Molecular Genetics Overview of Primary Mitochondrial Myopathies
Abstract
1. Introduction
2. General Aspects
3. Defects of MRC Complexes
3.1. Complex I and Assembly Factors
3.2. Complex II and Assembly Factors
3.3. Complex III and Assembly Factors
3.4. Complex IV and Assembly Factors
3.5. Complex V and Assembly Factors
4. Defect of Translational Apparatus
5. Defects of Electron Carriers
6. Defects of mtDNA Replication and Homeostasis
6.1. Rearrangements of mtDNA
6.2. Defects of mtDNA Mantainance
6.2.1. Defects of mtDNA Replication Apparatus
6.2.2. Defects of Mitochondrial Deoxyribonucleosides Pools
| Genes | Type of Article | Patients/Age at Onset | Muscle Manifestations | Other Clinical Features | References |
|---|---|---|---|---|---|
| SUCLG1 | CR | 1 pt 6mo | Marked muscular hypotonia, severe muscle atrophy | Failure to thrive | [134] |
| SUCLG1 | CR | 1 pt 17 h | Axial hypotonia, no head control, poor swallowing and muscle weakness | Severe metabolic acidosis, liver failure | [132] |
| SUCLA2 | CS | 50 pts Median age: 2 mo | Eyelid ptosis, ophthalmoplegia | Psychomotor retardation and failure to thrive, dystonia, hearing impairment, epilepsy | [132] |
| TK2 | CS | 92 pts Age range: birth–72 yo | Proximal muscle weakness, high CK, ptosis, PEO, dysphagia, dysarthria/dysphonia, | seizures, encephalopathy and cognitive impairment, sensorineural hearing loss | [135] |
| DGUOK | CS | 6 pts Age range: 20 yo–69 yo | PEO, ptosis, limb girdle weakness, myalgia, dysphonia, dysphagia | [129] | |
| DGUOK | CR | 1 pt 14 yo | Fatigue during exercise | Abdominal pain, reduced left ventricular systolic | [136] |
| TYMP | CS | 102 pts Age range: 11–59 yo | Bilateral ptosis, ophthalmoparesis, lower limb hyposthenia | Vomiting, abdominal pain, severe malnutrition | [14] |
| RRM2B | CS | 3 pts Age range: 8 we–27 mo | Muscle hypotonia and progressive weakness, poor head control and respiratory distress. High CK | Failure to thrive and diarrhea, persistent acidosis, sensorineural hearing loss, retinopathy | [137] |
| RRM2B | CS | 31 pts Age range: 0–6 mo | Muscular hypotonia, respiratorydistress | Failure to thrive, hearing loss, encephalopathy, seizures. Renal, eye and GI involvement. Anemia | [138] |
| POLG | CS | 95 pts Age range: 2–23 yo | Myopathy, ptosis and PEO, neck flexor weakness | Seizures, hepatopathy, lactic acidaemia, sensory ataxia, bradykinesia | [123] |
| Twinkle | CS | 4 pts Age range: 20s–30s | Ptosis and ophthalmoplegia, diplopia | Gait difficulty, stiffness, resting tremor, depression | [127] |
| SLC25A4 | CS | 25 pts Age range: 0–48 yo | Myopathy and muscle hypotonia, dysarthria, respiratory insufficiency | Ischemic stroke, hydrocephalus, insomnia, mental retardation, headache, cardiomyopathy, cataracts, scoliosis | [121] |
| MGME1 | CS | 6 pts Age range: NA | PEO, proximal weakness, generalized muscle wasting, respiratory failure, exercise intolerance | Mental retardation, depressive episodes, gastrointestinal symptoms, spinal deformities, ataxia, dilated cardiomyopathy and arrhythmias, | [128] |
| OPA 1 | CS | 8 pts Age range: birth–60 yo | Ptosis and ophthalmoplegia, exercise intolerance and myalgia, muscle weakness | Optic atrophy, hearing loss, pes cavus, feeding difficulties | [139] |
| SPG7 | CS | 9 pts Age range: Late20s–Mid60s | Ptosis and ophthalmoplegia, proximal myopathy, dysphagia, dysphonia, dysarthria | Spasticity, ataxia, bladder symptoms, mild cognitive impairment | [140] |
6.2.3. Defects of Mitochondrial Dynamics and Quality Control
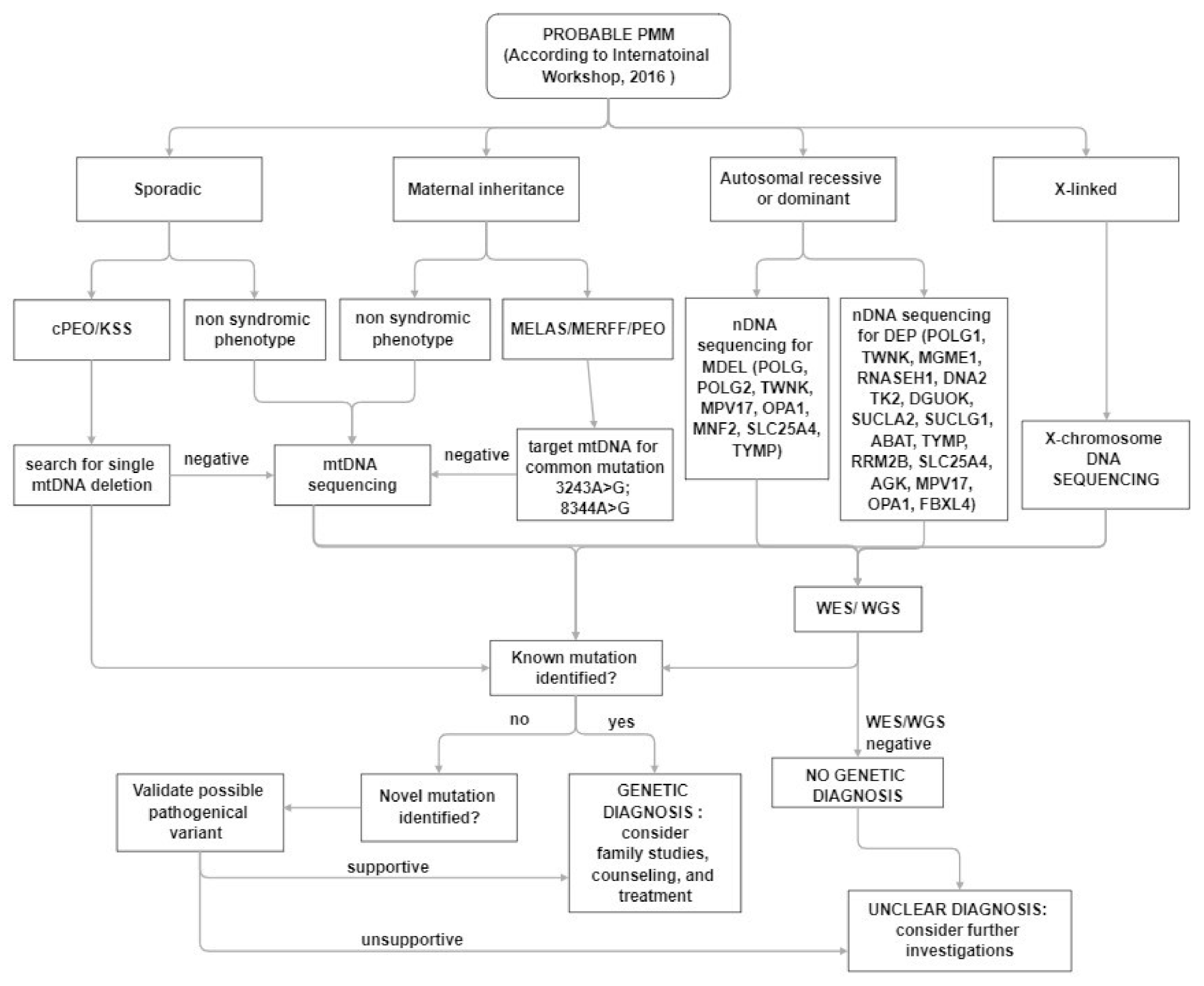
7. Genetic Diagnostic Approach in PMM
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mancuso, M.; Mcfarland, R.; Klopstock, T.; Hirano, M. International workshop: Outcome measures and clinical trial readiness in primary mitochondrial myopathies in children and adults. Consensus recommendations. 16–18 November 2016, Rome, Italy. Neuromuscul. Disord. 2017, 27, 1126–1137. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.S.; Bindoff, L.A.; Gorman, G.S.; Klopstock, T.; Kornblum, C.; Mancuso, M.; McFarland, R.; Sue, C.M.; Suomalainen, A.; Taylor, R.W.; et al. Mitochondrial disease in adults: Recent advances and future promise. Lancet Neurol. 2021, 20, 573–584. [Google Scholar] [CrossRef]
- Frazier, A.E.; Thorburn, D.R.; Compton, A.G. Mitochondrial energy generation disorders: Genes, mechanisms, and clues to pathology. J. Biol. Chem. 2019, 294, 5386–5395. [Google Scholar] [CrossRef] [PubMed]
- McCormick, E.M.; Zolkipli-Cunningham, Z.; Falk, M.J. Mitochondrial Disease Genetics Update Recent insights into the Molecular Diagnosis and Expanding Phenotype of Primary Mitochondrial Disease. Curr. Opin. Pediatr. 2018, 30, 714–724. [Google Scholar] [CrossRef]
- Chinnery, P.F.; Dimauro, P.S.; Shanske, S.; Schon, P.E.A.; Zeviani, M.; Mariotti, C.; Carrara, F.; Lombes, A.; Laforet, P.; Ogier, H.; et al. Risk of developing a mitochondrial DNA deletion disorder. Lancet 2004, 364, 592–596. [Google Scholar] [CrossRef]
- Lightowlers, R.N.; Taylor, R.W.; Turnbull, D.M. Mutations causing mitochondrial disease: What is new and what challenges remain? Science 2015, 349, 1494–1499. [Google Scholar] [CrossRef]
- Ghezzi, D.; Zeviani, M. Human diseases associated with defects in assembly of OXPHOS complexes. Essays Biochem. 2018, 62, 271–286. [Google Scholar]
- Fassone, E.; Rahman, S. Complex I deficiency: Clinical features, biochemistry and molecular genetics. J. Med. Genet. 2012, 49, 578–590. [Google Scholar] [CrossRef]
- Alston, C.L.; Rocha, M.C.; Lax, N.Z.; Turnbull, D.M.; Taylor, R.W. The genetics and pathology of mitochondrial disease. J. Pathol. 2017, 241, 236–250. [Google Scholar] [CrossRef]
