Cardiac Rhythm Monitoring Using Wearables for Clinical Guidance before and after Catheter Ablation
Abstract
:1. Introduction
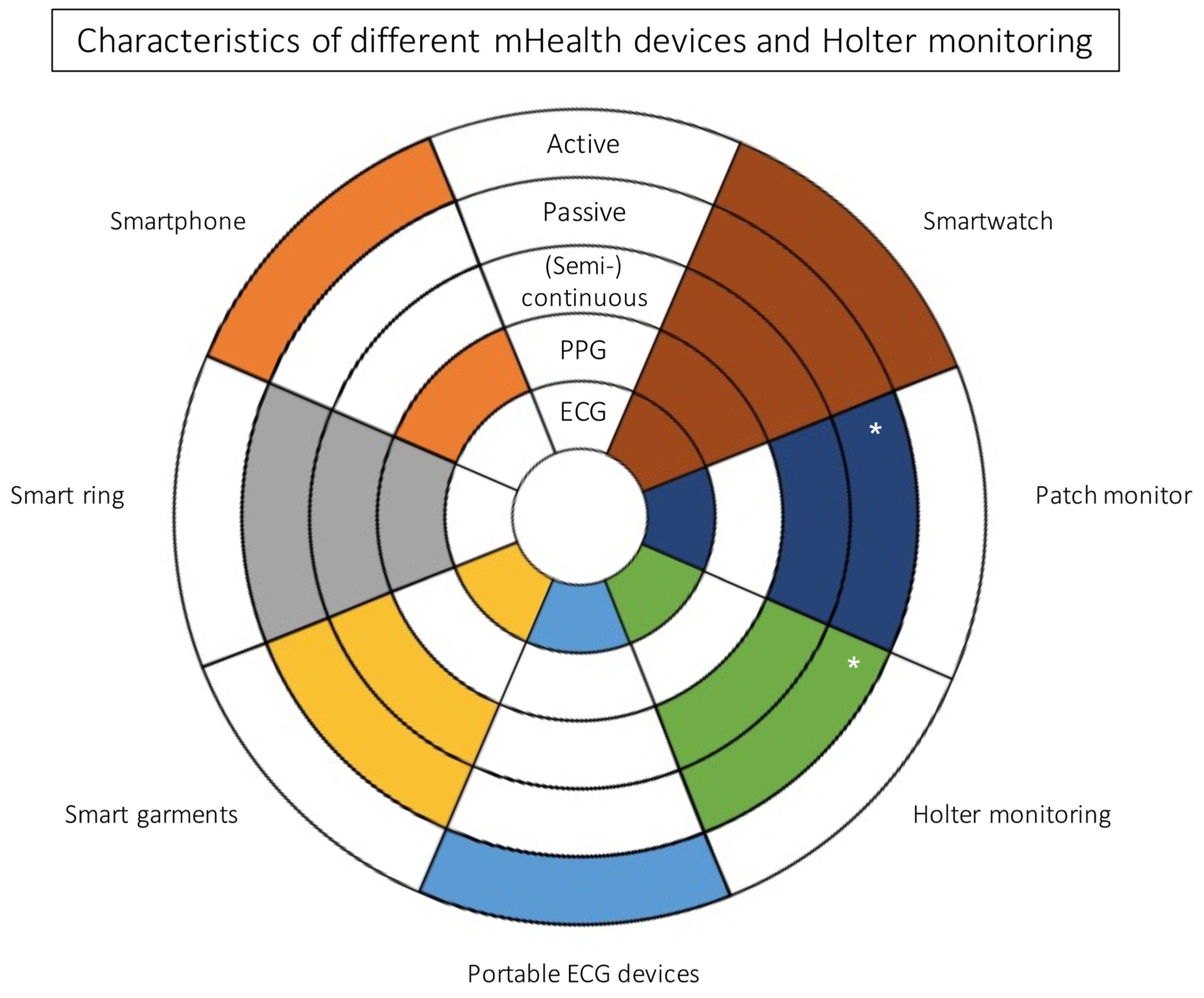
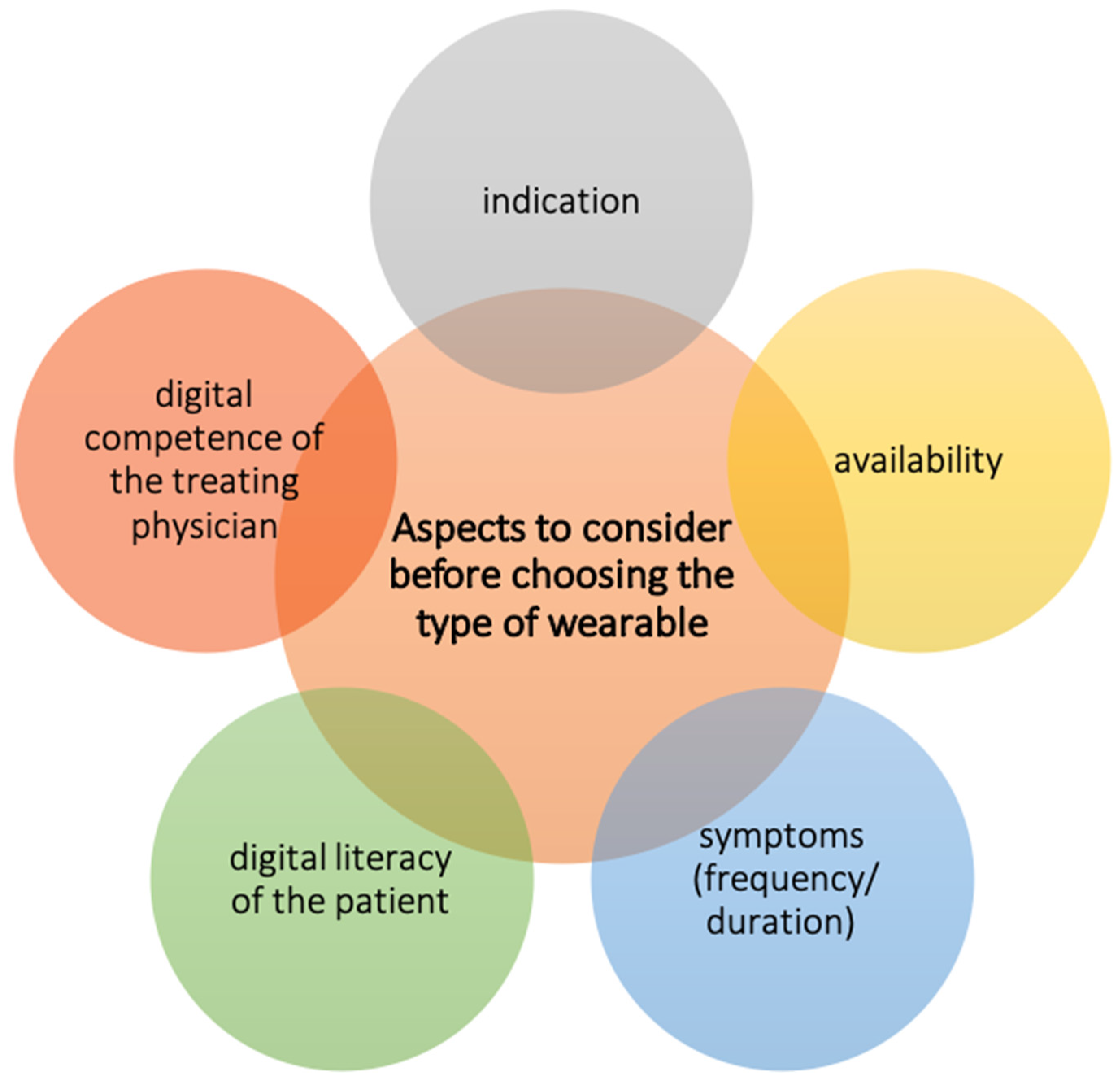
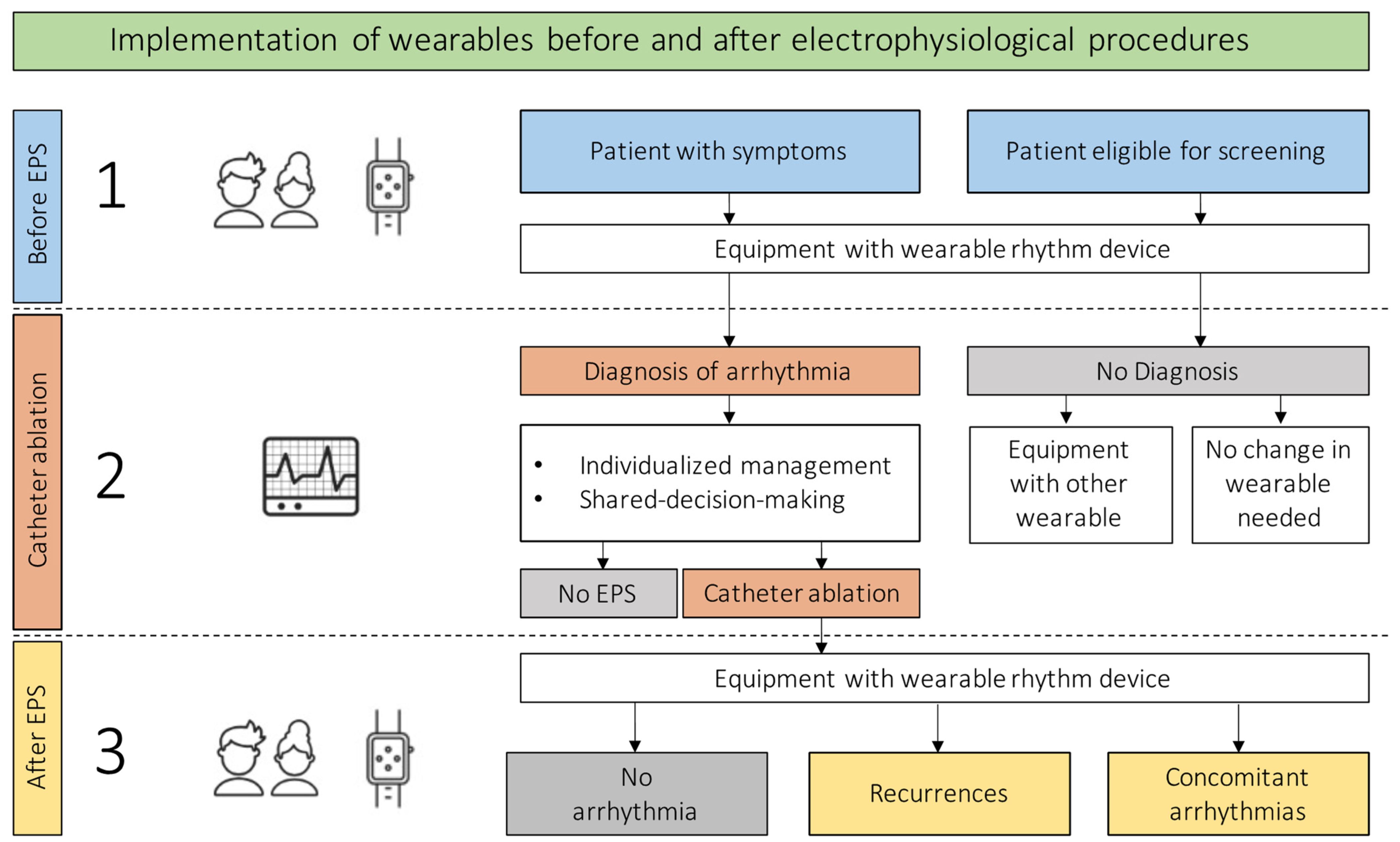
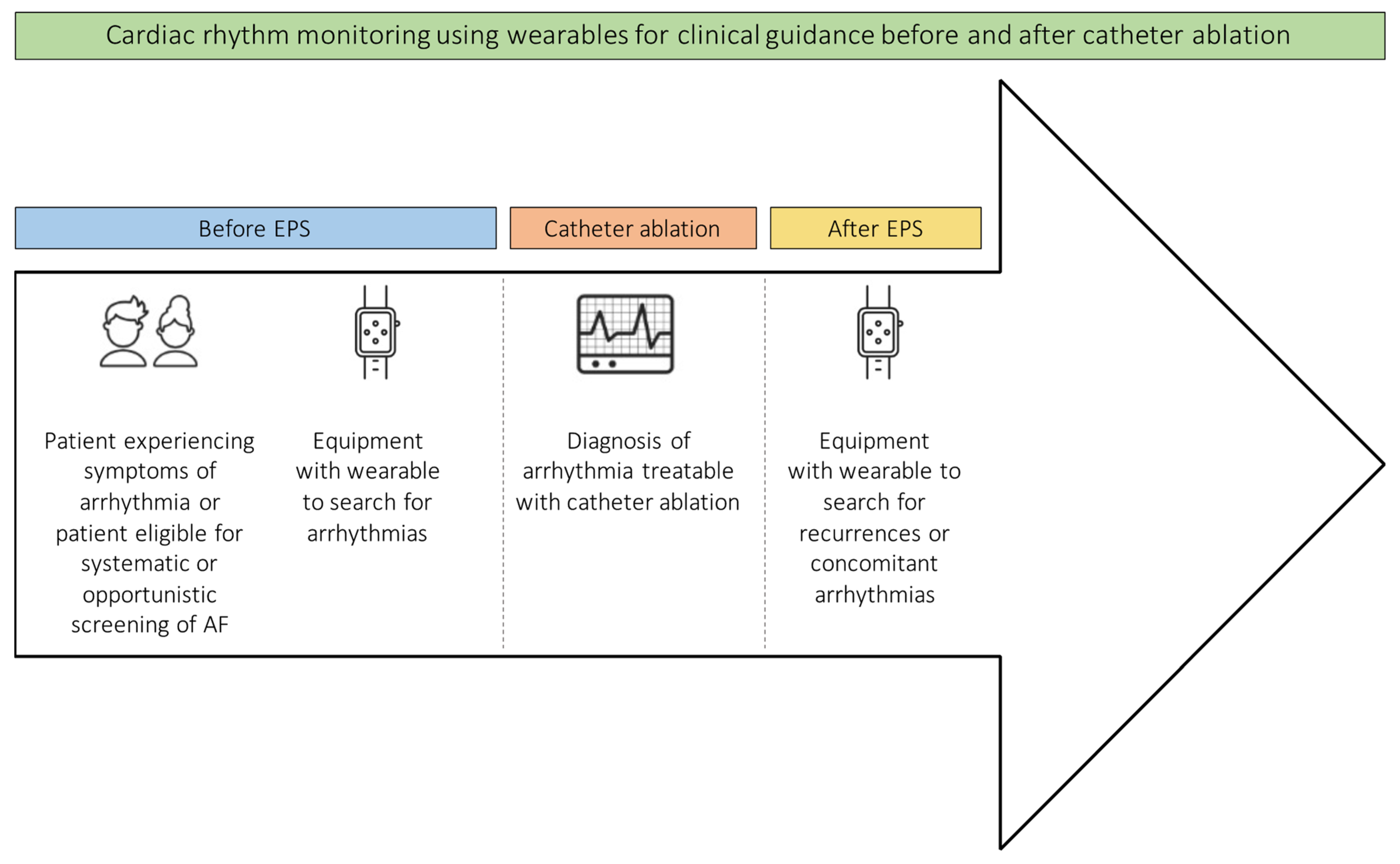
2. Clinical Use of Wearables
3. Confirmation of Diagnosis
4. Monitoring for Recurrences and Concomitant Arrhythmias
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hermans, A.N.L.; Gawalko, M.; Dohmen, L.; van der Velden, R.M.J.; Betz, K.; Duncker, D.; Verhaert, D.V.M.; Heidbuchel, H.; Svennberg, E.; Neubeck, L.; et al. Mobile health solutions for atrial fibrillation detection and management: A systematic review. Clin. Res. Cardiol. 2021, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Giebel, G.D.; Gissel, C. Accuracy of mHealth Devices for Atrial Fibrillation Screening: Systematic Review. JMIR mHealth uHealth 2019, 7, e13641. [Google Scholar] [CrossRef] [PubMed]
- Xintarakou, A.; Sousonis, V.; Asvestas, D.; Vardas, P.E.; Tzeis, S. Remote Cardiac Rhythm Monitoring in the Era of Smart Wearables: Present Assets and Future Perspectives. Front. Cardiovasc. Med. 2022, 9, 853614. [Google Scholar] [CrossRef]
- Bouzid, Z.; Al-Zaiti, S.S.; Bond, R.; Sejdić, E. Remote and wearable ECG devices with diagnostic abilities in adults: A state-of-the-science scoping review. Heart Rhythm 2022. [Google Scholar] [CrossRef]
- Duncker, D.; Ding, W.Y.; Etheridge, S.; Noseworthy, P.A.; Veltmann, C.; Yao, X.; Bunch, T.J.; Gupta, D. Smart Wearables for Cardiac Monitoring—Real-World Use beyond Atrial Fibrillation. Sensors 2021, 21, 2539. [Google Scholar] [CrossRef]
