Clinical Relevance of Drug Interactions with Cannabis: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
- Possible: The interaction was documented by results from less than three case reports.
- Probable: The interaction was documented by results from at least one observational study (cohort or case–control study) or a least three case reports.
- Defined: The interaction was documented by results from at least one meta-analysis, narrative systematic review, or randomized or non-randomized clinical trial.
3. Results
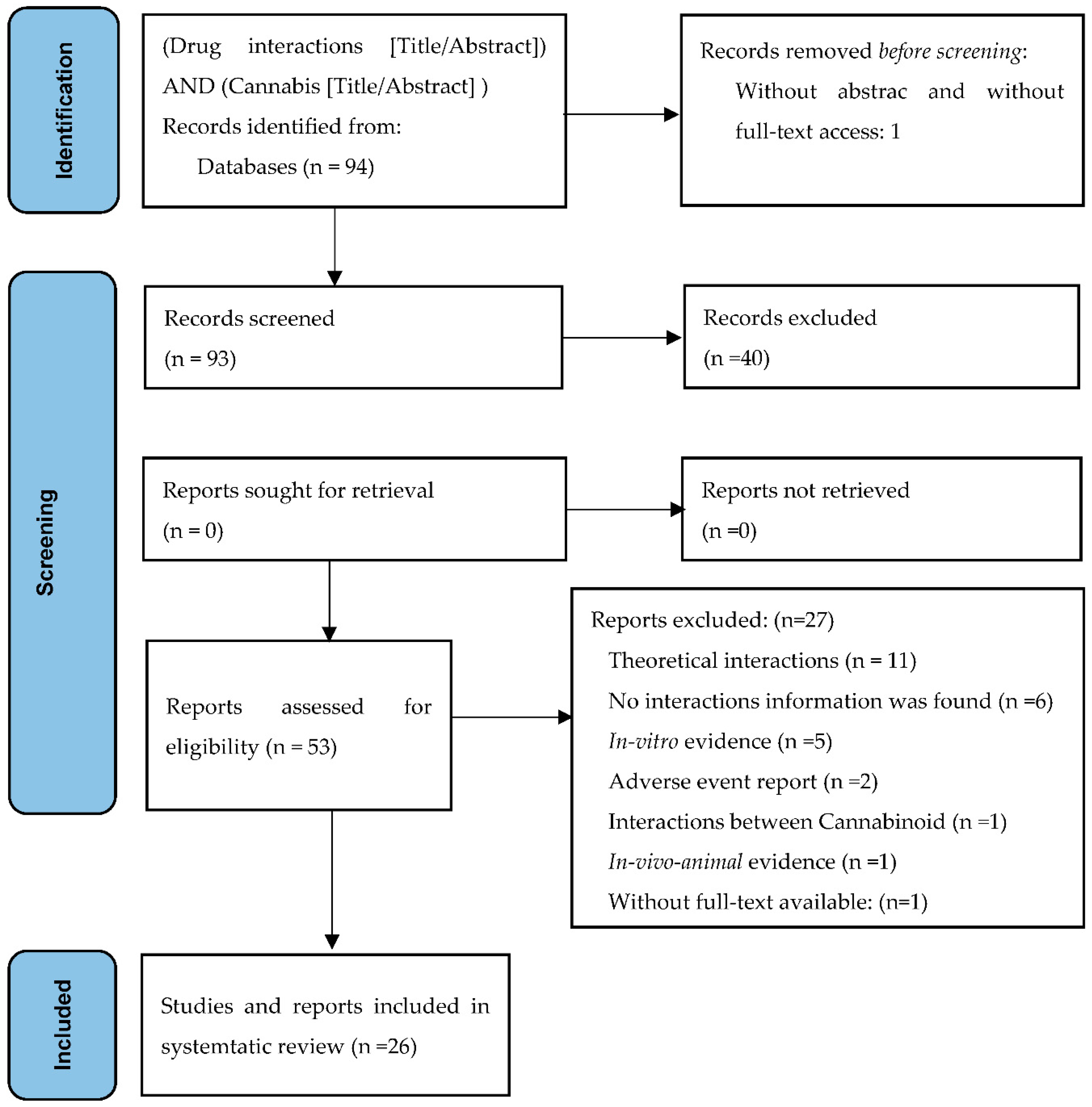
3.1. Results of the Search
3.2. Drug Interactions
4. Discussion
4.1. Evidence
4.2. Limitations of this Review
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Szaflarski, J.P.; Martina Bebin, E. Cannabis, cannabidiol, and epilepsy—From receptors to clinical response. Epilepsy Behav. 2014, 41, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.K. Cannabinergic Pain Medicine: A Concise Clinical Primer and Survey of Randomized-controlled Trial Results. Clin. J. Pain 2013, 29, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, B.; Baranowska-Kuczko, M.; Kicman, A.; Schlicker, E. Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19. Int. J. Mol. Sci. 2021, 22, 1986. [Google Scholar] [CrossRef] [PubMed]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the Treatment of Crohn’s Disease and Ulcerative Colitis: Evidence From Cochrane Reviews. Inflamm. Bowel Dis. 2020, 26, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, N.; Richert, L. Pharmacists and the future of cannabis medicine. J. Am. Pharm. Assoc. 2020, 60, 207–211. [Google Scholar] [CrossRef] [Green Version]
- Alsherbiny, M.; Li, C. Medicinal Cannabis—Potential Drug Interactions. Medicines 2018, 6, 3. [Google Scholar] [CrossRef] [Green Version]
- Han, B.H.; Sherman, S.; Mauro, P.M.; Martins, S.S.; Rotenberg, J.; Palamar, J.J. Demographic trends among older cannabis users in the United States, 2006-13: Cannabis use among older adults. Addiction 2017, 112, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Amariles, P.; Andrés Giraldo, N.; José Faus, M. Interacciones medicamentosas: Aproximación para establecer y evaluar su relevancia clínica. Med. Clin. 2007, 129, 27–35. [Google Scholar] [CrossRef]
- Nasrin, S.; Watson, C.J.W.; Perez-Paramo, Y.X.; Lazarus, P. Cannabinoid Metabolites as Inhibitors of Major Hepatic CYP450 Enzymes, with Implications for Cannabis–drugInteractions. Drug Metab. Dispos. 2021, 49, 1070–1080. [Google Scholar] [CrossRef]
- Amariles, P.; Madrigal-Cadavid, J.; Giraldo, N.A. Relevancia clínica de las interacciones medicamentosas: Propuesta de actualización de la clasificación, acorde con su gravedad y probabilidad. Rev. Chil. Infectol. 2021, 38, 304–305. [Google Scholar] [CrossRef] [PubMed]
- Cytochrome P450 Drug Interaction Table—Drug Interactions-The Flockhart Table. Available online: https://drug-interactions.medicine.iu.edu/MainTable.aspx (accessed on 20 November 2021).
