Diagnosis and Non-Invasive Treatment of Obesity in Adults with Type 2 Diabetes Mellitus: A Review of Guidelines
Abstract
1. Introduction
2. The Aim of the Review
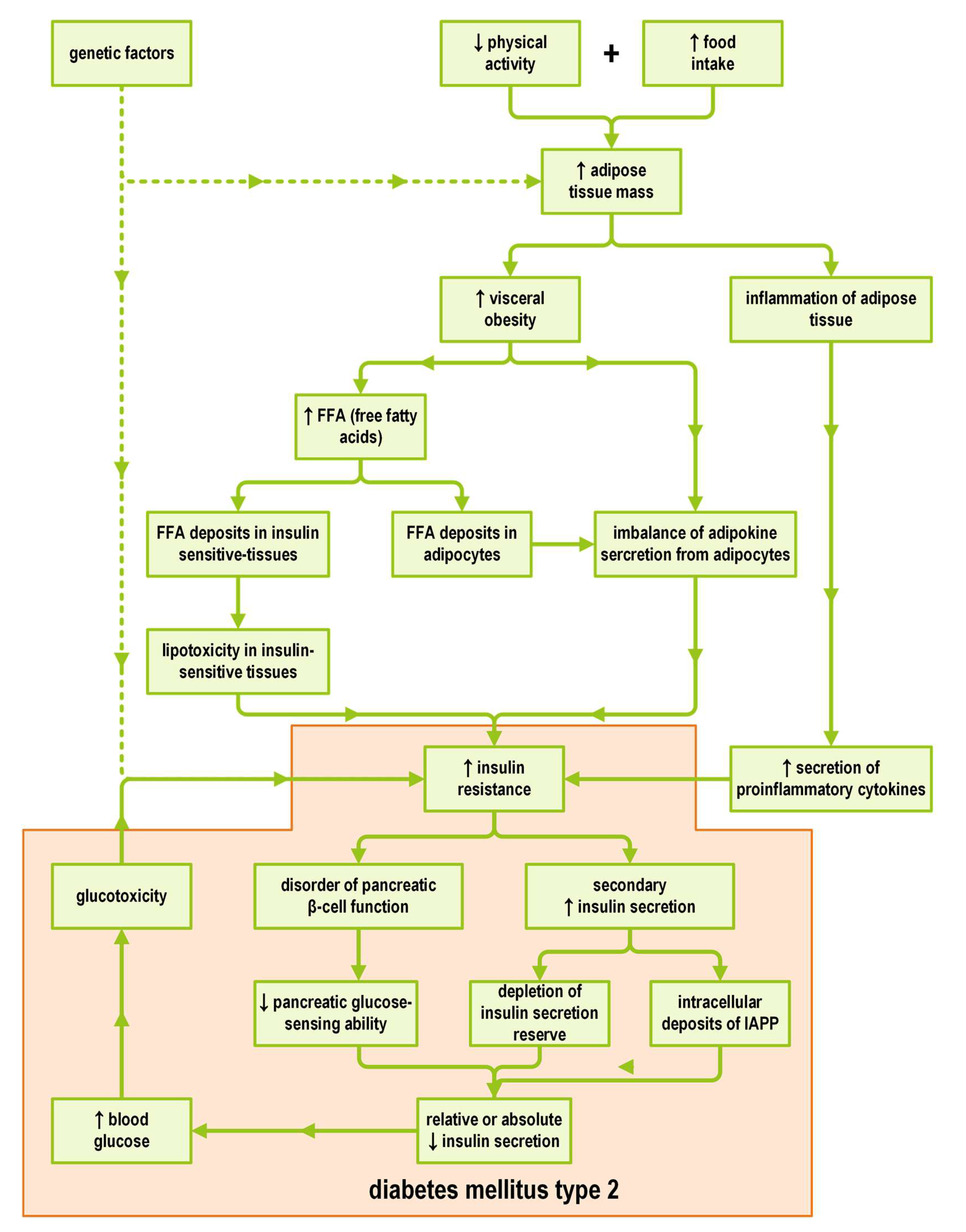
3. The Significance of the Interrelationship between Obesity and T2DM
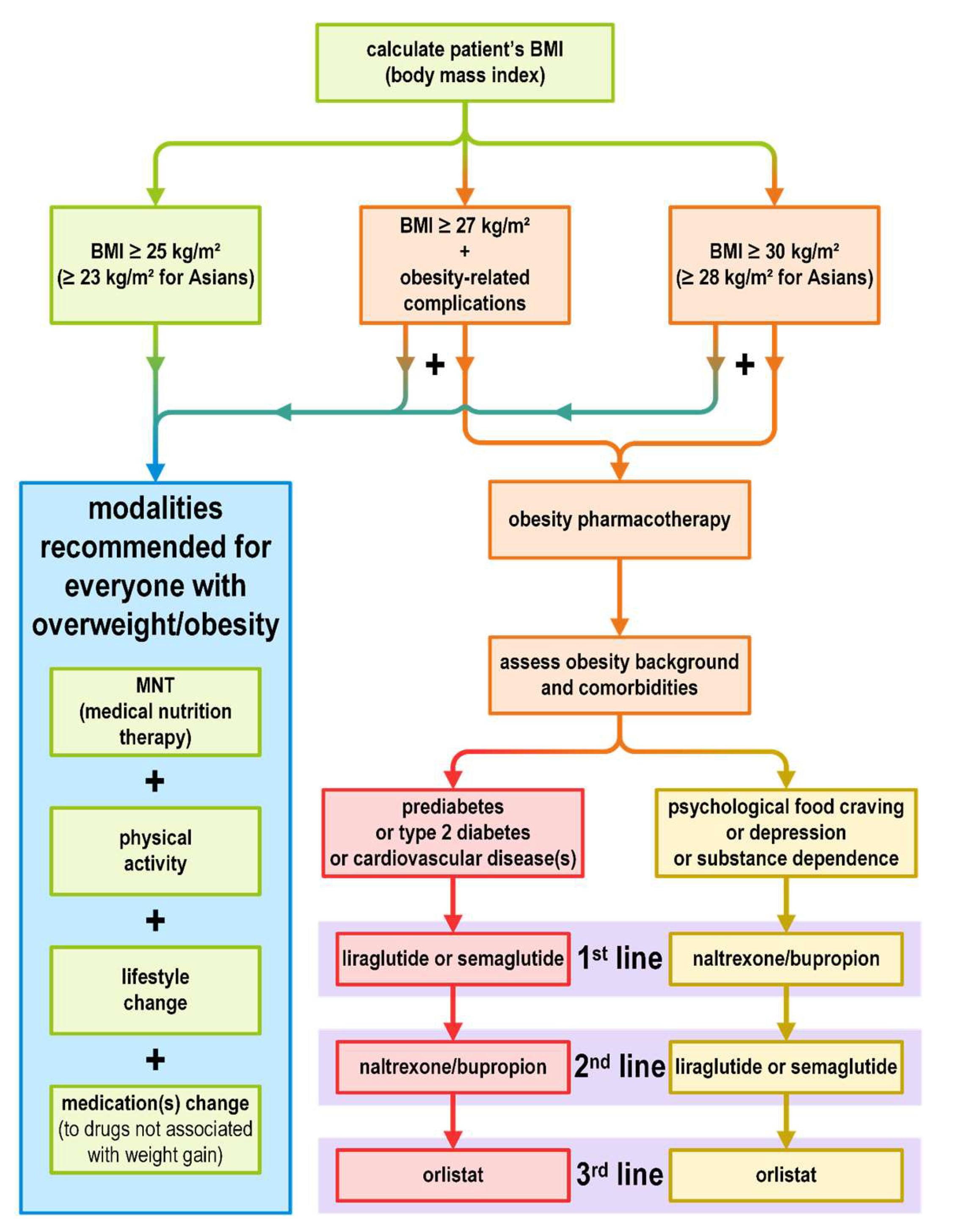
4. Obesity Diagnosis and Treatment Plan
5. Medical Nutrition Therapy (MNT) for Obesity Treatment in T2DM
6. Physical Activity in Obesity Therapy in T2DM
7. Obesity Pharmacotherapy and Antihyperglycemic Treatment
7.1. Antihyperglycemic Agents and Their Impact on Body Weight
7.2. Antihyperglycemic Agents Facilitating Body Weight Reduction
7.2.1. SGLT2is
7.2.2. GLP-1RAs
8. Weight Loss Medications
8.1. Liraglutide and Semaglutide
8.2. Orlistat
8.3. Naltrexone/Bupropion
8.4. Phentermine
8.5. Phentermine/Topiramate
9. Medications for Comorbidities and Their Impact on Body Weight
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vettor, R.; Conci, S. Obesity Pathogenesis. In Obesity: Pathogenesis, Diagnosis, and Treatment; Sbraccia, P., Finer, N., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 89–108. [Google Scholar]
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity and Overweight—Fact Sheets. 2020. Available online: https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 9 May 2023).
- CDC (Centers for Disease Control and Prevention). Defining Adult Overweight and Obesity|Overweight & Obesity. 2017. Available online: https://www.cdc.gov/obesity/adult/defining.html (accessed on 9 May 2023).
- Chiu, M.; Austin, P.C.; Manuel, D.G.; Shah, B.R.; Tu, J.V. Deriving ethnic-specific BMI cutoff points for assessing diabetes risk. Diabetes Care 2011, 34, 1741–1748. [Google Scholar] [CrossRef] [PubMed]