- Ledwoch, J.; Duncker, D. eHealth—Smart Devices revolutionieren die Kardiologie. Herzschrittmacherther. Elektrophysiol. 2020, 31, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, V.D.; Lee, S.-L.; Robertus, J.-L.; Nienaber, C.A.; Trayanova, N.A.; Ernst, S. Artificial intelligence in the diagnosis and management of arrhythmias. Eur. Heart J. 2021, 42, 3904–3916. [Google Scholar] [CrossRef]
- Manninger, M.; Kosiuk, J.; Zweiker, D.; Njeim, M.; Antolic, B.; Kircanski, B.; Larsen, J.M.; Svennberg, E.; Vanduynhoven, P.; Duncker, D. Role of wearable rhythm recordings in clinical decision making—The wEHRAbles project. Clin. Cardiol. 2020, 43, 1032–1039. [Google Scholar] [CrossRef]
- Veltmann, C.; Ehrlich, J.R.; Gassner, U.M.; Meder, B.; Möckel, M.; Radke, P.; Scholz, E.; Schneider, H.; Stellbrink, C.; Duncker, D. Wearable-basierte Detektion von Arrhythmien. Der Kardiol. 2021, 15, 341–353. [Google Scholar] [CrossRef]
- Betz, K.; van der Velden, R.; Gawalko, M.; Hermans, A.; Pluymaekers, N.; Hillmann, H.A.K.; Hendriks, J.; Duncker, D.; Linz, D. Interpretation of Photoplethysmography: A Step-by-Step Guide. Herzschrittmacherther. Elektrophysiol. 2021, 32, 406–411. [Google Scholar] [CrossRef]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 2020, 42, ehaa612. [Google Scholar] [CrossRef]
- Kuck, K.-H.; Lebedev, D.S.; Mikhaylov, E.N.; Romanov, A.; Gellér, L.; Kalējs, O.; Neumann, T.; Davtyan, K.; On, Y.K.; Popov, S.; et al. Catheter ablation or medical therapy to delay progression of atrial fibrillation: The randomized controlled atrial fibrillation progression trial (ATTEST). Europace 2020, 23, 362–369a. