How to Manage Metallosis: A Retrospective Cohort Analysis after Revision Hip Surgery
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Collection
2.2. Characteristics of Revision Prosthesis
2.3. Functional Evaluation
2.4. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. Complications and Implant Survivorship
3.3. Functional Evaluation
3.4. Serum Data
3.5. Microbiological Data
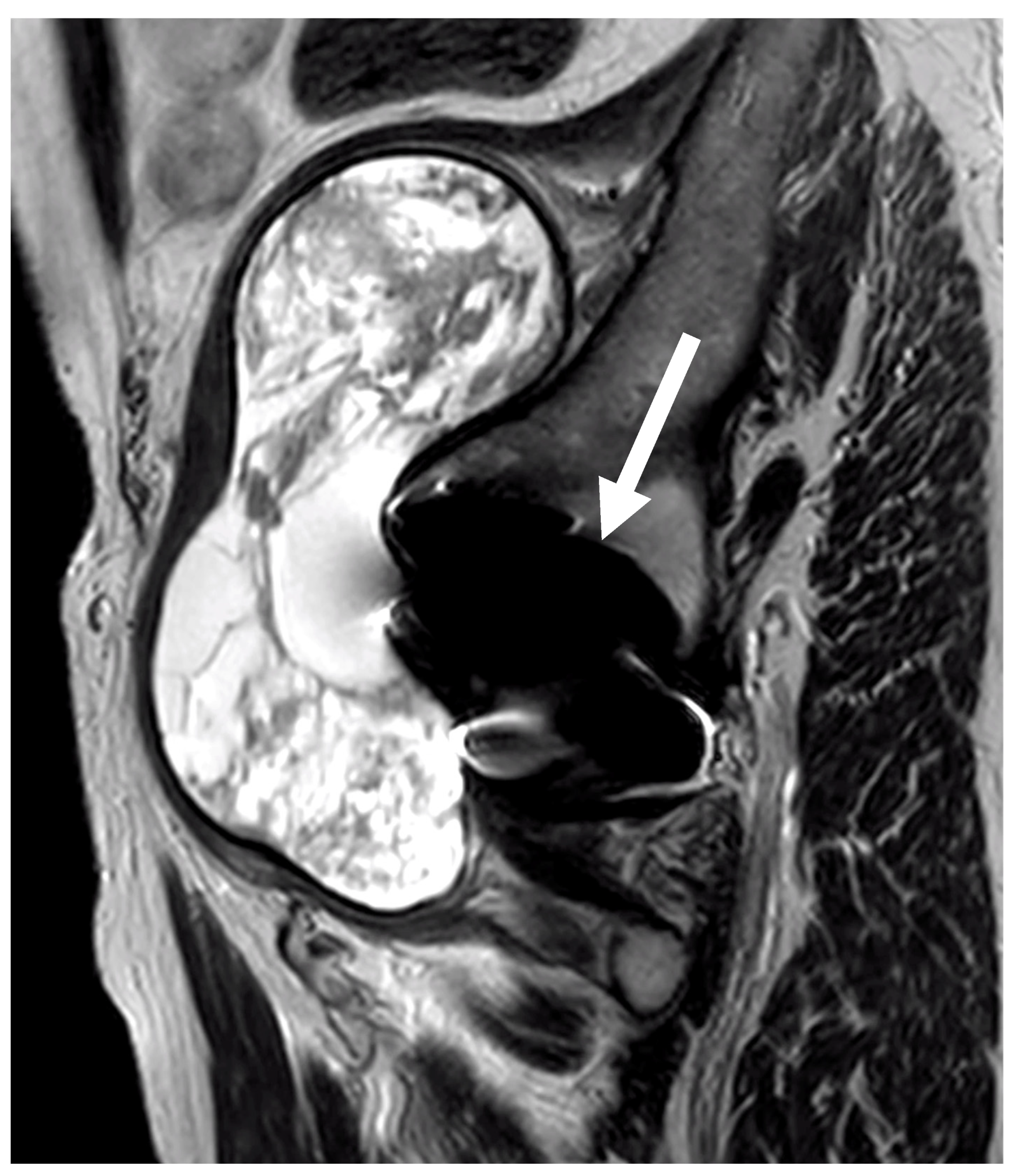
3.6. Imaging Data
4. Discussion
4.1. Aim of the Study
4.2. What Is Known in the Literature
4.3. Comparison between Literature and Our Data
4.4. Limitation of the Study and Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Learmonth, I.D.; Young, C.; Rorabeck, C. The operation of the century: Total hip replacement. Lancet 2007, 370, 1508–1519. [Google Scholar] [CrossRef]
- Blankstein, M.; Haimes, M.A.; Nelms, N.J. Selecting a Press-fit Stem for Total Hip Arthroplasty: The Rationale and Evolution of the Modern Femoral Prosthesis. J. Am. Acad. Orthop. Surg. 2022, 30, e1279–e1290. [Google Scholar] [CrossRef]
- Cho, M.R.; Choi, W.K.; Kim, J.J. Current Concepts of Using Large Femoral Heads in Total Hip Arthroplasty. Hip Pelvis 2016, 28, 134–141. [Google Scholar] [CrossRef]
- Smith, A.J.; Dieppe, P.; Vernon, K.; Porter, M.; Blom, A.W.; National Joint Registry of England and Wales. Failure rates of stemmed metal-on-metal hip replacements: Analysis of data from the National Joint Registry of England and Wales. Lancet 2012, 379, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- Morsi, E.M.Z.; Drwish, A.E.E.; Saber, A.M.; Nassar, I.M.; Zaki, A.E.M. The Use of Standard Cemented Femoral Stems in Total Hip Replacement After Failed Internal Fixation of Intertrochanteric Femoral Fractures. J. Arthroplast. 2020, 35, 2525–2528. [Google Scholar] [CrossRef]
- Philpott, A.; Weston-Simons, J.S.; Grammatopoulos, G.; Bejon, P.; Gill, H.S.; McLardy-Smith, P.; Gundle, R.; Murray, D.W.; Pandit, H. Predictive outcomes of revision total hip replacement—A consecutive series of 1176 patients with a minimum 10-year follow-up. Maturitas 2014, 77, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.M.; Fehring, T.K.; Lombardi, A.V.; Barnes, C.L.; Cabanela, M.E.; Jacobs, J.J. Risk stratification algorithm for management of patients with dual modular taper total hip arthroplasty: Consensus statement of the American Association of Hip and Knee Surgeons, the American Academy of Orthopaedic Surgeons and the Hip Society. J. Arthroplast. 2014, 29, 2060–2064. [Google Scholar] [CrossRef]
