PSMA PET/CT in Castration-Resistant Prostate Cancer: Myth or Reality?
Abstract
:1. Introduction
2. Materials and Methods
3. Results
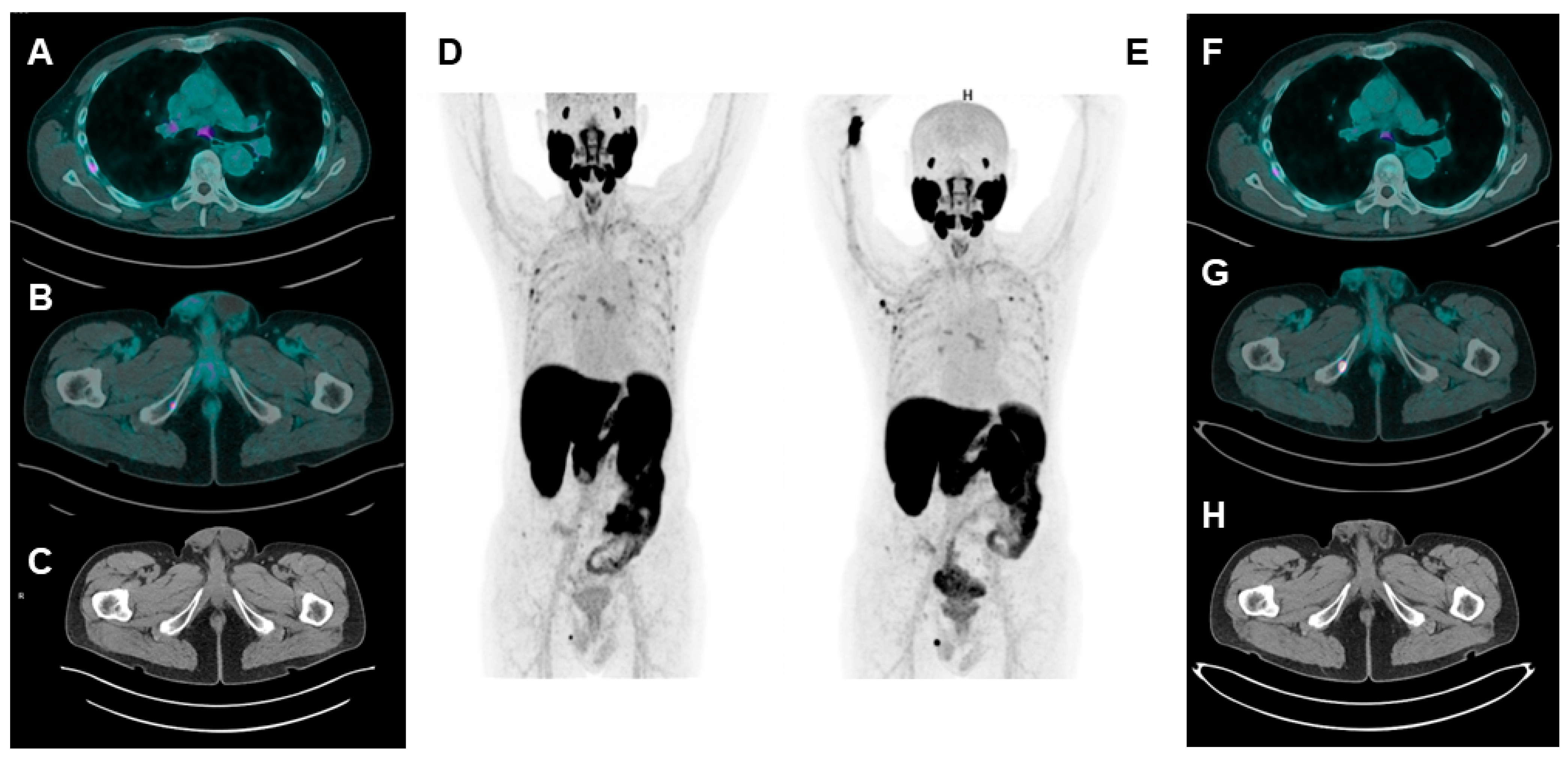
3.1. From M0 to M1 Castration-Resistant Prostate Cancer: Bargain or Lost Opportunity?
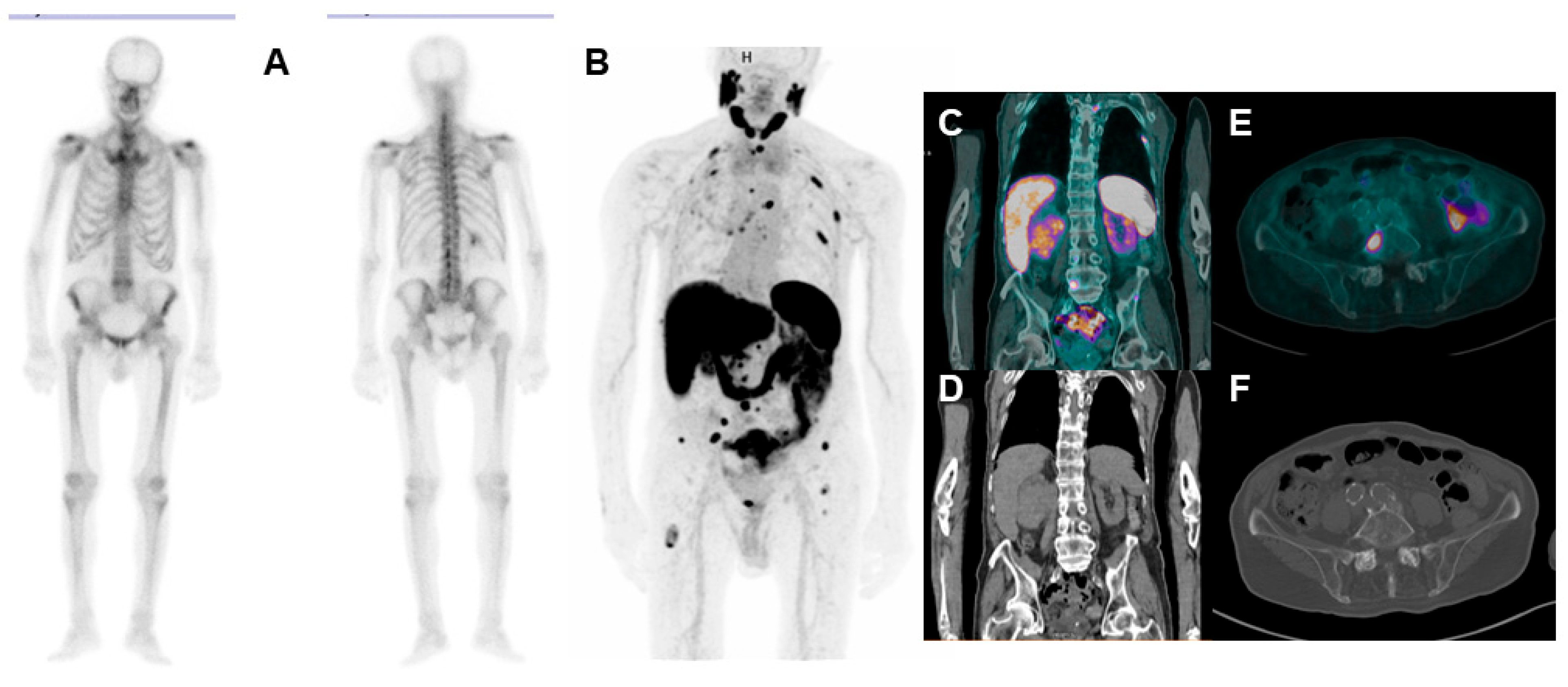
3.2. Unspecific Bone Uptake: Mind the Gap!
3.3. Dual-Tracer PSMA/18F-FDG: Is It a Must?
3.4. Second Primary Neoplasms: Is PSMA Really “Prostate Specific”?
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Scher, H.I.; Morris, M.J.; Stadler, W.M.; Higano, C.; Basch, E.; Fizazi, K.; Antonarakis, E.S.; Beer, T.M.; Carducci, M.A.; Chi, K.N.; et al. Trial design and objectives for castration-resistant prostate cancer: Updated recommendations from the prostate cancer clinical trials working group 3. J. Clin. Oncol. 2016, 34, 1402–1418. [Google Scholar] [CrossRef]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, G.; et al. EAU-EANM-ESTRO-ESUR-ISUP_SIOG Guidelines on Prostate Cancer 2022. Eur. Urol. 2022, 79, 243–262. [Google Scholar] [CrossRef] [PubMed]
- Sharova, E.; Maruzzo, M.; Del Bianco, P.; Cavallari, I.; Pierantoni, F.; Basso, U.; Ciminale, V.; Zagonel, V. Prognostic Stratification of Metastatic Prostate Cancer Patients Treated with Abiraterone and Enzalutamide through an Integrated Analysis of Circulating Free microRNAs and Clinical Parameters. Front. Oncol. 2021, 11, 626104. [Google Scholar] [CrossRef]
- Berruti, A.; Bracarda, S.; Caffo, O.; Cortesi, E.; D’Angelillo, R.; Del Re, M.; Facchini, G.; Pappagallo, G.; Procopio, G.; Sabbatini, R.; et al. nmCRPC, a look in the continuous care of prostate cancer patients: State of art and future perspectives. Cancer Treat. Rev. 2023, 115, 102525. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Bögemann, M.; Suzuki, K.; Shore, N. Treatment of nonmetastatic castration-resistant prostate cancer: Focus on second-generation androgen receptor inhibitors. Prostate Cancer Prostatic Dis. 2021, 24, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Eiber, M.; Beheshti, M.; Bomanji, J.; Calais, J.; Ceci, F.; Cho, S.Y.; Fanti, S.; Giesel, F.L.; Goffin, K.; et al. PSMA PET/CT: Joint EANM procedure guideline/SNMMI procedure standard for prostate cancer imaging 2.0. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 1466–1486. [Google Scholar] [CrossRef] [PubMed]
- Fourquet, A.; Aveline, C.; Cussenot, O.; Créhange, G.; Montravers, F.; Talbot, J.N.; Gauthé, M. 68Ga-PSMA-11 PET/CT in restaging castration-resistant nonmetastatic prostate cancer: Detection rate, impact on patients’ disease management and adequacy of impact. Sci. Rep. 2020, 10, 2104. [Google Scholar] [CrossRef]
- Farolfi, A.; Hirmas, N.; Gafita, A.; Weber, M.; Barbato, F.; Wetter, A.; Mei, R.; Pianori, D.; Hadaschik, B.; Herrmann, K.; et al. Identification of PCWG3 Target Populations Is More Accurate and Reproducible with PSMA PET than with Conventional Imaging: A Multicenter Retrospective Study. J. Nucl. Med. 2021, 62, 675–678. [Google Scholar] [CrossRef]
- Fendler, W.P.; Weber, M.; Iravani, A.; Hofman, M.S.; Calais, J.; Czernin, J.; Ilhan, H.; Saad, F.; Small, E.J.; Smith, M.R.; et al. Prostate-specific membrane antigen ligand positron emission tomography in men with nonmetastatic castration-resistant prostate cancer. Clin. Cancer Res. 2019, 25, 7448–7454. [Google Scholar] [CrossRef]
- Pyka, T.; Okamoto, S.; Dahlbender, M.; Tauber, R.; Retz, M.; Heck, M.; Tamaki, N.; Schwaiger, M.; Maurer, T.; Eiber, M. Comparison of bone scintigraphy and 68Ga-PSMA PET for skeletal staging in prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 2114–2121. [Google Scholar] [CrossRef]
- Zang, S.; Shao, G.; Cui, C.; Li, T.-N.; Huang, Y.; Yao, X.; Fan, Q.; Chen, Z.; Du, J.; Jia, R.; et al. 68Ga-PSMA-11 PET/CT for prostate cancer staging and risk stratification in Chinese patients. Oncotarget 2017, 8, 12247–12258. [Google Scholar] [CrossRef]