- Grayson, L.; Vines, B.; Nichol, K.; Szaflarski, J.P. An interaction between warfarin and cannabidiol, a case report. Epilepsy Behav. Case Rep. 2018, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Damkier, P.; Lassen, D.; Christensen, M.M.H.; Madsen, K.G.; Hellfritzsch, M.; Pottegård, A. Interaction between warfarin and cannabis. Basic Clin. Pharmacol. Toxicol. 2019, 124, 28–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, A.; Painter, N.A. Probable Interaction Between Warfarin and Inhaled and Oral Administration of Cannabis. J. Pharm. Pract. 2019, 33, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Yamreudeewong, W.; Wong, H.K.; Brausch, L.M.; Pulley, K.R. Probable Interaction Between Warfarin and Marijuana Smoking. Ann. Pharmacother. 2009, 43, 1347–1353. [Google Scholar] [CrossRef] [PubMed]
- Vierke, C.; Marxen, B.; Boettcher, M.; Hiemke, C.; Havemann-Reinecke, U. Buprenorphine–cannabis interaction in patients undergoing opioid maintenance therapy. Eur. Arch. Psychiatry Clin. Neurosci. 2021, 271, 847. [Google Scholar] [CrossRef] [Green Version]
- Hauser, N.; Sahai, T.; Richards, R.; Roberts, T. High on Cannabis and Calcineurin Inhibitors: A Word of Warning in an Era of Legalized Marijuana. Case Rep. Transplant. 2016, 2016, 4028492. [Google Scholar] [CrossRef] [Green Version]
- Leino, A.D.; Emoto, C.; Fukuda, T.; Privitera, M.; Vinks, A.A.; Alloway, R.R. Evidence of a clinically significant drug-drug interaction between cannabidiol and tacrolimus. Am. J. Transplant. 2019, 19, 2944–2948. [Google Scholar] [CrossRef]
- Cuñetti, L.; Manzo, L.; Peyraube, R.; Arnaiz, J.; Curi, L.; Orihuela, S. Chronic Pain Treatment with Cannabidiol in Kidney Transplant Patients in Uruguay. Transplant. Proc. 2018, 50, 461–464. [Google Scholar] [CrossRef]
- Zullino, D.F.; Delessert, D.; Eap, C.B.; Preisig, M.; Baumann, P. Tobacco and cannabis smoking cessation can lead to intoxication with clozapine or olanzapine. Int. Clin. Psychopharmacol. 2002, 17, 141–143. [Google Scholar] [CrossRef]
- Madden, K.; Tanco, K.; Bruera, E. Clinically Significant Drug-Drug Interaction Between Methadone and Cannabidiol. Pediatrics 2020, 145, e20193256. [Google Scholar] [CrossRef] [PubMed]
- Gaston, T.E.; Bebin, E.M.; Cutter, G.R.; Liu, Y.; Szaflarski, J.P.; UAB CBD Program. Interactions between cannabidiol and commonly used antiepileptic drugs. Epilepsia 2017, 58, 1586–1592. [Google Scholar] [CrossRef] [Green Version]
- Geffrey, A.L.; Pollack, S.F.; Bruno, P.L.; Thiele, E.A. Drug-drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia 2015, 56, 1246–1251. [Google Scholar] [CrossRef]
- Wheless, J.W.; Dlugos, D.; Miller, I.; Oh, D.A.; Parikh, N.; Phillips, S.; Renfroe, J.B.; Roberts, C.M.; Saeed, I.; Sparagana, S.P.; et al. Pharmacokinetics and Tolerability of Multiple Doses of Pharmaceutical-Grade Synthetic Cannabidiol in Pediatric Patients with Treatment-Resistant Epilepsy. CNS Drugs 2019, 33, 593–604. [Google Scholar] [CrossRef] [Green Version]
- Chetty, M.; Miller, R.; Moodley, S.V. Smoking and body weight influence the clearance of chlorpromazine. Eur. J. Clin. Pharmacol. 1994, 46, 523–526. [Google Scholar] [CrossRef]
- MacDonald, E.; Adams, A. The Use of Medical Cannabis with Other Medications: A Review of Safety and Guidelines—An Update [Internet]; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2019. [Google Scholar]