- Ikeda, S.; Kaku, N.; Hosoyama, T.; Tsumura, H. Survival rates of different bearing surfaces with the same model of stem in total hip arthroplasty: Predictive factors for revision surgery. Arch. Orthop. Trauma. Surg. 2022, 143, 4501–4510. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, S.; Ong, K.; Lau, E.; Mowat, F.; Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Jt. Surg. Am. 2007, 89, 780–785. [Google Scholar] [CrossRef]
- Grupp, T.M.; Weik, T.; Bloemer, W.; Knaebel, H.P. Modular titanium alloy neck adapter failures in hip replacement—Failure mode analysis and influence of implant material. BMC Musculoskelet. Disord. 2010, 11, 3. [Google Scholar] [CrossRef]
- Wright, G.; Sporer, S.; Urban, R.; Jacobs, J. Fracture of a modular femoral neck after total hip arthroplasty: A case report. J. Bone Jt. Surg. Am. 2010, 92, 1518–1521. [Google Scholar] [CrossRef]
- Filer, J.L.; Berstock, J.; Hughes-Roberts, Y.; Foote, J.; Sandhu, H. Haemorrhagic Pseudotumour Following Metal-on-Metal Hip Replacement. Cureus 2021, 13, e15541. [Google Scholar] [CrossRef] [PubMed]
- Langton, D.J.; Jameson, S.S.; Joyce, T.J.; Gandhi, J.N.; Sidaginamale, R.; Mereddy, P.; Lord, J.; Nargol, A.V. Accelerating failure rate of the ASR total hip replacement. J. Bone Jt. Surg. Br. 2011, 93, 1011–1016. [Google Scholar] [CrossRef]
- Pandit, H.; Glyn-Jones, S.; McLardy-Smith, P.; Gundle, R.; Whitwell, D.; Gibbons, C.L.; Ostlere, S.; Athanasou, N.; Gill, H.S.; Murray, D.W. Pseudotumours associated with metal-on-metal hip resurfacings. J. Bone Jt. Surg. Br. 2008, 90, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, I.; Galea, V.P.; Donahue, G.S.; Matuszak, S.J.; Muratoglu, O.; Malchau, H. The Cobalt/Chromium Ratio Provides Similar Diagnostic Value to a Low Cobalt Threshold in Predicting Adverse Local Tissue Reactions in Patients With Metal-on-Metal Hip Arthroplasty. J. Arthroplast. 2018, 33, 3020–3024. [Google Scholar] [CrossRef]
- Campbell, J.R.; Estey, M.P. Metal release from hip prostheses: Cobalt and chromium toxicity and the role of the clinical laboratory. Clin. Chem. Lab. Med. 2013, 51, 213–220. [Google Scholar] [CrossRef]
- Culliford, D.; Maskell, J.; Judge, A.; Cooper, C.; Prieto-Alhambra, D.; Arden, N.K.; Group, C.O.S. Future projections of total hip and knee arthroplasty in the UK: Results from the UK Clinical Practice Research Datalink. Osteoarthr. Cartil. 2015, 23, 594–600. [Google Scholar] [CrossRef]
- Katz, J.N.; Wright, E.A.; Wright, J.; Malchau, H.; Mahomed, N.N.; Stedman, M.; Baron, J.A.; Losina, E. Twelve-year risk of revision after primary total hip replacement in the U.S. Medicare population. J. Bone Jt. Surg. Am. 2012, 94, 1825–1832. [Google Scholar] [CrossRef]