- Mitsogianni, M.; Papatsoris, A.; Bala, V.M.; Issa, H.; Moussa, M.; Mitsogiannis, I. An overview of hormonal directed pharmacotherapy for the treatment of prostate cancer. Expert Opin. Pharmacother. 2023, 24, 1765–1774. [Google Scholar] [CrossRef]
- Cattrini, C.; Caffo, O.; Olmos, D.; Castro, E.; De Giorgi, U.; Mennitto, A.; Gennari, A. Apalutamide, Darolutamide and Enzalutamide for Nonmetastatic Castration-Resistant Prostate Cancer (nmCRPC): A Critical Review. Cancers 2022, 14, 1792. [Google Scholar] [CrossRef]
- Murthy, V.; Aggarwal, R.; Koo, P.J. The Emerging Role of Next-Generation Imaging in Prostate Cancer. Curr. Oncol. Rep. 2022, 24, 33–42. [Google Scholar] [CrossRef]
- Filippi, L.; Bagni, O.; Schillaci, O. Digital PET/CT with 18F-FACBC in early castration-resistant prostate cancer: Our preliminary results. Expert Rev. Med. Devices 2022, 19, 591–598. [Google Scholar] [CrossRef]
- Anttinen, M.; Ettala, O.; Malaspina, S.; Jambor, I.; Sandell, M.; Kajander, S.; Rinta-Kiikka, I.; Schildt, J.; Saukko, E.; Rautio, P.; et al. A Prospective Comparison of 18F-prostate-specific Membrane Antigen-1007 Positron Emission Tomography Computed Tomography, Whole-body 1.5 T Magnetic Resonance Imaging with Diffusion-weighted Imaging, and Single-photon Emission Computed Tomography/Computed Tomography with Traditional Imaging in Primary Distant Metastasis Staging of Prostate Cancer (PROSTAGE). Eur. Urol. Oncol. 2021, 4, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, C.; Wei, Y.; Meng, J.; Zhang, Y.; Gan, H.; Xu, X.; Wan, F.; Pan, J.; Ma, X.; et al. A prospective trial of 68Ga-PSMA and 18F-FDG PET/CT in nonmetastatic prostate cancer patients with an early PSA progression during castration. Clin. Cancer Res. 2020, 26, 4551–4558. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.; Kurek, C.; Barbato, F.; Eiber, M.; Maurer, T.; Nader, M.; Hadaschik, B.; Grünwald, V.; Herrmann, K.; Wetter, A.; et al. PSMA-Ligand PET for Early Castration-Resistant Prostate Cancer: A Retrospective Single-Center Study. J. Nucl. Med. 2021, 62, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Friedman, N.C.; Hines, E. The Will Rogers Phenomenon and PSMA PET/CT. J. Nucl. Med. 2022, 63, 966. [Google Scholar] [CrossRef]
- Zapatero, A.; Conde Moreno, A.J.; Barrado, L.; Arcos, M.; Zapatero, A.; Conde Moreno, A.J.; Barrado, M.; Arcos, L.; Aldave, D. Node Oligorecurrence in Prostate Cancer: A Challenge. Cancers 2023, 15, 4159. [Google Scholar] [CrossRef] [PubMed]
- Ingrosso, G.; Detti, B.; Fodor, A.; Caini, S.; Borghesi, S.; Triggiani, L.; Trippa, F.; Russo, D.; Bruni, A.; Francolini, G.; et al. Stereotactic ablative radiotherapy in castration-resistant prostate cancer patients with oligoprogression during androgen receptor-targeted therapy. Clin. Transl. Oncol. 2021, 23, 1577–1584. [Google Scholar] [CrossRef]
- Fendler, W.P.; Calais, J.; Eiber, M.; Flavell, R.R.; Mishoe, A.; Feng, F.Y.; Nguyen, H.G.; Reiter, R.E.; Rettig, M.B.; Okamoto, S.; et al. Assessment of 68Ga-PSMA-11 PET Accuracy in Localizing Recurrent Prostate Cancer: A Prospective Single-Arm Clinical Trial. JAMA Oncol. 2019, 5, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.S.; Lawrentschuk, N.; Francis, R.J.; Tang, C.; Vela, I.; Thomas, P.; Rutherford, N.; Martin, J.M.; Frydenberg, M.; Shakher, R.; et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): A prospective, randomised, multicentre study. Lancet 2020, 395, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Rauscher, I.; Krönke, M.; König, M.; Gafita, A.; Maurer, T.; Horn, T.; Schiller, K.; Weber, W.; Eiber, M. Matched-Pair Comparison of 68Ga-PSMA-11 PET/CT and 18F-PSMA-1007 PET/CT: Frequency of Pitfalls and Detection Efficacy in Biochemical Recurrence after Radical Prostatectomy. J. Nucl. Med. 2020, 61, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Sheikhbahaei, S.; Afshar-Oromieh, A.; Eiber, M.; Solnes, L.B.; Javadi, M.S.; Ross, A.E.; Pienta, K.J.; Allaf, M.E.; Haberkorn, U.; Pomper, M.G.; et al. Pearls and pitfalls in clinical interpretation of prostate-specific membrane antigen (PSMA)-targeted PET imaging. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 2117–2136. [Google Scholar] [CrossRef]