- Fernandez-Vizarra, E.; Bugiani, M.; Goffrini, P.; Carrara, F.; Farina, L.; Procopio, E.; Donati, A.; Uziel, G.; Ferrero, I.; Zeviani, M. Impaired complex III assembly associated with BCS1L gene mutations in isolated mitochondrial encephalopathy. Hum. Mol. Genet. 2007, 16, 1241–1252. [Google Scholar] [CrossRef]
- Brischigliaro, M.; Zeviani, M. Cytochrome c oxidase deficiency. Biochim. Biophys. Acta Bioenerg. 2021, 1862, 148335. [Google Scholar] [CrossRef]
- Rahman, S. Mitochondrial disease in children. J. Intern. Med. 2020, 287, 609–633. [Google Scholar] [CrossRef]
- Gandhi, V.V.; Samuels, D.C. A review comparing deoxyribonucleoside triphosphate (dNTP) concentrations in the mitochondrial and cytoplasmic compartments of normal and transformed cells. Nucleosides Nucleotides Nucleic Acids 2011, 30, 317–339. [Google Scholar] [CrossRef]
- Garone, C.; Tadesse, S.; Hirano, M. Clinical and genetic spectrum of mitochondrial neurogastrointestinal encephalomyopathy. Brain A J. Neurol. 2011, 134, 3326–3332. [Google Scholar] [CrossRef]
- Tilokani, L.; Nagashima, S.; Paupe, V.; Prudent, J. Mitochondrial dynamics: Overview of molecular mechanisms. Essays Biochem. 2018, 62, 341–360. [Google Scholar]
- Rusecka, J.; Kaliszewska, M.; Bartnik, E.; Tońska, K. Nuclear genes involved in mitochondrial diseases caused by instability of mitochondrial DNA. J. Appl. Genet. 2018, 59, 43–57. [Google Scholar] [CrossRef]
- Garcia, I.; Jones, E.; Ramos, M.; Innis-Whitehouse, W.; Gilkerson, R. The little big genome: The organization of mitochondrial DNA. Front. Biosci. 2017, 22, 710–721. [Google Scholar]
- Calvo, S.E.; Mootha, V. The mitochondrial proteome and human disease. Annu. Rev. Genom. Hum. Genet. 2010, 11, 25–44. [Google Scholar] [CrossRef]
- Sato, M.; Sato, K. Maternal inheritance of mitochondrial DNA by diverse mechanisms to eliminate paternal mitochondrial DNA. Biochim. Biophys. Acta—Mol. Cell Res. 2013, 1833, 1979–1984. [Google Scholar] [CrossRef]
- Schwartz, M.; Vissing, J. Paternal inheritance of mitochondrial DNA. N. Engl. J. Med. 2002, 347, 576–580. [Google Scholar] [CrossRef]
- Ballana, E.; Govea, N.; de Cid, R.; Garcia, C.; Arribas, C.; Rosell, J.; Xavier, E. Detection of Unrecognized Low-Level mtDNA Heteroplasmy May Explain the Variable Phenotypic Expressivity of Apparently Homoplasmic mtDNA Mutations. Hum. Mutat. 2008, 29, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, R.; Faustin, B.; Rocher, C.; Malgat, M.; Mazat, J.P.; Letellier, T. Mitochondrial threshold effects. Biochem. J. 2003, 370, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Andreu, A.L.; Hanna, M.G.; Reichmann, H.; Bruno, C.; Penn, A.S.; Tanji, K.; Pallotti, F.; Iwata, S.; Bonilla, E.; Lach, B.; et al. Exercise Intolerance Due to Mutations in the Cytochrome b Gene of Mitochondrial DNA. N. Engl. J. Med. 1999, 341, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S. Mitochondrial disease and epilepsy. Dev. Med. Child Neurol. 2012, 54, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Meyerson, C.; van Stavern, G.; McClelland, C. Leber hereditary optic neuropathy: Current perspectives. Clin. Ophthalmol. 2015, 9, 1165–1176. [Google Scholar] [PubMed]
- Mancuso, M.; Filosto, M.; Mootha, V.K.; Rocchi, A.; Pistolesi, S.; Murri, L.; DiMauro, S.; Siciliano, G. A novel mitochondrial tRNAPhe mutation causes MERRF syndrome. Neurology 2004, 62, 2119–2121. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.F. Mitochondrial disease in adults: What’s old and what’s new? EMBO Mol. Med. 2015, 7, 1503–1512. [Google Scholar] [CrossRef]
- Osellame, L.D.; Blacker, T.S.; Duchen, M.R. Cellular and molecular mechanisms of mitochondrial function. Best Pr. Res. Clin. Endocrinol. Metab. 2012, 26, 711–723. [Google Scholar] [CrossRef]
- Fiedorczuk, K.; Sazanov, L.A. Mammalian Mitochondrial Complex I Structure and Disease-Causing Mutations. Trends Cell Biol. 2018, 28, 835–867. [Google Scholar] [CrossRef]
- Lake, N.J.; Compton, A.G.; Rahman, S.; Thorburn, D.R. Leigh syndrome: One disorder, more than 75 monogenic causes. Ann. Neurol. 2016, 79, 190–203. [Google Scholar] [CrossRef]
- Rafiq, J.; Duno, M.; Ostergaard, E.; Ravn, K.; Vissing, C.R.; Wibrand, F.; Vissing, J. Exercise intolerance and myoglobinuria associated with a novel maternally iherited MT-ND1 mutation. JIMD Rep. 2015, 25, 65–70. [Google Scholar]
- Gorman, G.S.; Blakely, E.L.; Hornig-Do, H.T.; Tuppen, H.A.; Greaves, L.C.; He, L.; Baker, A.; Falkous, G.; Newman, J.; Trenell, M.I.; et al. Novel MTND1 mutations cause isolated exercise intolerance, complex I deficiency and increased assembly factor expression. Clin. Sci. 2015, 128, 895–904. [Google Scholar] [CrossRef]
- Zanolini, A.; Potic, A.; Carrara, F.; Lamantea, E.; Diodato, D.; Blasevich, F.; Marchet, S.; Mora, M.; Pallotti, F.; Morandi, L.; et al. Pure myopathy with enlarged mitochondria associated to a new mutation in MTND2 gene. Mol. Genet. Metab. Rep. 2017, 10, 24–27. [Google Scholar] [CrossRef]
- Andreu, A.L.; Tanji, K.; Bruno, C.; Hadjigeorgiou, G.M.; Sue, C.M.; Jay, C.; Ohnishi, T.; Shanske, S.; Bonilla, E.; DiMauro, S. Exercise intolerance due to a nonsense mutation in the mtDNA ND4 gene. Ann. Neurol. 1999, 45, 820–823. [Google Scholar] [CrossRef]
- Downham, E.; Winterthun, S.; Nakkestad, H.L.; Hirth, A.; Halvorsen, T.; Taylor, R.W.; Bindoff, L.A. A novel mitochondrial ND5 (MTND5) gene mutation giving isolated exercise intolerance. Neuromuscul. Disord. 2008, 18, 310–314. [Google Scholar] [CrossRef]
- Musumeci, O.; Andreu, A.L.; Shanske, S.; Bresolin, N.; Comi, G.P.; Rothstein, R.; Schon, E.A.; DiMauro, S. Intragenic inversion of mtDNA: A new type of pathogenic mutation in a patient with mitochondrial myopathy. Am. J. Hum. Genet. 2000, 66, 1900–1904. [Google Scholar] [CrossRef]
- Alston, C.L.; Morak, M.; Reid, C.; Hargreaves, I.P.; Pope, S.A.S.; Land, J.M.; Heales, S.J.; Horvath, R.; Mundy, H.; Taylor, R.W. A novel mitochondrial MTND5 frameshift mutation causing isolated complex I deficiency, renal failure and myopathy. Neuromuscul. Disord. 2010, 20, 131–135. [Google Scholar] [CrossRef]