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, P.; Camm, A.J.; Goette, A.; Brandes, A.; Eckardt, L.; Elvan, A.; Fetsch, T.; Van Gelder, I.C.; Haase, D.; Haegeli, L.M.; et al. Early Rhythm-Control Therapy in Patients with Atrial Fibrillation. N. Engl. J. Med. 2020, 383, 1305–1316. [Google Scholar] [CrossRef]
- Willems, S.; Borof, K.; Brandes, A.; Breithardt, G.; Camm, A.J.; Crijns, H.J.G.M.; Eckardt, L.; Gessler, N.; Goette, A.; Haegeli, L.M.; et al. Systematic, early rhythm control strategy for atrial fibrillation in patients with or without symptoms: The EAST-AFNET 4 trial. Eur. Heart J. 2021, ehab593. [Google Scholar] [CrossRef] [PubMed]
- Svennberg, E.; Tjong, F.; Goette, A.; Akoum, N.; Biaise, L.D.; Bordachar, P.; Boriani, G.; Burri, H.; Conte, G.; Deharo, J.-C.; et al. How to Use Digital Devices to Detect and Manage Arrhythmias: An EHRA Practical Guide. Europace 2022. [Google Scholar] [CrossRef]
- Gawałko, M.; Duncker, D.; Manninger, M.; van der Velden, R.M.; Hermans, A.N.L.; Verhaert, D.V.M.; Pison, L.; Pisters, R.; Hemels, M.; Sultan, A.; et al. The European TeleCheck-AF project on remote app-based management of atrial fibrillation during the COVID-19 pandemic: Centre and patient experiences. Europace 2021, 23, 1003–1015. [Google Scholar] [CrossRef]
- Chang, P.-C.; Wen, M.-S.; Chou, C.-C.; Wang, C.-C.; Hung, K.-C. Atrial fibrillation detection using ambulatory smartwatch photoplethysmography and validation with simultaneous holter recording. Am. Heart J. 2022, 247, 55–62. [Google Scholar] [CrossRef]