- Benowitz, N.L.; Nguyen, T.L.; Jones, R.T.; Herning, R.I.; Bachman, J. Metabolic and psychophysiologic studies of cannabidiol-hexobarbital interaction. Clin. Pharmacol. Ther. 1980, 28, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Abbott, K.L.; Flannery, P.C.; Gill, K.S.; Boothe, D.M.; Dhanasekaran, M.; Mani, S.; Pondugula, S.R. Adverse pharmacokinetic interactions between illicit substances and clinical drugs. Drug Metab. Rev. 2020, 52, 44–65. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, W.T.; Stewart, D.; Childress, D. Drug Interactions between Common Illicit Drugs and Prescription Therapies. Am. J. Drug Alcohol Abus. 2012, 38, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Kosel, B.W.; Aweeka, F.T.; Benowitz, N.L.; Shade, S.B.; Hilton, J.F.; Lizak, P.S.; Abrams, D.I. The effects of cannabinoids on the pharmacokinetics of indinavir and nelfinavir. AIDS 2002, 16, 543–550. [Google Scholar] [CrossRef]
- Stout, S.M.; Cimino, N.M. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: A systematic review. Drug Metab. Rev. 2014, 46, 86–95. [Google Scholar] [CrossRef] [Green Version]
- Stott, C.; White, L.; Wright, S.; Wilbraham, D.; Guy, G. A Phase I, open-label, randomized, crossover study in three parallel groups to evaluate the effect of Rifampicin, Ketoconazole, and Omeprazole on the pharmacokinetics of THC/CBD oromucosal spray in healthy volunteers. SpringerPlus 2013, 2, 236. [Google Scholar] [CrossRef] [Green Version]
- Morrison, G.; Crockett, J.; Blakey, G.; Sommerville, K. A Phase 1, Open-Label, Pharmacokinetic Trial to Investigate Possible Drug-Drug Interactions Between Clobazam, Stiripentol, or Valproate and Cannabidiol in Healthy Subjects. Clin. Pharmacol. Drug Dev. 2019, 8, 1009–1031. [Google Scholar] [CrossRef] [PubMed]
- Ben-Menachem, E.; Gunning, B.; Cabrera, C.M.A.; VanLandingham, K.; Crockett, J.; Critchley, D.; Wray, L.; Tayo, B.; Morrison, G.; Toledo, M. A Phase II Randomized Trial to Explore the Potential for Pharmacokinetic Drug-Drug Interactions with Stiripentol or Valproate when Combined with Cannabidiol in Patients with Epilepsy. CNS Drugs 2020, 34, 661–672. [Google Scholar] [CrossRef]
- Qian, Y.; Gurley, B.J.; Markowitz, J.S. The Potential for Pharmacokinetic Interactions Between Cannabis Products and Conventional Medications. J. Clin. Psychopharmacol. 2019, 39, 462–471. [Google Scholar] [CrossRef]
- Silva, D.A.; Pate, D.W.; Clark, R.D.; Davies, N.M.; El-Kadi, A.; Löbenberg, R. Phytocannabinoid drug-drug interactions and their clinical implications. Pharmacol. Ther. 2020, 215, 107621. [Google Scholar] [CrossRef]
- Gardner, M.J.; Tornatore, K.M.; Jusko, W.J.; Kanarkowski, R. Effects of tobacco smoking and oral contraceptive use on theophylline disposition. Br. J. Clin. Pharmacol. 1983, 16, 271–280. [Google Scholar] [CrossRef] [Green Version]
- Jusko, W.J.; Schentag, J.J.; Clark, J.H.; Gardner, M.; Yurchak, A.M. Enhanced biotransformation of theophylline in marihuana and tobacco smokers. Clin. Pharmacol. Ther. 1978, 24, 406–410. [Google Scholar] [CrossRef]
- Jusko, W.J.; Gardner, M.J.; Mangione, A.; Schentag, J.J.; Koup, J.R.; Vance, J.W. Factors affecting theophylline clearances: Age, tobacco, marijuana, cirrhosis, congestive heart failure, obesity, oral contraceptives, benzodiazepines, barbiturates, and ethanol. J. Pharm. Sci. 1979, 68, 1358–1366. [Google Scholar] [CrossRef]