- Jochumsen, M.R.; Bouchelouche, K. PSMA PET/CT for Primary Staging of Prostate Cancer—An Updated Overview. Semin. Nucl. Med. 2023, 37487824. [Google Scholar] [CrossRef]
- Arnfield, E.G.; Thomas, P.A.; Roberts, M.J.; Pelecanos, A.M.; Ramsay, S.C.; Lin, C.Y.; Latter, M.J.; Garcia, P.L.; Pattison, D.A. Clinical insignificance of [18F]PSMA-1007 avid non-specific bone lesions: A retrospective evaluation. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4495–4507. [Google Scholar] [CrossRef]
- Grünig, H.; Maurer, A.; Thali, Y.; Kovacs, Z.; Strobel, K.; Burger, I.A.; Müller, J. Focal unspecific bone uptake on [18F]-PSMA-1007 PET: A multicenter retrospective evaluation of the distribution, frequency, and quantitative parameters of a potential pitfall in prostate cancer imaging. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4483–4494. [Google Scholar] [CrossRef]
- Hoberück, S.; Löck, S.; Borkowetz, A.; Sommer, U.; Winzer, R.; Zöphel, K.; Fedders, D.; Michler, E.; Kotzerke, J.; Kopka, K.; et al. Intraindividual comparison of [68Ga]-Ga-PSMA-11 and [18F]-F-PSMA-1007 in prostate cancer patients: A retrospective single-center analysis. EJNMMI Res. 2021, 11, 109. [Google Scholar] [CrossRef]
- Seifert, R.; Telli, T.; Hadaschik, B.; Fendler, W.P.; Kuo, P.H.; Herrmann, K. Is 18F-FDG PET Needed to Assess 177Lu-PSMA Therapy Eligibility? A VISION-like, Single-Center Analysis. J. Nucl. Med. 2023, 64, 731–737. [Google Scholar] [CrossRef]
- Ninatti, G.; Pini, C.; Gelardi, F.; Ghezzo, S.; Mapelli, P.; Picchio, M.; Antunovic, L.; Briganti, A.; Montorsi, F.; Landoni, C.; et al. The potential role of osteoporosis in unspecific [18F]PSMA-1007 bone uptake. Eur. J. Nucl. Med. Mol. Imaging 2023, 1–8. [Google Scholar] [CrossRef]
- Eiber, M.; Kroenke, M.; Wurzer, A.; Ulbrich, L.; Jooß, L.; Maurer, T.; Horn, T.; Schiller, K.; Langbein, T.; Buschner, G.; et al. 18F-rhPSMA-7 PET for the Detection of Biochemical Recurrence of Prostate Cancer after Radical Prostatectomy. J. Nucl. Med. 2020, 61, 696. [Google Scholar] [CrossRef]
- Kroenke, M.; Mirzoyan, L.; Horn, T.; Peeken, J.C.; Wurzer, A.; Wester, H.J.; Makowski, M.; Weber, W.A.; Eiber, M.; Rauscher, I. Matched-Pair Comparison of 68Ga-PSMA-11 and 18F-rhPSMA-7 PET/CT in Patients with Primary and Biochemical Recurrence of Prostate Cancer: Frequency of Non–Tumor-Related Uptake and Tumor Positivity. J. Nucl. Med. 2021, 62, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Alberts, I.; Sachpekidis, C.; Fech, V.; Rominger, A.; Afshar-Oromieh, A. PSMA-negative prostate cancer and the continued value of choline-PET/CT. NuklearMedizin 2020, 59, 33–34. [Google Scholar] [CrossRef]
- Urso, L.; Lancia, F.; Ortolan, N.; Frapoli, M.; Rauso, M.; Artioli, P.; Cittanti, C.; Uccelli, L.; Frassoldati, A.; Evangelista, L.; et al. 18F-Choline PET/CT or PET/MR and the evaluation of response to systemic therapy in prostate cancer: Are we ready? Clin. Transl. Imaging 2022, 10, 687–695. [Google Scholar] [CrossRef] [PubMed]
- De Giorgi, U.; Caroli, P.; Scarpi, E.; Conteduca, V.; Burgio, S.L.; Menna, C.; Moretti, A.; Galassi, R.; Rossi, L.; Amadori, D.; et al. 18F-Fluorocholine PET/CT for early response assessment in patients with metastatic castration-resistant prostate cancer treated with enzalutamide. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1276–1283. [Google Scholar] [CrossRef]
