Can Measuring the ‘Dual Anchors of Aorta’ Enhance the Success Rate of TAVR?—A Single-Center Experience
Abstract
1. Introduction
2. Background
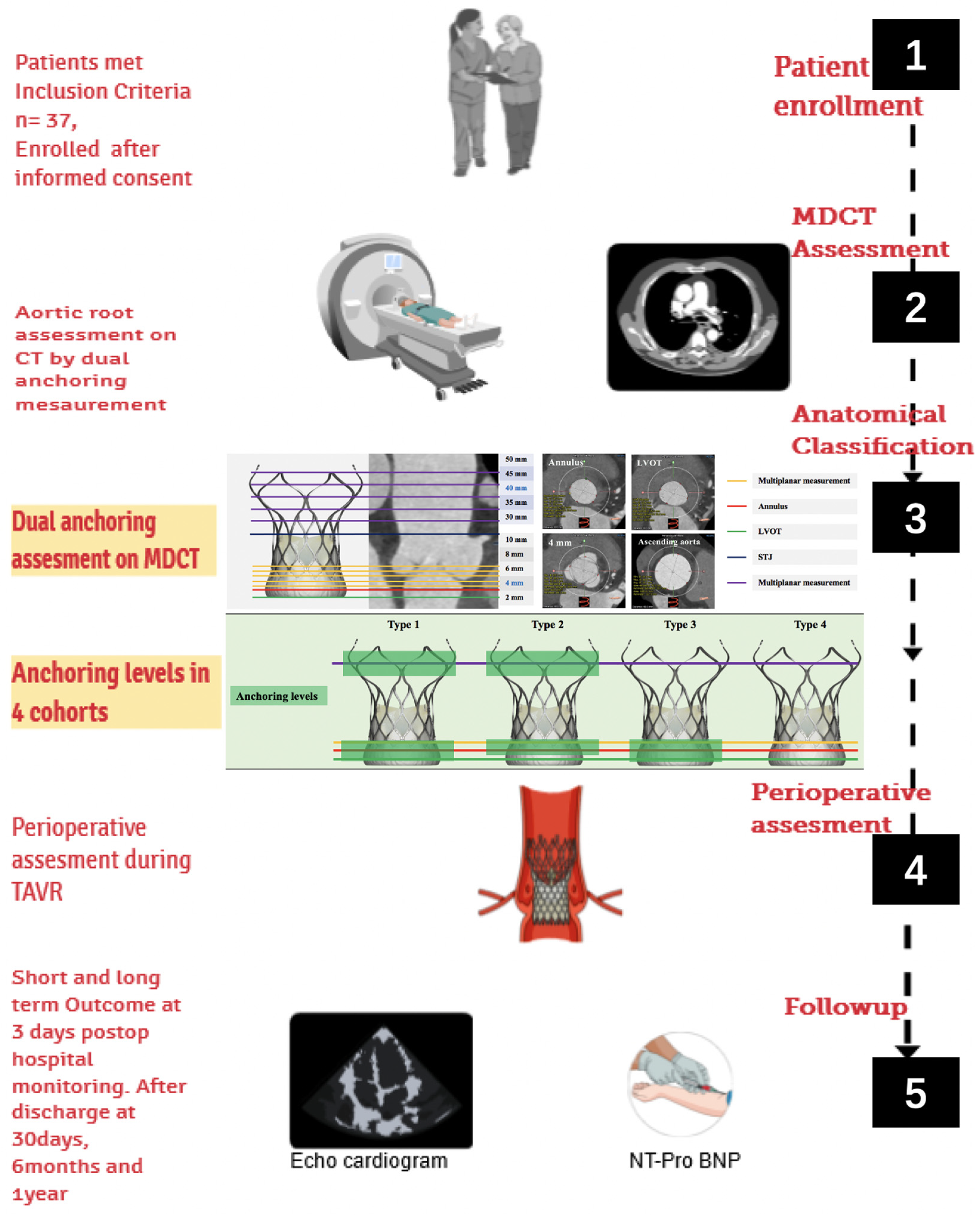
3. Materials and Methods
3.1. Study Population
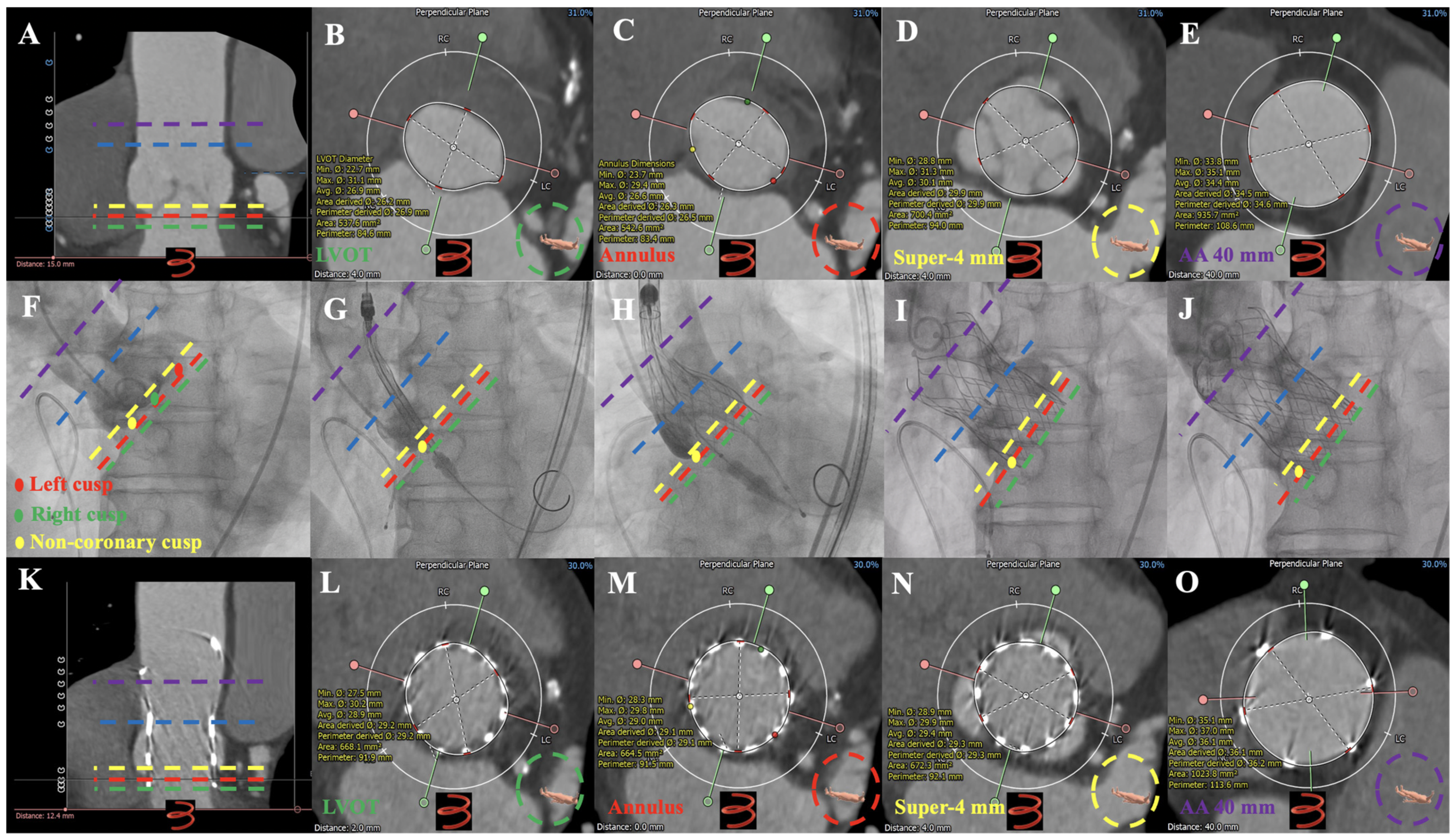
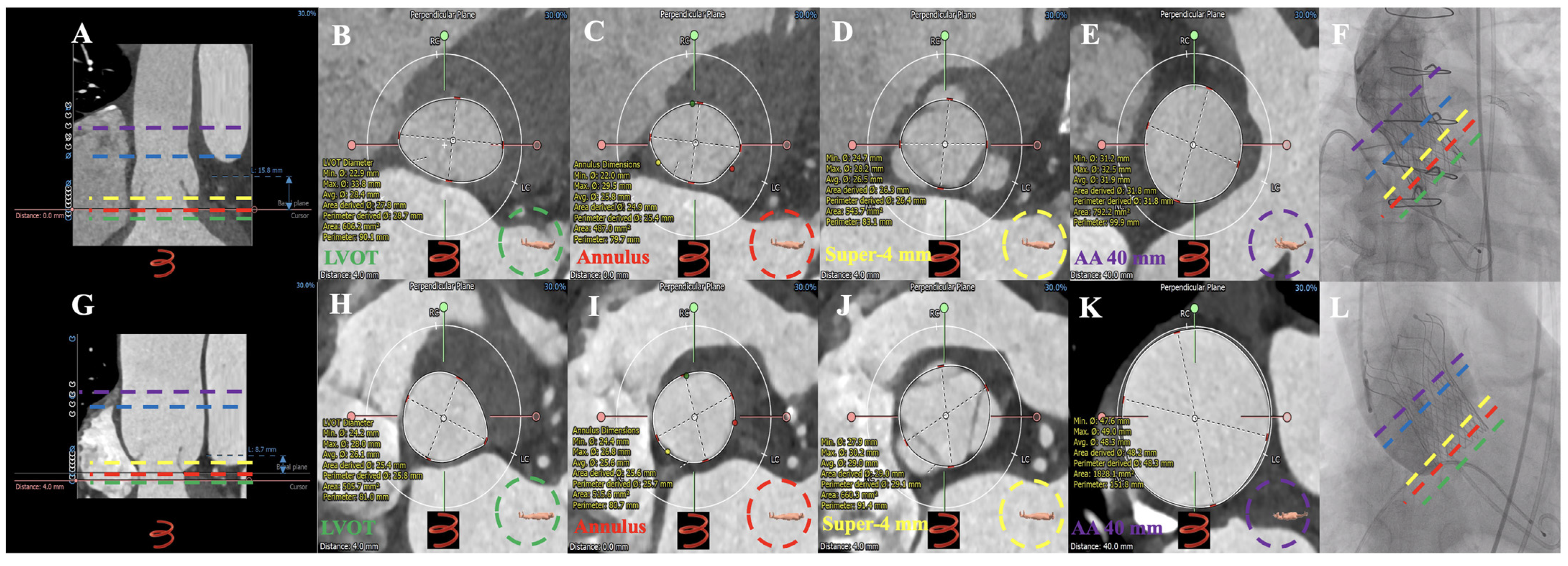
3.2. MDCT Evaluation and Anatomical Classification
3.3. Operation
3.4. Perioperative Outcome and Follow-Up
3.5. Statistical Analysis
4. Results
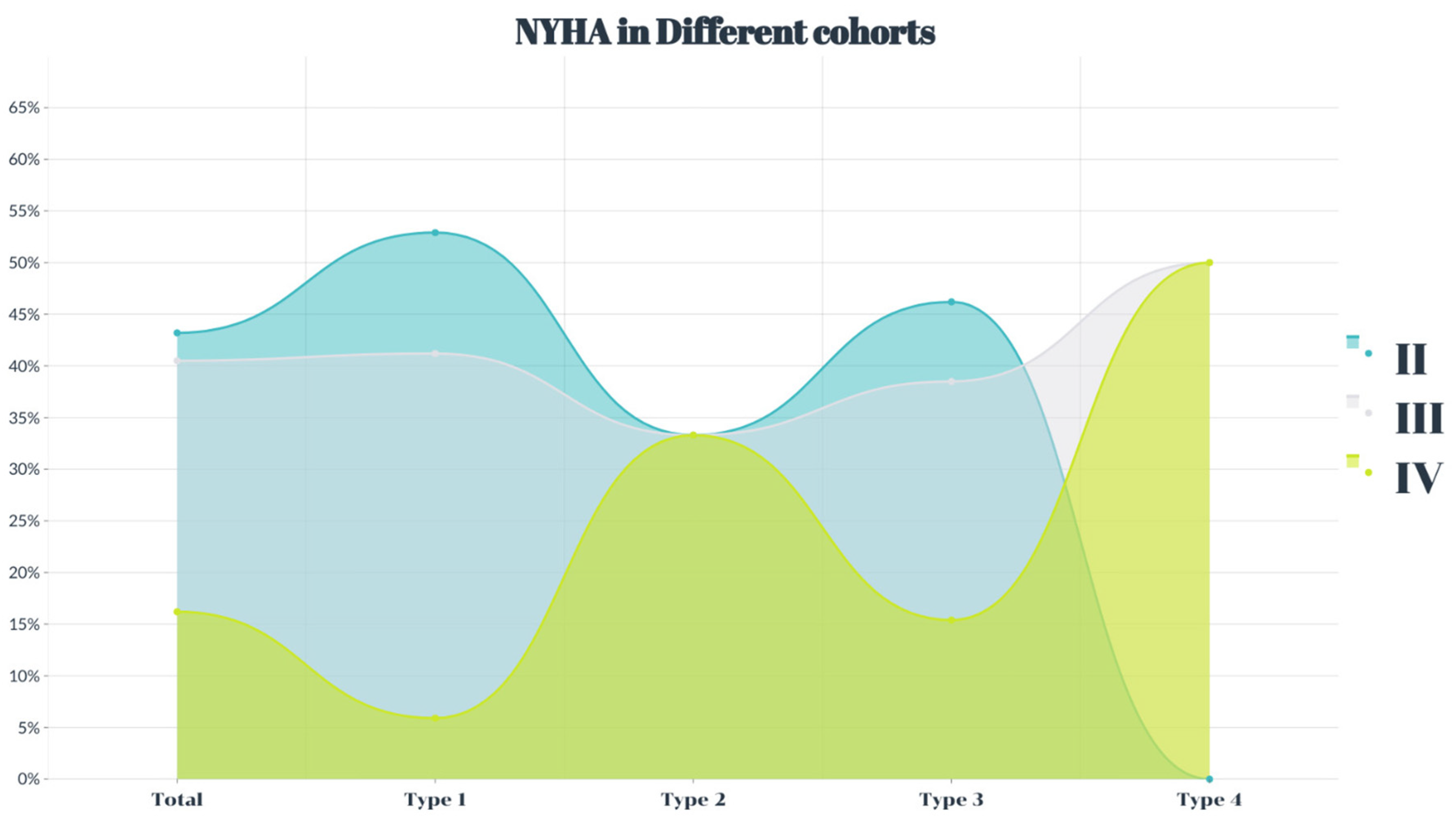
4.1. Basic Characteristics of Patients with Chronic Severe AR Undergoing TAVR Treatment
4.2. Preoperative MDCT Anatomical Characteristics
4.3. Operation Outcome and Device Failure Analysis
4.4. Follow-Up Outcomes
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, B.; Xu, H.; Zhang, H.; Liu, Q.; Ye, Y.; Hao, J.; Zhao, Q.; Qi, X.; Liu, S.; Zhang, E.; et al. Prognostic Value of N-Terminal Pro-B-Type Natriuretic Peptide in Elderly Patients With Valvular Heart Disease. J. Am. Coll. Cardiol. 2020, 75, 1659–1672. [Google Scholar] [CrossRef]
- Xu, H.; Liu, Q.; Cao, K.; Ye, Y.; Zhang, B.; Li, Z.; Hao, J.; Qi, X.; Zhao, Q.; Liu, S.; et al. Distribution, Characteristics, and Management of Older Patients With Valvular Heart Disease in China: China-DVD Study. JACC Asia 2022, 2, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, Z.; Chen, Z.; Wang, X.; Zhang, L.; Li, S.; Zheng, C.; Kang, Y.; Jiang, L.; Zhu, Z.; et al. Current status and etiology of valvular heart disease in China: A population-based survey. BMC Cardiovasc. Disord. 2021, 21, 339. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Zhou, D.; Cheng, L.; Ge, J. Aortic regurgitation is more prevalent than aortic stenosis in Chinese elderly population: Implications for transcatheter aortic valve replacement. Int. J. Cardiol. 2015, 201, 547–548. [Google Scholar] [CrossRef]
- Iung, B.; Baron, G.; Butchart, E.G.; Delahaye, F.; Gohlke-Bärwolf, C.; Levang, O.W.; Tornos, P.; Vanoverschelde, J.-L.; Vermeer, F.; Boersma, E.; et al. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. Eur. Heart J. 2003, 24, 1231–1243. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.T.; Enriquez-Sarano, M.; Michelena, H.I.; Nkomo, V.T.; Scott, C.G.; Bailey, K.R.; Oguz, D.; Ullah, M.W.; Pellikka, P.A. Predictors of Progression in Patients With Stage B Aortic Regurgitation. J. Am. Coll. Cardiol. 2019, 74, 2480–2492. [Google Scholar] [CrossRef] [PubMed]
- Carroll, J.D.; Mack, M.J.; Vemulapalli, S.; Herrmann, H.C.; Gleason, T.G.; Hanzel, G.; Deeb, G.M.; Thourani, V.H.; Cohen, D.J.; Desai, N.; et al. STS-ACC TVT Registry of Transcatheter Aortic Valve Replacement. J. Am. Coll. Cardiol. 2020, 76, 2492–2516. [Google Scholar] [CrossRef]
