Electrolyte Disturbances Related to Sodium and Potassium and Electroconvulsive Therapy: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Information Sources, Search Strategy, Selection Process
2.2. Data Collection Process, Data Items
2.3. Eligibility Criteria
2.4. Study Risk of Bias Assessment
2.5. Synthesis Method
3. Results
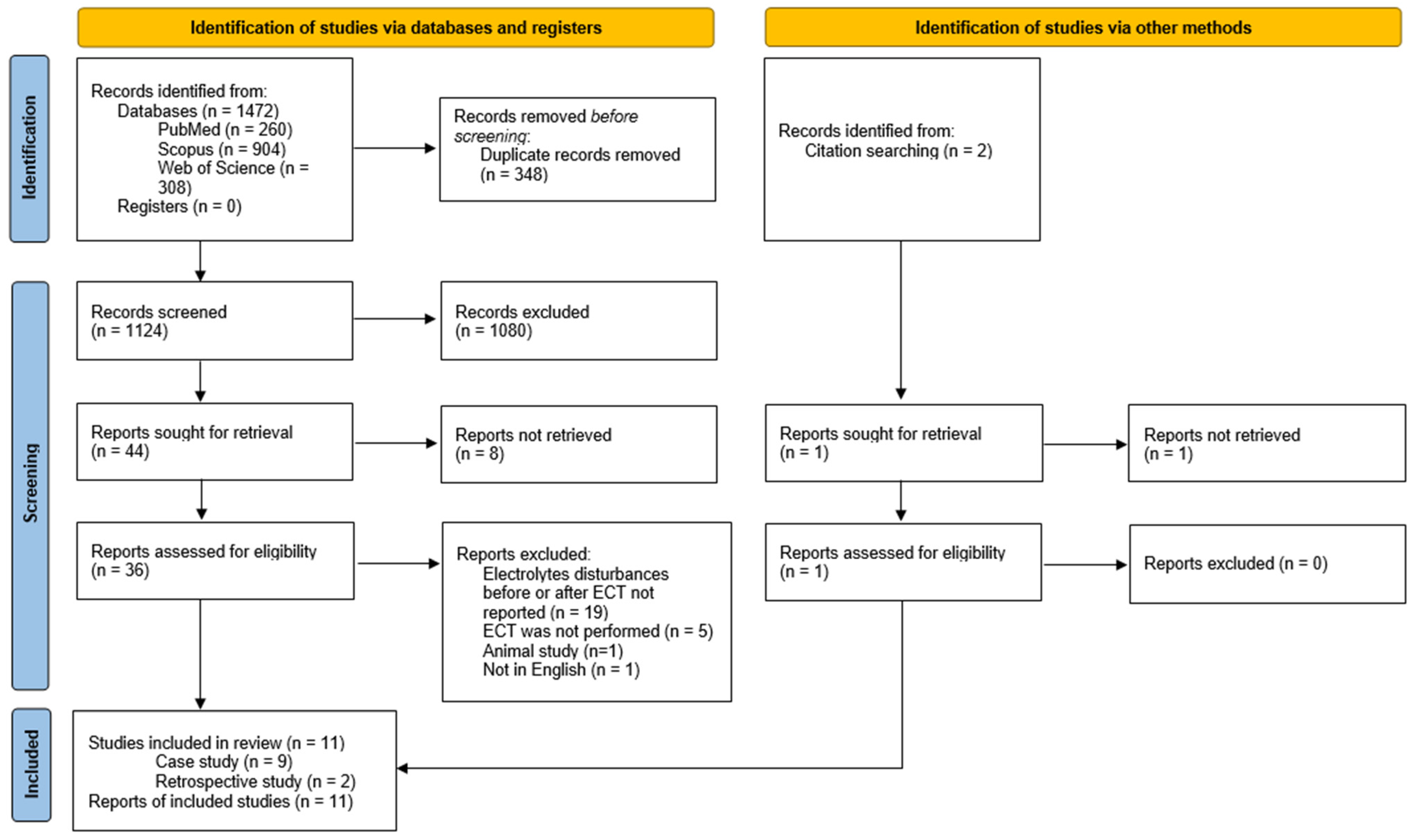
3.1. Study Selection and Characteristics
3.2. Risk of Bias in Studies
3.3. Results of Individual Studies and Syntheses
3.3.1. General Characteristic
3.3.2. ECT for Patients with Electrolytes Disturbances
Characteristic of Included Studies
Efficacy of Electroconvulsive Therapy for Patients with Electrolyte Disturbances
Safety of Electroconvulsive Therapy for Patients with Electrolytes Disturbances
3.3.3. Electrolytes Disturbances after ECT
Characteristic of Included Studies
Efficacy
Safety
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Pohl, H.R.; Wheeler, J.S.; Murray, H.E. Sodium and Potassium in Health and Disease. Met. Ions Life Sci. 2013, 13, 29–47. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Li, Z.; Jiang, L.; Wang, Y.; Xi, X. Risk Factors for Intensive Care Unit-Acquired Weakness: A Systematic Review and Meta-Analysis. Acta Neurol. Scand. 2018, 138, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Villar, S.; Poza-Hernández, P.; Freigang, S.; Zubizarreta-Ormazabal, I.; Paz-Martín, D.; Holl, E.; Pérez-Pardo, O.C.; Tovar-Doncel, M.S.; Wissa, S.M.; Cimadevilla-Calvo, B.; et al. Automatic Real-Time Analysis and Interpretation of Arterial Blood Gas Sample for Point-of-Care Testing: Clinical Validation. PLoS ONE 2021, 16, e0248264. [Google Scholar] [CrossRef]
- Ibrahim, S.L.; Alzubaidi, Z.F.; Al-Maamory, F.A.D. Electrolyte Disturbances in a Sample of Hospitalized Patients from Iraq. J. Med. Life 2022, 15, 1129–1135. [Google Scholar] [CrossRef]
- Stelmach, E.; Hołownia, O.; Słotwiński, M.; Gerhant, A.; Olajossy, M. Hiponatremia in the Practice of a Psychiatrist. Part 1: SIADH Syndrome and Drug-Induced Hyponatremia. Curr. Probl. Psychiatry 2017, 18, 120–137. [Google Scholar] [CrossRef]
- Gerhant, A.; Słotwiński, M.; Hołownia, O.; Stelmach, E.; Olajossy, M. Hiponatremia in the Practice of a Psychiatrist. Part 2: Psychogenic Polydipsia. Curr. Probl. Psychiatry 2017, 18, 138–152. [Google Scholar] [CrossRef]
- Li, Y.; Chen, X.; Shen, Z.; Wang, Y.; Hu, J.; Xu, J.; Shen, B.; Ding, X. Electrolyte and Acid-Base Disorders in Cancer Patients and Its Impact on Clinical Outcomes: Evidence from a Real-World Study in China. Ren. Fail. 2020, 42, 234–243. [Google Scholar] [CrossRef]
- Wang, S.-M.; Han, C.; Bahk, W.-M.; Lee, S.-J.; Patkar, A.A.; Masand, P.S.; Pae, C.-U. Addressing the Side Effects of Contemporary Antidepressant Drugs: A Comprehensive Review. Chonnam Med. J. 2018, 54, 101–112. [Google Scholar] [CrossRef]
- Mazhar, F.; Battini, V.; Pozzi, M.; Invernizzi, E.; Mosini, G.; Gringeri, M.; Capuano, A.; Scavone, C.; Radice, S.; Clementi, E.; et al. Hyponatremia Following Antipsychotic Treatment: In Silico Pharmacodynamics Analysis of Spontaneous Reports from the US Food and Drug Administration Adverse Event Reporting System Database and an Updated Systematic Review. Int. J. Neuropsychopharmacol. 2021, 24, 477–489. [Google Scholar] [CrossRef]
- Kellner, C.H.; Obbels, J.; Sienaert, P. When to Consider Electroconvulsive Therapy (ECT). Acta Psychiatr. Scand. 2020, 141, 304–315. [Google Scholar] [CrossRef]