- Filippi, L.; Urso, L.; Schillaci, O.; Evangelista, L. [18F]-FDHT PET for the Imaging of Androgen Receptor in Prostate and Breast Cancer: A Systematic Review. Diagnostics 2023, 13, 2613. [Google Scholar] [CrossRef] [PubMed]
- Beltran, H.; Romanel, A.; Conteduca, V.; Casiraghi, N.; Sigouros, M.; Franceschini, G.M.; Orlando, F.; Fedrizzi, T.; Ku, S.Y.; Dann, E.; et al. Circulating tumor DNA profile recognizes transformation to castration-resistant neuroendocrine prostate cancer. J. Clin. Investig. 2020, 130, 1653. [Google Scholar] [CrossRef]
- Bluemn, E.G.; Coleman, I.M.; Lucas, J.M.; Coleman, R.T.; Hernandez-Lopez, S.; Tharakan, R.; Bianchi-Frias, D.; Dumpit, R.F.; Kaipainen, A.; Corella, A.N.; et al. Androgen Receptor Pathway-Independent Prostate Cancer Is Sustained through FGF Signaling. Cancer Cell 2017, 32, 474–489.e6. [Google Scholar] [CrossRef]
- Bakht, M.K.; Lovnicki, J.M.; Tubman, J.; Stringer, K.F.; Chiaramonte, J.; Reynolds, M.R.; Derecichei, I.; Ferraiuolo, R.M.; Fifield, B.A.; Lubanska, D.; et al. Differential Expression of Glucose Transporters and Hexokinases in Prostate Cancer with a Neuroendocrine Gene Signature: A Mechanistic Perspective for 18F-FDG Imaging of PSMA-Suppressed Tumors. J. Nucl. Med. 2020, 61, 904–910. [Google Scholar] [CrossRef]
- Wang, H.T.; Yao, Y.H.; Li, B.G.; Tang, Y.; Chang, J.W.; Zhang, J. Neuroendocrine Prostate Cancer (NEPC) Progressing from conventional prostatic adenocarcinoma: Factors associated with time to development of nepc and survival from NEPC Diagnosis-A systematic review and pooled analysis. J. Clin. Oncol. 2014, 32, 3383–3390. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Wang, Y.; Zhu, Y.; Shi, Y.; Xu, L.; Huang, G.; Liu, J. The Added Value of 18F-FDG PET/CT Compared with 68Ga-PSMA PET/CT in Patients with Castration-Resistant Prostate Cancer. J. Nucl. Med. 2022, 63, 69–75. [Google Scholar] [CrossRef]
- Dondi, F.; Antonelli, A.; Suardi, N.; Guerini, A.E.; Albano, D.; Lucchini, S.; Camoni, L.; Treglia, G.; Bertagna, F. PET/CT and Conventional Imaging for the Assessment of Neuroendocrine Prostate Cancer: A Systematic Review. Cancers 2023, 15, 4404. [Google Scholar] [CrossRef] [PubMed]
- Bauckneht, M.; Bertagna, F.; Donegani, M.I.; Durmo, R.; Miceli, A.; De Biasi, V.; Laudicella, R.; Fornarini, G.; Berruti, A.; Baldari, S.; et al. The prognostic power of 18F-FDG PET/CT extends to estimating systemic treatment response duration in metastatic castration-resistant prostate cancer (mCRPC) patients. Prostate Cancer Prostatic Dis. 2021, 24, 1198–1207. [Google Scholar] [CrossRef] [PubMed]
- Bauckneht, M.; Marini, C.; Cossu, V.; Campi, C.; Riondato, M.; Bruno, S.; Orengo, A.M.; Vitale, F.; Carta, S.; Chiola, S.; et al. Gene’s expression underpinning the divergent predictive value of [18F]F-fluorodeoxyglucose and prostate-specific membrane antigen positron emission tomography in primary prostate cancer: A bioinformatic and experimental study. J. Transl. Med. 2023, 21, 3. [Google Scholar] [CrossRef]
- Wang, J.; Xu, W.; Wang, B.; Lin, G.; Wei, Y.; Abudurexiti, M.; Zhu, W.; Liu, C.; Qin, X.; Dai, B.; et al. GLUT1 is an AR target contributing to tumor growth and glycolysis in castration-resistant and enzalutamide-resistant prostate cancers. Cancer Lett. 2020, 485, 45–55. [Google Scholar] [CrossRef]
- Perez, P.M.; Hope, T.A.; Behr, S.C.; Van Zante, A.; Small, E.J.; Flavell, R.R. Intertumoral Heterogeneity of 18 F-FDG and 68Ga-PSMA Uptake in Prostate Cancer Pulmonary Metastases. Clin. Nucl. Med. 2019, 44, e28–e32. [Google Scholar] [CrossRef]
