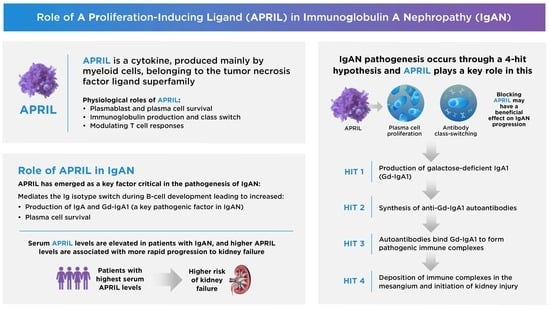
A PRoliferation-Inducing Ligand (APRIL) in the Pathogenesis of Immunoglobulin A Nephropathy: A Review of the Evidence
Abstract
1. Introduction
2. The Biological Roles of APRIL in Health and Disease
2.1. APRIL Production
2.2. Physiologic Functions
2.2.1. B-Cell Survival
2.2.2. Ig Class Switching
2.2.3. Downstream Effects on T Cells
2.3. Association with Disease
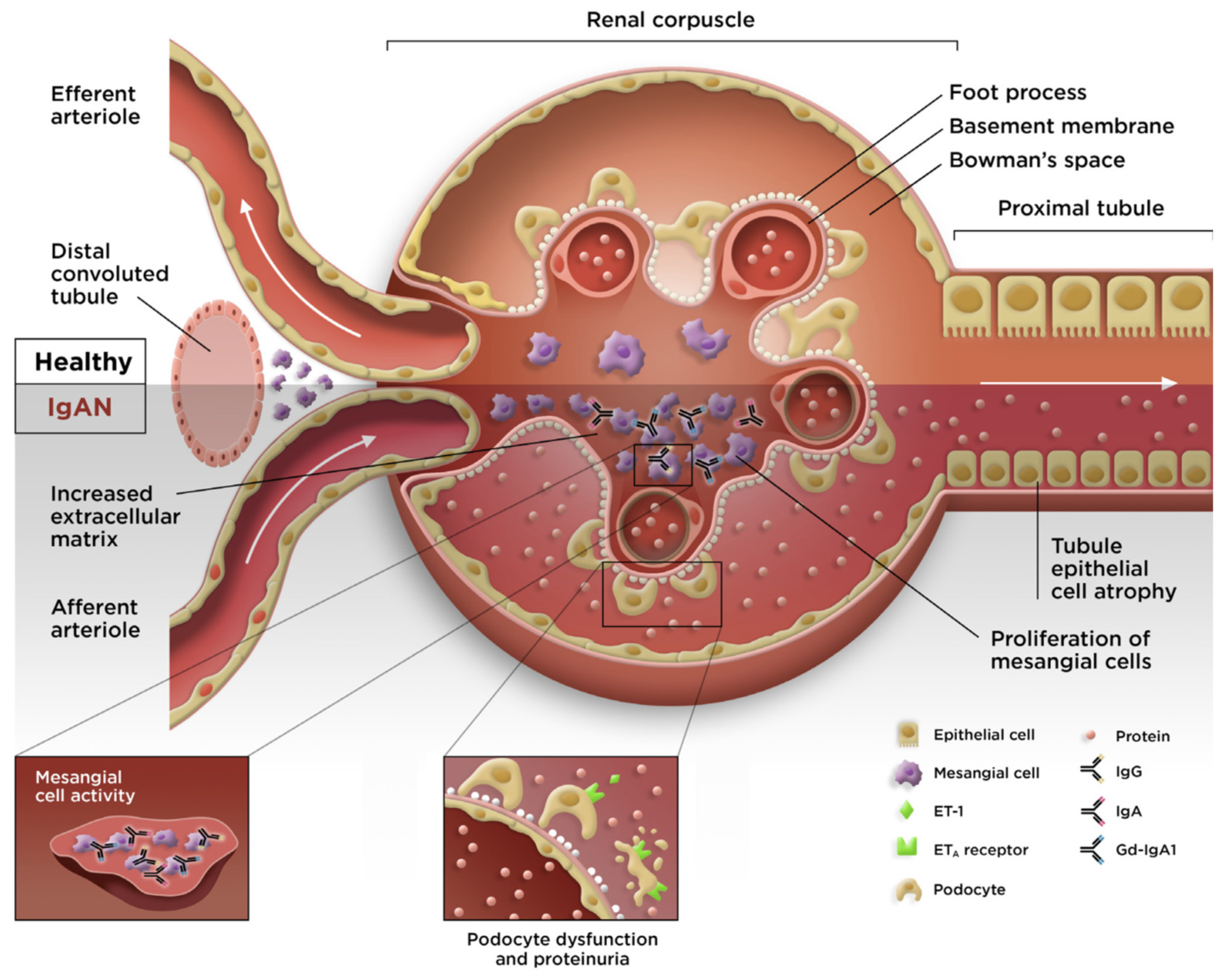
3. Overview of IgAN
4. Evidence Supporting the Involvement of APRIL in IgAN
4.1. Gut Mucosa–Kidney Axis in IgAN
4.2. APRIL Is Produced in GALT
4.3. Clinical Epidemiology
4.3.1. Debate 1: IgAN Pathology: APRIL, BAFF, or Both?
4.3.2. Debate 2: Does Nasopharyngeal Lymphoid Tissue Play a Major Role?
4.4. Insights from Genetic Studies
4.5. Experimental Evidence from Animal Models
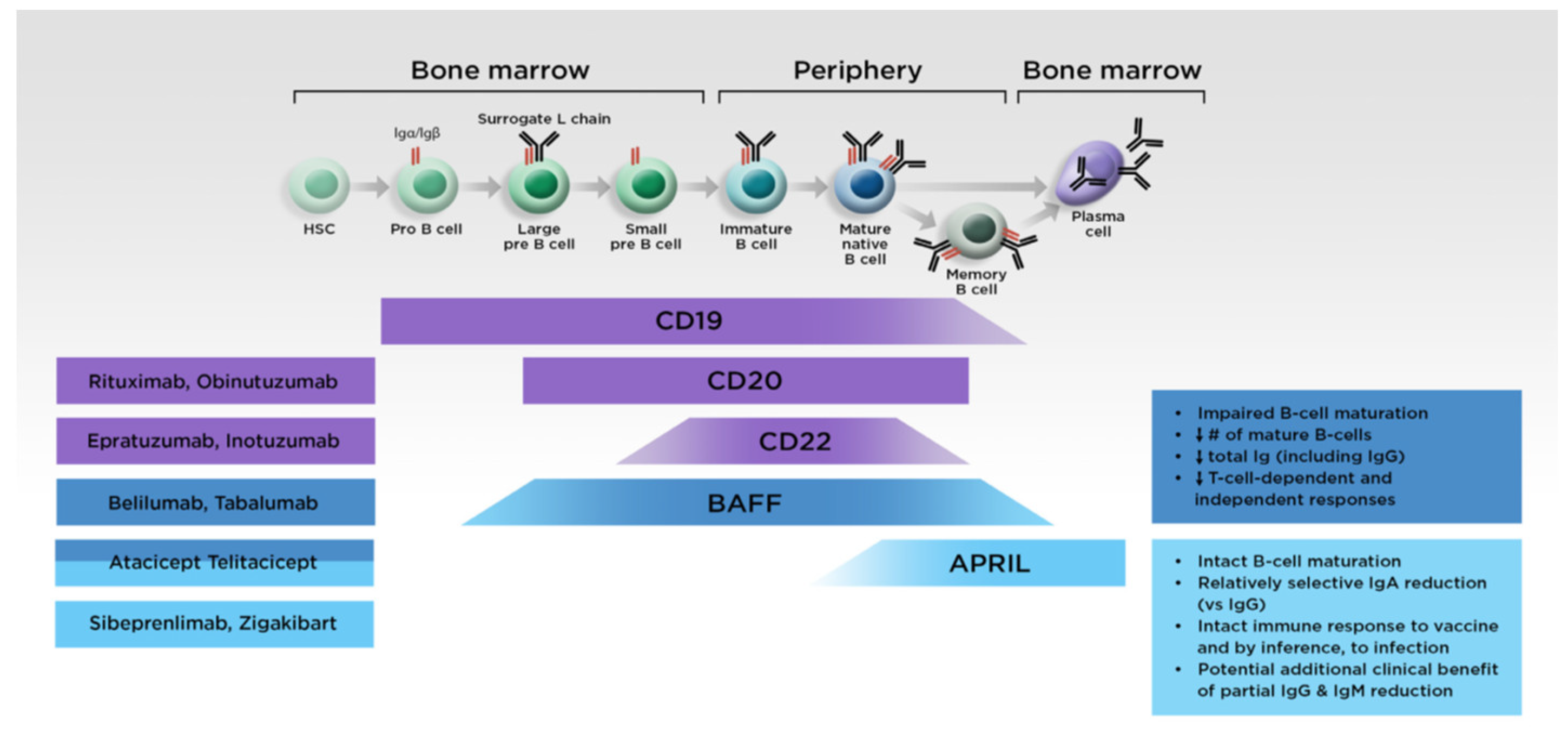
5. Treatment of IgAN and Development of Anti-APRIL Therapies
5.1. Symptom Reduction and Supportive Care
5.2. Targeted Treatments
5.2.1. Anti-APRIL Monoclonal Antibodies
5.2.2. Fusion Protein Antagonists Targeting Both APRIL and BAFF
6. Future Directions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hahne, M.; Kataoka, T.; Schroter, M.; Hofmann, K.; Irmler, M.; Bodmer, J.L.; Schneider, P.; Bornand, T.; Holler, N.; French, L.E.; et al. APRIL, a new ligand of the tumor necrosis factor family, stimulates tumor cell growth. J. Exp. Med. 1998, 188, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Wallweber, H.J.; Compaan, D.M.; Starovasnik, M.A.; Hymowitz, S.G. The crystal structure of a proliferation-inducing ligand, APRIL. J. Mol. Biol. 2004, 343, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Medema, J.P.; Planelles-Carazo, L.; Hardenberg, G.; Hahne, M. The uncertain glory of APRIL. Cell Death Differ. 2003, 10, 1121–1125. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.A.; Mackay, F. The BAFF-APRIL system in cancer. Cancers 2023, 15, 1791. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.Q.; Li, M.; Zhang, H.; Low, H.Q.; Wei, X.; Wang, J.Q.; Sun, L.D.; Sim, K.S.; Li, Y.; Foo, J.N.; et al. A genome-wide association study in Han Chinese identifies multiple susceptibility loci for IgA nephropathy. Nat. Genet. 2011, 44, 178–182. [Google Scholar] [CrossRef]
- Habas, E.; Ali, E.; Farfar, K.; Errayes, M.; Alfitori, J.; Habas, E.; Ghazouani, H.; Akbar, R.; Khan, F.; Al Dab, A.; et al. IgA nephropathy pathogenesis and therapy: Review & updates. Medicine 2022, 101, e31219. [Google Scholar] [CrossRef]
- Baert, L.; Manfroi, B.; Casez, O.; Sturm, N.; Huard, B. The role of APRIL—A PRoliferation Inducing Ligand—In autoimmune diseases and expectations from its targeting. J. Autoimmun. 2018, 95, 179–190. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Raab-Traub, N.; Casali, P.; Cerutti, A. EBV-encoded latent membrane protein 1 cooperates with BAFF/BLyS and APRIL to induce T cell-independent Ig heavy chain class switching. J. Immunol. 2003, 171, 5215–5224. [Google Scholar] [CrossRef]
- Moisini, I.; Davidson, A. BAFF: A local and systemic target in autoimmune diseases. Clin. Exp. Immunol. 2009, 158, 155–163. [Google Scholar] [CrossRef]
- Schweighoffer, E.; Tybulewicz, V.L. BAFF signaling in health and disease. Curr. Opin. Immunol. 2021, 71, 124–131. [Google Scholar] [CrossRef]
- Zhai, Y.L.; Zhu, L.; Shi, S.F.; Liu, L.J.; Lv, J.C.; Zhang, H. Increased APRIL expression induces IgA1 aberrant glycosylation in IgA nephropathy. Medicine 2016, 95, e3099. [Google Scholar] [CrossRef]
- Makita, Y.; Suzuki, H.; Kano, T.; Takahata, A.; Julian, B.A.; Novak, J.; Suzuki, Y. TLR9 activation induces aberrant IgA glycosylation via APRIL- and IL-6-mediated pathways in IgA nephropathy. Kidney Int. 2020, 97, 340–349. [Google Scholar] [CrossRef]
- Dillon, S.R.; Gross, J.A.; Ansell, S.M.; Novak, A.J. An APRIL to remember: Novel TNF ligands as therapeutic targets. Nat. Rev. Drug Discov. 2006, 5, 235–246. [Google Scholar] [CrossRef]
- Matthes, T.; Dunand-Sauthier, I.; Santiago-Raber, M.L.; Krause, K.H.; Donze, O.; Passweg, J.; McKee, T.; Huard, B. Production of the plasma-cell survival factor a proliferation-inducing ligand (APRIL) peaks in myeloid precursor cells from human bone marrow. Blood 2011, 118, 1838–1844. [Google Scholar] [CrossRef]