- Ng, Y.S.; Thompson, K.; Loher, D.; Hopton, S.; Falkous, G.; Hardy, S.A.; Schaefer, A.M.; Shaunak, S.; Roberts, M.E.; Lilleker, J.B.; et al. Novel MT-ND Gene Variants Causing Adult-Onset Mitochondrial Disease and Isolated Complex I Deficiency. Front. Genet. 2020, 11, 1–7. [Google Scholar] [CrossRef]
- Alston, C.L.; Compton, A.G.; Formosa, L.E.; Strecker, V.; Oláhová, M.; Haack, T.B.; Smet, J.; Stouffs, K.; Diakumis, P.; Ciara, E.; et al. Biallelic Mutations in TMEM126B Cause Severe Complex I Deficiency with a Variable Clinical Phenotype. Am. J. Hum. Genet. 2016, 99, 217–227. [Google Scholar] [CrossRef]
- Sánchez-Caballero, L.; Ruzzenente, B.; Bianchi, L.; Assouline, Z.; Barcia, G.; Metodiev, M.D.; Rio, M.; Funalot, B.; van den Brand, M.A.M.; Guerrero-Castillo, S.; et al. Mutations in Complex I Assembly Factor TMEM126B Result in Muscle Weakness and Isolated Complex I Deficiency. Am. J. Hum. Genet. 2016, 99, 208–216. [Google Scholar] [CrossRef]
- Repp, B.M.; Mastantuono, E.; Alston, C.L.; Schiff, M.; Haack, T.B.; Rötig, A.; Ardissone, A.; Lombès, A.; Catarino, C.B.; Diodato, D.; et al. Clinical, biochemical and genetic spectrum of 70 patients with ACAD9 deficiency: Is riboflavin supplementation effective? Orphanet J. Rare Dis. 2018, 13, 1–10. [Google Scholar] [CrossRef]
- Dumoulin, R.; Sagnol, I.; Ferlin, T.; Bozon, D.; Stepien, G.; Mousson, B. A novel gly290asp mitochondrial cytochrome b mutation linked to a complex III deficiency in progressive exercise intolerance. Mol. Cell Probes 1996, 10, 389–391. [Google Scholar] [CrossRef]
- Keightley, J.A.; Anitori, R.; Burton, M.D.; Quan, F.; Buist, N.R.M.; Kennaway, N.G. Mitochondrial encephalomyopathy and complex III deficiency associated with a stop-codon mutation in the cytochrome b gene. Am. J. Hum. Genet. 2000, 67, 1400–1410. [Google Scholar] [CrossRef]
- Bruno, C.; Santorelli, F.M.; Assereto, S.; Tonoli, E.; Tessa, A.; Traverso, M.; Scapolan, S.; Bado, M.; Tedeschi, S.; Minetti, C. Progressive exercise intolerance associated with a new muscle-restricted nonsense mutation (G142X) in the mitochondrial cytochrome b gene. Muscle Nerve 2003, 28, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Lamantea, E.; Carrara, F.; Mariotti, C.; Morandi, L.; Tiranti, V.; Zeviani, M. A novel nonsense mutation (Q352X) in the mitochondrial cytochrome b gene associated with a combined deficiency of complexes I and III. Neuromuscul. Disord. 2002, 12, 49–52. [Google Scholar] [CrossRef]
- Kollberg, G.; Moslemi, A.R.; Lindberg, C.; Holme, E.; Oldfors, A. Mitochondrial myopathy and rhabdomyolysis associated with a novel nonsense mutation in the gene encoding cytochrome c oxidase subunit I. J. Neuropathol. Exp. Neurol. 2005, 64, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Taanman, J.W.; Cooper, J.M.; Nelson, I.; Hargreaves, I.; Meunier, B.; Hanna, M.G.; García, J.J.; Capaldi, R.A.; Lake, B.D.; et al. A missense mutation of cytochrome oxidase subunit II causes defective assembly and myopathy. Am. J. Hum. Genet. 1999, 65, 1030–1039. [Google Scholar] [CrossRef]
- Roos, S.; Sofou, K.; Hedberg-Oldfors, C.; Kollberg, G.; Lindgren, U.; Thomsen, C.; Tulinius, M.; Oldfors, A. Mitochondrial complex IV deficiency caused by a novel frameshift variant in MT-CO2 associated with myopathy and perturbed acylcarnitine profile. Eur. J. Hum. Genet. 2018, 27, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Horváth, R.; Lochmüller, H.; Hoeltzenbein, M.; Müller-Höcker, J.; Schoser, B.G.; Pongratz, D.; Jaksch, M. Spontaneous recovery of a childhood onset mitochondrial myopathy caused by a stop mutation in the mitochondrial cytochrome c oxidase III gene. J. Med. Genet. 2004, 41, 1–5. [Google Scholar] [CrossRef][Green Version]
- Inoue, M.; Uchino, S.; Iida, A.; Noguchi, S.; Hayashi, S.; Takahashi, T.; Fujii, K.; Komaki, H.; Takeshita, E.; Nonaka, I.; et al. COX6A2 variants cause a muscle-specific cytochrome c oxidase deficiency. Ann. Neurol. 2019, 86, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Pitceathly, R.D.S.; Taanman, J.W.; Rahman, S.; Meunier, B.; Sadowski, M.; Cirak, S.; Hargreaves, I.; Land, J.M.; Nanji, T.; Polke, J.M.; et al. COX10 mutations resulting in complex multisystem mitochondrial disease that remains stable into adulthood. JAMA Neurol. 2013, 70, 1556–1561. [Google Scholar] [PubMed]
- Swalwell, H.; Kirby, D.M.; Blakely, E.L.; Mitchell, A.; Salemi, R.; Sugiana, C.; Compton, A.G.; Tucker, E.J.; Ke, B.X.; Lamont, P.J.; et al. Respiratory chain complex I deficiency caused by mitochondrial DNA mutations. Eur. J. Hum. Genet. 2011, 19, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Moreira, D.; Ugalde, C.; Smeets, R.; Rodenburg, R.J.T.; Lopez-Laso, E.; Ruiz-Falco, M.L.; Briones, P.; Martin, M.A.; Smeitink, J.A.M.; Arenas, J. X-linked NDUFA1 gene mutations associated with mitochondrial encephalomyopathy. Ann. Neurol. 2007, 61, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Van Rahden, V.A.; Fernandez-Vizarra, E.; Alawi, M.; Brand, K.; Fellmann, F.; Horn, D.; Zeviani, M.; Kutsche, K. Mutations in NDUFB11, encoding a complex i component of the mitochondrial respiratory chain, cause microphthalmia with linear skin defects syndrome. Am. J. Hum. Genet. 2015, 96, 640–650. [Google Scholar] [CrossRef]
- Pagniez-Mammeri, H.; Loublier, S.; Legrand, A.; Bénit, P.; Rustin, P.; Slama, A. Mitochondrial complex I deficiency of nuclear origin. I. Structural genes. Mol. Genet. Metab. 2012, 105, 163–172. [Google Scholar] [CrossRef]
- Fassone, E.; Duncan, A.J.; Taanman, J.; Pagnamenta, A.T.; Sadowski, M.I.; Holand, T.; Qasim, W.; Rutland, P.; Calvo, S.E.; Mootha, V.K.; et al. FOXRED1, encoding an FAD-dependent oxidoreductase complex-I-specific molecular chaperone, is mutated in infantile-onset mitochondrial encephalopathy. Hum. Mol. Genet. 2015, 24, 4183. [Google Scholar] [CrossRef]