- Arora, S.; Lahewala, S.; Zuzek, Z.; Thakkar, S.; Jani, C.; Jaswaney, R.; Singh, A.; Bhyan, P.; Arora, N.; Main, A.; et al. Transcatheter aortic valve replacement in aortic regurgitation: The U.S. experience. Catheter. Cardiovasc. Interv. 2021, 98, E153–E162. [Google Scholar] [CrossRef]
- Vahanian, A.; Beyersdorf, F.; Praz, F.; Milojevic, M.; Baldus, S.; Bauersachs, J.; Capodanno, D.; Conradi, L.; De Bonis, M.; De Paulis, R.; et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2022, 43, 561–632. [Google Scholar] [CrossRef]
- Writing Committee, M.; Otto, C.M.; Nishimura, R.A.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P., III; Gentile, F.; Jneid, H.; Krieger, E.V.; Mack, M.; et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2021, 77, e25–e197. [Google Scholar]
- Takagi, H.; Hari, Y.; Kawai, N.; Ando, T.; Group, A. Meta-Analysis and Meta-Regression of Transcatheter Aortic Valve Implantation for Pure Native Aortic Regurgitation. Heart Lung Circ. 2020, 29, 729–741. [Google Scholar] [CrossRef]
- Yoon, S.H.; Schmidt, T.; Bleiziffer, S.; Schofer, N.; Fiorina, C.; Munoz-Garcia, A.J.; Yzeiraj, E.; Amat-Santos, I.J.; Tchetche, D.; Jung, C.; et al. Transcatheter Aortic Valve Replacement in Pure Native Aortic Valve Regurgitation. J. Am. Coll. Cardiol. 2017, 70, 2752–2763. [Google Scholar] [CrossRef]
- Roy, D.A.; Schaefer, U.; Guetta, V.; Hildick-Smith, D.; Möllmann, H.; Dumonteil, N.; Modine, T.; Bosmans, J.; Petronio, A.S.; Moat, N.; et al. Transcatheter aortic valve implantation for pure severe native aortic valve regurgitation. J. Am. Coll. Cardiol. 2013, 61, 1577–1584. [Google Scholar] [CrossRef]
- Testa, L.; Latib, A.; Rossi, M.L.; De Marco, F.; De Carlo, M.; Fiorina, C.; Oreglia, J.; Petronio, A.S.; Ettori, F.; De Servi, S.; et al. CoreValve implantation for severe aortic regurgitation: A multicentre registry. EuroIntervention 2014, 10, 739–745. [Google Scholar] [CrossRef]
- Liu, L.; Chen, S.; Shi, J.; Qin, C.; Guo, Y. Transcatheter Aortic Valve Replacement in Aortic Regurgitation. Ann. Thorac. Surg. 2020, 110, 1959–1965. [Google Scholar] [CrossRef]
- Chen, S.; Zheng, F.; Li, M.; Hou, S.; Zhang, W.; Zhang, L.; Zhang, X.; Pan, W.; Zhou, D.; Ge, J.; et al. A study on correlation between preprocedural CT indexes and procedural success rate of transfemoral transcatheter aortic valve replacement with different self-expanding valves (VitaFlow or VenusA-Valve) in patients with pure native aortic regurgitation. Ann. Transl. Med. 2022, 10, 643. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhai, M.; Mao, Y.; Xu, C.; Ma, Y.; Li, L.; Jin, P.; Yang, J. Transcatheter aortic valve replacement in patients with quadricuspid aortic valve in a single center. Front. Cardiovasc. Med. 2022, 9, 1011466. [Google Scholar] [CrossRef]