- Samy, E.; Wax, S.; Huard, B.; Hess, H.; Schneider, P. Targeting BAFF and APRIL in systemic lupus erythematosus and other antibody-associated diseases. Int. Rev. Immunol. 2017, 36, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Fraga, M.; Fernandez, R.; Albar, J.P.; Hahne, M. Biologically active APRIL is secreted following intracellular processing in the Golgi apparatus by furin convertase. EMBO Rep. 2001, 2, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Treml, J.F.; Hao, Y.; Stadanlick, J.E.; Cancro, M.P. The BLyS family: Toward a molecular understanding of B cell homeostasis. Cell Biochem. Biophys. 2009, 53, 1–16. [Google Scholar] [CrossRef]
- Dostert, C.; Grusdat, M.; Letellier, E.; Brenner, D. The TNF family of ligands and receptors: Communication modules in the immune system and beyond. Physiol. Rev. 2019, 99, 115–160. [Google Scholar] [CrossRef] [PubMed]
- Belnoue, E.; Pihlgren, M.; McGaha, T.L.; Tougne, C.; Rochat, A.F.; Bossen, C.; Schneider, P.; Huard, B.; Lambert, P.H.; Siegrist, C.A. APRIL is critical for plasmablast survival in the bone marrow and poorly expressed by early-life bone marrow stromal cells. Blood 2008, 111, 2755–2764. [Google Scholar] [CrossRef]
- Litinskiy, M.B.; Nardelli, B.; Hilbert, D.M.; He, B.; Schaffer, A.; Casali, P.; Cerutti, A. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nat. Immunol. 2002, 3, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Park, S.R.; Kim, P.H.; Lee, K.S.; Lee, S.H.; Seo, G.Y.; Yoo, Y.C.; Lee, J.; Casali, P. APRIL stimulates NF-kappaB-mediated HoxC4 induction for AID expression in mouse B cells. Cytokine 2013, 61, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, J.; Zhang, Y.-M.; Zhang, X.-M.; Tao, J. Effect of TACI Signaling on Humoral Immunity and Autoimmune Diseases. J. Immunol. Res. 2015, 2015, 247426. [Google Scholar] [CrossRef]
- Myette, J.R.; Kano, T.; Suzuki, H.; Sloan, S.E.; Szretter, K.J.; Ramakrishnan, B.; Adari, H.; Deotale, K.D.; Engler, F.; Shriver, Z.; et al. A proliferation inducing ligand (APRIL) targeted antibody is a safe and effective treatment of murine IgA nephropathy. Kidney Int. 2019, 96, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Schiemann, B.; Gommerman, J.L.; Vora, K.; Cachero, T.G.; Shulga-Morskaya, S.; Dobles, M.; Frew, E.; Scott, M.L. An essential role for BAFF in the normal development of B cells through a BCMA-independent pathway. Science 2001, 293, 2111–2114. [Google Scholar] [CrossRef]
- Varfolomeev, E.; Kischkel, F.; Martin, F.; Seshasayee, D.; Wang, H.; Lawrence, D.; Olsson, C.; Tom, L.; Erickson, S.; French, D.; et al. APRIL-deficient mice have normal immune system development. Mol. Cell Biol. 2004, 24, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.V.; Lopez-Fraga, M.; Elustondo, F.A.; Carvalho-Pinto, C.E.; Rodriguez, D.; Gomez-Caro, R.; De Jong, J.; Martinez, A.C.; Medema, J.P.; Hahne, M. APRIL modulates B and T cell immunity. J. Clin. Investig. 2002, 109, 1587–1598. [Google Scholar] [CrossRef]
- Schrezenmeier, E.; Jayne, D.; Dorner, T. Targeting B cells and plasma cells in glomerular diseases: Translational perspectives. J. Am. Soc. Nephrol. 2018, 29, 741–758. [Google Scholar] [CrossRef]
- Castigli, E.; Wilson, S.A.; Scott, S.; Dedeoglu, F.; Xu, S.; Lam, K.P.; Bram, R.J.; Jabara, H.; Geha, R.S. TACI and BAFF-R mediate isotype switching in B cells. J. Exp. Med. 2005, 201, 35–39. [Google Scholar] [CrossRef]
- Reijmers, R.M.; Groen, R.W.; Kuil, A.; Weijer, K.; Kimberley, F.C.; Medema, J.P.; van Kuppevelt, T.H.; Li, J.P.; Spaargaren, M.; Pals, S.T. Disruption of heparan sulfate proteoglycan conformation perturbs B-cell maturation and APRIL-mediated plasma cell survival. Blood 2011, 117, 6162–6171. [Google Scholar] [CrossRef]
- Barone, F.; Patel, P.; Sanderson, J.D.; Spencer, J. Gut-associated lymphoid tissue contains the molecular machinery to support T-cell-dependent and T-cell-independent class switch recombination. Mucosal Immunol. 2009, 2, 495–503. [Google Scholar] [CrossRef]
- Cerutti, A. The regulation of IgA class switching. Nat. Rev. Immunol. 2008, 8, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Gorbacheva, V.; Ayasoufi, K.; Fan, R.; Baldwin, W.M., 3rd; Valujskikh, A. B cell activating factor (BAFF) and a proliferation inducing ligand (APRIL) mediate CD40-independent help by memory CD4 T cells. Am. J. Transpl. 2015, 15, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Castigli, E.; Scott, S.; Dedeoglu, F.; Bryce, P.; Jabara, H.; Bhan, A.K.; Mizoguchi, E.; Geha, R.S. Impaired IgA class switching in APRIL-deficient mice. Proc. Natl. Acad. Sci. USA 2004, 101, 3903–3908. [Google Scholar] [CrossRef] [PubMed]