- Haack, T.B.; Danhauser, K.; Haberberger, B.; Hoser, J.; Strecker, V.; Boehm, D.; Uziel, G.; Lamantea, E.; Invernizzi, F.; Poulton, J.; et al. Exome sequencing identifies ACAD9 mutations as a cause of complex I deficiency. Nat. Genet. 2010, 42, 1131–1134. [Google Scholar] [CrossRef]
- Saada, A.; Vogel, R.O.; Hoefs, S.J.; Van den Brand, M.A.; Wessels, H.J.; Willems, P.H.; Venselaar, H.; Shaag, A.; Barghuti, F.; Reish, O.; et al. Mutations in NDUFAF3 (C3ORF60), Encoding an NDUFAF4 (C6ORF66)-Interacting Complex I Assembly Protein, Cause Fatal Neonatal Mitochondrial Disease. Am. J. Hum. Genet. 2009, 84, 718–727. [Google Scholar] [CrossRef]
- Jain-Ghai, S.; Cameron, J.M.; al Maawali, A.; Blaser, S.; Mackay, N.; Robinson, B.; Raiman, J. Complex II deficiency-A case report and review of the literature. Am. J. Hum. Genet. Part A 2013, 161, 285–294. [Google Scholar] [CrossRef]
- Renkema, G.H.; Wortmann, S.B.; Smeets, R.J.; Venselaar, H.; Antoine, M.; Visser, G.; Ben-Omran, T.; van den Heuvel, L.P.; Timmers, H.J.; Smeitink, J.A.; et al. SDHA mutations causing a multisystem mitochondrial disease: Novel mutations and genetic overlap with hereditary tumors. Eur. J. Hum. Genet. 2015, 23, 202–209. [Google Scholar] [CrossRef]
- Alston, C.L.; Davison, J.E.; Meloni, F.; van der Westhuizen, F.H.; He, L.; Hornig-Do, H.T.; Peet, A.C.; Gissen, P.; Goffrini, P.; Ferrero, I.; et al. Recessive germline SDHA and SDHB mutations causing leukodystrophy and isolated mitochondrial complex II deficiency. J. Med. Genet. 2021, 49, 569–577. [Google Scholar] [CrossRef]
- Ohlenbusch, A.; Edvardson, S.; Skorpen, J.; Bjornstad, A.; Saada, A.; Elpeleg, O.; Gärtner, J.; Brockmann, K. Leukoencephalopathy with accumulated succinate is indicative of SDHAF1 related complex II deficiency. Orphanet J. Rare Dis. 2012, 7, 7–11. [Google Scholar] [CrossRef]
- Xia, D.; Esser, L.; Tang, W.K.; Zhou, F.; Zhou, Y.; Yu, L.; Yu, C.A. Structural analysis of cytochrome bc1 complexes: Implications to the mechanism of function. Biochim. Biophys. Acta Bioenerg. 2013, 1827, 1278–1294. [Google Scholar] [CrossRef]
- Fernández-Vizarra, E.; Zeviani, M. Nuclear gene mutations as the cause of mitochondrial complex III deficiency. Front. Genet. 2015, 6, 1–11. [Google Scholar] [CrossRef]
- Spiro, A.J.; Moore, C.L.; Prineas, J.W.; Strasberg, P.M.; Rapin, I. A Cytochrome-Related Inherited Disorder of the Nervous System and Muscle. Arch. Neurol. 1970, 23, 103–112. [Google Scholar] [CrossRef]
- Darley Usmar, V.M.; Kennaway, N.G.; Buist, N.R.M.; Capaldi, R.A. Deficiency in ubiquinone cytochrome c reductase in a patient with mitochondrial myopathy and lactic acidosis. Proc. Natl. Acad. Sci. USA 1983, 80, 5103–5106. [Google Scholar] [CrossRef]
- Hayes, D.J.; Lecky, B.R.F.; Landon, D.N.; Morgan Hughes, J.A.; Clark, J.B. A new Mitochondrial myopathy biochemical studies revealing a deficiency in the cytochrome b-c complex (complex III) of the respiratory chain. Brain 1984, 107, 1165–1177. [Google Scholar] [CrossRef]
- Fouad Bouzidi, M.; Schägger, H.; Collombet, J.M.; Carrier, H.; Flocard, F.; Quard, S.; Mousson, B.; Godinot, C. Decreased expression ofubiquinol-cytochrome c reductase subunits in patients exhibiting mitochondrial myopathy with progressive exercise intolerance. Neuromuscul. Disord. 1993, 3, 599–604. [Google Scholar] [CrossRef]
- De Coo, I.F.M.; Renier, W.O.; Ruitenbeek, W.; Ter Laak, H.J.; Bakker, M.; Schagger, H.; Van Oost, B.A.; Smeets, H.J. A 4–Base Pair Deletion in the Mitochondrial Cytochrome b Gene Associated with Parkinsonism/MELAS Overlap Syndrome. Ann. Neurol. 1999, 45, 130–133. [Google Scholar] [CrossRef]
- Wibrand, F.; Ravn, K.; Schwartz, M.; Rosenberg, T.; Horn, N.; Vissing, J. Multisystem Disorder Associated with a Missense Mutation in the Mitochondrial Cytochrome b gene. Ann. Neurol. 2001, 50, 540–543. [Google Scholar] [CrossRef]
- Schuelke, M.; Krude, H.; Finckh, B.; Mayatepek, E.; Janssen, A.; Schmelz, M.; Trefz, F.; Trijbels, F.; Smeitink, J. Septo-optic Dysplasia Associated with a New Mitochondrial Cytochrome b Mutation. Ann. Neurol. 2002, 51, 388–392. [Google Scholar] [CrossRef]
- Legros, F.; Chatzoglou, E.; Frachon, P.; Ogier De Baulny, H.; Laforêt, P.; Jardel, C.; Godinot, C.; Lombès, A. Functional characterization of novel mutations in the human cytochrome b gene. Eur. J. Hum. Genet. 2001, 9, 510–518. [Google Scholar] [CrossRef]
- Mancuso, M.; Filosto, M.; Stevens, J.C.; Patterson, M.; Shanske, S.; Krishna, S.; DiMauro, S. Mitochondrial myopathy and complex III deficiency in a patient with a new stop-codon mutation (G339X) in the cytochrome b gene. J. Neurol. Sci. 2003, 209, 61–63. [Google Scholar] [CrossRef]
- Pitceathly, R.D.S.; Rahman, S.; Wedatilake, Y.; Polke, J.M.; Cirak, S.; Foley, A.R.; Sailer, A.; Hurles, M.E.; Stalker, J.; Hargreaves, I.; et al. NDUFA4 Mutations Underlie Dysfunction of a Cytochrome c Oxidase Subunit Linked to Human Neurological Disease. Cell Rep. 2013, 3, 1795–1805. [Google Scholar] [CrossRef]
- Shoubridge, E.A. Cytochrome c oxidase deficiency. Am. J. Med. Genet. 2001, 106, 46–52. [Google Scholar] [CrossRef]
- Clark, K.M.; Taylor, R.W.; Johnson, M.A.; Chinnery, P.F.; Chrzanowska-Lightowlers, Z.M.A.; Andrews, R.M.; Nelson, I.P.; Wood, N.W.; Lamont, P.J.; Hanna, M.G.; et al. An mtDNA mutation in the initiation codon of the cytochrome C oxidase subunit II gene results in lower levels of the protein and a mitochondrial encephalomyopathy. Am. J. Hum. Genet. 1999, 64, 1330–1339. [Google Scholar] [CrossRef][Green Version]