- Chen, Y.; Lu, Z.; Yao, J.; Wang, M.; Niu, G.; Zhang, H.; Liu, Q.; Zhao, J.; Zhao, Z.; Perrin, N.; et al. A novel anatomic classification to guide transcatheter aortic valve replacement for pure aortic regurgitation. Echocardiography 2022, 39, 1571–1580. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.T.; Michelena, H.I.; Maleszewski, J.J.; Schaff, H.V.; Pellikka, P.A. Contemporary Etiologies, Mechanisms, and Surgical Approaches in Pure Native Aortic Regurgitation. Mayo Clin. Proc. 2019, 94, 1158–1170. [Google Scholar] [CrossRef]
- Le Polain De Waroux, J.B.; Pouleur, A.C.; Goffinet, C.; Vancraeynest, D.; Van Dyck, M.; Robert, A.; Gerber, B.L.; Pasquet, A.; El Khoury, G.; Vanoverschelde, J.-L.J. Functional anatomy of aortic regurgitation: Accuracy, prediction of surgical repairability, and outcome implications of transesophageal echocardiography. Circulation 2007, 116, I264–I269. [Google Scholar] [CrossRef]
- Koo, H.J.; Kang, J.-W.; A Kim, J.; Kim, J.B.; Jung, S.-H.; Choo, S.J.; Chung, C.H.; Lee, J.W.; Lim, T.-H.; Yang, D.H. Functional classification of aortic regurgitation using cardiac computed tomography: Comparison with surgical inspection. Int. J. Cardiovasc. Imaging 2018, 34, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Lu, Z.N.; Modine, T.; Jilaihawi, H.; Piazza, N.; Tang, Y.D.; Xu, J.-Z.; Peng, X.-P.; Wang, H.-P.; Li, C.-B.; et al. Evaluation of the safety and efficacy of a novel Anatomical classification and dUal anchoRing theory to Optimize the tavR strategy for pure severe Aortic regurgitation (AURORA): A prospective cohort study. BMC Cardiovasc. Disord. 2022, 22, 445. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Dong, H.; Li, H.W.; Zou, Y.B.; Jiang, X.J. Characteristics of four-limb blood pressure and brachial-ankle pulse wave velocity in Chinese patients with Takayasu arteritis. Blood Press. 2022, 31, 146–154. [Google Scholar] [CrossRef]
- Zoghbi, W.A.; Adams, D.; Bonow, R.O.; Enriquez-Sarano, M.; Foster, E.; Grayburn, P.A.; Hahn, R.T.; Han, Y.; Hung, J.; Lang, R.M.; et al. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. J. Am. Soc. Echocardiogr. 2017, 30, 303–371. [Google Scholar] [CrossRef]
- Yang, C.; Song, G.; Niu, G.; Wu, Y. Coronary protection for the small left coronary sinus during transcatheter aortic valve replacement: A case report. Eur. Heart J. Case Rep. 2022, 6, ytac011. [Google Scholar] [CrossRef]
- Pawade, T.; Clavel, M.-A.; Tribouilloy, C.; Dreyfus, J.; Mathieu, T.; Tastet, L.; Renard, C.; Gun, M.; Jenkins, W.S.A.; Macron, L.; et al. Computed Tomography Aortic Valve Calcium Scoring in Patients With Aortic Stenosis. Circ. Cardiovasc. Imaging 2018, 11, e007146. [Google Scholar] [CrossRef]