- Vincent, F.B.; Saulep-Easton, D.; Figgett, W.A.; Fairfax, K.A.; Mackay, F. The BAFF/APRIL system: Emerging functions beyond B cell biology and autoimmunity. Cytokine Growth Factor. Rev. 2013, 24, 203–215. [Google Scholar] [CrossRef]
- Knight, A.K.; Radigan, L.; Marron, T.; Langs, A.; Zhang, L.; Cunningham-Rundles, C. High serum levels of BAFF, APRIL, and TACI in common variable immunodeficiency. Clin. Immunol. 2007, 124, 182–189. [Google Scholar] [CrossRef]
- Kim, J.S.; Kim, S.J.; Cheong, J.W.; Kim, Y.; Hwang, D.Y.; Yoon, S.; Jang, J.; Hyun, S.Y.; Min, Y.H. Clinical significance of B cell-activating factor (BAFF) and a proliferation-inducing ligand (APRIL) in acute graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Korean J. Hematol. 2011, 46, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Camarena, D.C.; Ortiz-Lazareno, P.C.; Cruz, A.; Oregon-Romero, E.; Machado-Contreras, J.R.; Munoz-Valle, J.F.; Orozco-Lopez, M.; Marin-Rosales, M.; Palafox-Sanchez, C.A. Association of BAFF, APRIL serum levels, BAFF-R, TACI and BCMA expression on peripheral B-cell subsets with clinical manifestations in systemic lupus erythematosus. Lupus 2016, 25, 582–592. [Google Scholar] [CrossRef]
- Moura, R.A.; Cascao, R.; Perpetuo, I.; Canhao, H.; Vieira-Sousa, E.; Mourao, A.F.; Rodrigues, A.M.; Polido-Pereira, J.; Queiroz, M.V.; Rosario, H.S.; et al. Cytokine pattern in very early rheumatoid arthritis favours B-cell activation and survival. Rheumatology 2011, 50, 278–282. [Google Scholar] [CrossRef]
- Dechkhajorn, W.; Benjathummarak, S.; Glaharn, S.; Chaisri, U.; Viriyavejakul, P.; Maneerat, Y. The activation of BAFF/APRIL system in spleen and lymph nodes of Plasmodium falciparum infected patients. Sci. Rep. 2020, 10, 3865. [Google Scholar] [CrossRef] [PubMed]
- Gyllemark, P.; Forsberg, P.; Ernerudh, J.; Henningsson, A.J. Intrathecal Th17- and B cell-associated cytokine and chemokine responses in relation to clinical outcome in Lyme neuroborreliosis: A large retrospective study. J. Neuroinflammation 2017, 14, 27. [Google Scholar] [CrossRef]
- Alturaiki, W.; Mubarak, A.; Mir, S.A.; Afridi, A.; Premanathan, M.; Mickymaray, S.; Vijayakumar, R.; Alsagaby, S.A.; Almalki, S.G.; Alghofaili, F.; et al. Plasma levels of BAFF and APRIL are elevated in patients with asthma in Saudi Arabia. Saudi J. Biol. Sci. 2021, 28, 7455–7459. [Google Scholar] [CrossRef]
- Baert, L.; Ahmed, M.C.; Manfroi, B.; Huard, B. The number 13 of the family: A proliferation inducing ligand. Curr. Opin. Immunol. 2021, 71, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Chu, V.T.; Enghard, P.; Schurer, S.; Steinhauser, G.; Rudolph, B.; Riemekasten, G.; Berek, C. Systemic activation of the immune system induces aberrant BAFF and APRIL expression in B cells in patients with systemic lupus erythematosus. Arthritis Rheum. 2009, 60, 2083–2093. [Google Scholar] [CrossRef] [PubMed]
- Chu, V.T.; Enghard, P.; Riemekasten, G.; Berek, C. In vitro and in vivo activation induces BAFF and APRIL expression in B cells. J. Immunol. 2007, 179, 5947–5957. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, J.; Wang, L.; Zhou, W.; Zhang, Z. The role of a proliferation-inducing ligand (APRIL) in the pathogenesis of rheumatoid arthritis. Scand. J. Rheumatol. 2014, 43, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Gabay, C.; Krenn, V.; Bosshard, C.; Seemayer, C.A.; Chizzolini, C.; Huard, B. Synovial tissues concentrate secreted APRIL. Arthritis Res. Ther. 2009, 11, R144. [Google Scholar] [CrossRef]
- Pattrapornpisut, P.; Avila-Casado, C.; Reich, H.N. IgA nephropathy: Core curriculum 2021. Am. J. Kidney Dis. 2021, 78, 429–441. [Google Scholar] [CrossRef]
- Willey, C.J.; Coppo, R.; Schaefer, F.; Mizerska-Wasiak, M.; Mathur, M.; Schultz, M.J. The incidence and prevalence of IgA nephropathy in Europe. Nephrol. Dial. Transpl. 2023, 38, 2340–2349. [Google Scholar] [CrossRef] [PubMed]
- Kwon, C.S.; Daniele, P.; Forsythe, A.; Ngai, C. A systematic literature review of the epidemiology, health-related quality of life impact, and economic burden of immunoglobulin A nephropathy. J. Health Econ. Outcomes Res. 2021, 8, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, O.; Du, F.; Tang, Z.; Pareja, K.; Bhattacharjee, S. POS-304 a targeted literature review of prevalence and treatment patterns of iga nephropathy in mainland China, Taiwan, and South Korea. Kidney Int. Rep. 2022, 7, S136. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, H. The genetics of IgA nephropathy: An overview from China. Kidney Dis. 2015, 1, 27–32. [Google Scholar] [CrossRef]
