professionals/physician_gls/pdf/pancreatic_blocks.pdf (accessed on 20 February 2020).
- Buccheri, G.; Ferrigno, D.; Tamburini, M. Karnofsky and ECOG performance status scoring in lung cancer: A prospective, longitudinal study of 536 patients from a single institution. Eur. J. Cancer 1996, 32, 1135–1141. [Google Scholar] [CrossRef]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Evans, A.; Costello, E. The role of inflammatory cells in fostering pancreatic cancer cell growth and invasion. Front. Physiol. 2012, 3, 270. [Google Scholar] [CrossRef]
- Humphris, J.L.; Chang, D.K.; Johns, A.L.; Scarlett, C.J.; Pajic, M.; Jones, M.D.; Colvin, E.K.; Nagrial, A.; Chin, V.T.; Chantrill, L.A.; et al. The prognostic and predictive value of serum CA19.9 in pancreatic cancer. Ann. Oncol. 2012, 23, 1713–1722. [Google Scholar] [CrossRef]
- Cong, L.; Liu, Q.; Zhang, R.; Cui, M.; Zhang, X.; Gao, X.; Guo, J.; Dai, M.; Zhang, T.; Liao, Q.; et al. Tumor size classification of the 8th edition of TNM staging system is superior to that of the 7th edition in predicting the survival outcome of pancreatic cancer patients after radical resection and adjuvant chemotherapy. Sci. Rep. 2018, 8, 10383. [Google Scholar] [CrossRef]
- Ventriglia, J.; Petrillo, A.; Alváro, M.H.; Laterza, M.M.; Savastano, B.; Gambardella, V.; Tirino, G.; Pompella, L.; Diana, A.; Iovino, F.; et al. Neutrophil to Lymphocyte Ratio as a Predictor of Poor Prognosis in Metastatic Pancreatic Cancer Patients Treated with Nab-Paclitaxel plus Gemcitabine: A Propensity Score Analysis. Gastroenterol. Res. Pract. 2018, 2018, 2373868. [Google Scholar] [CrossRef] [PubMed]

| Stage at Diagnosis (Radiological Stage) | Number of Patients |
|---|---|
| 1 | 145 |
| 2 | 47 |
| 3 | 4 |
| Resectability | |
| Upfront resectable | 133 |
| Borderline | 26 |
| Locally advanced | 37 |
| Upfront treatment | |
| Upfront resection | 86 |
| NAC | 110 |
| NAC regimen | |
| Gemcitabine plus nab-paclitaxel | 41 |
| FOLFIRINOX | 60 |
| Other | 9 |
| Charleson comorbidity | |
| <2 | 36 |
| 2–5 | 117 |
| >5 | 41 |
| Unknown | 2 |
| Sex | |
| M | 104 |
| F | 92 |
| Inflammatory markers | Median (range) |
| NLR | 2.9 (0.2–17.6) |
| PLR | 181 (12.6–662) |
| LMR | 2.4 (0.4–18) |
| p-Value | Correlation Coefficient (Spearmen’s) | |
|---|---|---|
| ECOG | 0.102 | 0.158 |
| CCI | 0.165 | 0.134 |
| Weight Loss | 0.997 | 0 |
| Back pain | 0.276 | −0.106 |
| NLR > 5 | 0.875 | −0.015 |
| PLR > 150 | 0.03 * | 0.21 |
| LMR > 3 | 0.075 | −0.172 |
| mGPS | 0.436 | −0.93 |
| CA 19.9 > 1000 kU/L | 0.818 | 0.023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maloney, S.; Pavlakis, N.; Itchins, M.; Arena, J.; Mittal, A.; Hudson, A.; Colvin, E.; Sahni, S.; Diakos, C.; Chan, D.; et al. The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer. J. Clin. Med. 2023, 12, 1989. https://doi.org/10.3390/jcm12051989
Maloney S, Pavlakis N, Itchins M, Arena J, Mittal A, Hudson A, Colvin E, Sahni S, Diakos C, Chan D, et al. The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer. Journal of Clinical Medicine. 2023; 12(5):1989. https://doi.org/10.3390/jcm12051989
Chicago/Turabian StyleMaloney, Sarah, Nick Pavlakis, Malinda Itchins, Jennifer Arena, Anubhav Mittal, Amanda Hudson, Emily Colvin, Sumit Sahni, Connie Diakos, David Chan, and et al. 2023. "The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer" Journal of Clinical Medicine 12, no. 5: 1989. https://doi.org/10.3390/jcm12051989
APA StyleMaloney, S., Pavlakis, N., Itchins, M., Arena, J., Mittal, A., Hudson, A., Colvin, E., Sahni, S., Diakos, C., Chan, D., Gill, A. J., Samra, J., & Clarke, S. J. (2023). The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer. Journal of Clinical Medicine, 12(5), 1989. https://doi.org/10.3390/jcm12051989