- Song, G.; Jilaihawi, H.; Wang, M.; Chen, M.; Wang, J.; Wang, W.; Feng, Y.; Liu, X.; Zhang, R.; Kong, X.; et al. Severe Symptomatic Bicuspid and Tricuspid Aortic Stenosis in China: Characteristics and Outcomes of Transcatheter Aortic Valve Replacement with the Venus-A Valve. Struct. Heart 2018, 2, 60–68. [Google Scholar] [CrossRef]
- Zhou, D.; Pan, W.; Wang, J.; Wu, Y.; Chen, M.; Modine, T.; Mylotte, D.; Piazza, N.; Ge, J. VitaFlow transcatheter valve system in the treatment of severe aortic stenosis: One-year results of a multicenter study. Catheter. Cardiovasc. Interv. 2020, 95, 332–338. [Google Scholar] [CrossRef]
- Varc-3 Writing, C.; Généreux, P.; Piazza, N.; Alu, M.C.; Nazif, T.; Hahn, R.T.; Pibarot, P.; Bax, J.J.; A Leipsic, J.; Blanke, P.; et al. Valve Academic Research Consortium 3, updated endpoint definitions for aortic valve clinical research. Eur. Heart J. 2021, 42, 1825–1857. [Google Scholar] [CrossRef]
- Webb, J.G.; Htun, N. Transcatheter Options for the Treatment of Noncalcified Aortic Regurgitation. JACC Cardiovasc. Interv. 2015, 8, 1850–1853. [Google Scholar] [CrossRef]
- Bonow, R.O.; O’Gara, P.T. Left Ventricular Volume and Outcomes in Patients With Chronic Aortic Regurgitation. JAMA Cardiol. 2022, 7, 885–886. [Google Scholar] [CrossRef] [PubMed]
- Paraggio, L.; Burzotta, F.; Graziani, F.; Aurigemma, C.; Romagnoli, E.; Pedicino, D.; Locorotondo, G.; Mencarelli, E.; Lillo, R.; Bruno, P.; et al. Transcatheter aortic valve implantation in pure aortic regurgitation: Hemodynamic and echocardiographic findings in bioprosthesis vs. native valve. Catheter. Cardiovasc. Interv. 2022, 99, 1599–1608. [Google Scholar] [CrossRef]
- Graziani, F.; Mencarelli, E.; Burzotta, F.; Paraggio, L.; Aurigemma, C.; Romagnoli, E.; Pedicino, D.; Locorotondo, G.; Lombardo, A.; Leone, A.M.; et al. Early Hemodynamic and Structural Impact of Transcatheter Aortic Valve Replacement in Pure Aortic Regurgitation. JACC Cardiovasc. Interv. 2020, 13, 2582–2584. [Google Scholar] [CrossRef] [PubMed]




| Standards | Contents |
|---|---|
| Inclusion criteria | |
| Age ≥ 55 years | |
| Symptomatic severe AR | |
| American Association of Chest Physicians score > 4% | |
| New York Heart Association grades II–IV | |
| MDCT evaluation anatomy appropriate for TAVR treatment | |
| Exclusion criteria | |
| Acute myocardial infarction within 30 days | |
| Left ventricular ejection fraction ≤ 20% | |
| Life expectancy < 1 year | |
| Mild to moderate AR | |
| With moderate to severe aortic stenosis | |