- Yeo, S.C.; Goh, S.M.; Barratt, J. Is immunoglobulin A nephropathy different in different ethnic populations? Nephrology 2019, 24, 885–895. [Google Scholar] [CrossRef]
- Suzuki, Y.; Monteiro, R.C.; Coppo, R.; Suzuki, H. The phenotypic difference of IgA nephropathy and its race/gender-dependent molecular mechanisms. Kidney360 2021, 2, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Cheng, T.; Liu, C.; Zhu, T.; Guo, C.; Li, S.; Rao, X.; Li, J. IgA nephropathy: Current understanding and perspectives on pathogenesis and targeted treatment. Diagnostics 2023, 13, 303. [Google Scholar] [CrossRef]
- Pitcher, D.; Braddon, F.; Hendry, B.; Mercer, A.; Osmaston, K.; Saleem, M.A.; Steenkamp, R.; Wong, K.; Turner, A.N.; Wang, K.; et al. Long-term outcomes in IgA nephropathy. Clin. J. Am. Soc. Nephrol. 2023, 18, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Gentile, M.; Sanchez-Russo, L.; Riella, L.V.; Verlato, A.; Manrique, J.; Granata, S.; Fiaccadori, E.; Pesce, F.; Zaza, G.; Cravedi, P. Immune abnormalities in IgA nephropathy. Clin. Kidney J. 2023, 16, 1059–1070. [Google Scholar] [CrossRef]
- Selvaskandan, H.; Barratt, J.; Cheung, C.K. Immunological drivers of IgA nephropathy: Exploring the mucosa-kidney link. Int. J. Immunogenet. 2022, 49, 8–21. [Google Scholar] [CrossRef]
- Luvizotto, M.J.; Menezes-Silva, L.; Woronik, V.; Monteiro, R.C.; Camara, N.O.S. Gut-kidney axis in IgA nephropathy: Role on mesangial cell metabolism and inflammation. Front. Cell Dev. Biol. 2022, 10, 993716. [Google Scholar] [CrossRef] [PubMed]
- Nihei, Y.; Suzuki, H.; Suzuki, Y. Current understanding of IgA antibodies in the pathogenesis of IgA nephropathy. Front. Immunol. 2023, 14, 1165394. [Google Scholar] [CrossRef]
- Ohyama, Y.; Yamaguchi, H.; Ogata, S.; Chiurlia, S.; Cox, S.N.; Kouri, N.M.; Stangou, M.J.; Nakajima, K.; Hayashi, H.; Inaguma, D.; et al. Racial heterogeneity of IgA1 hinge-region O-glycoforms in patients with IgA nephropathy. iScience 2022, 25, 105223. [Google Scholar] [CrossRef]
- Martin-Penagos, L.; Fernandez-Fresnedo, G.; Benito-Hernandez, A.; Mazon, J.; de Cos, M.; Oviedo, M.V.; San Segundo, D.; Lopez-Hoyos, M.; Gomez-Roman, J.; Ruiz, J.C.; et al. Measurement of galactosyl-deficient IgA1 by the monoclonal antibody KM55 contributes to predicting patients with IgA nephropathy with high risk of long-term progression. Nefrología (Engl. Ed.) 2021, 41, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Hou, P.; Lv, J.; Moldoveanu, Z.; Li, Y.; Kiryluk, K.; Gharavi, A.G.; Novak, J.; Zhang, H. The level of galactose-deficient IgA1 in the sera of patients with IgA nephropathy is associated with disease progression. Kidney Int. 2012, 82, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Coppo, R. The gut-kidney axis in IgA nephropathy: Role of microbiota and diet on genetic predisposition. Pediatr. Nephrol. 2018, 33, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Yeo, S.C.; Cheung, C.K.; Barratt, J. New insights into the pathogenesis of IgA nephropathy. Pediatr. Nephrol. 2018, 33, 763–777. [Google Scholar] [CrossRef]
- Haniuda, K.; Gommerman, J.L.; Reich, H.N. The microbiome and IgA nephropathy. Semin. Immunopathol. 2021, 43, 649–656. [Google Scholar] [CrossRef]
- Tang, Y.; Xiao, Y.; He, H.; Zhu, Y.; Sun, W.; Hu, P.; Xu, X.; Liu, Z.; Yan, Z.; Wei, M. Aberrant gut microbiome contributes to barrier dysfunction, inflammation and local immune responses in IgA nephropathy. Kidney Blood Press. Res. 2023, 48, 261–276. [Google Scholar] [CrossRef]
- Hardenberg, G.; van Bostelen, L.; Hahne, M.; Medema, J.P. Thymus-independent class switch recombination is affected by APRIL. Immunol. Cell Biol. 2008, 86, 530–534. [Google Scholar] [CrossRef]
- Mora, J.R.; von Andrian, U.H. Differentiation and homing of IgA-secreting cells. Mucosal Immunol. 2008, 1, 96–109. [Google Scholar] [CrossRef]
- Han, S.S.; Yang, S.H.; Choi, M.; Kim, H.R.; Kim, K.; Lee, S.; Moon, K.C.; Kim, J.Y.; Lee, H.; Lee, J.P.; et al. The role of TNF superfamily member 13 in the progression of IgA nephropathy. J. Am. Soc. Nephrol. 2016, 27, 3430–3439. [Google Scholar] [CrossRef]
- Apeland, T.; Ushakova, A.; Mansoor, M.A.; Furriol, J.; Jonsson, G.; Marti, H.P. Association of redox and inflammation-related biomarkers with prognosis in IgA nephropathy: A prospective observational study. Free Radic. Biol. Med. 2022, 188, 62–70. [Google Scholar] [CrossRef]
- Currie, E.G.; Coburn, B.; Porfilio, E.A.; Lam, P.; Rojas, O.L.; Novak, J.; Yang, S.; Chowdhury, R.B.; Ward, L.A.; Wang, P.W.; et al. Immunoglobulin A nephropathy is characterized by anticommensal humoral immune responses. JCI Insight 2022, 7, e141289. [Google Scholar] [CrossRef]