- Corbel, M.J.; Tolari, F.; Yadava, V.K. Appropriate body-mass index for Asian populations and its implications. Lancet 2004, 363, 157–163. [Google Scholar]
- Misra, A. Ethnic-Specific Criteria for Classification of Body Mass Index: A Perspective for Asian Indians and American Diabetes Association Position Statement. Diabetes Technol. Ther. 2015, 17, 667. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.C.; Araneta, M.R.G.; Kanaya, A.M.; Chiang, J.L.; Fujimoto, W. BMI Cut Points to Identify At-Risk Asian Americans for Type 2 Diabetes Screening. Diabetes Care 2015, 38, 150. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.U.; Lee, J.H.; Kim, J.S.; Hwang, Y., II; Kim, T.H.; Lim, S.Y.; Yoo, K.H.; Jung, K.S.; Kim, Y.K.; Rhee, C.K. Comparison of World Health Organization and Asia-Pacific body mass index classifications in COPD patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 2465. [Google Scholar] [CrossRef]
- Engin, A. The Definition and Prevalence of Obesity and Metabolic Syndrome. In Obesity and Lipotoxicity; Engin, A.B., Engin, A., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–17. [Google Scholar]
- WHO/Europe. Body Mass Index—BMI. Available online: https://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 9 May 2023).
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic Report of a WHO Consultation (WHO Technical Report Series 894); World Health Organization: Geneva, Switzerland, 2000; Available online: https://www.who.int/nutrition/publications/obesity/WHO_TRS_894/en/ (accessed on 9 May 2023).
- AMA (American Medical Association). Resolutions. 2013 Annual Meeting. 2013, p. 37 (461). Available online: https://www.ama-assn.org/sites/ama-assn.org/files/corp/media-browser/public/hod/a13-resolutions_0.pdf (accessed on 9 May 2023).