- DiMauro, S.; Nicholson, J.F.; Hays, A.P.; Eastwood, A.B.; Papadimitriou, A.; Koenigsberger, R.; DeVivo, D.C. Benign infantile mitochondrial myopathy due to reversible cytochrome c oxidase deficiency. Ann. Neurol. 1983, 14, 226–234. [Google Scholar] [CrossRef]
- Horvath, R.; Kemp, J.P.; Tuppen, H.A.L.; Hudson, G.; Oldfors, A.; Marie, S.K.N.; Moslemi, A.-R.; Servidei, S.; Holme, E.; Shanske, S.; et al. Molecular basis of infantile reversible cytochrome c oxidase deficiency myopathy. Brain 2009, 132, 3165–3174. [Google Scholar] [CrossRef]
- Papadopoulou, L.C.; Sue, C.M.; Davidson, M.M.; Tanji, K.; Nishino, I.; Sadlock, J.E.; Krishna, S.; Walker, W.; Selby, J.; Glerum, D.M.; et al. Fatal infantile cardioencephalomyopathy with COX deficiency and mutations in SCO2, a COX assembly gene. Nat. Genet. 1999, 23, 333–337. [Google Scholar] [CrossRef]
- Salviati, L.; Sacconi, S.; Rasalan, M.M.; Kronn, D.F.; Braun, A.; Canoll, P.; Davidson, M.; Shanske, S.; Bonilla, E.; Hays, A.P.; et al. Cytochrome c oxidase deficiency due to a novel SCO2 mutation mimics Werdnig-Hoffmann disease. Arch. Neurol. 2002, 59, 862–865. [Google Scholar] [CrossRef] [PubMed]
- Barca, E.; Ganetzky, R.D.; Potluri, P.; Juanola-Falgarona, M.; Gai, X.; Li, D.; Jalas, C.; Hirsch, Y.; Emmanuele, V.; Tadesse, S.; et al. USMG5 Ashkenazi Jewish founder mutation impairs mitochondrial complex V dimerization and ATP synthesis. Hum. Mol. Genet. 2018, 27, 3305–3312. [Google Scholar] [CrossRef] [PubMed]
- Hejzlarová, K.; Mráček, T.; Vrbacký, M.; Kaplanová, V.; Karbanová, V.; Nůsková, H.; Pecina, P.; Houštěk, J. Nuclear genetic defects of mitochondrial ATP synthase. Physiol. Res. 2014, 63, S57–S71. [Google Scholar] [CrossRef] [PubMed]
- Holt, I.J.; Harding, A.E.; Petty, R.K.H.; Morgan-Hughes, J.A. A new mitochondrial disease associated with mitochondrial DNA heteroplasmy. Am. J. Hum. Genet. 1990, 46, 428–433. [Google Scholar]
- Ganetzky, R.D.; Stendel, C.; McCormick, E.M.; Zolkipli-Cunningham, Z.; Goldstein, A.C.; Klopstock, T.; Falk, M.J. MT-ATP6 mitochondrial disease variants: Phenotypic and biochemical features analysis in 218 published cases and cohort of 14 new cases. Hum. Mutat. 2019, 40, 499–515. [Google Scholar] [CrossRef]
- Gelfand, J.M.; Duncan, J.L.; Racine, C.A.; Gillum, L.A.; Chin, C.T.; Zhang, Y.; Zhang, Q.; Wong, L.J.C.; Roorda, A.; Green, A.J. Heterogeneous patterns of tissue injury in NARP syndrome. J. Neurol. 2011, 258, 440–448. [Google Scholar] [CrossRef]
- D’Aurelio, M.; Vives-Bauza, C.; Davidson, M.M.; Manfredi, G. Mitochondrial DNA background modifies the bioenergetics of NARP/MILS ATP6 mutant cells. Hum. Mol. Genet. 2009, 19, 374–386. [Google Scholar] [CrossRef]
- Kara, B.; Arikan, M.; Maraş, H.; Abaci, N.; Çakiris, A.; Üstek, D. Whole mitochondrial genome analysis of a family with NARP/MILS caused by m.8993T>C mutation in the MT-ATP6 gene. Mol. Genet. Metab. 2012, 107, 389–393. [Google Scholar] [CrossRef]
- Pitceathly, R.D.; Murphy, S.M.; Cottenie, E.; Chalasani, A.; Sweeney, M.G.; Woodward, C.; Mudanohwo, E.E.; Hargreaves, I.; Heales, S.; Land, J.; et al. Genetic dysfunction of MT-ATP6 causes axonal Charcot-Marie-Tooth disease. Neurology 2012, 79, 1145–1154. [Google Scholar] [CrossRef]
- Auré, K.; Dubourg, O.; Jardel, C.; Clarysse, L.; Sternberg, D.; Fournier, E.; Laforêt, P.; Streichenberger, N.; Petiot, P.; Gervais-Bernard, H.; et al. Episodic weakness due to mitochondrial DNA MT-ATP6/8 mutations. Neurology 2013, 81, 1810–1818. [Google Scholar] [CrossRef]
- Burrage, L.C.; Tang, S.; Wang, J.; Donti, T.R.; Walkiewicz, M.; Graham, B.H.; Wong, L.; Scaglia, F. Mitochondrial Myopathy, Lactic Acidosis, and Sideroblastic Anemia (MLASA) Plus Associated with a novel De Novo Mutation (m.8969G>A) in the Mitochondrial Encoded ATP6 gene. Mol. Genet. Metab. 2014, 113, 207–212. [Google Scholar] [CrossRef]
- Boczonadi, V.; Ricci, G.; Horvath, R. Mitochondrial DNA transcription and translation: Clinical syndromes. Essays Biochem. 2018, 62, 321–340. [Google Scholar]
- Yarham, J.W.; Al-Dosary, M.; Blakely, E.L.; Alston, C.L.; Taylor, R.W.; Elson, J.L.; Mcfarland, R. A comparative analysis approach to determining the pathogenicity of mitochondrial tRNA mutations. Hum. Mutat. 2011, 32, 1319–1325. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Momoi, M.Y.; Tominaga, K.; Momoi, T.; Nihei, K.; Yanagisawa, M.; Kagawa, Y.; Ohta, S. A Point Mutation in the mitochondrial tRNALeu(UUR) gene in MELAS (Mitochondrial myopathy, encephalopathy, lactic acidosis and stroke like episodes). Biochem. Biophys. Res. Commun. 1990, 173, 816–822. [Google Scholar] [CrossRef]
- Yatsuga, S.; Povalko, N.; Nishioka, J.; Katayama, K.; Kakimoto, N.; Matsuishi, T.; Kakuma, T.; Koga, Y. MELAS: A nationwide prospective cohort study of 96 patients in Japan. Biochim. Biophys. Acta—Gen. Subj. 2021, 1820, 619–624. [Google Scholar] [CrossRef]
- Mahale, R.R.; Gautham, J.; Mailankody, P.; Padmanabha, H.; Mathuranath, P.S. Isolated mitochondrial myopathy due to m.3243A > G mutation in MT-TL1 gene. Acta Neurol. Belg. 2021, 2–3. [Google Scholar] [CrossRef]
- Finsterer, J.; Zarrouk-Mahjoub, S.; Shoffner, J.M. MERRF Classification: Implications for Diagnosis and Clinical Trials. Pediatric Neurol. 2018, 80, 8–23. [Google Scholar] [CrossRef]
- Lu, Y.; Zhao, D.; Yao, S.; Wu, S.; Hong, D.; Wang, Q.; Liu, J.; Smeitink, J.A.M.; Yuan, Y.; Wang, Z. Mitochondrial tRNA genes are hotspots for mutations in a cohort of patients with exercise intolerance and mitochondrial myopathy. J. Neurol. Sci. 2017, 379, 137–143. [Google Scholar] [CrossRef]
- Lehmann, D.; Schubert, K.; Joshi, P.R.; Hardy, S.A.; Tuppen, H.A.L.; Baty, K.; Blakely, E.L.; Bamberg, C.; Zierz, S.; Deschauer, M.; et al. Pathogenic mitochondrial mt-tRNA Ala variants are uniquely associated with isolated myopathy. Eur. J. Hum. Genet. 2015, 23, 1735–1738. [Google Scholar] [CrossRef]