| Previous aortic valve replacement | |
| Inappropriate MDCT evaluation anatomy | |
| Other THV treatments |
| Characteristics (n, %) | Total N = 37 | Type 1 N = 17 | Type 2 N = 3 | Type 3 N = 13 | Type 4 N = 4 | p |
|---|---|---|---|---|---|---|
| Age | 73.1 ± 8.7 | 71.4 ± 7.2 | 67.7 ± 11.0 | 73.7 ± 9.9 | 82.3 ± 4.3 abc | 0.096 |
| Male | 23 (62.2) | 10 (58.8) | 3 (100.0) | 7 (53.8) | 3 (75.0) | 0.463 |
| STS score (%) | 8.6 ± 2.1 | 7.9 ± 1.9 | 7.7 ± 0.8 | 9.2 ± 2.2 | 10.2 ± 2.5 | 0.123 |
| Coronary artery disease | 14 (37.8) | 6 (35.3) | 3 (100.0) | 4 (30.8) | 1 (25.0) | 0.137 |
| Previous PCI | 3 (8.1) | 2 (11.8) | 1 (33.3) | 0 | 0 | 0.224 |
| Previous CABG | 2 (5.4) | 1 (5.9) | 1 (33.3) | 0 | 0 | 0.135 |
| Atrial fibrillation | 13 (35.1) | 6 (35.5) | 1 (33.3) | 4 (30.8) | 2 (50.0) | 0.919 |
| Previous PPI | 2 (5.4) | 1 (5.9) | 0 | 1 (7.7) | 0 | 0.910 |
| hypertension | 23 (62.2) | 9 (52.9) | 2 (66.7) | 11 (84.6) | 1 (25.0) | 0.123 |
| Hyperlipidemia | 21 (56.8) | 9 (52.9) | 2 (66.7) | 8 (61.5) | 2 (50.0) | 0.937 |
| Diabetes mellitus | 7 (18.9) | 7 (11.8) | 2 (66.7) | 2 (15.4) | 1 (25.0) | 0.156 |
| COPD | 3 (8.1) | 2 (11.8) | 0 | 0 | 1 (25.0) | 0.355 |
| Cerebral vascular disease | 2 (5.4) | 1 (5.9) | 1 (33.3) | 0 | 0 | 0.135 |
| Hepatic insufficiency | 2 (5.4) | 1 (5.9) | 1 (33.3) | 0 | 0 | 0.135 |
| Peripheral vascular disease | 2 (5.4) | 1 (5.9) | 0 | 1 (7.7) | 0 | 0.910 |
| Characteristics (n, %) | Total N = 37 | Type 1 N = 17 | Type 2 N = 3 | Type 3 N = 13 | Type 4 N = 4 | p |
|---|---|---|---|---|---|---|
| Valvular classification | ||||||
| Tricuspid valve | 33 (89.2) | 16 (94.1) | 3 (100.0) | 11 (84.6) | 3 (75.0) | 0.574 |
| Quadricuspid valve | 2 (5.4) | 1 (5.9) | 0 | 1 (7.7) | 0 | |
| Bicuspid valve | 2 (5.4) | 0 | 0 | 1 (7.7) | 1 (25.1) | |
| Measurement (diameter calculated by the perimeter, mm) | ||||||
| Annular circumference | 81.7 ± 6.8 | 80.6 ± 5.2 | 85.3 ± 4.7 | 80.8 ± 7.1 | 86.9 ± 11.3 | 0.280 |
| Annulus | 26.0 ± 2.2 | 25.6 ± 1.7 | 17.1 ± 1.5 | 15.7 ± 2.3 | 27.7 ± 3.6 | 0.284 |
| LVOT | 26.9 ± 2.9 | 26.2 ± 2.0 | 28.8 ± 2.4 | 26.5 ± 2.8 | 30.1 ± 4.9 ac | 0.053 |
| STJ Height | 25.67 ± 5.4 | 24.0 ± 3.2 | 30.9 ± 4.7 a | 27.5 ± 7.1 | 23.2 ± 4.0 | 0.078 |
| STJ | 35.7 ± 4.9 | 32.4 ± 2.7 | 35.1 ± 4.9 | 39.9 ± 4.7 a | 36.3 ± 2.8 | 0.000 |
| AA widest | 41.9 ± 6.2 | 38.6 ± 4.6 | 38.0 ± 4.7 | 47.2 ± 5.3 ab | 41.8 ± 5.5 | 0.000 |
| Right coronary sinus | 36.9 ± 4.5 | 35.2 ± 3.2 | 39.5 ± 7.9 | 38.6 ± 4.5 a | 36.6 ± 5.8 | 0.156 |
| Left coronary sinus | 37.6 ± 4.3 | 35.5 ± 2.9 | 42.1 ± 4.3 a | 39.4 ± 4.3 a | 37.5 ± 6.0 | 0.019 |
| Non-coronary sinus | 38.2 ± 4.1 | 36.3 ± 2.8 | 41.4 ± 5.8 a | 40.1 ± 4.4 a | 37.8 ± 3.7 | 0.037 |