- Care, D.; Suppl, S.S. Obesity management for the treatment of type 2 diabetes: Standards of medical care in diabetes—2020. Diabetes Care 2020, 43 (Suppl. S1), S89–S97. [Google Scholar] [CrossRef]
- American Diabetes Association. Addendum. 8. Obesity Management for the Treatment of Type 2 Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care 2020, 43 (Suppl. S1), S89–S97. [Google Scholar] [CrossRef]
- Lipscombe, L.; Booth, G.; Butalia, S.; Dasgupta, K.; Eurich, D.T.; Goldenberg, R.; Khan, N.; MacCallum, L.; Shah, B.R.; Simpson, S. Pharmacologic Glycemic Management of Type 2 Diabetes in Adults. Can. J. Diabetes 2018, 42, S88–S103. [Google Scholar] [CrossRef]
- Lipscombe, L.; Butalia, S.; Dasgupta, K.; Eurich, D.T.; MacCallum, L.; Shah, B.R.; Simpson, S.; Senior, P.A. Pharmacologic Glycemic Management of Type 2 Diabetes in Adults: 2020 Update. Can. J. Diabetes 2020, 44, 575–591. [Google Scholar] [CrossRef]
- Senior, P.A.; Houlden, R.L.; Kim, J.; Mackay, D.; Nagpal, S.; Rabi, D.; Sherifali, D.; Bajaj, H.S. Pharmacologic Glycemic Management of Type 2 Diabetes in Adults: 2020 Update—The User’s Guide. Can. J. Diabetes 2020, 44, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; D’Alessio, D.A.; Fradkin, J.; Kernan, W.N.; Mathieu, C. Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2018, 61, 2461–2498. [Google Scholar] [CrossRef]
- Professional Practice Committee. Addressing Obesity in Diabetes. Association of Diabetes Care and Education Specialists. 2018, p. 1. Available online: https://www.diabeteseducator.org/docs/default-source/practice/practice-documents/practice-papers/addressing-obesity-in-diabetes.pdf?sfvrsn=0 (accessed on 9 May 2023).
- Wharton, S.; Lau, D.C.W.; Vallis, M.; Sharma, A.M.; Biertho, L.; Campbell-Scherer, D.; Adamo, K.; Alberga, A.; Bell, R.; Boulé, N.; et al. Obesity in adults: A clinical practice guideline. Cmaj 2020, 192, E875–E891. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Jablonski, K.A.; Fujimoto, W.Y.; Barrett-Connor, E.; Haffner, S.; Hanson, R.L.; Hill, J.O.; Hubbard, V.; Kriska, A.; Stamm, E.; et al. Relation of central adiposity and body mass index to the development of diabetes in the Diabetes Prevention Program. Am. J. Clin. Nutr. 2008, 87, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Williamson, D.F.; Liu, S. Weight change and diabetes incidence: Findings from a national cohort of US adults. Am. J. Epidemiol. 1997, 146, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Holecki, M.; Żak-Gołąb, A.; Zahorska-Markiewicz, B. Otyłość a Cukrzyca. Wzajemna Zależność Między Otyłością a Cukrzycą. [Obesity and diabetes. Interaction between obesity and diabetes]. In Cukrzyca [Diabetes], 3rd ed.; Sieradzki, J., Ed.; Via Medica: Gdańsk, Poland, 2019; pp. 223–237. [Google Scholar]
- Anderwald, C.; Bernroider, E.; Krššák, M.; Stingl, H.; Brehm, A.; Bischof, M.G.; Nowotny, P.; Roden, M.; Waldhäusl, W. Effects of insulin treatment in type 2 diabetic patients on intracellular lipid content in liver and skeletal muscle. Diabetes 2002, 51, 3025–3032. [Google Scholar] [CrossRef]
- Muoio, D.M.; Newgard, C.B. Mechanisms of disease: Molecular and metabolic mechanisms of insulin resistance and β-cell failure in type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 193–205. [Google Scholar] [CrossRef]
- Zierath, J.R.; Krook, A.; Wallberg-Henriksson, H. Insulin action in skeletal muscle from patients with NIDDM. Mol. Cell. Biochem. 1998, 182, 153–160. [Google Scholar] [CrossRef]
- Mahgoub, M.O.; Ali, I.I.; Adeghate, J.O.; Kal, H.; Adeghate, E.A. An Update on the Molecular and Cellular Basis of Pharmacotherapy in Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2023, 24, 9328. [Google Scholar] [CrossRef]
- Goodyear, L.J.; Smith, R.J.; Dohm, G.L.; Goodyear, L.J.; Giorgino, F.; Sherman, L.A.; Carey, J.; Smith, R.J.; Dohm, G.L. Insulin receptor phosphorylation, insulin receptor substrate-1 phosphorylation, and phosphatidylinositol 3-kinase activity are decreased in intact skeletal muscle strips from obese subjects. J. Clin. Investig. 1995, 95, 2195–2204. [Google Scholar] [CrossRef]
- Bjornholm, M.; Kawano, Y.; Lehtihet, M.; Zierath, J.R. Insulin Receptor Substrate-1 Phosphorylation and Phosphatidylinositol 3-Kinase Activity in Skeletal Muscle From NIDDM Subjects After In Vivo Insulin Stimulation. Diabetes 1997, 46, 524–527. [Google Scholar] [CrossRef] [PubMed]
- Zaid, H.; Antonescu, C.N.; Randhawa, V.K.; Klip, A. Insulin action on glucose transporters through molecular switches, tracks and tethers. Biochem. J. 2008, 413, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Shulman, G.I.; Shulman, G.I. Cellular mechanisms of insulin resistance. J. Clin. Investig. 2020, 106, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Herzberg-Schäfer, S.; Heni, M.; Stefan, N.; Häring, H.-U.; Fritsche, A. Impairment of GLP1-induced insulin secretion: Role of genetic background, insulin resistance and hyperglycaemia. Diabetes Obes. Metab. 2012, 14 (Suppl. S3), 85–90. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; McDaniel, M.L. Identifying the links between obesity, insulin resistance and β-cell function: Potential role of adipocyte-derived cytokines in the pathogenesis of type 2 diabetes. Eur. J. Clin. Investig. 2002, 32, 24–34. [Google Scholar] [CrossRef]
- Unger, R.H. Lipotoxic Diseases. Annu. Rev. Med. 2002, 53, 319–336. [Google Scholar] [CrossRef]
- Adeghate, E. An update on the biology and physiology of resistin. Cell. Mol. Life Sci. C 2004, 61, 2485–2496. [Google Scholar] [CrossRef]
- Scheen, A.J. Pathophysiology of type 2 diabetes. Acta Clin. Belg. 2003, 58, 335–341. [Google Scholar] [CrossRef]