- Cardaioli, E.; da Pozzo, P.; Malfatti, E.; Gallus, G.N.; Rubegni, A.; Malandrini, A.; Gaudiano, C.; Guidi, L.; Serni, G.; Berti, G.; et al. Chronic progressive external ophthalmoplegia: A new heteroplasmic tRNALeu(CUN) mutation of mitochondrial DNA. J. Neurol. Sci. 2008, 272, 106–109. [Google Scholar] [CrossRef]
- Karadimas, C.L.; Salviati, L.; Sacconi, S.; Chronopoulou, P.; Shanske, S.; Bonilla, E.; de Vivo, D.C.; DiMauro, S. Mitochondrial myopathy and ophthalmoplegia in a sporadic patient with the G12315A mutation in mitochondrial DNA. Neuromuscul. Disord. 2002, 12, 865–868. [Google Scholar] [CrossRef]
- Soldath, P.; Madsen, K.L.; Buch, A.E.; Duno, M.; Wibrand, F.; Vissing, J. Pure exercise intolerance and ophthalmoplegia associated with the m.12,294G > A mutation in the MT-TL2 gene: A case report. BMC Musculoskelet. Disord. 2017, 18, 1–6. [Google Scholar] [CrossRef]
- Hellebrekers, D.M.E.I.; Blakely, E.L.; Hendrickx, A.T.M.; Hardy, S.A.; Hopton, S.; Falkous, G.; de Coo, I.F.M.; Smeets, H.J.M.; van der Beek, N.M.E.; Taylor, R.W. A novel mitochondrial m.4414T>C MT-TM gene variant causing progressive external ophthalmoplegia and myopathy. Neuromuscul. Disord. 2019, 29, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Visuttijai, K.; Hedberg-Oldfors, C.; Lindgren, U.; Nordström, S.; Elíasdóttir, Ó.; Lindberg, C.; Oldfors, A. Progressive external ophthalmoplegia associated with novel MT-TN mutations. Acta Neurol. Scand. 2021, 143, 103–108. [Google Scholar] [CrossRef]
- Darin, N.; Hedberg-Oldfors, C.; Kroksmark, A.K.; Moslemi, A.R.; Kollberg, G.; Oldfors, A. Benign mitochondrial myopathy with exercise intolerance in a large multigeneration family due to a homoplasmic m.3250T>C mutation in MTTL1. Eur. J. Neurol. 2017, 24, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Mimaki, M.; Hatakeyama, H.; Komaki, H.; Yokoyama, M.; Arai, H.; Kirino, Y.; Suzuki, T.; Nishino, I.; Nonaka, I.; Goto, Y.I. Reversible infantile respiratory chain deficiency: A clinical and molecular study. Ann. Neurol. 2010, 68, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M.; Brady, L.; MacNeil, L. Myasthenia graves-like symptoms associated with rare mitochondrial mutation (m.5728T>C). Mitochondrion 2019, 47, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, I.P. Coenzyme Q10 in Mitochondrial and Lysosomal Disorders. J. Clin. Med. 2021, 10, 1970. [Google Scholar] [CrossRef] [PubMed]
- Quinzii, C.M.; Lopez, L.C.; Naini, A.; DiMauro, S.; Hirano, M. Human CoQ10 deficiencies. Biofactors 2008, 32, 113–118. [Google Scholar] [CrossRef]
- Ogasahara, S.; Engel, A.G.; Frens, D.; Mack, D. Muscle coenzyme Q deficiency in familial mitochondrial encephalomyopathy. Proc. Natl. Acad. Sci. USA 1989, 86, 2379–2382. [Google Scholar] [CrossRef]
- Salviati, L.; Trevisson, E.; Doimo, M.; Navas, P. Primary Coenzyme Q10 Deficiency. In GeneReviews® [Internet]; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Lalani, S.R.; Vladutiu, G.D.; Plunkett, K.; Lotze, T.E.; Adesina, A.M.; Scaglia, F. Isolated mitochondrial myopathy associated with muscle coenzyme Q10 deficiency. Arch. Neurol. 2005, 62, 317–320. [Google Scholar] [CrossRef]
- Horvath, R.; Schneiderat, P.; Schoser, B.G.H.; Gempel, K.; Neuen-Jacob, E.; Plöger, H.; Müller-Höcker, J.; Pongratz, D.E.; Naini, A.; DiMauro, S.; et al. Coenzyme Q10 deficiency and isolated myopathy. Neurology 2006, 66, 253–255. [Google Scholar] [CrossRef]
- Gempel, K.; Topaloglu, H.; Talim, B.; Schneiderat, P.; Schoser, B.G.; Hans, V.H.; Pálmafy, B.; Kale, G.; Tokatli, A.; Quinzii, C.; et al. The myopathic form of coenzyme Q10 deficiency is caused by mutations in the electron-transferring-flavoprotein dehydrogenase (ETFDH) gene. Brain 2007, 130, 2037–2044. [Google Scholar] [CrossRef]
- Schon, E.A.; Rizzuto, R.; Moraes, C.T.; Nakase, H.; Zeviani, M.; DiMauro, S. A direct repeat is a hotspot for large-scale deletion of human mitochondrial DNA. Science 1989, 244, 346–349. [Google Scholar] [CrossRef]
- Mancuso, M.; Orsucci, D.; Angelini, C.; Bertini, E.; Carelli, V.; Comi, G.P.; Donati, M.A.; Federico, A.; Minetti, C.; Moggio, M.; et al. Redefining phenotypes associated with mitochondrial DNA single deletion. J. Neurol. 2015, 262, 1301–1309. [Google Scholar] [CrossRef]
- Grady, J.P.; Campbell, G.; Ratnaike, T.; Blakely, E.L.; Falkous, G.; Nesbitt, V.; Schaefer, A.M.; McNally, R.J.; Gorman, G.S.; Taylor, R.W.; et al. Disease progression in patients with single, large-scale mitochondrial DNA deletions. Brain 2014, 137, 323–334. [Google Scholar] [CrossRef]
- Wild, K.T.; Goldstein, A.C.; Muraresku, C.; Ganetzky, R.D. Broadening the phenotypic spectrum of Pearson syndrome: Five new cases and a review of the literature. Am. J. Med. Genet. Part A 2019, 182, 365–373. [Google Scholar] [CrossRef]
- Viscomi, C.; Zeviani, M. MtDNA-maintenance defects: Syndromes and genes. J. Inherit. Metab. Dis. 2017, 40, 587–599. [Google Scholar] [CrossRef]
- Deschauer, M.; Hudson, G.; Muller, T.; Taylor, R.W.; Chinnery, P.F.; Zierz, S. A novel ANT1 gene mutation with probable germline mosaicism in autosomal dominant progressive external ophthalmoplegia. Neuromuscul. Disord. 2005, 15, 311–315. [Google Scholar] [CrossRef]
- Kaukonen, J.; Juselius, J.K.; Tiranti, V.; Kyttala, A.; Zeviani, M.; Comi, G.P.; Keranen, S.; Peltonen, L.; Suomalainen, A. Role of adenine nucleotide translocator 1 in mtDNA maintenance. Science 2000, 289, 782–785. [Google Scholar] [CrossRef]
- Finsterer, J.; Zarrouk-Mahjoub, S. Phenotypic spectrum of SLC25A4 mutations. Biomed. Rep. 2018, 9, 119–122. [Google Scholar]
- Van Goethem, G.; Dermaut, B.; Lofgren, A.; Martin, J.J.; Van Broeckhoven, C. Mutation of POLG is associated with progressive external ophthalmoplegia characterized by mtDNA deletions. Nat. Genet. 2001, 28, 211–212. [Google Scholar] [CrossRef]