- Perino, A.C.; Gummidipundi, S.E.; Lee, J.; Hedlin, H.; Garcia, A.; Ferris, T.; Balasubramanian, V.; Gardner, R.M.; Cheung, L.; Hung, G.; et al. Arrhythmias Other Than Atrial Fibrillation in Those with an Irregular Pulse Detected with a Smartwatch: Findings from the Apple Heart Study. Circ. Arrhythm. Electrophysiol. 2021, 14, e010063. [Google Scholar] [CrossRef]
- Behzadi, A.; Shamloo, A.S.; Mouratis, K.; Hindricks, G.; Arya, A.; Bollmann, A. Feasibility and Reliability of SmartWatch to Obtain 3-Lead Electrocardiogram Recordings. Sensors 2020, 20, 5074. [Google Scholar] [CrossRef]
- Spaccarotella, C.A.M.; Polimeni, A.; Migliarino, S.; Principe, E.; Curcio, A.; Mongiardo, A.; Sorrentino, S.; De Rosa, S.; Indolfi, C. Multichannel Electrocardiograms Obtained by a Smartwatch for the Diagnosis of ST-Segment Changes. JAMA Cardiol. 2020, 5, 1176–1180. [Google Scholar] [CrossRef]
- Wegner, F.K.; Kochhäuser, S.; Frommeyer, G.; Lange, P.S.; Ellermann, C.; Leitz, P.; Müller, P.; Köbe, J.; Eckardt, L.; Dechering, D.G. Prospective blinded evaluation of smartphone-based ECG for differentiation of supraventricular tachycardia from inappropriate sinus tachycardia. Clin. Res. Cardiol. 2021, 110, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Samol, A.; Bischof, K.; Luani, B.; Pascut, D.; Wiemer, M.; Kaese, S. Single-Lead ECG Recordings Including Einthoven and Wilson Leads by a Smartwatch: A New Era of Patient Directed Early ECG Differential Diagnosis of Cardiac Diseases? Sensors 2019, 19, 4377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasai, Y.; Kasai, J.; Sekiguchi, Y.; Asano, S.; Fukunaga, H.; Nagase, T.; Nitta, J. Apple Watch® facilitates single-session catheter ablation of coexisting atrioventricular nodal reentrant tachycardia and atrioventricular reentrant tachycardia. Clin. Case Rep. 2021, 9, e04702. [Google Scholar] [CrossRef] [PubMed]