- Bastard, J.-P.; Maachi, M.; Lagathu, C.; Kim, M.J.; Caron, M.; Vidal, H.; Capeau, J.; Feve, B. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur. Cytokine Netw. 2006, 17, 4–12. [Google Scholar]
- Rehman, K.; Akash, M.S.H. Mechanisms of inflammatory responses and development of insulin resistance: How are they interlinked? J. Biomed. Sci. 2016, 23, 87. [Google Scholar] [CrossRef]
- Nauck, M.; Stöckmann, F.; Ebert, R.; Creutzfeldt, W. Reduced incretin effect in type 2 (non-insulin-dependent) diabetes. Diabetologia 1986, 29, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Fehmann, H.C.; Göke, R.; Göke, B. Cell and molecular biology of the incretin hormones glucagon-like peptide-I and glucose-dependent insulin releasing polypeptide. Endocr. Rev. 1995, 16, 390–410. [Google Scholar] [CrossRef] [PubMed]
- Polonsky, K.S. The β-cell in diabetes: From molecular genetics to clinical research. Diabetes 1995, 44, 705–717. [Google Scholar] [CrossRef] [PubMed]
- Surampudi, P.N.; John-Kalarickal, J.; Fonseca, V.A. Emerging concepts in the pathophysiology of type 2 diabetes mellitus. Mt. Sinai J. Med. 2009, 76, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Westermark, P.; Johnson, K.H.; O’Brien, T.D.; Betsholtz, C. Islet amyloid polypeptide--a novel controversy in diabetes research. Diabetologia 1992, 35, 297–303. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Maleki, M.; Sathyapalan, T.; Jamialahmadi, T.; Sahebkar, A. Pathophysiology of Physical Inactivity-Dependent Insulin Resistance: A Theoretical Mechanistic Review Emphasizing Clinical Evidence. J. Diabetes Res. 2021, 2021, 7796727. [Google Scholar] [CrossRef]
- Rodin, J. Insulin levels, hunger, and food intake: An example of feedback loops in body weight regulation. Health Psychol. 1985, 4, 1–24. [Google Scholar] [CrossRef]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of cardiology/American Heart Association task force on practice guidelines and the obesity society. Circulation 2014, 129, 102–141. [Google Scholar] [CrossRef]
- Luig, T.; Anderson, R.; Sharma, A.M.; Campbell-Scherer, D.L. Personalizing obesity assessment and care planning in primary care: Patient experience and outcomes in everyday life and health. Clin. Obes. 2018, 8, 411–423. [Google Scholar] [CrossRef]
- Koliaki, C.; Spinos, T.; Spinou, Μ.; Brinia, Μ.-E.; Mitsopoulou, D.; Katsilambros, N. Defining the Optimal Dietary Approach for Safe, Effective and Sustainable Weight Loss in Overweight and Obese Adults. Healthcare 2018, 6, 73. [Google Scholar] [CrossRef]
- Warren, J.; Smalley, B.; Barefoot, N. Higher motivation for weight loss in African American than caucasian rural patients with hypertension and/or diabetes. Ethn. Dis. 2016, 26, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Davidson, P.G.; Burns, H.; Nadelson, M.D.H.; Mesznik, S.; Uhley, V.; Moloney, L. Medical Nutrition Therapy and Weight Loss Questions for the Evidence Analysis Library Prevention of Type 2 Diabetes Project: Systematic Reviews. J. Acad. Nutr. Diet. 2017, 117, 1578–1611. [Google Scholar] [CrossRef] [PubMed]
- Mabire, L.; Mani, R.; Liu, L.; Mulligan, H.; Baxter, D. The Influence of Age, Sex and Body Mass Index on the Effectiveness of Brisk Walking for Obesity Management in Adults: A Systematic Review and Meta-Analysis. J. Phys. Act. Health 2017, 14, 389–407. [Google Scholar] [CrossRef] [PubMed]
- Wadden, T.A. Eight-year weight losses with an intensive lifestyle intervention: The look AHEAD study. Obesity 2014, 22, 5–13. [Google Scholar] [CrossRef]
- Pastors, J.G.; Warshaw, H.; Daly, A.; Franz, M.; Kulkarni, K. The evidence for the effectiveness of medical nutrition therapy in diabetes management. Diabetes Care 2002, 25, 608–613. [Google Scholar] [CrossRef]
- Rothberg, A.E.; McEwen, L.N.; Kraftson, A.T.; Ajluni, N.; Fowler, C.E.; Nay, C.K.; Miller, N.M.; Burant, C.F.; Herman, W.H. Impact of weight loss on waist circumference and the components of the metabolic syndrome. BMJ Open Diabetes Res. Care 2017, 5, 3–8. [Google Scholar] [CrossRef]
- Care, D.; Suppl, S.S. Glycemic targets: Standards of medical care in diabetes-2020. Diabetes Care 2020, 43, S66–S76. [Google Scholar] [CrossRef]
- Baum, A.; Scarpa, J.; Bruzelius, E.; Tamler, R.; Basu, S.; Faghmous, J. Targeting weight loss interventions to reduce cardiovascular complications of type 2 diabetes: A machine learning-based post-hoc analysis of heterogeneous treatment effects in the Look AHEAD trial. Lancet Diabetes Endocrinol. 2017, 5, 808–815. [Google Scholar] [CrossRef]
- Lean, M.E.J.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019, 7, 344–355. [Google Scholar] [CrossRef]
- Wadden, T.A. The look AHEAD study: A description of the lifestyle intervention and the evidence supporting it. Obesity 2006, 14, 737–752. [Google Scholar] [CrossRef]
- Pi-Sunyer, X. The Look AHEAD Trial: A Review and Discussion of Its Outcomes. Curr. Nutr. Rep. 2014, 3, 387–391. [Google Scholar] [CrossRef]
- Hamdy, O.; Ganda, O.P.; Maryniuk, M.; Gabbay, R.A. Chapter 2. Clinical nutrition guideline for overweight and obese adults with type 2 diabetes (T2D) or prediabetes, or those at high risk for developing T2D. Am. J. Manag. Care 2018, 24, SP226–SP231. [Google Scholar] [PubMed]
- Esposito, K.; Maiorino, M.I.; Petrizzo, M.; Bellastella, G.; Giugliano, D. The effects of a Mediterranean diet on the need for diabetes drugs and remission of newly diagnosed type 2 diabetes: Follow-up of a randomized trial. Diabetes Care 2014, 37, 1824–1830. [Google Scholar] [CrossRef]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y. Weight Loss with a Low-Carbohydrate, Mediterranean, or Low-Fat Diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef]