- Martin-Penagos, L.; Benito-Hernandez, A.; San Segundo, D.; Sango, C.; Azueta, A.; Gomez-Roman, J.; Fernandez-Fresnedo, G.; Lopez-Hoyos, M.; Ruiz, J.C.; Rodrigo, E. A proliferation-inducing ligand increase precedes IgA nephropathy recurrence in kidney transplant recipients. Clin. Transpl. 2019, 33, e13502. [Google Scholar] [CrossRef]
- Liang, Y.; Zeng, Q.; Wang, X.H.; Yan, L.; Yu, R.H. Mechanism of yiqi yangying heluo formula in the treatment of IgA nephropathy by affecting Gd-IgA1 based on BAFF molecular level and T lymphocyte immunity. Biomed. Res. Int. 2023, 2023, 5124034. [Google Scholar] [CrossRef]
- Sallustio, F.; Curci, C.; Chaoul, N.; Fonto, G.; Lauriero, G.; Picerno, A.; Divella, C.; Di Leo, V.; De Angelis, M.; Ben Mkaddem, S.; et al. High levels of gut-homing immunoglobulin A+ B lymphocytes support the pathogenic role of intestinal mucosal hyperresponsiveness in immunoglobulin A nephropathy patients. Nephrol. Dial. Transpl. 2021, 36, 452–464. [Google Scholar] [CrossRef]
- Maixnerova, D.; El Mehdi, D.; Rizk, D.V.; Zhang, H.; Tesar, V. New treatment strategies for IgA nephropathy: Targeting plasma cells as the main source of pathogenic antibodies. J. Clin. Med. 2022, 11, 2810. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.; Xu, D.; Fang, J.; Van Vollenhoven, R.; Zhang, F. OPO137: Efficacy and safety of telitacicept, a novel BLyS/APRIL dual inhibitor, in patients with systemic lupus erythematosus: A phase 3, randomized, placebo-controlled 52-week study. Ann. Rheum. Dis. 2023, 82, 90–91. [Google Scholar]
- Kano, T.; Suzuki, H.; Makita, Y.; Nihei, Y.; Fukao, Y.; Nakayama, M.; Lee, M.; Kato, R.; Aoki, R.; Yamada, K.; et al. Mucosal immune system dysregulation in the pathogenesis of IgA nephropathy. Biomedicines 2022, 10, 3027. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, H.; Goto, S.; Takahashi, N.; Tsuchida, M.; Watanabe, H.; Yamamoto, S.; Kaneko, Y.; Higashi, K.; Mori, H.; Nakamura, Y.; et al. Aberrant mucosal immunoreaction to tonsillar microbiota in immunoglobulin A nephropathy. Nephrol. Dial. Transpl. 2021, 36, 75–86. [Google Scholar] [CrossRef]
- Nagasawa, Y.; Misaki, T.; Ito, S.; Naka, S.; Wato, K.; Nomura, R.; Matsumoto-Nakano, M.; Nakano, K. Title IgA nephropathy and oral bacterial species related to dental caries and periodontitis. Int. J. Mol. Sci. 2022, 23, 725. [Google Scholar] [CrossRef]
- Horie, A.; Hiki, Y.; Odani, H.; Yasuda, Y.; Takahashi, M.; Kato, M.; Iwase, H.; Kobayashi, Y.; Nakashima, I.; Maeda, K. IgA1 molecules produced by tonsillar lymphocytes are under-O-glycosylated in IgA nephropathy. Am. J. Kidney Dis. 2003, 42, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Muto, M.; Manfroi, B.; Suzuki, H.; Joh, K.; Nagai, M.; Wakai, S.; Righini, C.; Maiguma, M.; Izui, S.; Tomino, Y.; et al. Toll-like receptor 9 stimulation induces aberrant expression of a proliferation-inducing ligand by tonsillar germinal center B cells in IgA nephropathy. J. Am. Soc. Nephrol. 2017, 28, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Wang, L.N.; Jiang, Y.; Yao, L.; Dong, L.P.; Li, Z.L.; Li, X.L. Tonsillectomy for IgA nephropathy: A meta-analysis. Am. J. Kidney Dis. 2015, 65, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Hotta, O.; Miyazaki, M.; Furuta, T.; Tomioka, S.; Chiba, S.; Horigome, I.; Abe, K.; Taguma, Y. Tonsillectomy and steroid pulse therapy significantly impact on clinical remission in patients with IgA nephropathy. Am. J. Kidney Dis. 2001, 38, 736–743. [Google Scholar] [CrossRef]
- Moriyama, T.; Karasawa, K.; Miyabe, Y.; Akiyama, K.; Iwabuchi, Y.; Ogura, S.; Takabe, T.; Sugiura, N.; Seki, M.; Hanafusa, N.; et al. Long-term beneficial effects of tonsillectomy on patients with immunoglobulin A nephropathy. Kidney360 2020, 1, 1270–1283. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wan, Q.; Lan, Z.; Xia, M.; Liu, H.; Chen, G.; He, L.; Wang, C.; Liu, H. Efficacy and indications of tonsillectomy in patients with IgA nephropathy: A retrospective study. PeerJ 2022, 10, e14481. [Google Scholar] [CrossRef]
- Feehally, J.; Coppo, R.; Troyanov, S.; Bellur, S.S.; Cattran, D.; Cook, T.; Roberts, I.S.; Verhave, J.C.; Camilla, R.; Vergano, L.; et al. Tonsillectomy in a European cohort of 1,147 patients with IgA nephropathy. Nephron 2016, 132, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Rovin, B.H.; Adler, S.G.; Barratt, J.; Bridoux, F.; Burdge, K.A.; Chan, T.M.; Cook, H.T.; Fervenza, F.C.; Gibson, K.L.; Glassock, R.J.; et al. Executive summary of the KDIGO 2021 Guideline for the Management of Glomerular Diseases. Kidney Int. 2021, 100, 753–779. [Google Scholar] [CrossRef] [PubMed]