| Left coronary height | 14.4 ± 4.0 | 14.3 ± 3.6 | 20.4 ± 4.2 a | 13.7 ± 3.7 | 12.7 ± 4.7 | 0.045 |
| Right coronary height | 18.6 ± 3.4 | 18.1 ± 3.0 | 22.3 ± 3.5 | 18.4 ± 4.0 | 19.0 ± 2.6 | 0.268 |
| Annulus angle | 49.7 ± 12.4 | 48.2 ± 9.0 | 49.3 ± 3.5 | 51.6 ± 17.4 | 50.3 ± 12.3 | 0.910 |
| >60° | 7 (18.9) | 1 (5.9) | 0 | 5 (38.5) | 1 (25.0) | 0.092 |
| 50°–60° | 12 (32.4) | 4 (23.5) | 1 (33.3) | 6 (46.2) | 1 (25.0) | |
| <50° | 18 (48.6) | 12 (70.6) | 2 (66.7) | 2 (15.4) | 2 (50.0) | |
| Dual-anchoring multiplane measurement (diameter calculated by the perimeter, mm) | ||||||
| Sup-2 mm | 26.2 ± 2.6 | 25.7 ± 1.7 | 27.5 ± 1.7 | 25.5 ± 2.3 | 29.3 ± 5.0 ac | 0.040 |
| Sup-2 mm | 26.7 ± 2.0 | 26.4 ± 1.6 | 27.9 ± 1.4 | 26.7 ± 2.3 | 26.8 ± 2.9 | 0.715 |
| Sup-4 mm | 28.3 ± 2.1 | 28.2 ± 1.8 | 29.4 ± 2.0 | 28.4 ± 2.5 | 27.0 ± 2.4 | 0.516 |
| Sup-6 mm | 29.6 ± 2.2 | 29.4 ± 2.0 | 31.1 ± 2.0 | 29.8 ± 2.2 | 28.0 ± 2.8 | 0.295 |
| Sup-8 mm | 30.5 ± 2.4 | 30.1 ± 1.7 | 31.8 ± 4.5 | 31.1 ± 2.5 | 28.8 ± 2.9 | 0.254 |
| Sup-10 mm | 31.2 ± 2.6 | 30.6 ± 1.8 | 32.4 ± 5.0 | 32.2 ± 2.6 | 30.0 ± 3.0 | 0.212 |
| Sup-min | 26.6 ± 2.0 | 26.4 ± 1.7 | 27.9 ± 1.4 | 26.7 ± 2.3 | 26.2 ± 2.9 | 0.686 |
| AA-30 mm | 37.7 ± 5.4 | 34.2 ± 2.8 | 36.1 ± 5.8 | 42.5 ± 5.2 ab | 38.5 ± 2.6 | 0.000 |
| AA-35 mm | 38.7 ± 5.4 | 35.3 ± 2.9 | 35.3 ± 4.0 | 43.6 ± 5.4 ab | 39.0 ± 2.7 c | 0.000 |
| AA-40 mm | 39.7 ± 5.4 | 36.6 ± 3.1 | 36.2 ± 3.8 | 44.6 ± 5.3 ab | 39.5 ± 3.4 | 0.000 |
| AA-45 mm | 40.6 ± 5.6 | 37.5 ± 3.6 | 36.9 ± 3.9 | 45.5 ± 5.4 ab | 40.7 ± 3.2 | 0.000 |
| AA-50 mm | 41.6 ± 6.1 | 38.0 ± 4.1 | 37.7 ± 3.8 | 46.3 ± 5.4 ab | 44.6 ± 5.9 a | 0.000 |
| AA-min | 37.5 ± 5.2 | 34.2 ± 2.8 | 34.9 ± 4.7 | 42.2 ± 4.8 ab | 37.9 ± 2.3 | 0.000 |
| Characteristics (n, %) | Total N = 37 | Type 1 N = 17 | Type 2 N = 3 | Type 3 N = 13 | Type 4 N = 4 | p |
|---|---|---|---|---|---|---|
| Retrospective cases | 7 (18.9) | 0 | 0 | 3 (23.1) | 4 (100.0) | 0.000 |
| Prospective cases | 30 (81.1) | 17 (100.0) | 3 (100.0) | 10 (76.9) | 0 | |
| General anesthesia | 21 (56.8) | 10 (58.8) | 3 (100.0) | 7 (53.8) | 1 (25.0) | 0.261 |
| Local anesthesia * | 16 (43.2) | 7 (41.2) | 0 | 6 (46.2) | 3 (75.0) | |
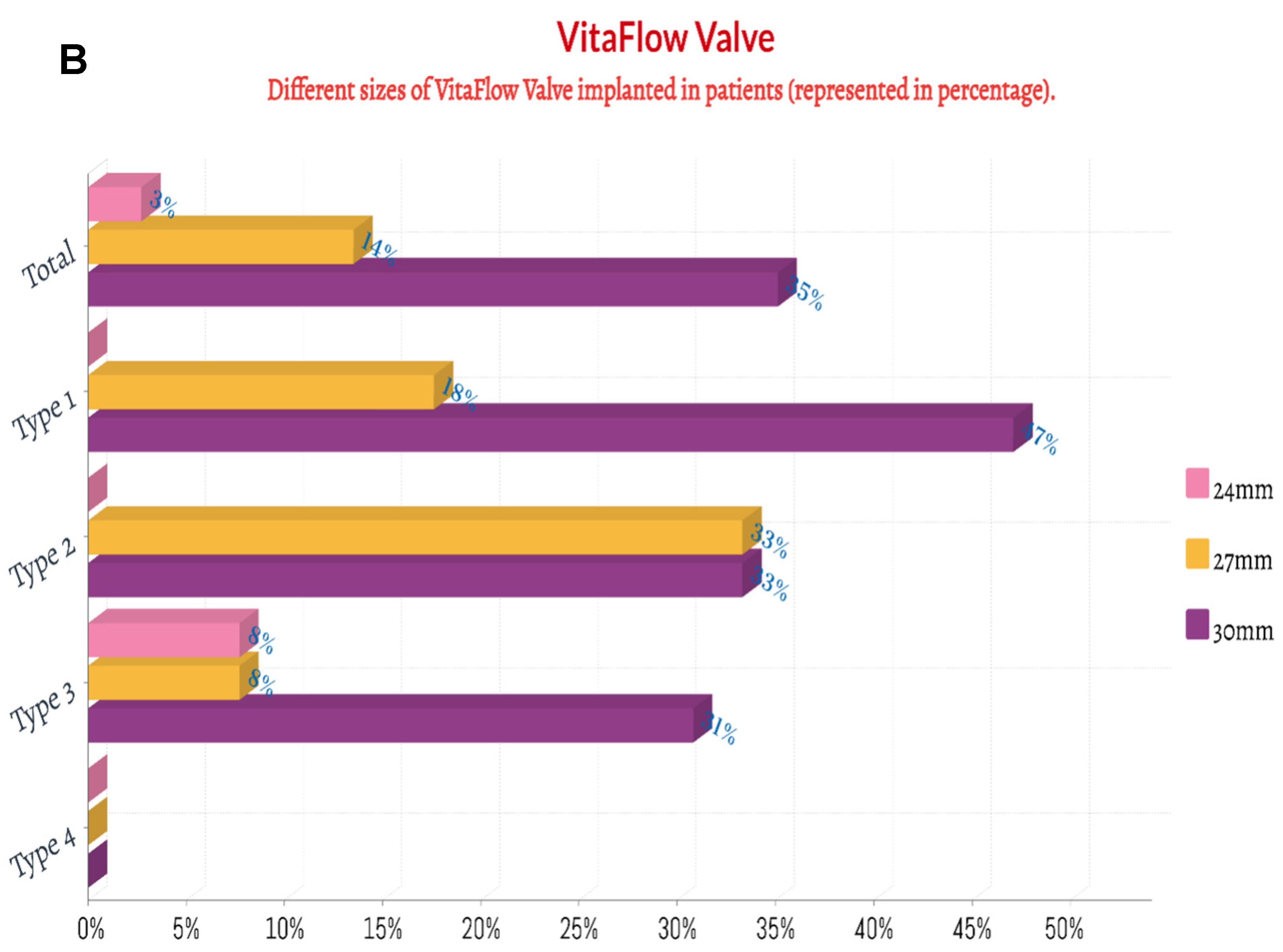
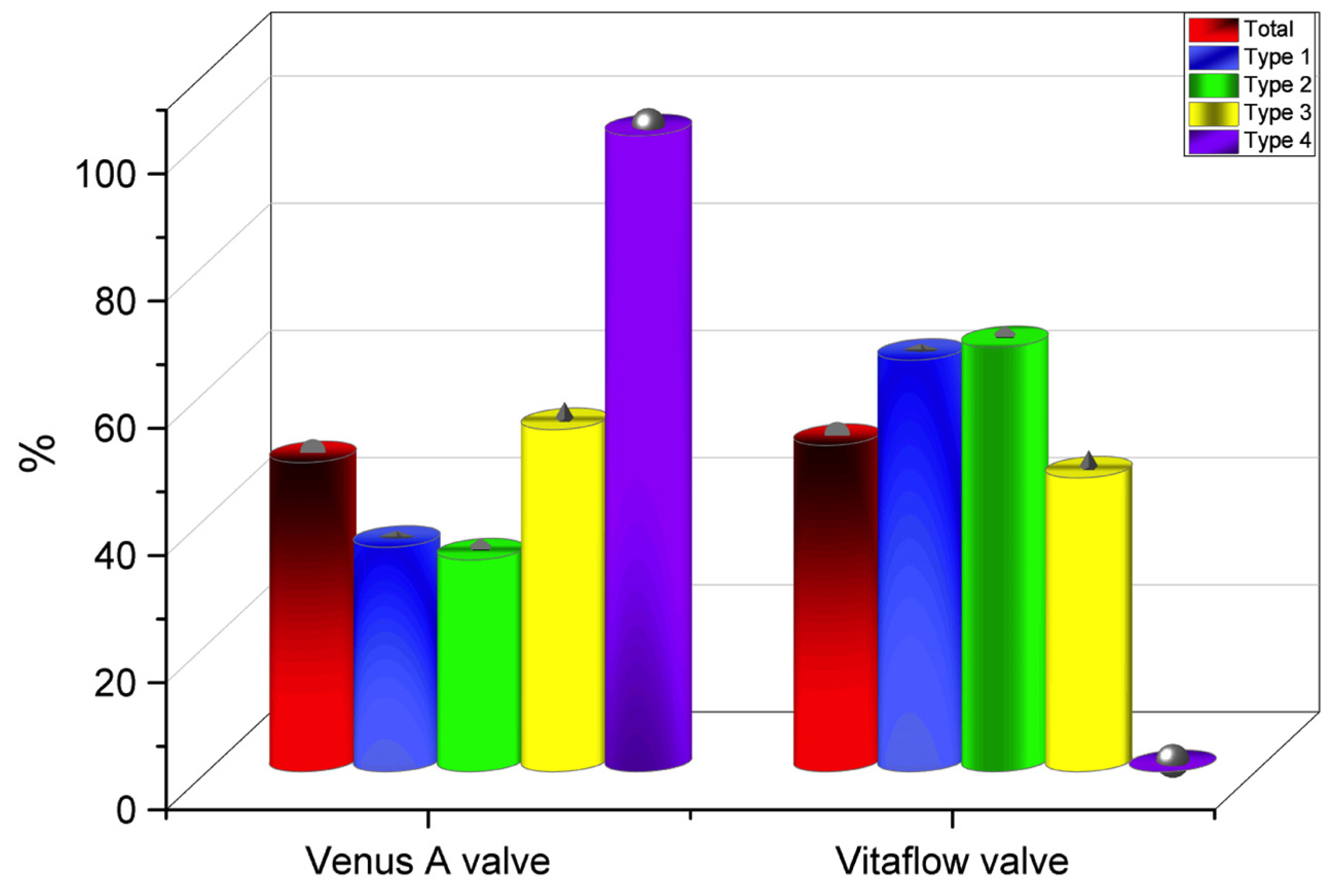
| First-generation THV | 28 (75.7) | 12 (70.6) | 3 (100.0) | 9 (69.2) | 4 (100.0) | 0.426 |
| Second-generation THV # | 9 (24.3) | 5 (29.4) | 0 | 4 (30.8) | 0 | |
| Oversize (%) | ||||||
| Annulus | 17.4 ± 7.9 | 20.4 ± 8.7 | 10.6 ± 1.7 a | 18.0 ± 4.8 | 8.0 ± 5.2 a | 0.009 |
| Narrowest super-annulus | 14.7 ± 7.4 | 16.7 ± 6.7 | 7.7 ± 4.5 | 13.7 ± 7.1 | 13.9 ± 11.0 | 0.240 |