- Sainsbury, E.; Kizirian, N.V.; Partridge, S.R.; Gill, T.; Colagiuri, S.; Gibson, A.A. Effect of dietary carbohydrate restriction on glycemic control in adults with diabetes: A systematic review and meta-analysis. Diabetes Res. Clin. Pract. 2018, 139, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Fard, N.R.P.; Karimi, M.; Baghaei, M.H.; Surkan, P.J.; Rahimi, M.; Esmaillzadeh, A.; Willett, W.C. Effects of the Dietary Approaches to Stop Hypertension (DASH) eating plan on cardiovascular risks among type 2 diabetic patients: A randomized crossover clinical trial. Diabetes Care 2011, 34, 55–57. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Barnard, N.D.; Levin, S.M.; Watanabe, M. Vegetarian diets and glycemic control in diabetes: A systematic review and meta-analysis. Cardiovasc. Diagn. Ther. 2014, 4, 373–382. [Google Scholar] [CrossRef]
- Johansson, K.; Neovius, M.; Hemmingsson, E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 99, 14–23. [Google Scholar] [CrossRef]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): An open-label, cluster-randomised trial. Lancet 2018, 391, 541–551. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Barquera, S.; Duhamel, J.-F.; Hardinsyah, R.; Haslam, D.; Lafontan, M. Recommendations for healthier hydration: Addressing the public health issues of obesity and type 2 diabetes. Clin. Obes. 2012, 2, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Missbach, B.; Dias, S.; König, J.; Hoffmann, G. Impact of different training modalities on glycaemic control and blood lipids in patients with type 2 diabetes: A systematic review and network meta-analysis. Diabetologia 2014, 57, 1789–1797. [Google Scholar] [CrossRef]
- Chudyk, A.; Petrella, R.J. Effects of exercise on cardiovascular risk factors in type 2 diabetes: A meta-analysis. Diabetes Care 2011, 34, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Boulé, N.; Prud’homme, D.; Physical Activity in Obesity Management. Canadian Adult Obesity Clinical Practice Guidelines. 2011, pp. 1–23. Available online: https://obesitycanada.ca/guidelines/physicalactivity (accessed on 9 May 2023).
- Washburn, R.A.; Szabo, A.N.; Lambourne, K.; Willis, E.A.; Ptomey, L.T.; Honas, J.J.; Herrmann, S.D.; Donnelly, J.E. Does the method of weight loss effect long-term changes in weight, body composition or chronic disease risk factors in overweight or obese adults? A systematic review. PLoS ONE 2014, 9, e109849. [Google Scholar] [CrossRef] [PubMed]
- Borek, A.J.; Abraham, C.; Greaves, C.J.; Tarrant, M. Group-Based Diet and Physical Activity Weight-Loss Interventions: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Appl. Psychol. Health Well-Being 2018, 10, 62–86. [Google Scholar] [CrossRef] [PubMed]
- Lemes, Í.R.; Turi-Lynch, B.C.; Cavero-Redondo, I.; Linares, S.N.; Monteiro, H.L. Aerobic training reduces blood pressure and waist circumference and increases HDL-c in metabolic syndrome: A systematic review and meta-analysis of randomized controlled trials. J. Am. Soc. Hypertens. 2018, 12, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Umpierre, D.; Ribeiro, P.A.; Kramer, C.K. Physical Activity Advice Only or Structured Exercise Training and Association with HbA1c Levels in Type 2 Diabetes: A Systematic Review and Meta-analysis. JAMA—J. Am. Med. Assoc. Am. Med. Assoc. 2011, 305, 1790–1799. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. Fam. Med. Prim. Care Rev. 2006, 174, 801–809. [Google Scholar] [CrossRef]
- Madjd, A.; Taylor, M.A.; Neek, L.S.; Delavari, A.; Malekzadeh, R.; MacDonald, I.A.; Farshchi, H.R. Effect of weekly physical activity frequency on weight loss in healthy overweight and obese women attending a weight loss program: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 104, 1202–1208. [Google Scholar] [CrossRef]
- Yang, Z.; Scott, C.A.; Mao, C.; Tang, J.; Farmer, A.J. Resistance exercise versus aerobic exercise for type 2 diabetes: A systematic review and meta-analysis. Sport. Med. 2014, 44, 487–499. [Google Scholar] [CrossRef]
- Dunstan, D.W.; Kingwell, B.A.; Larsen, R.; Healy, G.N.; Cerin, E.; Hamilton, M.T.; Shaw, J.E.; Bertovic, D.A.; Zimmet, P.Z.; Salmon, J.; et al. Breaking up prolonged sitting reduces postprandial glucose and insulin responses. Diabetes Care 2012, 35, 976–983. [Google Scholar] [CrossRef]
- American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: Standards of medical care in diabetes―2020. Diabetes Care 2020, 43 (Suppl. S1), S98–S110. [Google Scholar] [CrossRef]
- Matyjaszek-Matuszek, B.; Szafraniec, A.; Porada, D. Pharmacotherapy of obesity—State of the art. Endokrynol. Pol. 2018, 69, 448–457. [Google Scholar] [CrossRef]
- American Diabetes Association. Addendum. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2020. Diabetes Care 2020, 43 (Suppl. S1), S98–S110. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Zhu, Q.Q.; Chen, Y.H.; Li, X.L.; Chen, F.; Huang, J.A.; Xu, B. Cardiovascular safety, long-term noncardiovascular safety, and efficacy of sodium-glucose cotransporter 2 inhibitors in patients with type 2 diabetes mellitus: A systemic review and meta-analysis with trial sequential analysis. J. Am. Heart Assoc. 2018, 7, e007165. [Google Scholar] [CrossRef]
- FDA. FDA Approves New Treatment for a Type of Heart Failure. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatment-type-heart-failure (accessed on 9 May 2023).
- FDA. FDA Approves Treatment for Chronic Kidney Disease. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-treatment-chronic-kidney-disease (accessed on 9 May 2023).