- Huang, P.; Lyons, M.; O'Sullivan, M. The Infection Rate of Metal-on-Metal Total Hip Replacement Is Higher When Compared to Other Bearing Surfaces as Documented by the Australian Orthopaedic Association National Joint Replacement Registry. HSS J. 2018, 14, 99–105. [Google Scholar] [CrossRef]
- Judd, K.T.; Noiseux, N. Concomitant infection and local metal reaction in patients undergoing revision of metal on metal total hip arthroplasty. Iowa Orthop. J. 2011, 31, 59–63. [Google Scholar]
- Treacy, R.B.; McBryde, C.W.; Pynsent, P.B. Birmingham hip resurfacing arthroplasty. A minimum follow-up of five years. J. Bone Jt. Surg. Br. 2005, 87, 167–170. [Google Scholar] [CrossRef]
- Anwar, H.A.; Aldam, C.H.; Visuvanathan, S.; Hart, A.J. The effect of metal ions in solution on bacterial growth compared with wear particles from hip replacements. J. Bone Jt. Surg. Br. 2007, 89, 1655–1659. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bozic, K.J.; Kurtz, S.M.; Lau, E.; Ong, K.; Vail, T.P.; Berry, D.J. The epidemiology of revision total hip arthroplasty in the United States. J. Bone Jt. Surg. Am. 2009, 91, 128–133. [Google Scholar] [CrossRef]
- Rao, B.M.; Stokey, P.; Tanios, M.; Liu, J.; Ebraheim, N.A. A systematic review of the surgical outcomes of interprosthetic femur fractures. J. Orthop. 2022, 33, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Han, K.Y.; Nam, Y.J.; Kim, K.W. Result of Modular Necks in Primary Total Hip Arthroplasty with a Average Follow-up of Four Years. Hip Pelvis 2016, 28, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Jauch, S.Y.; Huber, G.; Hoenig, E.; Baxmann, M.; Grupp, T.M.; Morlock, M.M. Influence of material coupling and assembly condition on the magnitude of micromotion at the stem-neck interface of a modular hip endoprosthesis. J. Biomech. 2011, 44, 1747–1751. [Google Scholar] [CrossRef] [PubMed]
- Gill, I.P.; Webb, J.; Sloan, K.; Beaver, R.J. Corrosion at the neck-stem junction as a cause of metal ion release and pseudotumour formation. J. Bone Jt. Surg. Br. 2012, 94, 895–900. [Google Scholar] [CrossRef]
- Ishida, T.; Tateiwa, T.; Takahashi, Y.; Nishikawa, Y.; Shishido, T.; Masaoka, T.; Yamamoto, K. Adverse Tissue Reactions and Metal Ion Behavior After Small-Head Metasul Hip Arthroplasty: A Long-Term Follow-Up Study. Orthop. Surg. 2022, 14, 264–273. [Google Scholar] [CrossRef]
- Martinez Villen, G.; Rodriguez Nogue, L.; Garcia Gonzalez, E. Postoperative assessment and management of metallosis and periprosthetic osteolysis in patients treated with metal-on-polyethylene total wrist prostheses. J. Hand Surg. Eur. Vol. 2022, 47, 952–958. [Google Scholar] [CrossRef]