- McNamara, N.A.; Dang, L.T.; Sturza, J.; Ziobro, J.M.; Fedak Romanowski, E.M.; Smith, G.C.; Joshi, S.M.; Leber, S.M.; Carlson, M.; Robertson, P.; et al. Thrombocytopenia in pediatric patients on concurrent cannabidiol and valproic acid. Epilepsia 2020, 61, e85–e89. [Google Scholar] [CrossRef] [PubMed]
- Ge, B.; Zhang, Z.; Zuo, Z. Updates on the Clinical Evidenced Herb-Warfarin Interactions. Evid. -Based Complement. Altern. Med. 2014, 2014, 957362. [Google Scholar] [CrossRef] [Green Version]
- Greger, J.; Bates, V.; Mechtler, L.; Gengo, F. A Review of Cannabis and Interactions with Anticoagulant and Antiplatelet Agents. J. Clin. Pharmacol. 2020, 60, 432–438. [Google Scholar] [CrossRef]
- Vázquez, M.; Guevara, N.; Maldonado, C.; Guido, P.C.; Schaiquevich, P. Potential Pharmacokinetic Drug-Drug Interactions between Cannabinoids and Drugs Used for Chronic Pain. BioMed Res. Int. 2020, 2020, 3902740. [Google Scholar] [CrossRef] [PubMed]
- Abrams, D.I.; Couey, P.; Shade, S.B.; Kelly, M.E.; Benowitz, N.L. Cannabinoid–Opioid Interaction in Chronic Pain. Clin. Pharm. 2011, 90, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Schaiquevich, P. Farmacología clínica de cannabidiol en epilepsias refractarias. Farm. Hosp. 2020, 44, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Taha, T.; Meiri, D.; Talhamy, S.; Wollner, M.; Peer, A.; Bar-Sela, G. Cannabis Impacts Tumor Response Rate to Nivolumab in Patients with Advanced Malignancies. Oncologist 2019, 24, 549–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Severity | Probability | ||
|---|---|---|---|
| Defined | Probable | Possible | |
| Severe | 1 (very high risk) | 1 (very high risk) | 2 (high risk) |
| Moderate | 2 (high risk) | 2 (high risk) | 3 (medium risk) |
| Minor | 3 (medium risk) | 3 (medium risk) | 4 (low risk) |
| Lack of severity | 5 (riskless) | N/A | N/A |
| Pharmacological Group | Number of Interactions | Level of Relevance | Number by the Level of Relevance (%) |
|---|---|---|---|
| Nervous system | 13 | 2 | 1 (8) |
| 3 | 9 (69) | ||
| 5 | 3 (23) | ||
| Blood and hematopoietic organs | 1 | 1 | 1 (100) |
| Anti-infectives for systemic use | 4 | 3 | 3 (75) |
| 5 | 1 (25) | ||
| antineoplastic agents and immunomodulators | 1 | 2 | 1 (100) |
| Total of pairs of cannabis–drug interactions | 19 | 1 | 1 (5) |
| 2 | 2 (11) | ||
| 3 | 12 (63) | ||
| 5 | 4 (21) |
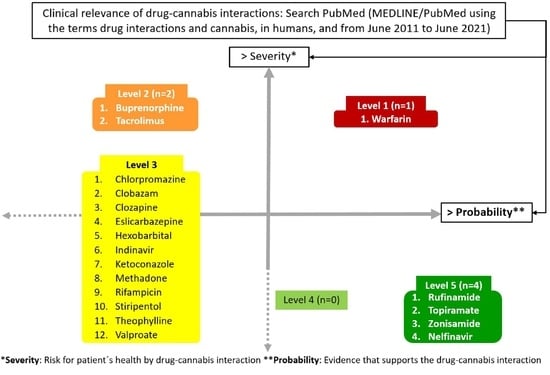
| Drug | Severity | Probability | Mechanism | Comments and Recommendations a |
|---|---|---|---|---|
| LEVEL 1 | ||||
| Warfarin [12,13,14,15] | Severe | Probable | CYP2CP inhibition | No clinical trials were found; nevertheless, 4 case reports were found, where the INR, whose therapeutic range was between 2 and 3, increased to 6.9 b, 4.6 b, 7.2 b, 11.6 b with the concomitant administration of CBD in the first case and with inhaled cannabis (recreational) in the three other cases. Symptoms such as gastrointestinal bleeding were observed. The use of cannabis with warfarin could be contraindicated. If necessary, adjust warfarin dosage and closely monitor the patient’s INR. Advise the patient to avoid using cannabis recreationally. |
| LEVEL 2 | ||||