- McMurray, J.J.V.; Solomon, S.D.; Inzucchi, S.E.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; Bělohlávek, J.; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef]
- Solomon, S.D.; McMurray, J.J.V.; Claggett, B.; de Boer, R.A.; DeMets, D.; Hernandez, A.F.; Inzucchi, S.E.; Kosiborod, M.N.; Lam, C.S.P.; Martinez, F.; et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N. Engl. J. Med. 2022, 387, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef]
- Jabbour, S.; Seufert, J.; Scheen, A.; Bailey, C.J.; Karup, C.; Langkilde, A.M. Dapagliflozin in patients with type 2 diabetes mellitus: A pooled analysis of safety data from phase IIb/III clinical trials. Diabetes Obes. Metab. 2018, 20, 620–628. [Google Scholar] [CrossRef]
- Yin, N.; Zhang, H.; Ye, R.; Dong, M.; Lin, J.; Zhou, H.; Huang, Y.; Chen, L.; Jiang, X.; Nagaoka, K.; et al. Fluvastatin sodium ameliorates obesity through brown fat activation. Int. J. Mol. Sci. 2019, 20, 1622. [Google Scholar] [CrossRef]
- FDA. FDA Approves First Oral GLP-1 Treatment for Type 2 Diabetes. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-oral-glp-1-treatment-type-2-diabetes (accessed on 9 May 2023).
- Thrasher, J. Pharmacologic Management of Type 2 Diabetes Mellitus: Available Therapies. Am. J. Med. 2017, 130, S4–S17. [Google Scholar] [CrossRef]
- Pi-Sunyer, X.; Astrup, A.; Fujioka, K.; Greenway, F.; Halpern, A.; Krempf, M.; Lau, D.C.W.; le Roux, C.W.; Violante Ortiz, R.; Jensen, C.B.; et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. N. Engl. J. Med. 2015, 373, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.; Manjoo, P.; Wharton, S. Canadian Adult Obesity Clinical Practice Guidelines: Pharmacotherapy in Obesity Management. Available online: https://obesitycanada.ca/guidelines/pharmacotherapy (accessed on 9 May 2023).
- Pratley, R.E.; Aroda, V.R.; Lingvay, I.; Lüdemann, J.; Andreassen, C.; Navarria, A.; Viljoen, A. Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): A randomised, open-label, phase 3b trial. Lancet Diabetes Endocrinol. 2018, 6, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Htike, Z.Z.; Zaccardi, F.; Papamargaritis, D.; Webb, D.R.; Khunti, K.; Davies, M.J. Efficacy and safety of glucagon-like peptide-1 receptor agonists in type 2 diabetes: A systematic review and mixed-treatment comparison analysis. Diabetes Obes. Metab. 2017, 19, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Wynne, A.G.; Matyjaszek-Matuszek, B.; Bartaskova, D.; Cox, D.A.; Woodward, B.; Li, Y.G.; Tham, L.S.; Milicevic, Z. Efficacy and safety of an expanded dulaglutide dose range: A phase 2, placebo-controlled trial in patients with type 2 diabetes using metformin. Diabetes Obes. Metab. 2019, 21, 2048–2057. [Google Scholar] [CrossRef]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L.; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.; Brown-Frandsen, K. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef]
- Gerstein, H.C.; Colhoun, H.M.; Dagenais, G.R.; Diaz, R.; Lakshmanan, M.; Pais, P.; Probstfield, J.; Riesmeyer, J.S.; Riddle, M.C.; Rydén, L.; et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): A double-blind, randomised placebo-controlled trial. Lancet 2019, 394, 121–130. [Google Scholar] [CrossRef]
- Storgaard, H.; Cold, F.; Gluud, L.L.; Vilsbøll, T.; Knop, F.K. Glucagon-like peptide-1 receptor agonists and risk of acute pancreatitis in patients with type 2 diabetes. Diabetes Obes. Metab. 2017, 19, 906–908. [Google Scholar] [CrossRef]
- Monami, M.; Nreu, B.; Scatena, A.; Cresci, B. Safety issues with glucagon-like peptide-1 receptor agonists: Pancreatitis, pancreatic cancer, and cholelithiasis. Data from randomised controlled trials. Diabetes Obes. Metab. 2017, 19, 1233–1241. [Google Scholar] [CrossRef]
- EMA (European Medicines Agency). Saxenda: EPAR—Product Information. 2022. Available online: https://www.ema.europa.eu/documents/product-information/saxenda-epar-product-information_en.pdf (accessed on 15 June 2023).
- Knudsen, L.B.; Madsen, L.W.; Andersen, S.; Almholt, K.; De Boer, A.S.; Drucker, D.J.; Gotfredsen, C.; Egerod, F.L.; Hegelund, A.C.; Jacobsen, H.; et al. Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation. Endocrinology 2010, 151, 1473–1486. [Google Scholar] [CrossRef]
- Bezin, J.; Gouverneur, A.; Pénichon, M.; Mathieu, C.; Garrel, R.; Hillaire-Buys, D.; Pariente, A.; Faillie, J.-L. GLP-1 Receptor Agonists and the Risk of Thyroid Cancer. Diabetes Care 2022, 46, 384–390. [Google Scholar] [CrossRef]
- Thompson, C.A.; Stüurmer, T. Putting GLP-1 RAs and Thyroid Cancer in Context: Additional Evidence and Remaining Doubts. Diabetes Care 2023, 46, 249–251. [Google Scholar] [CrossRef]
- Chiu, W.Y.; Shih, S.R.; Tseng, C.H. A review on the association between glucagon-like peptide-1 receptor agonists and thyroid cancer. Exp. Diabetes Res. 2012, 2012, 924168. [Google Scholar] [CrossRef] [PubMed]
- EMA (European Medicines Agency). Wegovy: EPAR—Product Information. 2023. Available online: https://www.ema.europa.eu/documents/overview/wegovy-epar-medicine-overview_en.pdf (accessed on 15 June 2023).
- FDA. FDA Approves New Drug Treatment for Chronic Weight Management, First Since 2014. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-treatment-chronic-weight-management-first-2014 (accessed on 9 May 2023).