- Siddeek, H.; Fisher, K.; McMakin, S.; Bass, J.L.; Cortez, D. AVNRT captured by Apple Watch Series 4: Can the Apple watch be used as an event monitor? Ann. Noninvasive Electrocardiol. 2020, 25, e12742. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Li, C.; Lin, J.; Weng, C.; Lin, W.; Hsieh, Y. Detecting supraventricular tachycardia with smartwatches facilitates the decision for catheter ablation: A case series. Pacing Clin. Electrophysiol. 2021, 45, 157–159. [Google Scholar] [CrossRef]
- Aljuaid, M.; Marashly, Q.; AlDanaf, J.; Tawhari, I.; Barakat, M.; Barakat, R.; Zobell, B.; Cho, W.; Chelu, M.G.; Marrouche, N.F. Smartphone ECG Monitoring System Helps Lower Emergency Room and Clinic Visits in Post-Atrial Fibrillation Ablation Patients. Clin. Med. Insights Cardiol. 2020, 14, 1179546820901508. [Google Scholar] [CrossRef] [Green Version]
- Kaitani, K.; Inoue, K.; Kobori, A.; Nakazawa, Y.; Ozawa, T.; Kurotobi, T.; Morishima, I.; Miura, F.; Watanabe, T.; Masuda, M.; et al. Efficacy of Antiarrhythmic Drugs Short-Term Use After Catheter Ablation for Atrial Fibrillation (EAST-AF) trial. Eur. Heart J. 2016, 37, 610–618. [Google Scholar] [CrossRef] [Green Version]
- Willems, S.; Khairy, P.; Andrade, J.G.; Hoffmann, B.A.; Levesque, S.; Verma, A.; Weerasooriya, R.; Novak, P.; Arentz, T.; Deisenhofer, I.; et al. Redefining the Blanking Period After Catheter Ablation for Paroxysmal Atrial Fibrillation: Insights From the ADVICE (Adenosine Following Pulmonary Vein Isolation to Target Dormant Conduction Elimination) Trial. Circ. Arrhythm. Electrophysiol. 2016, 9, e003909. [Google Scholar] [CrossRef] [Green Version]