- Michalski, K.; Ruf, J.; Goetz, C.; Seitz, A.K.; Buck, A.K.; Lapa, C.; Hartrampf, P.E. Prognostic implications of dual tracer PET/CT: PSMA ligand and [18F]FDG PET/CT in patients undergoing [177Lu]PSMA radioligand therapy. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2024–2030. [Google Scholar] [CrossRef]
- Güzel, Y.; Kömek, H.; Can, C.; Kaplan, İ.; Akdeniz, N.; Kepenek, F.; Gündoğan, C. Role of volumetric parameters obtained from 68Ga-PSMA PET/CT and 18F-FDG PET/CT in predicting overall survival in patients with mCRPC receiving taxane therapy. Ann. Nucl. Med. 2023, 37, 517–527. [Google Scholar] [CrossRef]
- Adnan, A.; Basu, S. Concept proposal for a six-tier integrated dual tracer PET-CT (68Ga-PSMA and FDG) image scoring system (“Pro-PET” score) and examining its potential implications in metastatic castration-resistant prostate carcinoma theranostics and prognosis. Nucl. Med. Commun. 2021, 42, 566–574. [Google Scholar] [CrossRef]
- Hofman, M.S.; Violet, J.; Hicks, R.J.; Ferdinandus, J.; Ping Thang, S.; Akhurst, T.; Iravani, A.; Kong, G.; Ravi Kumar, A.; Murphy, D.G.; et al. [177 Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): A single-centre, single-arm, phase 2 study. Lancet Oncol. 2018, 19, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Ferdinandus, J.; Violet, J.; Sandhu, S.; Hicks, R.J.; Ravi Kumar, A.S.; Iravani, A.; Kong, G.; Akhurst, T.; Thang, S.P.; Murphy, D.G.; et al. Prognostic biomarkers in men with metastatic castration-resistant prostate cancer receiving [177Lu]-PSMA-617. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2322–2327. [Google Scholar] [CrossRef] [PubMed]
- Pouliot, F.; Beauregard, J.M.; Saad, F.; Trudel, D.; Richard, P.O.; Turcotte, É.; Rousseau, É.; Probst, S.; Kassouf, W.; Anidjar, M.; et al. The Triple-Tracer strategy against Metastatic PrOstate cancer (3TMPO) study protocol. BJU Int. 2022, 130, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Jadvar, H. Is There Utility for FDG PET in Prosate Cancer? Semin. Nucl. Med. 2016, 46, 502. [Google Scholar] [CrossRef]
- Filippi, L.; Urso, L.; Bianconi, F.; Palumbo, B.; Marzola, M.C.; Evangelista, L.; Schillaci, O. Radiomics and theranostics with molecular and metabolic probes in prostate cancer: Toward a personalized approach. Expert Rev. Mol. Diagn. 2023, 23, 243–255. [Google Scholar] [CrossRef]
- Chang, S.S.; Reuter, V.E.; Heston, W.D.W.; Bander, N.H.; Grauer, L.S.; Gaudin, P.B. Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res. 1999, 59, 3192–3198. [Google Scholar]
- Rizzo, A.; Dall’Armellina, S.; Pizzuto, D.A.; Perotti, G.; Zagaria, L.; Lanni, V.; Treglia, G.; Racca, M.; Annunziata, S. PSMA Radioligand Uptake as a Biomarker of Neoangiogenesis in Solid Tumours: Diagnostic or Theragnostic Factor? Cancers 2022, 14, 4039. [Google Scholar] [CrossRef]
- Chang, S.S.; Reuter, V.E.; Heston, W.D.W.; Gaudin, P.B. Metastatic renal cell carcinoma neovasculature expresses prostate-specific membrane antigen. Urology 2001, 57, 801–805. [Google Scholar] [CrossRef]
- Urso, L.; Castello, A.; Rocca, G.C.; Lancia, F.; Panareo, S.; Cittanti, C.; Uccelli, L.; Florimonte, L.; Castellani, M.; Ippolito, C.; et al. Role of PSMA-ligands imaging in Renal Cell Carcinoma management: Current status and future perspectives. J. Cancer Res. Clin. Oncol. 2022, 148, 1299–1311. [Google Scholar] [CrossRef]
- Udovicich, C.; Callahan, J.; Bressel, M.; Ong, W.L.; Perera, M.; Tran, B.; Azad, A.; Haran, S.; Moon, D.; Chander, S.; et al. Impact of Prostate-specific Membrane Antigen Positron Emission Tomography/Computed Tomography in the Management of Oligometastatic Renal Cell Carcinoma. Eur. Urol. Open Sci. 2022, 44, 60. [Google Scholar] [CrossRef]
- Muoio, B.; Albano, D.; Dondi, F.; Bertagna, F.; Garibotto, V.; Kunikowska, J.; Piccardo, A.; Annunziata, S.; Espeli, V.; Migliorini, D.; et al. Diagnostic Accuracy of PET/CT or PET/MRI Using PSMA-Targeting Radiopharmaceuticals in High-Grade Gliomas: A Systematic Review and a Bivariate Meta-Analysis. Diagnostics 2022, 12, 1665. [Google Scholar] [CrossRef]