| Buprenorphine [16] | Moderate | Probable | CYP3A4 inhibition | A retrospective analysis with 32 patients reported concentrations of buprenorphine 170% b higher for those who consume cannabis (recreationally) concomitant with buprenorphine. In addition, in one case report, a patient experiment reported a 95% c decrease in serum levels of buprenorphine when stopping the use of cannabis. Avoid the use of buprenorphine with cannabis. If the combination is necessary, adjust buprenorphine dosage and monitor plasma levels of buprenorphine. Advise the patient to avoid using cannabis recreationally. |
| Tacrolimus [17,18,19] | Moderate | Probable | CYP3A4 inhibition | Two case reports and one clinical trial with 6 persons, where only one patient showed changes, with reported increases of 358% c, 200% b, and 77% c in the plasmatic level of this immunosuppressant with the use of CBD. Avoid the use of tacrolimus with cannabis. If the combination is necessary, adjust tacrolimus dosage and monitor plasma levels of tacrolimus. Individualize usage of CBD and assess each case before dosing. |
| LEVEL 3 | ||||
| Clozapine [20] | Moderate | Possible | CYP1A2 induction | In one case report, a patient stopped the consumption of cannabis and cigarettes, and the plasma levels of clozapine increased by 230% c. During this increase, the patient hallucinated. Adjust clozapine dosage and, if it is possible, monitor plasma levels. |
| Methadone [21] | Moderate | Possible | CYP3A4 and CYP2C19 inhibition | A case report evidences the administration of CBD oil to a patient having methadone treatment. Methadone levels increased by 117% c, and somnolence and fatigue were reported. Adjust methadone dosage and, if it is possible, monitor plasma levels. |
| Clobazam [22,23,24] | Minor | Defined | CYP2C19 inhibition | In 3 clinical trials, clobazam concentration increased by 25% c, 60% b, and 20% b in patients receiving different doses of clobazam and CBD. In the 3 studies, the antiepileptic doses were reduced when it was necessary to reduce the adverse events. Adjust clobazam dosage and, if it is possible, monitor plasma levels. |
| Chlorpromazine [25] | Minor | Defined | Possible CYP1A2 induction | A clinical trial showed an increase of 50% b in clearance in subjects who consume recreational cannabis. Adjust chlorpromazine dosage and, if it is possible, monitor plasma levels. |
| Eslicarbazepine [22,26] | Minor | Defined | Unknown. Perhaps the delivery vehicle (sesame oil) in this formulation of CBD could contribute to this interaction | In a clinical trial, with a concomitant administration of CBD (Epidiolex), an increase of 24% c was evidenced. This change was statistically significant, but it was evaluated in only 4 subjects. Adjust eslicarbazepine dosage and, if it is possible, monitor plasma levels. |
| Hexobarbital [27] | Minor | Defined | Possible CYP3A4 inhibition | In a clinical trial, hexobarbital clearance was 35% c lower when CBD was administered, compared to when it was not administrated, in subjects who consume recreational cannabis regularly. Adjust hexobarbital dosage and, if it is possible, monitor plasma levels. |
| Indinavir [28,29,30] | Minor | Defined | Possible CYP3A4 inhibition | In a clinical trial, the maximum concentration of indinavir decreased by 14.1% b using THC cigarettes in patients who use indinavir. There were no statistically significant changes with dronabinol usage. Adjust indinavir dosage and, if it is possible, monitor plasma levels. |