- Imada, A.O.; Painter, B.M.; Clinger, B.; Decker, M.M. A Rare Case of Metallosis Co-occurring With Chronic Prosthetic Joint Infection in Total Hip Arthroplasty. Cureus 2022, 14, e21515. [Google Scholar] [CrossRef]
- Favetti, F.; Mazzotta, G.; Papalia, M.; Panegrossi, G.; Casella, F.; Falez, F. Contamination of revision procedures in patients with adverse tissues reaction to metal on metal implant. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 86–93. [Google Scholar] [CrossRef]
- Sabah, S.A.; Mitchell, A.W.; Henckel, J.; Sandison, A.; Skinner, J.A.; Hart, A.J. Magnetic resonance imaging findings in painful metal-on-metal hips: A prospective study. J. Arthroplast. 2011, 26, 71–76.e2. [Google Scholar] [CrossRef]
- Hjorth, M.H.; Mechlenburg, I.; Soballe, K.; Romer, L.; Jakobsen, S.S.; Stilling, M. The anatomical location of cystic pseudotumors and muscle atrophy in metal-on-metal resurfacing hip arthroplasty is related to the surgical approach used for implantation. A subgroup analyses of a randomized controlled trial. Arch. Orthop. Trauma. Surg. 2023, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hauptfleisch, J.; Pandit, H.; Grammatopoulos, G.; Gill, H.S.; Murray, D.W.; Ostlere, S. A MRI classification of periprosthetic soft tissue masses (pseudotumours) associated with metal-on-metal resurfacing hip arthroplasty. Skelet. Radiol. 2012, 41, 149–155. [Google Scholar] [CrossRef]
- Higuchi, M.; Watanabe, N. Determination of the erythrocyte sedimentation rate using the hematocrit-corrected aggregation index and mean corpuscular volume. J. Clin. Lab. Anal. 2023, 37, e24877. [Google Scholar] [CrossRef] [PubMed]
- Markozannes, G.; Koutsioumpa, C.; Cividini, S.; Monori, G.; Tsilidis, K.K.; Kretsavos, N.; Theodoratou, E.; Gill, D.; Ioannidis, J.P.; Tzoulaki, I. Global assessment of C-reactive protein and health-related outcomes: An umbrella review of evidence from observational studies and Mendelian randomization studies. Eur. J. Epidemiol. 2021, 36, 11–36. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, A.O.; Parker, R.; Farnsworth, C.; Law, C.; Johnson-Davis, K.L. Method validation of multi-element panel in whole blood by inductively coupled plasma mass spectrometry (ICP-MS). J. Mass. Spectrom. Adv. Clin. Lab. 2023, 27, 33–39. [Google Scholar] [CrossRef]
- Farrar, J.T.; Young, J.P., Jr.; LaMoreaux, L.; Werth, J.L.; Poole, M.R. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 2001, 94, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Pivec, R.; Johnson, A.J.; Mears, S.C.; Mont, M.A. Hip arthroplasty. Lancet 2012, 380, 1768–1777. [Google Scholar] [CrossRef]