- APA Committee on Electroconvulsive Therapy. The Practice of Electroconvulsive Therapy: Recommendations for Treatment, Training, and Privileging, 2nd ed.; American Psychiatric Association: Washington, DC, USA, 2001. [Google Scholar]
- Nardone, R.; Brigo, F.; Trinka, E. Acute Symptomatic Seizures Caused by Electrolyte Disturbances. J. Clin. Neurol. 2016, 12, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Halawa, I.; Andersson, T.; Tomson, T. Hyponatremia and Risk of Seizures: A Retrospective Cross-Sectional Study. Epilepsia 2011, 52, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Littmann, L.; Gibbs, M.A. Electrocardiographic Manifestations of Severe Hyperkalemia. J. Electrocardiol. 2018, 51, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Hartnett, S.; Rex, S.; Sienaert, P. Asystole During Electroconvulsive Therapy: Does Electrode Placement Matter? A Systematic Review. J. ECT 2023, 39, 3–9. [Google Scholar] [CrossRef]
- Nazemroaya, B.; Ghosouri, A.; Honarmand, A.; Hashemi, S.T. Comparison of Hemodynamic Changes and Serum Potassium Levels in the Use of Succinylcholine and Cisatracurium in Electroconvulsive Therapy. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2021, 26, 106. [Google Scholar] [CrossRef]
- Aggarwal, R.; Katyal, S.; Singh, A.; Kaul, T.K.; Grewal, A.; Narula, N. Changes in Serum Potassium after Electro Convulsive Therapy (ECT). J. Anaesthesiol. Clin. Pharmacol. 2002, 18, 35–39. [Google Scholar]
- Aaron, O.I.; Faponle, A.F.; Bolaji, B.O.; Mosaku, S.K.; Adenekan, A.T.; Oginni, O.A. Modified Electroconvulsive Therapy in a Resource-Challenged Setting: Comparison of Two Doses (0.5 Mg/Kg and 1 Mg/Kg) of Suxamethonium Chloride. Saudi J. Anaesth. 2020, 14, 487–492. [Google Scholar] [CrossRef]
- Bali, I.M.; Dundee, J.W.; Doggart, J.R. The Source of Increased Plasma Potassium Following Succinylcholine. Anesth. Analg. 1975, 54, 680–686. [Google Scholar] [CrossRef]
- McCleane, G.J.; Howe, J.P. Electroconvulsive Therapy and Serum Potassium. Ulster Med. J. 1989, 58, 172–174. [Google Scholar]
- Gaffar, S.; Eskander, J.P.; Beakley, B.D.; McClure, B.P.; Amenta, P.; Pierre, N. A Case of Central Diabetes Insipidus after Ketamine Infusion during an External to Internal Carotid Artery Bypass. J. Clin. Anesth. 2017, 36, 72–75. [Google Scholar] [CrossRef]
- Swenson, K.L.; Badre, S.E.; Morsette, D.J.; Sladek, C.D. N-Methyl-D-Aspartic Acid Stimulation of Vasopressin Release: Role in Osmotic Regulation and Modulation by Gonadal Steroids. J. Neuroendocrinol. 1998, 10, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Hatab, S.Z.; Singh, A.; Felner, E.I.; Kamat, P. Transient Central Diabetes Insipidus Induced by Ketamine Infusion. Ann. Pharmacother. 2014, 48, 1642–1645. [Google Scholar] [CrossRef] [PubMed]
- García-Soler, P.; Amores-Torres, M.; Sanchiz-Cárdenas, S.; González-Gómez, J.M.; Dayaldasani, A.; Milano-Manso, G. Thiopental and False Hypernatremia: Check Your Analyzer. Med. Intensiv. 2017, 41, 573–574. [Google Scholar] [CrossRef] [PubMed]
- Feyen, B.F.E.; Coenen, D.; Jorens, P.G.; Wouters, K.; Maas, A.I.R.; Van Hoof, V.; Verbrugghe, W. Falsely Elevated Sodium Levels during Thiopental Treatment in the ICU: Technical Interference on a Laboratory Device with Important Clinical Relevance. Neurocrit. Care 2013, 18, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Prandota, J.; Iwanczak, F. Long Q-T Syndrome Precipitated by Atropine and Hypokalemia. Dev. Pharmacol. Ther. 1983, 6, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Gelmont, D.M.; Balmes, J.R.; Yee, A. Hypokalemia Induced by Inhaled Bronchodilators. Chest 1988, 94, 763–766. [Google Scholar] [CrossRef]