- Saka, S.; Hirawa, N.; Oka, A.; Yatsu, K.; Hirukawa, T.; Yamamoto, R.; Matsusaka, T.; Imai, E.; Narita, I.; Endoh, M.; et al. Genome-wide association study of IgA nephropathy using 23,465 microsatellite markers in a Japanese population. J. Hum. Genet. 2015, 60, 573–580. [Google Scholar] [CrossRef][Green Version]
- Feehally, J.; Farrall, M.; Boland, A.; Gale, D.P.; Gut, I.; Heath, S.; Kumar, A.; Peden, J.F.; Maxwell, P.H.; Morris, D.L.; et al. HLA has strongest association with IgA nephropathy in genome-wide analysis. J. Am. Soc. Nephrol. 2010, 21, 1791–1797. [Google Scholar] [CrossRef]
- Kiryluk, K.; Li, Y.; Scolari, F.; Sanna-Cherchi, S.; Choi, M.; Verbitsky, M.; Fasel, D.; Lata, S.; Prakash, S.; Shapiro, S.; et al. Discovery of new risk loci for IgA nephropathy implicates genes involved in immunity against intestinal pathogens. Nat. Genet. 2014, 46, 1187–1196. [Google Scholar] [CrossRef]
- Tian, C.; Hromatka, B.S.; Kiefer, A.K.; Eriksson, N.; Noble, S.M.; Tung, J.Y.; Hinds, D.A. Genome-wide association and HLA region fine-mapping studies identify susceptibility loci for multiple common infections. Nat. Commun. 2017, 8, 599. [Google Scholar] [CrossRef]
- Osman, W.; Okada, Y.; Kamatani, Y.; Kubo, M.; Matsuda, K.; Nakamura, Y. Association of common variants in TNFRSF13B, TNFSF13, and ANXA3 with serum levels of non-albumin protein and immunoglobulin isotypes in Japanese. PLoS ONE 2012, 7, e32683. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.G.; Alvarez, M.; Suzuki, H.; Hirose, S.; Izui, S.; Tomino, Y.; Huard, B.; Suzuki, Y. Pathogenic role of a proliferation-inducing ligand (APRIL) in murine IgA nephropathy. PLoS ONE 2015, 10, e0137044. [Google Scholar] [CrossRef] [PubMed]
- Maixnerova, D.; Tesar, V. Emerging role of monoclonal antibodies in the treatment of IgA nephropathy. Expert. Opin. Biol. Ther. 2023, 23, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.C.; Toto, R.D.; Stefansson, B.V.; Jongs, N.; Chertow, G.M.; Greene, T.; Hou, F.F.; McMurray, J.J.V.; Pecoits-Filho, R.; Correa-Rotter, R.; et al. A pre-specified analysis of the DAPA-CKD trial demonstrates the effects of dapagliflozin on major adverse kidney events in patients with IgA nephropathy. Kidney Int. 2021, 100, 215–224. [Google Scholar] [CrossRef]
- Kim, D.; Wong, M.G. Corticosteroid therapy in IgA nephropathy: A friend or foe? Kidney Blood Press. Res. 2023, 48, 392–404. [Google Scholar] [CrossRef]
- Lv, J.; Zhang, H.; Wong, M.G.; Jardine, M.J.; Hladunewich, M.; Jha, V.; Monaghan, H.; Zhao, M.; Barbour, S.; Reich, H.; et al. Effect of oral methylprednisolone on clinical outcomes in patients with IgA nephropathy: The TESTING randomized clinical trial. JAMA 2017, 318, 432–442. [Google Scholar] [CrossRef]
- Lv, J.; Wong, M.G.; Hladunewich, M.A.; Jha, V.; Hooi, L.S.; Monaghan, H.; Zhao, M.; Barbour, S.; Jardine, M.J.; Reich, H.N.; et al. Effect of oral methylprednisolone on decline in kidney function or kidney failure in patients with IgA nephropathy: The TESTING randomized clinical trial. JAMA 2022, 327, 1888–1898. [Google Scholar] [CrossRef] [PubMed]
- Barratt, J.; Lafayette, R.A.; Rovin, B.H.; Fellstrom, B. Budesonide delayed-release capsules to reduce proteinuria in adults with primary immunoglobulin A nephropathy. Expert. Rev. Clin. Immunol. 2023, 19, 699–710. [Google Scholar] [CrossRef]
- Smerud, H.K.; Barany, P.; Lindstrom, K.; Fernstrom, A.; Sandell, A.; Pahlsson, P.; Fellstrom, B. New treatment for IgA nephropathy: Enteric budesonide targeted to the ileocecal region ameliorates proteinuria. Nephrol. Dial. Transpl. 2011, 26, 3237–3242. [Google Scholar] [CrossRef]
- Barratt, J.; Lafayette, R.; Kristensen, J.; Stone, A.; Cattran, D.; Floege, J.; Tesar, V.; Trimarchi, H.; Zhang, H.; Eren, N.; et al. Results from part A of the multi-center, double-blind, randomized, placebo-controlled NefIgArd trial, which evaluated targeted-release formulation of budesonide for the treatment of primary immunoglobulin A nephropathy. Kidney Int. 2023, 103, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.Y. Sparsentan: First approval. Drugs 2023, 83, 563–568. [Google Scholar] [CrossRef]