- Cooper, H.J.; Della Valle, C.J.; Berger, R.A.; Tetreault, M.; Paprosky, W.G.; Sporer, S.M.; Jacobs, J.J. Corrosion at the head-neck taper as a cause for adverse local tissue reactions after total hip arthroplasty. J. Bone Jt. Surg. Am. 2012, 94, 1655–1661. [Google Scholar] [CrossRef]
- Bellamy, N.; Buchanan, W.W.; Goldsmith, C.H.; Campbell, J.; Stitt, L.W. Validation study of WOMAC: A health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 1988, 15, 1833–1840. [Google Scholar]
- Salaffi, F.; Leardini, G.; Canesi, B.; Mannoni, A.; Fioravanti, A.; Caporali, R.; Lapadula, G.; Punzi, L.; GOnarthrosis and Quality Of Life Assessment (GOQOLA) Study Group. Reliability and validity of the Western Ontario and McMaster Universities (WOMAC) Osteoarthritis Index in Italian patients with osteoarthritis of the knee. Osteoarthr. Cartil. 2003, 11, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Nilsdotter, A.; Bremander, A. Measures of hip function and symptoms: Harris Hip Score (HHS), Hip Disability and Osteoarthritis Outcome Score (HOOS), Oxford Hip Score (OHS), Lequesne Index of Severity for Osteoarthritis of the Hip (LISOH), and American Academy of Orthopedic Surgeons (AAOS) Hip and Knee Questionnaire. Arthritis Care Res. 2011, 63 (Suppl. S11), S200–S207. [Google Scholar] [CrossRef]
- Dettoni, F.; Pellegrino, P.; La Russa, M.R.; Bonasia, D.E.; Blonna, D.; Bruzzone, M.; Castoldi, F.; Rossi, R. Validation and cross cultural adaptation of the Italian version of the Harris Hip Score. Hip Int. 2015, 25, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Hsu, A.R.; Gross, C.E.; Levine, B.R. Pseudotumor from modular neck corrosion after ceramic-on-polyethylene total hip arthroplasty. Am. J. Orthop. 2012, 41, 422–426. [Google Scholar]
- Biant, L.C.; Bruce, W.J.; Assini, J.B.; Walker, P.M.; Walsh, W.R. The anatomically difficult primary total hip replacement: Medium- to long-term results using a cementless odular stem. J. Bone Jt. Surg. Br. 2008, 90, 430–435. [Google Scholar] [CrossRef]
- Kong, X.; Chai, W.; Yang, M.; Ong, A.; Chen, J.; Wang, Y.; Zhou, Y. Modular stem in total hip arthroplasty for patients with trochanter valgus deformity: Surgical technique and case series. BMC Musculoskelet. Disord. 2020, 21, 124. [Google Scholar] [CrossRef]
- Spitzer, A.I. The S-ROM cementless femoral stem: History and literature review. Orthopedics 2005, 28, s1117–s1124. [Google Scholar] [CrossRef]
- Archibeck, M.J.; Cummins, T.; Carothers, J.; Junick, D.W.; White, R.E., Jr. A comparison of two implant systems in restoration of hip geometry in arthroplasty. Clin. Orthop. Relat. Res. 2011, 469, 443–446. [Google Scholar] [CrossRef]