- Cairns, C.J.S.; Thomas, B.; Fletcher, S.; Parr, M.J.A.; Finfer, S.R. Life-Threatening Hyperkalaemia following Therapeutic Barbiturate Coma. Intensive Care Med. 2002, 28, 1357–1360. [Google Scholar] [CrossRef]
- Machata, A.M.; Gonano, C.; Bîrsan, T.; Zimpfer, M.; Spiss, C.K. Rare but Dangerous Adverse Effects of Propofol and Thiopental in Intensive Care. J. Trauma 2005, 58, 643–645. [Google Scholar] [CrossRef]
- Ng, S.Y.; Chin, K.J.; Kwek, T.K. Dyskalaemia Associated with Thiopentone Barbiturate Coma for Refractory Intracranial Hypertension: A Case Series. Intensive Care Med. 2011, 37, 1285–1289. [Google Scholar] [CrossRef]
- Weintraub, H.D.; Heisterkamp, D.V.; Cooperman, L.H. Changes in Plasma Potassium Concentration after Depolarizing Blockers in Anaesthetized Man. Br. J. Anaesth. 1969, 41, 1048–1052. [Google Scholar] [CrossRef]
- Yentis, S.M. Suxamethonium and Hyperkalaemia. Anaesth. Intensive Care 1990, 18, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Mazze, R.I.; Escue, H.M.; Houston, J.B. Hyperkalemia and Cardiovascular Collapse following Administration of Succinylcholine to the Traumatized Patient. Anesthesiology 1969, 31, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Gronert, G.A.; Theye, R.A. Pathophysiology of Hyperkalemia Induced by Succinylcholine. Anesthesiology 1975, 43, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 Explanation and Elaboration: Updated Guidance and Exemplars for Reporting Systematic Reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Moola, S.; Munn, Z.; Tufanarau, C.; Aromataris, E.; Sears, K.; Sfetcu, R.; Currie, M.; Qureshi, R.; Mattis, P.; Lisy, K.; et al. Chapter 7: Systematic Reviews of Etiology and Risk. In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020. [Google Scholar]
- Mashimo, K.; Sato, Y.; Yamauchi, T. Effective Electroconvulsive Therapy for Stupor in the High Risk Patient: A Report of Two Cases. Psychiatry Clin. Neurosci. 1996, 50, 129–131. [Google Scholar] [CrossRef]
- Koster, T.D.; Kooistra, W.E.; Tuinman, A.G. Near Miss with Succinylcholine for Electroconvulsive Therapy: A Case Report. Eur. J. Anaesthesiol. 2014, 31, 441–443. [Google Scholar] [CrossRef]
- Finlayson, A.J.; Vieweg, W.V.; Wilkey, W.D.; Cooper, A.J. Hyponatremic Seizure Following ECT. Can. J. Psychiatry. 1989, 34, 463–464. [Google Scholar] [CrossRef]
- Kern, M.; Singleton, A.; Brenes, G.; Arias, L.; Gligorovic, P. Successful and Rapid Response to Electroconvulsive Therapy of a Patient with Obsessive Compulsive Disorder and an Incidental Cyst. J. ECT 2020, 36, 2–3. [Google Scholar] [CrossRef]
- Cooper, R.C.; Baumann, P.L.; McDonald, W.M. An Unexpected Hyperkalemic Response to Succinylcholine during Electroconvulsive Therapy for Catatonic Schizophrenia. Anesthesiology 1999, 91, 574–575. [Google Scholar] [CrossRef]
- Hudcova, J.; Schumann, R. Electroconvulsive Therapy Complicated by Life-Threatening Hyperkalemia in a Catatonic Patient. Gen. Hosp. Psychiatry 2006, 28, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Greer, R.A.; Stewart, R.B. Hyponatremia and ECT. Am. J. Psychiatry 1993, 150, 1272. [Google Scholar] [PubMed]