- Lawhn-Heath, C.; Yom, S.S.; Liu, C.; Villanueva-Meyer, J.E.; Aslam, M.; Smith, R.; Narwal, M.; Juarez, R.; Behr, S.C.; Pampaloni, M.H.; et al. Gallium-68 prostate-specific membrane antigen ([68Ga]Ga-PSMA-11) PET for imaging of thyroid cancer: A feasibility study. EJNMMI Res. 2020, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.; Racca, M.; Dall’Armellina, S.; Delgado Bolton, R.C.; Albano, D.; Dondi, F.; Bertagna, F.; Annunziata, S.; Treglia, G. Potential Role of PSMA-Targeted PET in Thyroid Malignant Disease: A Systematic Review. Diagnostics 2023, 13, 564. [Google Scholar] [CrossRef] [PubMed]
- Vuijk, F.A.; Kleiburg, F.; Noortman, W.A.; Heijmen, L.; Feshtali Shahbazi, S.; van Velden, F.H.P.; Baart, V.M.; Bhairosingh, S.S.; Windhorst, B.D.; Hawinkels, L.J.A.C.; et al. Prostate-Specific Membrane Antigen Targeted Pet/CT Imaging in Patients with Colon, Gastric and Pancreatic Cancer. Cancers 2022, 14, 6209. [Google Scholar] [CrossRef] [PubMed]
- Van Boxtel, W.; Lütje, S.; Van Engen-Van Grunsven, I.C.H.; Verhaegh, G.W.; Schalken, J.A.; Jonker, M.A.; Nagarajah, J.; Gotthardt, M.; Van Herpen, C.M.L. 68Ga-PSMA-HBED-CC PET/CT imaging for adenoid cystic carcinoma and salivary duct carcinoma: A phase 2 imaging study. Theranostics 2020, 10, 2273–2283. [Google Scholar] [CrossRef]
- Tariq, A.; McCart Reed, A.E.; Morton, A.; Porten, S.; Vela, I.; Williams, E.D.; Yaxley, J.W.; Black, P.C.; Roberts, M.J. Urothelial Carcinoma and Prostate-specific Membrane Antigen: Cellular, Imaging, and Prognostic Implications. Eur. Urol. Focus 2022, 8, 1256–1269. [Google Scholar] [CrossRef]
- Sathekge, M.; Lengana, T.; Modiselle, M.; Vorster, M.; Zeevaart, J.R.; Maes, A.; Ebenhan, T.; Van de Wiele, C. 68Ga-PSMA-HBED-CC PET imaging in breast carcinoma patients. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 689–694. [Google Scholar] [CrossRef]
- Bertagna, F.; Albano, D.; Cerudelli, E.; Gazzilli, M.; Tomasini, D.; Bonù, M.; Giubbini, R.; Treglia, G. Radiolabelled PSMA PET/CT in breast cancer. A systematic review. Nucl. Med. Rev. 2020, 23, 32–35. [Google Scholar]
- Kunikowska, J.; Korzeniowski, K.; Pełka, K.; Lamparski, K.; Patkowski, W. [68Ga]Ga-PSMA-11 in diagnosis and follow-up after transarterial chemoembolization in hepatocellular carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2023, 1–2. [Google Scholar] [CrossRef]
- Löffeler, S.; Weedon-Fekjaer, H.; Wang-Hansen, M.S.; Sebakk, K.; Hamre, H.; Haug, E.S.; Fosså, S.D. “Natural course” of disease in patients with metastatic castrate-resistant prostate cancer: Survival and prognostic factors without life-prolonging treatment. Scand. J. Urol. 2015, 49, 440–445. [Google Scholar] [CrossRef]
- van Soest, R.J.; Efstathiou, J.A.; Sternberg, C.N.; Tombal, B. The Natural History and Outcome Predictors of Metastatic Castration-resistant Prostate Cancer. Eur. Urol. Focus 2016, 2, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Vassilev, Z.P.; Gabarró, M.S.; Kaye, J.A.; Saltus, C.W.; Riedel, O.; Scholle, O.; Mehtälä, J.; Korhonen, P.; Garbe, E.; Zong, J. Incidence of second primary malignancies in metastatic castration-resistant prostate cancer: Results from observational studies in three countries. Future Oncol. 2020, 16, 1889–1901. [Google Scholar] [CrossRef] [PubMed]
- Mehtälä, J.; Zong, J.; Vassilev, Z.; Brobert, G.; Gabarró, M.S.; Stattin, P.; Khanfir, H. Overall survival and second primary malignancies in men with metastatic prostate cancer. PLoS ONE 2020, 15, e0227552. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Zheng, G.; Hemminki, O.; Försti, A.; Sundquist, K.; Hemminki, K. Prostate cancer survivors: Risk and mortality in second primary cancers. Cancer Med. 2018, 7, 5752. [Google Scholar] [CrossRef] [PubMed]