- Duwelius, P.J.; Hartzband, M.A.; Burkhart, R.; Carnahan, C.; Blair, S.; Wu, Y.; Grunkemeier, G.L. Clinical results of a modular neck hip system: Hitting the “bull’s-eye” more accurately. Am. J. Orthop. 2010, 39, 2–6. [Google Scholar]
- Kouzelis, A.; Georgiou, C.S.; Megas, P. Dissociation of modular total hip arthroplasty at the neck-stem interface without dislocation. J. Orthop. Traumatol. 2012, 13, 221–224. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sporer, S.M.; DellaValle, C.; Jacobs, J.; Wimmer, M. A case of disassociation of a modular femoral neck trunion after total hip arthroplasty. J. Arthroplast. 2006, 21, 918–921. [Google Scholar] [CrossRef]
- Lam, L.O.; Stoffel, K.; Kop, A.; Swarts, E. Catastrophic failure of 4 cobalt-alloy Omnifit hip arthroplasty femoral components. Acta Orthop. 2008, 79, 18–21. [Google Scholar] [CrossRef]
- Vatani, N.; Comando, D.; Acuna, J.; Prieto, D.; Caviglia, H. Faulty design increases the risk of neck fracture in a hip prosthesis. Acta Orthop. Scand. 2002, 73, 513–517. [Google Scholar] [CrossRef]
- Gilbert, J.L.; Buckley, C.A.; Jacobs, J.J. In vivo corrosion of modular hip prosthesis components in mixed and similar metal combinations. The effect of crevice, stress, motion, and alloy coupling. J. Biomed. Mater. Res. 1993, 27, 1533–1544. [Google Scholar] [CrossRef] [PubMed]
- Inoue, D.; Restrepo, C.; Nourie, B.; Hozack, W.J. Clinical Results of Revision Hip Arthroplasty for Neck-Taper Corrosion and Adverse Local Tissue Reactions Around a Modular Neck Stem. J. Arthroplast. 2020, 35, S289–S293. [Google Scholar] [CrossRef] [PubMed]
- Crackau, M.; Martens, N.; Harnisch, K.; Berth, A.; Doring, J.; Lohmann, C.H.; Halle, T.; Bertrand, J. In vivo corrosion and damages in modular shoulder prostheses. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 1764–1778. [Google Scholar] [CrossRef]
- Pisanu, F.; Andreozzi, M.; Fiori, E.; Altamore, F.; Bartoli, M.; Caggiari, G.; Ortu, S.; Rios, M.; Manunta, A.F.; Doria, C. Surgical management of hip prosthetic failure in metallosis: A case series and literature review. J. Orthop. 2021, 28, 10–20. [Google Scholar] [CrossRef]
- Levine, B.R.; Hsu, A.R.; Skipor, A.K.; Hallab, N.J.; Paprosky, W.G.; Galante, J.O.; Jacobs, J.J. Ten-year outcome of serum metal ion levels after primary total hip arthroplasty: A concise follow-up of a previous report. J. Bone Jt. Surg. Am. 2013, 95, 512–518. [Google Scholar] [CrossRef]