- Grover, S.; Kattharaghatta Girigowda, V.; Aggarwal, M.; Malhotra, N. Catatonia Associated with Hyponatremia Treated with Electroconvulsive Therapy. J. ECT 2012, 28, e33–e34. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, H.L.; Gibson, J.C.; Steiner, M. Hyponatremia and Depression. J. Nerv. Ment. Dis. 1990, 178, 720–722. [Google Scholar] [CrossRef]
- Lafferty, J.E.; North, C.S.; Spitznagel, E.; Isenberg, K. Laboratory Screening Prior to ECT. J. ECT 2001, 17, 158–165. [Google Scholar] [CrossRef]
- Rasmussen, K.G.; Mohan, A.; Stevens, S.R. Serum Sodium Does Not Correlate with Seizure Length or Seizure Threshold in Electroconvulsive Therapy. J. ECT 2007, 23, 175–176. [Google Scholar] [CrossRef]
- Brent, R.H.; Chodroff, C. ECT as a Possible Treatment for SIADH: Case Report. J. Clin. Psychiatry 1982, 43, 73–74. [Google Scholar]
- de Leon, J. Polydipsia—A Study in a Long-Term Psychiatric Unit. Eur. Arch. Psychiatry Clin. Neurosci. 2003, 253, 37–39. [Google Scholar] [CrossRef]
- Lange-Asschenfeldt, C.; Kojda, G.; Cordes, J.; Hellen, F.; Gillmann, A.; Grohmann, R.; Supprian, T. Epidemiology, Symptoms, and Treatment Characteristics of Hyponatremic Psychiatric Inpatients. J. Clin. Psychopharmacol. 2013, 33, 799–805. [Google Scholar] [CrossRef]
- Burst, V. Etiology and Epidemiology of Hyponatremia. Front. Horm. Res. 2019, 52, 24–35. [Google Scholar] [CrossRef]
- Adrogué, H.J.; Tucker, B.M.; Madias, N.E. Diagnosis and Management of Hyponatremia: A Review. JAMA 2022, 328, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Ilardi, A. Diagnostic and Therapeutic Approach to Hypernatremia. Diagnosis 2022, 9, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Mazhar, F.; Pozzi, M.; Gentili, M.; Scatigna, M.; Clementi, E.; Radice, S.; Carnovale, C. Association of Hyponatraemia and Antidepressant Drugs: A Pharmacovigilance-Pharmacodynamic Assessment through an Analysis of the US Food and Drug Administration Adverse Event Reporting System (FAERS) Database. CNS Drugs 2019, 33, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Revol, R.; Rault, C.; Polard, E.; Bellet, F.; Guy, C. Hyponatremia associated with SSRI/NRSI: Descriptive and comparative epidemiological study of the incidence rates of the notified cases from the data of the French National Pharmacovigilance Database and the French National Health Insurance. Encephale 2018, 44, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Thanki, M.V.; Baliga, S.P.; Parameshwaran, S.; Rao, N.P.; Mehta, U.M.; Thirthalli, J. Safe Administration of Low Frequency RTMS in a Patient with Depression with Recurrent Antidepressant-Associated Hyponatremic Seizures. Brain Stimul. 2020, 13, 1168–1169. [Google Scholar] [CrossRef] [PubMed]
- Belz, M.; Methfessel, I.; Spang, M.; Besse, M.; Folsche, T.; Stephani, C.; Zilles, D. Overlooking the Obvious? Influence of Electrolyte Concentrations on Seizure Quality Parameters in Electroconvulsive Therapy. Eur. Arch. Psychiatry Clin. Neurosci. 2020, 270, 263–269. [Google Scholar] [CrossRef]
- Shah, P.J.; Dubey, K.P.; Watti, C.; Lalwani, J. Effectiveness of Thiopentone, Propofol and Midazolam as an Ideal Intravenous Anaesthetic Agent for Modified Electroconvulsive Therapy: A Comparative Study. Indian J. Anaesth. 2010, 54, 296–301. [Google Scholar] [CrossRef]
- Hovgaard, H.L.; Juhl-Olsen, P. Suxamethonium-Induced Hyperkalemia: A Short Review of Causes and Recommendations for Clinical Applications. Crit. Care Res. Pract. 2021, 2021, 6613118. [Google Scholar] [CrossRef]