- Saltus, C.W.; Vassilev, Z.P.; Zong, J.; Calingaert, B.; Andrews, E.B.; Soriano-Gabarró, M.; Kaye, J.A. Incidence of Second Primary Malignancies in Patients with Castration-Resistant Prostate Cancer: An Observational Retrospective Cohort Study in the United States. Prostate Cancer 2019, 2019, 4387415. [Google Scholar] [CrossRef]
- Chakraborty, S.; Tarantolo, S.R.; Batra, S.K.; Hauke, R.J. Incidence and Prognostic Significance of Second Primary Cancers in Renal Cell Carcinoma. Am. J. Clin. Oncol. 2013, 36, 132. [Google Scholar] [CrossRef]
- Chahinian, R.; El-Amine, A.; Matar, S.; Annan, M.; Shamseddine, A.; Haidar, M. 68Ga–Prostate-Specific Membrane Antigen, A Potential Radiopharmaceutical in PET/CT To detect primary Cholangiocarcinoma. Asia Ocean. J. Nucl. Med. Biol. 2020, 8, 136. [Google Scholar] [CrossRef]
- Norouzi, G.; Rezaei, A.; Adinehpour, Z.; Amini, H.; Vali, R. 68Ga-PSMA PET/CT Scan Leading to Diagnosis of PSMA-Positive Rectal Adenocarcinoma in a Patient With Prostate Cancer. Clin. Nucl. Med. 2022, 47, e323–e324. [Google Scholar] [CrossRef]
- Dhiantravan, N.; Hovey, E.; Bosco, A.; Wegner, E.A. Concomitant Prostate Carcinoma and Follicular Lymphoma: “flip-Flop” Appearances on PSMA and FDG PET/CT Scans. Clin. Nucl. Med. 2019, 44, 797–798. [Google Scholar] [CrossRef]
- Chalikandy, A.; Yadav, S.; Basu, S. Differentiation of Discordant Lesions on Dual-Tracer PET/CT (68Ga-PSMA-11 and 18F-FDG) in Prostate Carcinoma: Diagnosis of Second Primary Malignancies. J. Nucl. Med. Technol. 2023, 51, 265779. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, P.; Dai, W. Heterogeneous uptake of 18F-FDG and 18F-PSMA-1007 PET/CT in lung cancer and lymph node metastasis. BMC Pulm. Med. 2023, 23, 73. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P. 68Ga-PSMA-Avid Small Cell Lung Cancer on PET/CT: Incidental Second Malignancy in Treated Prostate Cancer. Clin. Nucl. Med. 2020, 45, 1016–1017. [Google Scholar] [CrossRef] [PubMed]
- Siva, S.; Callahan, J.; Pryor, D.; Martin, J.; Lawrentschuk, N.; Hofman, M.S. Utility of 68Ga prostate specific membrane antigen—positron emission tomography in diagnosis and response assessment of recurrent renal cell carcinoma. J. Med. Imaging Radiat. Oncol. 2017, 61, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Rowe, S.P.; Gorin, M.A.; Hammers, H.J.; Som Javadi, M.; Hawasli, H.; Szabo, Z.; Cho, S.Y.; Pomper, M.G.; Allaf, M.E. Imaging of metastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann. Nucl. Med. 2015, 29, 877–882. [Google Scholar] [CrossRef]

| Reference | Location/Year/ | Study | N. of Patients | Primary Endpoint | PSMA Ligands | Comment |
|---|---|---|---|---|---|---|
| Fendler et al. [9] | Germany/USA 2019 | Retrospective, investigator-initiated, multicenter | 200 | Detection rate of lesions, on a per-patient basis, by PSMA-PET. | 68Ga-PSMA-11/ 18F-DCFPyL | Almost all cases showed positive findings in spite of negative conventional imaging: 55% of patients had distant metastases (M1). |
| Fourquet et al. [7] | France/2020 | Retrospective | 31 | Impact of PSMA PET in the restaging of nmCRPC patients. | 68Ga-PSMA-11 | PSMA-PET detected at least 1 focus of tracer uptake in 90% of cases and changed clinical management in 87% of cases. |
| Wang et al. [17] | China/2021 | Prospective, observational | 37 | To assess metabolic heterogeneity (PSMA+/FDG− disease) in early progressive nmCRPC. | 68Ga-PSMA-11/ [18F]FDG | A total of 114 lesions were detected among 29 out of 37 nmCRPC patients. N+/M+ disease was detected in 73% of patients. |
| Weber et al. [18] | Germany/ 2021 | Retrospective | 55 | To investigate the ability of PSMA PET to detect metastatic lesions in early CRPC. | 68Ga-PSMA-11 | PSMA PET resulted in positive results in 75% of patients, of whom 45% had M1 status. |