- Tang, S.; Wang, J.; Lee, N.C.; Milone, M.; Halberg, M.C.; Schmitt, E.S.; Craigen, W.J.; Zhang, W.; Wong, L.J. Mitochondrial DNA polymerase gamma mutations: An ever expanding molecular and clinical spectrum. J. Med. Genet. 2011, 48, 669–681. [Google Scholar] [CrossRef]
- Walter, M.C.; Czermin, B.; Muller-Ziermann, S.; Bulst, S.; Stewart, J.D.; Hudson, G.; Schneiderat, P.; Abicht, A.; Holinski-Feder, E.; Lochmuller, H.; et al. Late-onset ptosis and myopathy in a patient with a heterozygous insertion in POLG2. J. Neurol. 2010, 257, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Van Maldergem, L.; Besse, A.; De Paepe, B.; Blakely, E.L.; Appadurai, V.; Humble, M.M.; Piard, J.; Craig, K.; He, L.; Hella, P.; et al. POLG2 deficiency causes adult-onset syndromic sensory neuropathy, ataxia and parkinsonism. Ann. Clin. Transl. Neurol. 2016, 4, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Spelbrink, J.N.; Li, F.Y.; Tiranti, V.; Nikali, K.; Yuan, Q.P.; Tariq, M.; Wanrooij, S.; Garrido, N.; Comi, G.; Morandi, L.; et al. Human mitochondrial DNA deletions associated with mutations in the gene encoding Twinkle, a phage T7 gene 4-like protein localized in mitochondria. Nat. Genet. 2001, 29, 100. [Google Scholar] [CrossRef] [PubMed]
- Baloh, R.H.; Salavaggione, E.; Milbrandt, J.; Pestronk, A. Familial parkinsonism and ophthalmoplegia from a mutation in the mitochondrial DNA helicase twinkle. Arch. Neurol. 2007, 64, 998–1000. [Google Scholar] [CrossRef] [PubMed]
- Kornblum, C.; Nicholls, T.J.; Haack, T.B.; Scholer, S.; Peeva, V.; Danhauser, K.; Hallmann, K.; Zsurka, G.; Rorbach, J.; Iuso, A.; et al. Loss-of-function mutations in MGME1 impair mtDNA replication and cause multisystemic mitochondrial disease. Nat. Genet. 2013, 45, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Ronchi, D.; Garone, C.; Bordoni, A.; Gutierrez Rios, P.; Calvo, S.E.; Ripolone, M.; Ranieri, M.; Rizzuti, M.; Villa, L.; Magri, F.; et al. Next-generation sequencing reveals DGUOK mutations in adult patients with mitochondrial DNA multiple deletions. Brain 2012, 135, 3404–3415. [Google Scholar] [CrossRef]
- Reyes, A.; Melchionda, L.; Nasca, A.; Carrara, F.; Lamantea, E.; Zanolini, A.; Lamperti, C.; Fang, M.; Zhang, J.; Ronchi, D.; et al. RNASEH1 mutations impair mtDNA replication and cause adult-onset mitochondrial encephalomyopathy. Am. J. Hum. Genet. 2015, 97, 186–193. [Google Scholar] [CrossRef]
- El-Hattab, A.W.; Scaglia, F. Mitochondrial DNA depletion syndromes: Review and updates of genetic basis, manifestations, and therapeutic options. Neurotherapeutics 2013, 10, 186–198. [Google Scholar] [CrossRef]
- Carrozzo, R.; Dionisi-Vici, C.; Steuerwald, U.; Lucioli, S.; Deodato, F.; Di Giandomenico, S.; Bertini, E.; Franke, B.; Kluijtmans, L.A.; Meschini, M.C.; et al. SUCLA2 mutations are associated with mild methylmalonic aciduria, Leigh-like encephalomyopathy, dystonia and deafness. Brain 2007, 130, 862–874. [Google Scholar] [CrossRef] [PubMed]
- El-Hattab, A.W.; Scaglia, F. SUCLG1-Related Mitochondrial DNA Depletion Syndrome, Encephalomyopathic Form with Methylmalonic Aciduria. In GeneReviews® [Internet]; 1993–2021; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mirzaa, G., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 2017. [Google Scholar]
- Carrozzo, R.; Verrigni, D.; Rasmussen, M.; de Coo, R.; Amartino, H.; Bianchi, M.; Buhas, D.; Mesli, S.; Naess, K.; Born, A.P.; et al. Succinate-CoA ligase deficiency due to mutations in SUCLA2 and SUCLG1: Phenotype and genotype correlations in 71 patients. J. Inherit. Metab. Dis. 2016, 39, 243–252. [Google Scholar] [CrossRef]
- Garone, C.; Taylor, R.; Nascimento, A.; Poulton, J.; Fratter, C.; Domínguez-González, C.; Evans, J.C.; Loos, M.; Isohanni, P.; Suomalainen, A.; et al. Retrospective natural history of thymidine kinase 2 deficiency. J. Med. Genet. 2018, 55, 515–521. [Google Scholar] [CrossRef]
- Buchaklian, A.H.; Helbling, D.; Ware, S.M.; Dimmock, D.P. Recessive deoxyguanosine kinase deficiency causes juvenile onset mitochondrial myopathy. Mol. Genet. Metab. 2012, 107, 92–94. [Google Scholar] [CrossRef]
- Bornstein, B.; Area, E.; Flanigan, K.M.; Ganesh, J.; Jayakar, P.; Swoboda, K.J.; Coku, J.; Naini, A.; Shanske, S.; Tanji, K.; et al. Mitochondrial DNA depletion syndrome due to mutations in the RRM2B gene. Neuromuscul. Disord. 2008, 18, 453–459. [Google Scholar] [CrossRef]
- Keshavan, N.; Abdenur, J.; Anderson, G.; Assouline, Z.; Barcia, G.; Bouhikbar, L.; Chakrapani, A.; Cleary, M.; Cohen, M.C.; Feillet, F.; et al. The natural history of infantile mitochondrial DNA depletion syndrome due to RRM2B deficiency. Genet. Med. 2020, 22, 199–209. [Google Scholar] [CrossRef]
- Amati-Bonneau, P.; Valentino, M.L.; Reynier, P.; Gallardo, M.E.; Bornstein, B.; Boissière, A.; Campos, Y.; Rivera, H.; de la Aleja, J.G.; Carroccia, R.; et al. OPA1 mutations induce mitochondrial DNA instability and optic atrophy ‘plus’ phenotypes. Brain 2008, 131, 338–351. [Google Scholar] [CrossRef]
- Pfeffer, G.; Gorman, G.S.; Griffin, H.; Kurzawa-Akanbi, M.; Blakely, E.L.; Wilson, I.; Sitarz, K.; Moore, D.; Murphy, J.L.; Alston, C.L.; et al. Mutations in the SPG7 gene cause chronic progressive external ophthalmoplegia through disordered mitochondrial DNA maintenance. Brain 2014, 137, 1323–1336. [Google Scholar] [CrossRef]
- Lopez-Gomez, C.; Levy, R.J.; Sanchez-Quintero, M.J.; Juanola-Falgarona, M.; Barca, E.; Garcia-Diaz, B.; Tadesse, S.; Garone, C.; Hirano, M. Deoxycytidine and Deoxythymidine Treatment for Thymidine Kinase 2 Deficiency. Ann. Neurol. 2017, 81, 641–652. [Google Scholar] [CrossRef]
- El-Hattab, A.W.; Scaglia, F.; Wong, L.J. Deoxyguanosine Kinase Deficiency. In GeneReviews® [Internet]; 1993–2021; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Mirzaa, G., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 2009. [Google Scholar]