| Ketoconazole [31,32] | Minor | Defined | CYP3A4 inhibition | In a clinical trial, the concomitant administration of ketoconazole with Sativex increased the AUC of THC by 82% c and the AUC of CBD by 84% c. Adjust ketoconazole dosage and, if it is possible, monitor plasma levels. |
| Rifampicin [31,32] | Minor | Defined | CYP3A4 induction | In a clinical trial, the concomitant administration of rifampicin with Sativex decreased the AUC of THC by 24% c and the AUC of CBD by 84% c. Adjust rifampicin dosage and, if it is possible, monitor plasma levels. |
| Stiripentol [33,34,35] | Minor | Defined | CYP2C19 inhibition | In a phase 1 study, stiripentol AUC increased by 60% b with the concomitant administration of CBD (Epidiolex). In another phase 2 study, the increase was 30% b. Adjust stiripentol dosage and, if it is possible, monitor plasma levels. |
| Theophylline [36,37,38,39] | Minor | Defined | CYP1A2 induction | There is evidence from two clinical trials and one retrospective study. In these, subjects used recreational cannabis and smoked cigarettes. Clearance was calculated, and it increased by 40% b, 42% c, and 48% c. Adjust theophylline dosage and, if it is possible, monitor plasma levels. |
| Valproate [22,40] | Minor | Defined | Possible UGT1A9 y UGT2B7 inhibition | In a clinical trial, after increasing the CBD (Epidiolex) dose, the valproate level did not change. Nevertheless, an increase in ALT and AST levels by 49% c and 55% c, respectively, was noted. In another clinical trial, 39% c of the patients taking CBD and valproate developed thrombocytopenia, but the results were not statistically significant. Assess liver function before starting CBD and monitor liver function. |
| LEVEL 5 | ||||
| Rufinamide [22] | Lack | Defined | Co-administration with CBD (Epidiolex) does not lead to significant changes in rufinamide levels. | |
| Topiramate [22] | Lack | Defined | Co-administration with CBD (Epidiolex) does not lead to significant changes in topiramate levels. | |
| Zonisamide [22] | Lack | Defined | Co-administration with CBD (Epidiolex) does not lead to significant changes in zonisamide levels. | |
| Nelfinavir [28,29,30] | Lack | Defined | Co-administration with THC cigarettes does not lead to significant changes in nelfinavir levels and AUC. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopera, V.; Rodríguez, A.; Amariles, P. Clinical Relevance of Drug Interactions with Cannabis: A Systematic Review. J. Clin. Med. 2022, 11, 1154. https://doi.org/10.3390/jcm11051154
Lopera V, Rodríguez A, Amariles P. Clinical Relevance of Drug Interactions with Cannabis: A Systematic Review. Journal of Clinical Medicine. 2022; 11(5):1154. https://doi.org/10.3390/jcm11051154
Chicago/Turabian StyleLopera, Valentina, Adriana Rodríguez, and Pedro Amariles. 2022. "Clinical Relevance of Drug Interactions with Cannabis: A Systematic Review" Journal of Clinical Medicine 11, no. 5: 1154. https://doi.org/10.3390/jcm11051154
APA StyleLopera, V., Rodríguez, A., & Amariles, P. (2022). Clinical Relevance of Drug Interactions with Cannabis: A Systematic Review. Journal of Clinical Medicine, 11(5), 1154. https://doi.org/10.3390/jcm11051154