- Heerspink, H.J.L.; Radhakrishnan, J.; Alpers, C.E.; Barratt, J.; Bieler, S.; Diva, U.; Inrig, J.; Komers, R.; Mercer, A.; Noronha, I.L.; et al. Sparsentan in patients with IgA nephropathy: A prespecified interim analysis from a randomised, double-blind, active-controlled clinical trial. Lancet 2023, 401, 1584–1594. [Google Scholar] [CrossRef] [PubMed]
- Lafayette, R.A.; Canetta, P.A.; Rovin, B.H.; Appel, G.B.; Novak, J.; Nath, K.A.; Sethi, S.; Tumlin, J.A.; Mehta, K.; Hogan, M.; et al. A randomized, controlled trial of rituximab in IgA nephropathy with proteinuria and renal dysfunction. J. Am. Soc. Nephrol. 2017, 28, 1306–1313. [Google Scholar] [CrossRef]
- Hofmann, K.; Clauder, A.K.; Manz, R.A. Targeting B Cells and plasma cells in autoimmune diseases. Front. Immunol. 2018, 9, 835. [Google Scholar] [CrossRef]
- Mei, H.E.; Frolich, D.; Giesecke, C.; Loddenkemper, C.; Reiter, K.; Schmidt, S.; Feist, E.; Daridon, C.; Tony, H.P.; Radbruch, A.; et al. Steady-state generation of mucosal IgA+ plasmablasts is not abrogated by B-cell depletion therapy with rituximab. Blood 2010, 116, 5181–5190. [Google Scholar] [CrossRef]
- Matsuzaki, K.; Suzuki, Y.; Nakata, J.; Sakamoto, N.; Horikoshi, S.; Kawamura, T.; Matsuo, S.; Tomino, Y. Nationwide survey on current treatments for IgA nephropathy in Japan. Clin. Exp. Nephrol. 2013, 17, 827–833. [Google Scholar] [CrossRef]
- Reich, H.N.; Floege, J. How I treat IgA nephropathy. Clin. J. Am. Soc. Nephrol. 2022, 17, 1243–1246. [Google Scholar] [CrossRef]
- Suzuki, H.; Novak, J. IgA glycosylation and immune complex formation in IgAN. Semin. Immunopathol. 2021, 43, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Mathur, M.; Barratt, J.; Suzuki, Y.; Engler, F.; Pasetti, M.F.; Yarbrough, J.; Sloan, S.; Oldach, D. Safety, tolerability, pharmacokinetics, and pharmacodynamics of VIS649 (sibeprenlimab), an APRIL-neutralizing IgG(2) monoclonal antibody, in healthy volunteers. Kidney Int. Rep. 2022, 7, 993–1003. [Google Scholar] [CrossRef]
- Barratt, J.; Hour, B.; Kooienga, L.; Roy, S.; Schwartz, B.; Siddiqui, A.; Tolentino, J.; Iyer, S.P.; Stromatt, C.; Endsley, A.; et al. POS-109 Interim results of phase 1 and 2 trials to investigate the safety, tolerability, pharmacokinetics, pharmacodynamics, and clinical activity oF BION-1301 in patients with IgA nephropathy. Kidney Int. Rep. 2022, 7, S46. [Google Scholar] [CrossRef]
- Barratt, J.; Tumlin, J.; Suzuki, Y.; Kao, A.; Aydemir, A.; Pudota, K.; Jin, H.; Guhring, H.; Appel, G.; JANUS study investigators. Randomized phase II JANUS study of atacicept in patients with IgA nephropathy and persistent proteinuria. Kidney Int. Rep. 2022, 7, 1831–1841. [Google Scholar] [CrossRef]
- Lafayette, R.A.; Maes, B.; Lin, C.; Barbour, S.; Phoon, R.; Kim, S.G.; Tesar, V.; Floege, J.; Jha, V.; Barratt, J. #3848 ORIGIN trial: 24-wk primary analysis of a randomized, double-blind, placebo-controlled Ph2b study of atacicept in patients with IgAN. Nephrol. Dial. Transpl. 2023, 38 (Suppl. S1), i23. [Google Scholar] [CrossRef]
- Lv, J.; Liu, L.; Hao, C.; Li, G.; Fu, P.; Xing, G.; Zheng, H.; Chen, N.; Wang, C.; Luo, P.; et al. Randomized phase 2 trial of telitacicept in patients with IgA nephropathy with persistent proteinuria. Kidney Int. Rep. 2023, 8, 499–506. [Google Scholar] [CrossRef]
- Harrison, P.; Lickliter, J.; Dillon, S.; Manjarrez, K.; Smith, A.; Lessig, M.; Sanderson, R.; Chunyk, A.; Zayed, H.; Peng, S. PO.6.131 A randomized, double-blind, placebo controlled study of the safety, tolerability, pharmacokinetics, and pharmacodynamics of ALPN-303, a potent dual BAFF/APRIL inhibitor, in adult healthy volunteers. Lupus Sci. Med. 2022, 9 (Suppl. S2), A99–A100. [Google Scholar] [CrossRef]
- Kooienga, L.; Suzuki, Y.; Chacko, B.; Wong, M.G.; Barratt, J.; Oh, C.; Sahay, M.; Mathur, M.; Wang, X.; Yarbrough, J.; et al. Interim Analysis (IA) of a Global Phase 2 RCT of Sibeprenlimab (VIS649), an APRIL-Neutralizing Monoclonal Antibody, in IgA Nephropathy. Poster TH-PO991, Presented at ASN Kidney Week, Orlando, FL, USA, 3–6 November 2022. Available online: https://visterrainc.com/wp-content/uploads/11.3.22-Interim-Analysis-IA-of-a-Global-Phase-2-Randomized-Controlled-Trial-of-Sibeprenlimab-VIS649-an-APRIL-Neutralizing-Monoclonal-Antibody-in-IgA-Nephropathy.pdf (accessed on 2 June 2023).
- Chan, D.T.M.; Kanjanabuch, T.; Liew, M.; Mathur, M.; Yarbrough, J.; Wang, X.; Suzuki, Y. WCN23-0684 Interim biomarker analysis from a randomized, double-blind, placebo-controlled, phase 2 trial of sibeprenlimab (VIS649) in participants with immunoglobulin A nephropathy. Kidney Int. Rep. 2023, 8, S76–S77. [Google Scholar] [CrossRef]