- Antosik-Wójcińska, A.; Dominiak, M.; Goetz, Z.; Gędek, A.; Braniecka, A.; Święcicki, Ł.; Mierzejewski, P. Analysis of the Efficacy and Safety of Electroconvulsive Therapy in Elderly Patients: A Retrospective Study in the Polish Population. Psychiatr. Pol. 2022, 56, 767–785. [Google Scholar] [CrossRef]
- Antosik-Wójcińska, A.; Święcicki, Ł. Electroconvulsive therapy—An effective and safe alternative for ineffective pharmacotherapy. Psychiatria 2014, 11, 166–170. [Google Scholar]
- Andrade, C.; Arumugham, S.S.; Thirthalli, J. Adverse Effects of Electroconvulsive Therapy. Psychiatr. Clin. N. Am. 2016, 39, 513–530. [Google Scholar] [CrossRef] [PubMed]
- McClintock, S.M.; Choi, J.; Deng, Z.-D.; Appelbaum, L.G.; Krystal, A.D.; Lisanby, S.H. Multifactorial Determinants of the Neurocognitive Effects of Electroconvulsive Therapy. J. ECT 2014, 30, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Dominiak, M.; Antosik-Wójcińska, A.Z.; Goetz, Z.; Sikorska, O.; Stefanowski, B.; Gorostiza, D.; Święcicki, Ł. Efficacy, Safety and Tolerability of Formula-Based Unilateral vs Bilateral Electroconvulsive Therapy in the Treatment of Major Depression: A Randomized Open Label Controlled Trial. J. Psychiatr. Res. 2021, 133, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.C.; Glasziou, P.P.; Boutron, I.; Milne, R.; Perera, R.; Moher, D.; Altman, D.G.; Barbour, V.; Macdonald, H.; Johnston, M.; et al. Better Reporting of Interventions: Template for Intervention Description and Replication (TIDieR) Checklist and Guide. BMJ 2014, 348, g1687. [Google Scholar] [CrossRef]
- Chantebel, R.; Chesneau, A.; Tavernier, E.; El-Hage, W.; Caille, A. Completeness of Descriptions of Repetitive Transcranial Magnetic Stimulation Intervention: A Systematic Review of Randomized Controlled Trials of RTMS in Depression. J. ECT 2019, 35, 7–13. [Google Scholar] [CrossRef]
| Electrolytes Disturbances | Degrees of Severity | Drug Used during ECT | ||
|---|---|---|---|---|
| Mild (mEq/L) | Moderate (mEq/L) | Severe (mEq/L) | ||
| Hyponatremia | 130–134 | 125–129 | <125 | Not reported |
| Hypernatremia | 145–149 | 150–169 | >170 | Ketamine [21,22,23], Thiopental (false hypernatremia) [24,25] |
| Hypokalemia | 3.5–2.5 | 2.5–3.0 | <2.5 | Atropine [26,27], Propofol [28,29,30], Thiopental [28,29,30] |
| Hyperkalemia | 5.5–6.0 | 6.0–7.0 | >7.0 | Succinylcholine [31,32,33,34], Propofol [28,29,30], Thiopental [28,29,30] |
| Author (Year) | Domain 1 | Domain 2 | Domain 3 | Domain 4 | Domain 5 | Domain 6 | Domain 7 | Domain 8 | Overall |
|---|---|---|---|---|---|---|---|---|---|
| MacMillan (1990) [46] | yes | yes | yes | yes | unclear | yes | yes | yes | 7/8 |
| Greer and Stewart (1993) [44] | yes | unclear | yes | unclear | yes | no | no | yes | 4/8 |
| Mashimo (1996) [38] | yes | yes | yes | yes | yes | yes | unclear | yes | 7/8 |
| Grover (2012) [45] | yes | yes | yes | yes | unclear | yes | unclear | yes | 7/8 |
| Kern (2020) [41] | yes | unclear | unclear | unclear | yes | yes | yes | no | 4/8 |
| Finlayson (1989) [40] | yes | yes | yes | yes | unclear | yes | yes | yes | 7/8 |
| Cooper (1999) [42] | yes | yes | yes | yes | unclear | unclear | yes | yes | 6/8 |
| Hudcova (2006) [43] | yes | unclear | yes | yes | unclear | yes | yes | yes | 6/8 |
| Koster (2014) [39] | yes | yes | yes | yes | unclear | yes | yes | yes | 7/8 |