| Malignant Findings | UBU Findings |
|---|---|
| Sclerotic/blasting change at any follow-up imaging | PSA < 0.1 ng/mL after curative surgical treatment |
| SUVmax increase (e.g., ≥30%) on follow-up 18F-PSMA-1007 PET, independent from any treatments | Unchanged uptake on follow-up 18F-PSMA-1007 PET with or without therapy after >6 months |
| Treatment-related changes (e.g., reduction in size or increased sclerosis) on follow-up | No longer present on follow-up 18F-PSMA-1007 |
| Appearance of metastatic lesion on different imaging techniques (e.g., 68Ga-PSMA-11 PET, MRI, bone scan, CT) | Benign aspect on a different imaging modality |
| Association with typical symptoms of malignancy | Managed as likely benign after clinical evaluation |
| Association with blood biomarkers (i.e., PSA and ALP increasing/decreasing) | PSA stable or undetectable PSA |
| Reference | Location Year | Study Type | N. of Patients | Primary Endpoint | Radiotracers | Comment |
|---|---|---|---|---|---|---|
| Michalski et al. [48] | Germany 2021 | Retrospective bicenter | 54 | Assessment of mismatched lesions in mCRPC patients before RLT. | [68Ga]-PSMA-11 [18F]FDG | 33% of mCRPC patients show mismatched PSMA- [18F]FDG+ lesions and these patients present shorter OS. |
| Seifert et al. [30] | Germany, USA 2022 | Retrospective | 89 | Assessment of mismatched lesions in mCRPC patients before RLT. | [68Ga]-PSMA-11 [18F]-PSMA-1007 [18F]FDG | 18% of patients had mismatched lesions between PSMA and [18F]FDG PET, however only 3% of patients had mismatch findings not detected using only PSMA PET. |
| Chen et al. [42] | China 2022 | Retrospective | 56 | To assess metabolic heterogeneity of mCRPC patients | [68Ga]-PSMA-11 [18F]FDG | [68Ga]-PSMA-11 PET/CT showed higher detection rate than [18F]FDG PET/CT (75% vs. 51.8%). However, 23.2% of patients showed at least 1 mismatched PSMA- [18F]FDG+ lesion. |
| Güzel et al. [49] | Turkey 2023 | Retrospective | 71 | To investigate the prognostic role of dual-tracer PET imaging in [18F]FDG+ mCRPC treated with chemotherapy | 68Ga-PSMA-11 [18F]FDG | Volumetric parameters and Pro-PET scores obtained from dual-tracer PET/CT imaging predict OS in patients with mCRPC treated with taxane chemotherapy. Dual-tracer imaging should be performed in these patients as 78.9% of visceral metastases were PSMA−/FDG+ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urso, L.; Filippi, L.; Castello, A.; Marzola, M.C.; Bartolomei, M.; Cittanti, C.; Florimonte, L.; Castellani, M.; Zucali, P.; Bruni, A.; et al. PSMA PET/CT in Castration-Resistant Prostate Cancer: Myth or Reality? J. Clin. Med. 2023, 12, 7130. https://doi.org/10.3390/jcm12227130
Urso L, Filippi L, Castello A, Marzola MC, Bartolomei M, Cittanti C, Florimonte L, Castellani M, Zucali P, Bruni A, et al. PSMA PET/CT in Castration-Resistant Prostate Cancer: Myth or Reality? Journal of Clinical Medicine. 2023; 12(22):7130. https://doi.org/10.3390/jcm12227130
Chicago/Turabian StyleUrso, Luca, Luca Filippi, Angelo Castello, Maria Cristina Marzola, Mirco Bartolomei, Corrado Cittanti, Luigia Florimonte, Massimo Castellani, Paolo Zucali, Alessio Bruni, and et al. 2023. "PSMA PET/CT in Castration-Resistant Prostate Cancer: Myth or Reality?" Journal of Clinical Medicine 12, no. 22: 7130. https://doi.org/10.3390/jcm12227130
APA StyleUrso, L., Filippi, L., Castello, A., Marzola, M. C., Bartolomei, M., Cittanti, C., Florimonte, L., Castellani, M., Zucali, P., Bruni, A., Sabbatini, R., Dominici, M., Panareo, S., & Evangelista, L. (2023). PSMA PET/CT in Castration-Resistant Prostate Cancer: Myth or Reality? Journal of Clinical Medicine, 12(22), 7130. https://doi.org/10.3390/jcm12227130