- Mccafferty, K.; Follman, K.; Pasetti, M.; Schachter, A.; Mathur, M.; Ma, D.; Yarbrough, J.; Shriver, M.; Engler, F.; Oldach, D.; et al. #3347 COVID vaccine responses during sibeprenlimab treatment of IgA nephropathy (IgAN): An interim analysis. Nephrol. Dial. Transpl. 2023, 39 (Suppl. S1), i17. [Google Scholar] [CrossRef]
- Kaegi, C.; Steiner, U.C.; Wuest, B.; Crowley, C.; Boyman, O. Systematic review of safety and efficacy of atacicept in treating immune-mediated disorders. Front. Immunol. 2020, 11, 433. [Google Scholar] [CrossRef]
- Isenberg, D.A.; Lin, C.J.F. Atacicept-it’s not over until the wolf-lady sings (or maybe howls). Rheumatology 2023, 62, 1359–1361. [Google Scholar] [CrossRef] [PubMed]
- Kappos, L.; Hartung, H.P.; Freedman, M.S.; Boyko, A.; Radu, E.W.; Mikol, D.D.; Lamarine, M.; Hyvert, Y.; Freudensprung, U.; Plitz, T.; et al. Atacicept in multiple sclerosis (ATAMS): A randomised, placebo-controlled, double-blind, phase 2 trial. Lancet Neurol. 2014, 13, 353–363. [Google Scholar] [CrossRef]
- Dhillon, S. Telitacicept: First approval. Drugs 2021, 81, 1671–1675. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.S.; Lewis, K.E.; DeMonte, D.; Bhandari, J.G.; Garrett, L.B.; Kuijper, J.L.; Ardourel, D.; Wolfson, M.F.; Debrot, S.; Mudri, S.; et al. Povetacicept, an enhanced dual APRIL/BAFF antagonist that modulates B lymphocytes and pathogenic autoantibodies for the treatment of lupus and other B cell-related autoimmune diseases. Arthritis Rheumatol. 2023, 75, 1187–1202. [Google Scholar] [CrossRef] [PubMed]
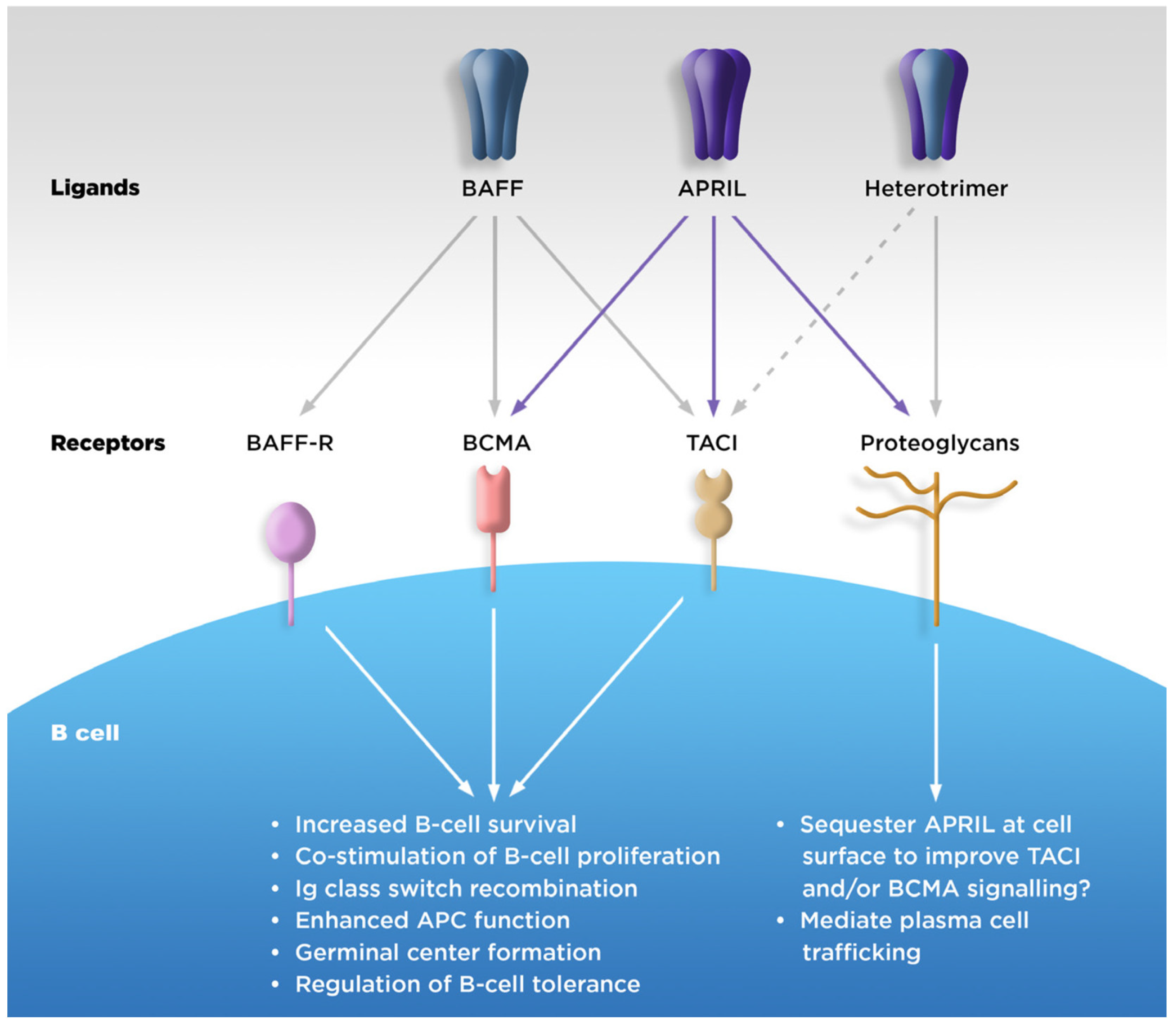

| Function | APRIL | BAFF | Receptors Mediating These Functions | |||
|---|---|---|---|---|---|---|
| TACI | BCMA | BAFF-R | Proteoglycans | |||
| B-cell survival and maturation | X | X | X | X | ||
| Plasma blast and plasma cell differentiation and survival | X | X | ||||
| Maintenance of B1 cell | X | X | ||||
| T-cell-independent antibody response | X | X | X | X | X | |
| Antibody class switch and recombination | X | X | ||||
| Plasma cell trafficking | X | X | ||||
| Potential Treatment (Sponsor) | Drug Type | IgAN Clinical Trials | Status |
|---|---|---|---|
| Sibeprenlimab (Otsuka Pharmaceutical Co., Ltd./Visterra Inc.) | Anti-APRIL monoclonal antibody | NCT03719443 | Phase I complete [110] |
| NCT04287985 | Phase II complete | ||
| NCT05248646 | Phase III recruiting | ||
| NCT05248659 | Phase II/III enrolling | ||
| Zigakibart (Chinook Therapeutics, Inc./Novartis) | Anti-APRIL monoclonal antibody | NCT05508204 | Phase I complete [111] |
| NCT03945318/NCT04684745 | Phase I/II ongoing [111] | ||
| NCT05852938 | Phase III enrolling | ||
| Atacicept (Vera Therapeutics/Merck KGaA) | TACI-IgG fusion protein | NCT02808429 | Phase II complete [112] |
| NCT04716231 | Phase II ongoing [113] Phase III recruiting | ||
| Telitacicept (Yantai Rongchang Pharmaceuticals, Ltd./RemeGen Co., Ltd.) | TACI receptor-IgG fusion protein | NCT04291781 | Phase II complete [114] |
| NCT04905212 | Phase II recruiting | ||
| NCT05596708 | Phase II/III planned | ||
| NCT05799287 | Phase III planned | ||
| Povetacicept (Alpine Immune Sciences, Inc.) | TACI-Ig fusion protein | NCT05034484 | Phase I complete [115] |
| NCT05732402 | Phase I recruiting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mathur, M.; Chan, T.M.; Oh, K.-H.; Kooienga, L.; Zhuo, M.; Pinto, C.S.; Chacko, B. A PRoliferation-Inducing Ligand (APRIL) in the Pathogenesis of Immunoglobulin A Nephropathy: A Review of the Evidence. J. Clin. Med. 2023, 12, 6927. https://doi.org/10.3390/jcm12216927
Mathur M, Chan TM, Oh K-H, Kooienga L, Zhuo M, Pinto CS, Chacko B. A PRoliferation-Inducing Ligand (APRIL) in the Pathogenesis of Immunoglobulin A Nephropathy: A Review of the Evidence. Journal of Clinical Medicine. 2023; 12(21):6927. https://doi.org/10.3390/jcm12216927
Chicago/Turabian StyleMathur, Mohit, Tak Mao Chan, Kook-Hwan Oh, Laura Kooienga, Min Zhuo, Cibele S. Pinto, and Bobby Chacko. 2023. "A PRoliferation-Inducing Ligand (APRIL) in the Pathogenesis of Immunoglobulin A Nephropathy: A Review of the Evidence" Journal of Clinical Medicine 12, no. 21: 6927. https://doi.org/10.3390/jcm12216927
APA StyleMathur, M., Chan, T. M., Oh, K.-H., Kooienga, L., Zhuo, M., Pinto, C. S., & Chacko, B. (2023). A PRoliferation-Inducing Ligand (APRIL) in the Pathogenesis of Immunoglobulin A Nephropathy: A Review of the Evidence. Journal of Clinical Medicine, 12(21), 6927. https://doi.org/10.3390/jcm12216927