| Author (Year) | Domain 1 | Domain 2 | Domain 3 | Domain 4 | Domain 5 | Domain 6 | Overall |
|---|---|---|---|---|---|---|---|
| Lafferty (2001) [47] | 1 | 1 | 1 | 0 | 1 | 0 | 4/6 |
| Rasmussen (2007) [48] | 1 | 1 | 1 | 1 | 1 | 0 | 5/6 |
| Author (Year) | Study Design | Number of Patients/Age/Gender (M/F) | Electrolyte Disturbance: (1) Type of Disturbance (2) Measurements before Treatment | Clinical Picture: (1) Psychiatric Diagnosis (2) Treatment before ECT (3) Other Conditions | ECT Characteristics: (1) Electrode Placement (2) Anesthetics Drugs (3) Number of Treatments | Outcomes: (1) ECT Influence on Clinical Symptoms (2) Electrolyte Levels after ECT |
|---|---|---|---|---|---|---|
| MacMillan et al. (1990) [46] | Case report | 1/72/M | (1) Hyponatremia (2) ECT was administered when serum sodium amounted to 130 mmol/L; on admission it was 127 mmol/L, the lowest value during hospitalization was 120 mmol/L | (1) Treatment resistant depression—major depressive episode (2) Doxepin, nortriptyline, tranylcypromine, alprazolam (3) Syndrome of inappropriate antidiuretic hormone secretion (SIADH), Chronic obstructive lung disease | (1) Unilateral right ECT (2) No info (3) 12 | (1) The improvement in clinical symptoms was observed after 12 ECT treatment; ECT treatment was safe (2) In following tests, with unrestricted fluid intake, serum sodium did not fall below 135 mmol/L |
| Greer and Stewart (1993) [44] | Case report | 1/83/F | (1) Hyponatremia (2) On admission serum sodium amounted to 121 mmol/L; during hospitalization it range between 117–128 mmol/L | (1) Treatment resistant depression (2) Diltiazem, digoxin, lorazepam, propoxyphene napsylate, two trials of antidepressants, demeclocycline (3) Several hip dislocations 1 year previously, coronary artery disease, hypertension, myocardial infraction, drunk large quantities of water | (1) Bilateral ECT (2) methohexital (35 mg), succinylcholine (60 mg), esmolol (80 mg) (3) 5 | (1) Data not available (2) Two days after first ECT treatment sodium level increased to 132 mmol/L and 136 mmol/L after fourth ECT treatments; |
| Kern et al. (2020) [41] | Case report | 1/65/F | (1) Hyponatremia (2) On admission serum sodium amounted to 129 mmol/L; during hospitalization it range between 130–132 mmol | (1) Depression, OCD, anorexia (2) Data not available (3) Chronic hyponatremia, Intraparenchymal lateral ventricular cyst (6.3 cm) and periventricular edema | (1) Bilateral ECT (2) Succinylcholine (60 mg), methohexital (50 mg), nitrogliceryne, labetalol (3) 5 in first series; 12 lifetime | (1) The significant improvement in depressive symptoms was observed (2) Hyponatremia remained stable during treatments |
| Mashimo et al. (1996) [38] | Case report | 1/52/F | (1) Hypernatremia (2) On admission serum sodium amounted to 166 mEq/L | (1) Depression and obsessive concern for her handicapped hand, (2) Diazepam, clomipramine (3) Trauma (lost four fingers), dehydration, stupor | (1) Bilateral ECT (2) Amobarbital sodium (200 mg) (3) 6 | (1) After six ECT treatments dramatic improvement in stupor symptoms was observed (2) Hypernatremia was corrected before ECT, however clinical symptoms persist; thus the psychogenic factor was considered to contribute to the presence of stupor |