- Rubino, D.M.; Greenway, F.L.; Khalid, U.; O’Neil, P.M.; Rosenstock, J.; Sørrig, R.; Wadden, T.A.; Wizert, A.; Garvey, W.T. Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults With Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA 2022, 327, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.G.; Park, C.Y.; Kang, J.H.; Park, Y.W.; Park, S.W. Randomized controlled trial to investigate the effects of a newly developed formulation of phentermine diffuse-controlled release for obesity. Diabetes Obes. Metab. 2010, 12, 876–882. [Google Scholar] [CrossRef]
- Allison, D.B.; Gadde, K.M.; Garvey, W.T.; Peterson, C.A.; Schwiers, M.L.; Najarian, T.; Tam, P.Y.; Troupin, B.; Day, W.W. Controlled-release phentermine/topiramate in severely obese adults: A randomized controlled trial (EQUIP). Obesity 2012, 20, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Elhag, W.; El Ansari, W.; Razaq, S.; Elsherif, M.; Mustafa, I. Lorcaserin vs. Phentermine among non-surgical and surgical obese patients: Anthropometric, glycemic, lipid, safety and cost outcomes. Ann. Med. Surg. 2019, 45, 75–81. [Google Scholar] [CrossRef]
- Kahan, S.; Fujioka, K. Obesity pharmacotherapy in patients with type 2 diabetes. Diabetes Spectr. 2017, 30, 250–257. [Google Scholar] [CrossRef]
- EMA (European Medicines Agency). Xenical: EPAR—Product Information. 2023. Available online: https://www.ema.europa.eu/documents/product-information/xenical-epar-product-information_en.pdf (accessed on 15 June 2023).
- EMA (European Medicines Agency). Mysimba: EPAR—Product Information. 2023. Available online: https://www.ema.europa.eu/documents/product-information/mysimba-epar-product-information_en.pdf (accessed on 15 June 2023).
- Heck, A.M.; Yanovski, J.A.; Calis, K.A. Orlistat, a new lipase inhibitor for the management of obesity. Pharmacotherapy 2000, 20, 270–279. [Google Scholar] [CrossRef]
- Sjöström, L. Analysis of the XENDOS study (Xenical in the Prevention of Diabetes in Obese Subjects). Endocr. Pract. 2006, 12, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Torgerson, J.; Hauptman, J.; Boldrin, M.; Sjöström, L. Xenical in the prevention of diabetes in obese subjects (XENDOS) study. Diabetes Care 2004, 27, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Ryan, D.H. Medical therapy for the patient with obesity. Circulation 2012, 125, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Billes, S.K.; Sinnayah, P.; Cowley, M.A. Naltrexone/bupropion for obesity: An investigational combination pharmacotherapy for weight loss. Pharmacol. Res. 2014, 84, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Greenway, F.L.; Whitehouse, M.J.; Guttadauria, M.; Anderson, J.W.; Atkinson, R.L.; Fujioka, K.; Gadde, K.M.; Gupta, A.K.; O’Neil, P.; Schumacher, D.; et al. Rational design of a combination medication for the treatment of obesity. Obesity 2009, 17, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Bjornsson, T.D.; Callaghan, J.T.; Einolf, H.J.; Fischer, V.; Gan, L.; Grimm, S.; Kao, J.; King, S.P.; Miwa, G.; Ni, L.; et al. The conduct of in vitro and in vivo drug-drug interaction studies: A PhRMA perspective. J. Clin. Pharmacol. 2003, 43, 443–469. [Google Scholar] [CrossRef] [PubMed]
- Rothman, R.B.; Baumann, M.H.; Dersch, C.M.; Romero, D.V.; Rice, K.C.; Carroll, F.I.V.Y.; Partilla, J.S. Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 2001, 39, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.; Picard, F.; Lemieux, C.; Lalonde, J.; Samson, P.; Deshaies, Y. The effects of topiramate and sex hormones on energy balance of male and female rats. Int. J. Obes. 2002, 26, 344–353. [Google Scholar] [CrossRef]
- Domecq, J.P.; Prutsky, G.; Leppin, A.; Sonbol, M.B.; Altayar, O.; Undavalli, C.; Wang, Z.; Elraiyah, T.; Brito, J.P.; Mauck, K.F.; et al. Drugs commonly associated with weight change: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2015, 100, 363–370. [Google Scholar] [CrossRef]
- Keith, S.W.; Redden, D.T.; Katzmarzyk, P.T.; Boggiano, M.M.; Hanlon, E.C.; Benca, R.M.; Ruden, D.; Pietrobelli, A.; Barger, J.L.; Fontaine, K.R.; et al. Putative contributors to the secular increase in obesity: Exploring the roads less traveled. Int. J. Obes. 2006, 30, 1585–1594. [Google Scholar] [CrossRef]
- Rynn, M.; Russell, J.; Erickson, J.; Detke, M.J.; Ball, S.; Dinkel, J.; Rickels, K.; Raskin, J. Efficacy and safety of duloxetine in the treatment of generalized anxiety disorder: A flexible-dose, progressive-titration, placebo-controlled trial. Depress. Anxiety 2008, 25, 182–189. [Google Scholar] [CrossRef]
- Nemeroff, C.B.; Thase, M.E. A double-blind, placebo-controlled comparison of venlafaxine and fluoxetine treatment in depressed outpatients. J. Psychiatr. Res. 2007, 41, 351–359. [Google Scholar] [CrossRef] [PubMed]
| Classification | BMI Value (kg/m2) | WC (cm) | Risk of Developing Body Mass Related Diseases | |||
|---|---|---|---|---|---|---|