| LVOT | 13.9 ± 11.3 | 17.9 ± 10.8 | 4.3 ± 5.0 a | 15.2 ± 9.6 | −0.4 ± 7.5 ac | 0.007 |
| 2 mm below the annulus | 17.1 ± 9.9 | 20.3 ± 9.6 | 9.3 ± 3.3 a | 19.0 ± 6.8 | 2.6 ± 8.2 a | 0.002 |
| Ascending aorta | 8.3 ± 15.2 | 17.5 ± 9.9 | 19.8 ± 13.3 | −5.1 ± 11.2 ab | 4.0 ± 14.3 ab | 0.000 |
| Narrowest AA | 14.7 ± 16.6 | 25.8 ± 20.7 | 25.0 ± 18.4 | −0.0 ± 11.5 ab | 7.8 ± 10.9 a | 0.000 |
| Outcome | ||||||
| THV downward | 6 (16.2) | 1 (5.9) | 2 (66.7) | 2 (15.4) | 1 (25.0) | 0.066 |
| THV upward | 8 (21.6) | 1 (5.9) | 0 | 5 (38.5) | 2 (50.0) | 0.060 |
| THV release into the artery | 5 (13.5) | 1 (2.7) | 0 | 3 (23.1) | 1 (25.0) | 0.426 |
| THV pulled out of the body | 1 (2.7) | 0 | 0 | 1 (7.7) | 0 | 0.594 |
| THV-in-THV implantation | 8 (21.6) | 1 (5.9) | 2 (66.7) | 3 (23.1) | 2 (50.0) | 0.046 |
| PPI | 9 (24.3) | 7 (41.2) | 0 | 2 (15.4) | 0 | 0.142 |
| Annular rupture | 0 | 0 | 0 | 0 | 0 | - |
| Descending aortic dissection | 2 (5.4) | 0 | 0 | 2 (15.4) | 0 | 0.272 |
| Death | 0 | 0 | 0 | 0 | 0 | - |
| Moderate-to-severe PVL | 5 (13.5) | 0 | 0 | 4 (30.8) | 1 (25.0) | 0.076 |
| Device success | 25 (67.6) | 16 (94.1) | 1 (33.3) | 6 (46.2) | 2 (50.0) | 0.016 |
| Operation success | 32 (86.5) | 17 (100.0) | 3 (100.0) | 9 (69.2) | 3 (75.0) | 0.076 |
| Characteristics (n,%) | Total N = 34 | Type 1 N = 17 | Type 2 N = 3 | Type 3 N = 11 | Type 4 N = 3 | p |
|---|---|---|---|---|---|---|
| Left ventricular ejection fraction (%) | ||||||
| Preoperative | 53.1 ± 12.1 | 53.9 ± 10.1 | 41.3 ± 11.0 | 57.9 ± 12.3 b | 46.3 ± 16.3 | 0.120 |
| Postoperative | 48.9 ± 10.4 | 49.3 ± 11.0 | 44.0 ± 14.4 | 52.1 ± 8.2 | 42.5 ± 9.3 | 0.382 |
| 1 month | 52.9 ± 9.5 | 54.7 ± 9.5 | 52.0 ± 13.5 | 51.4 ± 10.2 | 49.0 ± 7.2 | 0.701 |
| 3 months | 52.4 ± 10.8 | 54.0 ± 10.2 | 47.0 ± 13.9 | 51.6 ± 13.0 | 52.7 ± 7.5 | 0.804 |
| 6 months | 57.9 ± 8.8 n | 59.7 ± 5.2 n | 54.3 ± 15.7 | 57.0 ± 9.0 | 56.3 ± 15.3 | 0.798 |
| 1 year | 59.0 ± 9.2 mnoq | 61.1 ± 7.5 mn | 54.3 ± 12.9 | 55.7 ± 10.1 | 61.7 ± 11.6 n | 0.511 |
| Interventricular septal thickness (mm) | ||||||
| Preoperative | 10.2 ± 1.6 | 9.9 ± 1.5 | 8.7 ± 1.5 | 11.2 ± 1.2 ab | 10.0 ± 1.4 | 0.046 |
| Postoperative | 10.7 ± 1.7 | 10.7 ± 1.5 | 10.7 ± 2.9 | 11.6 ± 1.3 | 9.0 ± 1.8 c | 0.079 |
| 1 month | 10.8 ± 2.4 | 10.4 ± 2.0 | 10.7 ± 3.1 | 12.0 ± 3.3 | 10.3 ± 1.0 | 0.466 |
| 3 months | 10.7 ± 2.2 | 10.5 ± 2.1 | 10.0 ± 1.0 | 11.5 ± 3.0 | 9.7 ± 0.6 | 0.580 |
| 6 months | 10.8 ± 1.8 | 10.5 ± 1.5 | 10.0 ± 2.0 | 11.5 ± 2.1 | 11.0 ± 2.6 | 0.655 |
| 1 year | 10.3 ± 2.1 | 9.8 ± 1.4 | 8.7 ± 2.1 | 11.8 ± 3.0 b | 10.7 ± 1.5 | 0.133 |
| Left ventricular end-diastolic diameter (mm) | ||||||
| Preoperative | 62.5 ± 7.3 | 61.4 ± 8.6 | 69.3 ± 4.0 | 62.7 ± 5.8 | 62.0 ± 4.7 | 0.393 |