- Gaztañaga, L.; Frankel, D.S.; Kohari, M.; Kondapalli, L.; Zado, E.S.; Marchlinski, F.E. Time to recurrence of atrial fibrillation influences outcome following catheter ablation. Heart Rhythm 2013, 10, 2–9. [Google Scholar] [CrossRef]
- Linz, D.; Pluymaekers, N.A.H.A.; Hendriks, J.M. TeleCheck-AF for COVID-19. Eur. Heart J. 2020, 41, 1954–1955. [Google Scholar] [CrossRef]
- Pluymaekers, N.A.H.A.; Hermans, A.N.L.; van der Velden, R.M.J.; Gawałko, M.; Den Uijl, D.W.; Buskes, S.; Vernooy, K.; Crijns, H.J.G.M.; Hendriks, J.M.; Linz, D. Implementation of an on-demand app-based heart rate and rhythm monitoring infrastructure for the management of atrial fibrillation through teleconsultation: TeleCheck-AF. Europace 2020, 23, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Hermans, A.N.L.; Van Der Velden, R.M.J.; Gawalko, M.; Verhaert, D.V.M.; Desteghe, L.; Duncker, D.; Manninger, M.; Heidbuchel, H.; Pisters, R.; Hemels, M.; et al. On-demand mobile health infrastructures to allow comprehensive remote atrial fibrillation and risk factor management through teleconsultation. Clin. Cardiol. 2020, 43, 1232–1239. [Google Scholar] [CrossRef]
- Hermans, A.N.L.; Gawałko, M.; Hillmann, H.A.K.; Sohaib, A.; van der Velden, R.M.J.; Betz, K.; Verhaert, D.; Scherr, D.; Meier, J.; Sultan, A.; et al. Self-Reported Mobile Health-Based Risk Factor and CHA2DS2-VASc-Score Assessment in Patients with Atrial Fibrillation: TeleCheck-AF Results. Front. Cardiovasc. Med. 2022, 8, 757587. [Google Scholar] [CrossRef] [PubMed]