- Donaldson, J.R.; Miles, J.; Sri-Ram, K.; Poullis, C.; Muirhead-Allwood, S.; Skinner, J. The relationship between the presence of metallosis and massive infection in metal-on-metal hip replacements. Hip Int. 2010, 20, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Grammatopoulos, G.; Munemoto, M.; Inagaki, Y.; Tanaka, Y.; Athanasou, N.A. The Diagnosis of Infection in Metal-on-Metal Hip Arthroplasties. J. Arthroplast. 2016, 31, 2569–2573. [Google Scholar] [CrossRef] [PubMed]
- Simoni, G.; Maccauro, G.; Fenga, D.; De Santis, V.; Orani, R.A.; Centofanti, F.; Rosa, M.A. Arthrodesis of the ankle joint in septic osteoarthritis: Six years long term outcomes in authors’ personal experience. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Howie, D.W.; Cain, C.M.; Cornish, B.L. Pseudo-abscess of the psoas bursa in failed double-cup arthroplasty of the hip. J. Bone Jt. Surg. Br. 1991, 73, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Manfreda, F.; Bufi, E.; Florio, E.F.; Ceccarini, P.; Rinonapoli, G.; Caraffa, A.; Antinolfi, P. Osteolysis in total hip arthroplasty in relation to metal ion release: Comparison between monolithic prostheses and different modularities. World J. Orthop. 2021, 12, 768–780. [Google Scholar] [CrossRef] [PubMed]
- Pozzuoli, A.; Berizzi, A.; Crimi, A.; Belluzzi, E.; Frigo, A.C.; Conti, G.; Nicolli, A.; Trevisan, A.; Biz, C.; Ruggieri, P. Metal Ion Release, Clinical and Radiological Outcomes in Large Diameter Metal-on-Metal Total Hip Arthroplasty at Long-Term Follow-Up. Diagnostics 2020, 10, 941. [Google Scholar] [CrossRef]
- Lainiala, O.S.; Moilanen, T.P.S.; Hart, A.J.; Huhtala, H.S.A.; Sabah, S.A.; Eskelinen, A.P. Higher Blood Cobalt and Chromium Levels in Patients With Unilateral Metal-on-Metal Total Hip Arthroplasties Compared to Hip Resurfacings. J. Arthroplast. 2016, 31, 1261–1266. [Google Scholar] [CrossRef]
- Barlow, B.T.; Boles, J.W.; Lee, Y.Y.; Ortiz, P.A.; Westrich, G.H. Short-Term Outcomes and Complications After Rejuvenate Modular Total Hip Arthroplasty Revision. J. Arthroplast. 2016, 31, 857–862. [Google Scholar] [CrossRef]
- Dimitriou, D.; Liow, M.H.; Tsai, T.Y.; Leone, W.A.; Li, G.; Kwon, Y.M. Early Outcomes of Revision Surgery for Taper Corrosion of Dual Taper Total Hip Arthroplasty in 187 Patients. J. Arthroplast. 2016, 31, 1549–1554. [Google Scholar] [CrossRef]

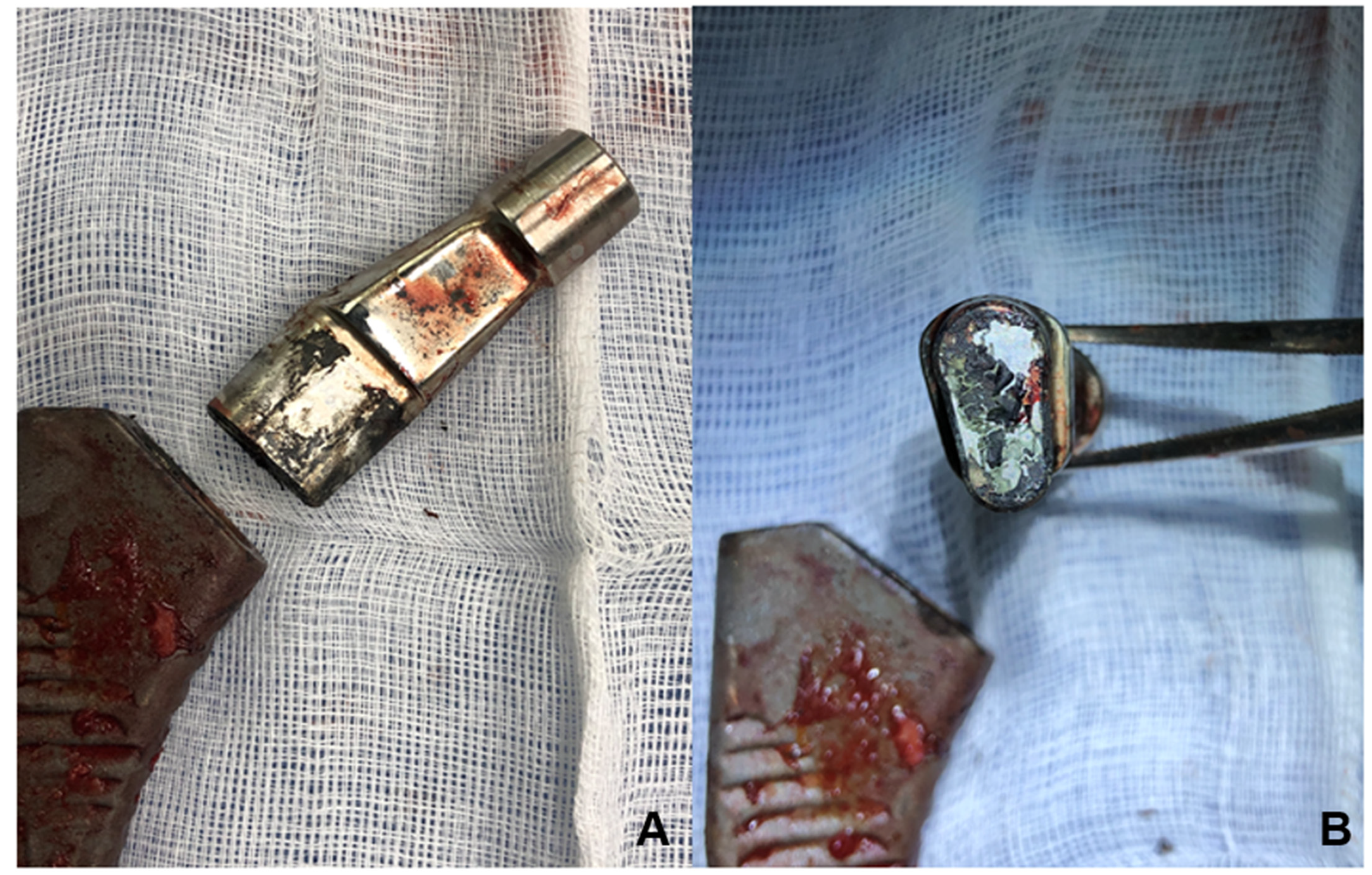

| Number of Patients | 43 |
| Age (y) | 67.1 (range of 30–86) |
| Gender (F/M) | 30/13 |
| Side (L/R) | 24/19 |
| Revision (months) | 35.3 ± 8.4 |
| Height (cm) | 157.8 ± 5.7 |
| Weight (kg) | 84.6 ± 15.4 |
| BMI (kg/m2) | 23.6 (range of 18–35) |
| Mean interval from initial THA surgery to revision (months) | 30.8 ± 14.5 (range of 10–60) |
| Mean follow-up period after revision surgery (months) | 40.2 ± 19.4 (range of 20–75) |