| Postoperative | 56.4 ± 7.5 m | 54.7 ± 8.7 m | 60.3 ± 7.6 m | 56.4 ± 4.5 m | 61.3 ± 7.0 | 0.339 |
| 1 month | 54.3 ± 6.9 m | 52.6 ± 6.5 m | 59.7 ± 6.0 m | 54.1 ± 4.9 m | 58.3 ± 11.0 | 0.251 |
| 3 months | 52.9 ± 5.5 mn | 51.3 ± 4.1 m | 57.7 ± 3.2 m | 53.6 ± 6.7 m | 53.3 ± 8.5 | 0.338 |
| 6 months | 50.1 ± 6.1 mno | 48.6 ± 6.8 mn | 56.7 ± 3.2 m | 49.5 ± 4.6 mn | 49.3 ± 6.7 m | 0.250 |
| 1 year | 49.9 ± 6.4 mno | 48.8 ± 7.2 mn | 53.0 ± 5.2 m | 49.2 ± 6.0 mn | 53.0 ± 5.2 | 0.623 |
| N-terminal pro–B-type natriuretic peptide (pg/mL) | ||||||
| Preoperative | 3481.4 ± 6419.5 | 2647.0 ± 5601.6 | 4594.7 ± 5298.8 | 1267.2 ± 1234.7 | 11,728.6 ± 12,265.7 ac | 0.034 |
| Postoperative | 1495.1 ± 1275.0 m | 1457.6 ± 1345.8 | 1932.3 ± 1580.9 | 1083.2 ± 863.0 | 2356.5 ± 1602.1 m | 0.369 |
| 1 month | 1605.0 ± 1446.0 m | 755.1 ± 1145.0 | 2360.3 ± 3533.2 | 863.0 ± 817.0 | 1609.1 ± 1045.2 m | 0.294 |
| 3 months | 934.4 ± 1293.8 m | 748.1 ± 1047.2 | 2050.0 ± 3026.0 | 710.2 ± 600.5 | 1070.5 ± 936.9 | 0.468 |
| 6 months | 1038.3 ± 1862.9 m | 671.0 ± 762.7 | 2786.4 ± 4512.5 | 760.3 ± 1133.5 | 703.3 ± 679.0 m | 0.396 |
| 1 year | 672.0 ± 929.3 m | 712.6 ± 1127.7 | 875.2 ± 1162.8 | 323.5 ± 342.6 m | 1383.5 ± 1130.7 | 0.539 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Ferdous, M.M.; Kottu, L.; Zhao, J.; Zhang, H.-L.; Wang, M.-Y.; Niu, G.-N.; Liu, Q.-R.; Zhou, Z.; Zhao, Z.-Y.; et al. Can Measuring the ‘Dual Anchors of Aorta’ Enhance the Success Rate of TAVR?—A Single-Center Experience. J. Clin. Med. 2023, 12, 1157. https://doi.org/10.3390/jcm12031157
Chen Y, Ferdous MM, Kottu L, Zhao J, Zhang H-L, Wang M-Y, Niu G-N, Liu Q-R, Zhou Z, Zhao Z-Y, et al. Can Measuring the ‘Dual Anchors of Aorta’ Enhance the Success Rate of TAVR?—A Single-Center Experience. Journal of Clinical Medicine. 2023; 12(3):1157. https://doi.org/10.3390/jcm12031157
Chicago/Turabian StyleChen, Yang, Md Misbahul Ferdous, Lakshme Kottu, Jie Zhao, Hong-Liang Zhang, Mo-Yang Wang, Guan-Nan Niu, Qing-Rong Liu, Zheng Zhou, Zhen-Yan Zhao, and et al. 2023. "Can Measuring the ‘Dual Anchors of Aorta’ Enhance the Success Rate of TAVR?—A Single-Center Experience" Journal of Clinical Medicine 12, no. 3: 1157. https://doi.org/10.3390/jcm12031157
APA StyleChen, Y., Ferdous, M. M., Kottu, L., Zhao, J., Zhang, H.-L., Wang, M.-Y., Niu, G.-N., Liu, Q.-R., Zhou, Z., Zhao, Z.-Y., Zhang, Q., Feng, D.-J., Zhang, B., Li, Z.-A., Merkus, D., Lv, B., Xu, H.-Y., Song, G.-Y., & Wu, Y.-J. (2023). Can Measuring the ‘Dual Anchors of Aorta’ Enhance the Success Rate of TAVR?—A Single-Center Experience. Journal of Clinical Medicine, 12(3), 1157. https://doi.org/10.3390/jcm12031157