- Brugada, J.; Katritsis, D.G.; Arbelo, E.; Arribas, F.; Bax, J.J.; Blomström-Lundqvist, C.; Calkins, H.; Corrado, D.; Deftereos, S.G.; Diller, G.-P.; et al. 2019 ESC Guidelines for the management of patients with supraventricular tachycardia. Eur. Heart J. 2019, 41, 655–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manninger, M.; Zweiker, D.; Svennberg, E.; Chatzikyriakou, S.; Pavlovic, N.; Zaman, J.A.B.; Kircanski, B.; Lenarczyk, R.; Vanduynhoven, P.; Kosiuk, J.; et al. Current perspectives on wearable rhythm recordings for clinical decision-making: The wEHRAbles 2 survey. Europace 2021, 23, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Gruwez, H.; Evens, S.; Proesmans, T.; Duncker, D.; Linz, D.; Heidbuchel, H.; Manninger, M.; Vandervoort, P.; Haemers, P.; Pison, L. Accuracy of Physicians Interpreting Photoplethysmography and Electrocardiography Tracings to Detect Atrial Fibrillation: INTERPRET-AF. Front. Cardiovasc. Med. 2021, 8, 734737. [Google Scholar] [CrossRef] [PubMed]
- van der Velden, R.M.J.; Verhaert, D.V.M.; Hermans, A.N.L.; Duncker, D.; Manninger, M.; Betz, K.; Gawalko, M.; Desteghe, L.; Pisters, R.; Hemels, M.; et al. The photoplethysmography dictionary: Practical guidance on signal interpretation and clinical scenarios from TeleCheck-AF. Eur. Heart J.-Digit. Health 2021, 2, 363–373. [Google Scholar] [CrossRef]
| Advantages | Disadvantages |
|---|---|
| May reduce time to diagnosis | Can imply costs for the patient |
| High availability | Lack of reimbursement for the treating physician |
| Possibility to improve arrhythmia management | Data overload |
| Remote monitoring option | Limited experience of physicians |
| Author | Device (Specification) | Technology | FDA Approved | Number of Patients | Timing | Arrhythmia Ablated |