| Grover et al. (2012) [45] | Case report | 1/48/F | (1) Hyponatremia (2) Serum sodium ranged between 109–120 mmol/L with occasional reading of normal sodium level in multiple measurements | (1) Catatonia secondary to hyponatremia (in the background of vomiting and fever) (2) Amisulpiride, iron, hydrocortisone, lorazepam (3) Adrenal insufficiency, anemia | (1) Bilateral ECT (2) Data not available (3) 6 | (1) The improvement in catatonic symptoms was observed after 6 ECT treatments. (2) Hyponatremia was corrected before ECT, but clinical symptoms persisted; patient was again found to have hyponatremia after a third ECT, after which she was started on fludrocortisone tablets—after this her sodium level stabilized |
| Rasmussen et al. (2007) [48] | Retrospective study | 207/55.6 (18.9)/59% F | (1) Hyponatremia and hypernatremia (2) Hyponatremia was defined as serum sodium level below 135 mmol/L; hypernatremia was defined as a serum sodium level greater than 145 mmol/L; measurements were performed maximum a week before treatment | (1) ECT treated inpatients | (1) ECT bitemporal (n = 84), bifrontal (n = 68), right unilateral (n = 40) (2) glycopyrrolate, thiopental, remifentanil, etomidate, succinylcholine (3) 207 | (1) ECT treatments were safe in all cases of hyponatremia (n = 12) and hypernatremia (n = 4). Prolonged seizures were not observed. |
| Author | Study Design | Number of Patient/Age/Gender (M/F) | Electrolyte Disturbance: (1) Type of Disturbance (2) Measurements after Treatment | Clinical Picture: (1) Psychiatric Diagnosis (2) Treatment before ECT (3) Other Conditions | ECT Characteristics: (1) Electrode Placement (2) Anesthetics drugs (3) Number of Treatments | Outcomes: (1) ECT Influence on Clinical Symptoms (2) Electrolyte Levels after ECT |
|---|---|---|---|---|---|---|
| Lafferty et al. (2001) [47] | Retrospective study | 484/54.1(19.1)/70%F | (1) Hyponatremia, hypernatremia, hypokalemia, hyperkalemia (2) Data not available | (1) Patients received ECT (2) Data not available (3) Data not available | (1) Data not available (2) Data not available (3) Data not available | (1) Abnormal sodium levels were associated with hospital admission after ECT, but without impact on ECT complications. Both re-admissions and complications were not associated with potassium abnormalities. (2) The proportion of Na+ and K+ abnormalities in ECT patients was similar to that in those without ECT. |
| Finlayson et al. (1989) [40] | Case report | 1/55/F | (1) Hyponatremia (2) 106 mmol/L | (1) Depression (2) Trazodone, L-tryptophan, triazolam, flupethixol, thioridazine; past treatment: tricyclic and monoamine oxidase inhibiting antidepressants, neuroleptics, benzodiazepines, lithium (3) Agitation, insomnia, poor appetite, somatic complaints of abdominal burning, loss of concentration and memory, polidypsia | (1) Unilateral right ECT (2) Data not available (3) 10 | (1) After eight treatments a decrease in agitation and slight improvement in affect was noted. (2) After 10 ECT treatment patient developed spontaneous seizures with serum sodium 106 mmol/L 50 min after the seizures. She drank 5–10 L of water per day during the hospitalization. |
| Grover et al. (2012) [45] | Case report | 1/48/F | (1) Hyponatremia (2) Data not available | (1) Catatonia secondary to hyponatremia (in the background of vomiting and fever) (2) Amisulpiride, iron, hydrocortisone, lorazepam Adrenal insufficiency, anemia | (1) Bilateral ECT (2) Data not available (3) 6 | (1) The improvement in catatonic symptoms was observed after 6 ECT treatments. (2) Hyponatremia was corrected before ECT, but clinical symptoms caused by it remained; patient was again found to have hyponatremia after a third ECT, after which she was started on fludrocortisone tablets—after this her sodium level stabilized |