| Non-Asian | Asian | Non-Asian | Asian | |||
| underweight | <18.5 | <17.5 | nd * | nd * | ↑ | |
| normal weight | 18.5–24.9 | 17.5–22.9 | nd * | nd * | N | |
| overweight | 25.0–29.9 | 23.0–27.9 | nd * | nd * | ↑ | |
| obesity | class I | 30.0–34.9 | 28.0–32.4 | ♀ > 88 * ♂ > 102 * | ♀ > 80 * ♂ > 90 * | ↑↑ |
| class I | 35.0–39.9 | 32.5–37.4 | nd * | nd * | ↑↑↑ | |
| class III | ≥40.0 | ≥37.5 | nd * | nd * | ↑↑↑↑ | |
| Drug Class | Mechanism of Action | Drug Sub-Class | Drugs | Weight Change | HbA1c Decrease [mmol/mol] | Cost | |
|---|---|---|---|---|---|---|---|
| biguanide | ↑ insulin sensitivity ↓ hepatic glucose production | metformin (conventional or extended-release) | NC | 10.9 | $ | ||
| incretin | ↑ satiety (only GLP1-RA), ↑ glucose-dependent insulin secretion ↓ stomach emptying ↓ glucagon secretion | GLP1 receptor agonist | short-acting: exenatide, lixisenatide, liraglutide † long-acting: dulaglutide, exenatide extended-release, semaglutide † | ↓ 1.1–4.4 kg | 6.6–15.3 | $$$$ | |
| DPP4 inhibitor | sitagliptin, vildagliptin, saxagliptin, linagliptin, alogliptin | NC | 5.5–7.6 | $$$ | |||
| SGLT2 inhibitor | ↓ renal glucose resorption | canagliflozin, dapagliflozin, empagliflozin, ertugliflozin | ↓ 2.0–3.0 kg | 5.5–7.6 | $$$ | ||
| α-glucosidase inhibitor | ↓ carbohydrate digestion and absorption (↓ action of α-glucosidase and α-amylase) | acarbose, miglitol | NC | 7.6–8.7 | $$ | ||
| insulin | direct substitution of endogenous insulin action | rapid-acting | aspart (conventional and fast-acting), glulisine, lispro | ↑ * | ≥ 9.8 * | $–$$$$ * | |
| intermediate-acting | human NPH | ||||||
| long-acting | degludec, detemir, glargine | ||||||
| insulin + GLP1-RA | (see GLP1-RA and insulin sections) | degludec/liraglutide, glargine/lixisenatide | NC | ND | $$$$ | ||
| insulin secretagogue | ↑ insulin secretion (glucose-independent) | sulfonylurea | glibenclamide (glyburide), glipizide, gliclazide, glimepiride | ↑ 1.2–3.2 kg | 6.6–13.1 | $ | |
| meglitinide | repaglinide, nateglinide | ↑ 1.4–3.3 kg | 7.6–12.0 | $$ | |||
| TZD | ↑ insulin sensitivity (activation of PPAR-γ receptors) | pioglitazone, rosiglitazone | ↑ 2.0–2.5 kg | 7.6–9.8 | $$$ | ||
| Orlistat | Liraglutide, Semaglutide † | Naltrexone/Bupropion | Phentermine/Topiramate | Phentermine ‡ | |||
|---|---|---|---|---|---|---|---|
| mechanism of action [20] | gastric/pancreatic lipase inhibitor | GLP1 receptor agonist | opioid receptor antagonist + NDRI | sympathomimetic + CAI and GABA/amine modulator | sympathomimetic | ||
| route of administration | oral | subcutaneous | oral | oral | oral | ||
| target dose | 120 mg | 3.0 mg (lirglutide) 2.4 mg (semaglutide) | 16 mg/180 mg | 15 mg/92 mg | 15 mg | ||
| dosage frequency | three times daily | once daily (liraglutide)once weekly (semaglutide) | twice daily | once daily | once daily | ||
| long-term weight change vs. placebo | % | −4.0% [14], −2.9% [95] | liraglutide: −4.0% [14], −5.4% [95], from −6.0% to −8.0% [20] semaglutide: −15.8% * [111] | −4.2% [14], −4.8% [95], from −8.2% to −11.5% [20] | −8.6% [14], −7.3% [112], −9.4% [113] §, from −5.1% to −10.9% [20] | −4.9% [14] (short-term effect) | |
| kg | −2.8 kg, −3.0 kg [20] | liraglutide: −4.2 kg; −6.8 kg * [111] semaglutide: −15.3 kg * [111] | ND | −6.4 kg [112] | ND | ||
| change in T2DM development risk | ↓ (−37.3%) | ↓ (−79.0%) | ND | ND | ND | ||
| effect on HbA1c [mmol/mol] | −4.4 | −10.9 | −5.5 | ND | NC/NS [114] | ||
| effect on lipids | TC | ↓ | ↓ | ND | ↓ [112,113] | NC/NS [114] | |
| LDL | ↓ | ↓ | ↓ | ↓ [112,113] | NC/NS [114] | ||
| non-HDL | ND | ↓ | ND | ND | ND | ||
| HDL | NC/NS | ↑ | ↑ | NC/NS [112]/↑ [113] | NC/NS [114] | ||
| TG | NC/NS | ↓ | ↓ | NC/NS [112]/↓ [113] | NC/NS [114] | ||
| cost of therapy (per month) | $$ | $$$$ | $$$ | $$ | $ | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łuniewski, M.; Matyjaszek-Matuszek, B.; Lenart-Lipińska, M. Diagnosis and Non-Invasive Treatment of Obesity in Adults with Type 2 Diabetes Mellitus: A Review of Guidelines. J. Clin. Med. 2023, 12, 4431. https://doi.org/10.3390/jcm12134431
Łuniewski M, Matyjaszek-Matuszek B, Lenart-Lipińska M. Diagnosis and Non-Invasive Treatment of Obesity in Adults with Type 2 Diabetes Mellitus: A Review of Guidelines. Journal of Clinical Medicine. 2023; 12(13):4431. https://doi.org/10.3390/jcm12134431
Chicago/Turabian StyleŁuniewski, Michał, Beata Matyjaszek-Matuszek, and Monika Lenart-Lipińska. 2023. "Diagnosis and Non-Invasive Treatment of Obesity in Adults with Type 2 Diabetes Mellitus: A Review of Guidelines" Journal of Clinical Medicine 12, no. 13: 4431. https://doi.org/10.3390/jcm12134431
APA StyleŁuniewski, M., Matyjaszek-Matuszek, B., & Lenart-Lipińska, M. (2023). Diagnosis and Non-Invasive Treatment of Obesity in Adults with Type 2 Diabetes Mellitus: A Review of Guidelines. Journal of Clinical Medicine, 12(13), 4431. https://doi.org/10.3390/jcm12134431