|---|---|---|---|---|---|---|
| Kasai et al. [23] | Smartwatch (Apple Watch SE) | PPG (heart rate) | Yes | 1 | Preprocedural | AVNRT, AVRT * |
| Siddeek et al. [24] | Smartwatch (Apple Watch Series 4) | ECG (single-lead) | Yes | 1 | Preprocedural | AVNRT |
| Wu et al. [25] | Smartwatch (Apple Watch—no detailed differentiation) | ECG (single-lead) | Yes | 3 | Preprocedural | AVRT, AVNRT |
| Aljuaid et al. [26] | Portable heart monitor (ECG check cardiac designs) | ECG (single-lead) | Yes | 45 | Postprocedural after AF ablation | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hillmann, H.A.K.; Soltani, S.; Mueller-Leisse, J.; Hohmann, S.; Duncker, D. Cardiac Rhythm Monitoring Using Wearables for Clinical Guidance before and after Catheter Ablation. J. Clin. Med. 2022, 11, 2428. https://doi.org/10.3390/jcm11092428
Hillmann HAK, Soltani S, Mueller-Leisse J, Hohmann S, Duncker D. Cardiac Rhythm Monitoring Using Wearables for Clinical Guidance before and after Catheter Ablation. Journal of Clinical Medicine. 2022; 11(9):2428. https://doi.org/10.3390/jcm11092428
Chicago/Turabian StyleHillmann, Henrike Aenne Katrin, Samira Soltani, Johanna Mueller-Leisse, Stephan Hohmann, and David Duncker. 2022. "Cardiac Rhythm Monitoring Using Wearables for Clinical Guidance before and after Catheter Ablation" Journal of Clinical Medicine 11, no. 9: 2428. https://doi.org/10.3390/jcm11092428
APA StyleHillmann, H. A. K., Soltani, S., Mueller-Leisse, J., Hohmann, S., & Duncker, D. (2022). Cardiac Rhythm Monitoring Using Wearables for Clinical Guidance before and after Catheter Ablation. Journal of Clinical Medicine, 11(9), 2428. https://doi.org/10.3390/jcm11092428