| Cooper et al. (1999) [42] | Case report | 1/40/F | (1) Hyperkalemia (2) 4.0–6.9 mEq | (1) Catatonia, schizophrenia (2) Amantadine, dantrolene, bromocriptine | (1) Data not available (2) Atropine (0.4 mg), Curare (3 mg), labetalol (10 mg), methohexital (80 mg), succinylcholine (120 mg) (3) 4 | (1) Data not available (2) Potassium level was 5.4 mEq after second, 5.5 mEq after third (4.2 before treatment), 6.9 mEq after fourth ECT treatment (4.0 before); the patients was noted to have wide-complex arrythmia after 2nd treatment, and peaking T waves after 4th. Succinylcholine was substitute to mivacurium and potassium level was normal after next treatments. |
| Hudcova and Schumann (2006) [43] | Case report | 1/34/F | (1) Hyperkalemia (2) 6.4 mEq/L | (1) Catatonia, schizophrenia, bipolar disorder (2) Haloperidol, bromocriptine, lorazepam (3) Obesity, fever, pulmonary embolism, intubation and mechanical ventilation | (1) Data not available (2) Succinylcholine (140 mg), etomidate (20 mg), glycopyrrolate (0.4 mg) (3) 3 | (1) ECT improved her psychiatric condition (2) Potassium level was 6.4 mEq/L for third ECT treatment and ventricular tachycardia was noted. Then succinylcholine was substitute to atracurium/mivacurium. No electrolyte abnormalities were noted during the following outpatient treatment |
| Koster et al. (2014) [39] | Case report | 1/42/F | (1) Hyperkalemia (2) 11.6 mmol/L−1 | (1) Neuroleptic malignant syndrome after drug overdose (2) Duloxetine, quetiapine, flurazepam, insulin, activated charcoal | (1) Data not available (2) Succinylcholine (1.2 mg kg−1) and etomidate—first 4 sessions; rocuronium (1.2 mg kg−1) and etomidate—next 7 sessions (3) 11 | (1) In total, patient received 11 ECT sessions with improvement in terms of rigidity, fever and creatine kinase (2) At the four session arterial blood sample showed a potassium of 11.6 mmol/L−1 (the patient developed VFib, treated with cardiopulmonary resuscitation; calcium gluconate was administered); in next sessions with rocuronium hyperkalemia was not reported |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gędek, A.; Materna, M.; Majewski, P.; Antosik, A.Z.; Dominiak, M. Electrolyte Disturbances Related to Sodium and Potassium and Electroconvulsive Therapy: A Systematic Review. J. Clin. Med. 2023, 12, 6677. https://doi.org/10.3390/jcm12206677
Gędek A, Materna M, Majewski P, Antosik AZ, Dominiak M. Electrolyte Disturbances Related to Sodium and Potassium and Electroconvulsive Therapy: A Systematic Review. Journal of Clinical Medicine. 2023; 12(20):6677. https://doi.org/10.3390/jcm12206677
Chicago/Turabian StyleGędek, Adam, Michał Materna, Paweł Majewski, Anna Z. Antosik, and Monika Dominiak. 2023. "Electrolyte Disturbances Related to Sodium and Potassium and Electroconvulsive Therapy: A Systematic Review" Journal of Clinical Medicine 12, no. 20: 6677. https://doi.org/10.3390/jcm12206677
APA StyleGędek, A., Materna, M., Majewski, P., Antosik, A. Z., & Dominiak, M. (2023). Electrolyte Disturbances Related to Sodium and Potassium and Electroconvulsive Therapy: A Systematic Review. Journal of Clinical Medicine, 12(20), 6677. https://doi.org/10.3390/jcm12206677