WOMAC at admission for revision surgery (mean ± standard deviation)
| 54.6 ± 19.6 51.2 ± 17.4 |
HHS at admission for revision surgery (mean ± standard deviation)
| 36.5 ± 5.4 41.4 ± 3.6 |
WOMAC at one year follow-up (mean ± standard deviation)
| 11.2 ± 16.4 10.2 ± 14.8 |
HHS at one year follow-up (mean ± standard deviation)
| 75.4 ± 5.5 78.7 ± 6.4 |
| Asymptomatic | Typical Symptomatic | Atypical Symptomatic | p-Value | |
|---|---|---|---|---|
| 17 | 16 | 10 | ||
| Cobalt [μg/L] | 3.2 (0–7.4) | 4.3 (0.2–11.7) | 5.3 (0.2–14.8) | 0.45 |
| Chromium [μg/L] | 1.4 (0–7.0) | 1.6 (0.2–8.2) | 1.6 (0.4–4.3) | 0.96 |
| ESR [mm/h] | 15.5 (2–45) | 14.7 (1–61) | 13.4 (1–61) | 0.67 |
| CRP [mg/dL] | 2.0 (0.5–16.6) | 2.1 (0.1–6.1) | 2.2 (0.1–7.2) | 0.89 |
| Age | Sex | Time from Initial Surgery to Revision | ARMD | Microbiology |
|---|---|---|---|---|
| 55 | Female | 35 months | Metallosis | S. epidermidis |
| 72 | Female | 31 months | Metallosis | E. faecalis |
| 64 | Male | 34 months | Metallosis | K. pneumoniae |
| Asymptomatic | Typical Symptomatic | Atypical Symptomatic | p-Value | ||
|---|---|---|---|---|---|
| Number of patients | 30 | 5 (17%) | 15 (50%) | 10 (33%) | |
| MARS/MRI | Muscle changes | 1 (3%) | 8 (26%) | 5 (17%) | 0.001 |
| Fluid accumulations | 2 (6%) | 4 (13%) | 4 (13%) | 0.001 | |
| Mass | 1 (3%) | 2 (6%) | 0 | 0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruschetta, A.; Palco, M.; Fenga, D.; Giuca, G.; Holzer, L.A.; Alito, A.; Cacciola, G.; De Meo, F.; Cavaliere, P. How to Manage Metallosis: A Retrospective Cohort Analysis after Revision Hip Surgery. J. Clin. Med. 2023, 12, 4809. https://doi.org/10.3390/jcm12144809
Bruschetta A, Palco M, Fenga D, Giuca G, Holzer LA, Alito A, Cacciola G, De Meo F, Cavaliere P. How to Manage Metallosis: A Retrospective Cohort Analysis after Revision Hip Surgery. Journal of Clinical Medicine. 2023; 12(14):4809. https://doi.org/10.3390/jcm12144809
Chicago/Turabian StyleBruschetta, Antongiulio, Michelangelo Palco, Domenico Fenga, Gabriele Giuca, Lukas A. Holzer, Angelo Alito, Giorgio Cacciola, Federico De Meo, and Pietro Cavaliere. 2023. "How to Manage Metallosis: A Retrospective Cohort Analysis after Revision Hip Surgery" Journal of Clinical Medicine 12, no. 14: 4809. https://doi.org/10.3390/jcm12144809
APA StyleBruschetta, A., Palco, M., Fenga, D., Giuca, G., Holzer, L. A., Alito, A., Cacciola, G., De Meo, F., & Cavaliere, P. (2023). How to Manage Metallosis: A Retrospective Cohort Analysis after Revision Hip Surgery. Journal of Clinical Medicine, 12(14), 4809. https://doi.org/10.3390/jcm12144809