- Franzolin, E.; Salata, C.; Bianchi, V.; Rampazzo, C. The Deoxynucleoside Triphosphate Triphosphohydrolase Activity of SAMHD1 Protein Contributes to the Mitochondrial DNA Depletion Associated with Genetic Deficiency of Deoxyguanosine Kinase. J. Biol. Chem. 2015, 290, 25986–25996. [Google Scholar] [CrossRef]
- Nishino, I.; Spinazzola, A.; Hirano, M. MNGIE: From nuclear DNA to mitochondrial DNA. Neuromuscul. Disord. 2001, 11, 7–10. [Google Scholar] [CrossRef]
- Halter, J.P.; Michael, W.; Schüpbach, M.; Mandel, H.; Casali, C.; Orchard, K.; Collin, M.; Valcarcel, D.; Rovelli, A.; Filosto, M.; et al. Allogeneic haematopoietic stem cell transplantation for mitochondrial neurogastrointestinal encephalomyopathy. Brain 2015, 138, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Torres-Torronteras, J.; Cabrera-Pérez, R.; Vila-Julià, F.; Viscomi, C.; Cámara, Y.; Hirano, M.; Zeviani, M.; Martí, R. Long-Term Sustained Effect of Liver-Targeted Adeno-Associated Virus Gene Therapy for Mitochondrial Neurogastrointestinal Encephalomyopathy. Hum. Gene Ther. 2018, 29, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Besse, A.; Wu, P.; Bruni, F.; Donti, T.; Graham, B.H.; Craigen, W.J.; McFarland, R.; Moretti, P.; Lalani, S.; Scott, K.L.; et al. The GABA transaminase, ABAT, is essential for mitochondrial nucleoside metabolism. Cell Metab. 2015, 21, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Besse, A.; Petersen, A.K.; Hunter, J.V.; Appadurai, V.; Lalani, S.R.; Bonnen, P.E. Personalized medicine approach confirms a milder case of ABAT deficiency. Mol. Brain 2016, 9, 93. [Google Scholar] [CrossRef]
- Bourdon, A.; Minai, L.; Serre, V.; Jais, J.P.; Sarzi, E.; Aubert, S.; Chrétien, D.; de Lonlay, P.; Paquis-Flucklinger, V.; Arakawa, H.; et al. Mutation of RRM2B, encoding p53-controlled ribonucleotide reductase (p53R2), causes severe mitochondrial DNA depletion. Nat. Genet. 2007, 39, 776–780. [Google Scholar] [CrossRef]
- Fratter, C.; Raman, P.; Alston, C.L.; Blakely, E.L.; Craig, K.; Smith, C.; Evans, J.; Seller, A.; Czermin, B.; Hanna, M.G.; et al. RRM2B mutations are frequent in familial PEO with multiple mtDNA deletions. Neurology 2011, 76, 2032–2034. [Google Scholar] [CrossRef]
- Takata, A.; Kato, M.; Nakamura, M.; Yoshikawa, T.; Kanba, S.; Sano, A.; Kato, T. Exome sequencing identifies a novel missense variant in RRM2B associated with autosomal recessive progressive external ophthalmoplegia. Genome Biol. 2011, 28, R92. [Google Scholar] [CrossRef]
- Meyer, J.N.; Leuthner, T.C.; Luz, A.L. Mitochondrial fusion, fission, and mitochondrial toxicity. Toxicology 2017, 391, 42–53. [Google Scholar] [CrossRef]
- Lenaers, G.; Hamel, C.; Delettre, C.; Amati-Bonneau, P.; Procaccio, V.; Bonneau, D.; Reynier, P.; Milea, D. Dominant optic atrophy. Orphanet J. Rare Dis. 2012, 7, 46. [Google Scholar] [CrossRef]
- Yu-Wai-Man, P.; Griffiths, P.G.; Gorman, G.S.; Lourenco, C.M.; Wright, A.F.; Auer-Grumbach, M.; Toscano, A.; Musumeci, O.; Valentino, M.L.; Caporali, L.; et al. Multi-system neurological disease is common in patients with OPA1 mutations. Brain 2010, 133, 771–786. [Google Scholar] [CrossRef] [PubMed]
- Wedding, I.M.; Koht, J.; Tran, G.T.; Misceo, D.; Selmer, K.K.; Holmgren, A.; Frengen, E.; Bindoff, L.; Tallaksen, C.M.; Tzoulis, C. Spastic paraplegia type 7 is associated with multiple mitochondrial DNA deletions. PLoS ONE 2014, 9, 86340. [Google Scholar]
- Barca, E.; Long, Y.; Cooley, V.; Schoenaker, R.; Emmanuele, V.; DiMauro, S.; Cohen, B.H.; Karaa, A.; Vladutiu, G.D.; Haas, R.; et al. Mitochondrial diseases in North America: An analysis of the NAMDC Registry. Neurol. Genet. 2020, 6, e402. [Google Scholar] [CrossRef] [PubMed]
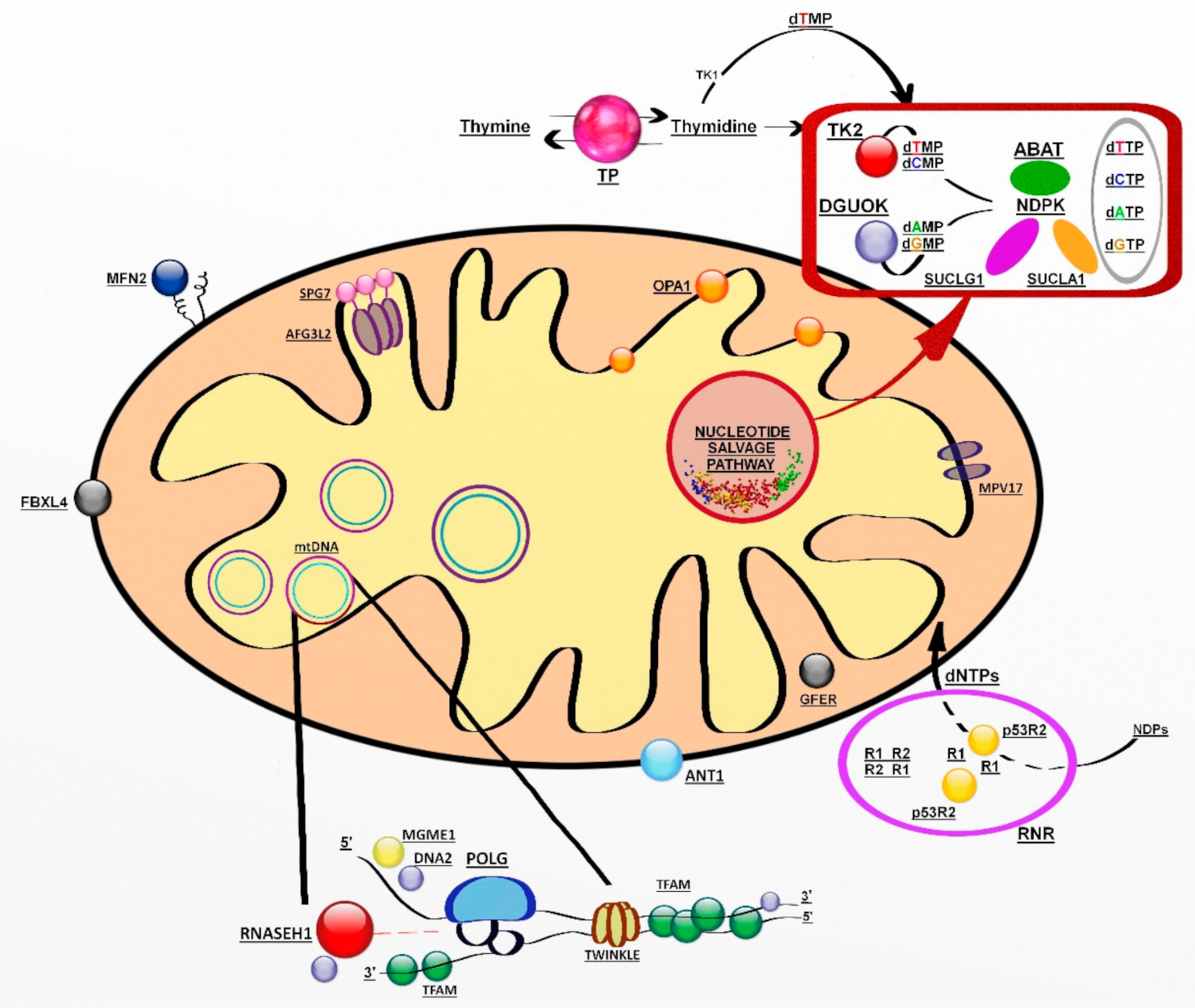
| Mitochondrial Dysfunction | Involved Gene | Muscle Phenotype |
|---|---|---|
| Defects of MRC Complexes | ||
| Complex I | Mitochondrial-encoded subunits: Mt-ND1, Mt-ND2, Mt-ND3, Mt-ND4, Mt-ND4L, Mt-ND5, Mt-ND6 Nuclear-encoded subunits: NDUFA1, NDUFA2, NDUFA9, NDUFA10, NDUFA11, NDUFA12, NDUFA13, NDUFB3, NDUFB9, NDUFB10, NDUFB11, NDUFS1, NDUFS2, NDUFS3, NDUFS4, NDUFS6, NDUFS7, NDUFS8, NDUFV1, NDUFV2 Assembly factors: ACAD9, FOXRED1, NDUFAF1, NDUFAF2, NDUFAF3, NDUFAF4, NDUFAF5, NDUFAF6, NUBPL, TIMMDC1, TMEM126B | Isolated myopathy, multisystem involvement, MELAS |
| Complex III | Mitochondrial-encoded subunits: Mt-CYB Nuclear-encoded subunits: CYC1, UBCRB, UQCRC2 Assembly factors: BCSIL, LYRM7, TTC19, UQCC2 | Isolated myopathy, multisystem involvement |
| Complex IV | Mitochondrial-encoded subunits: Mt-CO1, Mt-CO2, Mt-CO3 Nuclear-encoded subunits: COX41, COX412, NDUFA4 Assembly factors: COA3, COA5, COA6, COA7, COX10, COX14, SCO1, SCO2, COX15, COX20, PET100, APOFT1, SURF1, PET 11 | Isolated myopathy, multisystem involvement |
| Synthesis of electron carriers | ||
| COQ2, COQ4, COQ5, COQ6, COQ7, COQ8A, COQ8B, COQ9, PDSS1, PDSS2 CYCS, HCCS | Isolated myopathy, nephropathy, cardiomiopathy | |
| mtDNA replication and maintenance | ||
| mtDNA homeostasis | DNA2, MGME1, POLG, POLG2, RNASEH1, TWNK | cPEO, cPEO plus |
| Maintenance of mitochondrial nucleotide pools | ABAT, DGUOK, MPV17, RRM2B, SAMHD1, SUCLA2, SUCLG1, TK2, TYMP | cPEO, cPEO plus, isolated myopathy, MNGIE, MDDS |
| Disorders of mitochondrial dynamics and quality control | ||
| Mitochondrial Membrane Phospholipid Metabolism andProtein Import Machinery TAZ, TIMM8A XL AGK, CHKB, DNAJC19, GFER, PAM16, SERAC1, PLA2G6, TIMM22, TIMM50, TIMMDC1 Mitochondrial Membrane Phospholipid Metabolism and Protein Import Machinery TAZ, TIMM8A XL AGK, CHKB, DNAJC19, GFER, PAM16, SERAC1, PLA2G6, TIMM22, TIMM50, TIMMDC1 DNM1L, MFN2, OPA1, GDAP1, MSTO1 AD/AR MFF, STAT2, TRAK1, MIEF2 DNM1L, MFN2, OPA1, GDAP1, MSTO1, AFG32, SPG7 | cPEO | |
| Title | Main Focus | Reference |
|---|---|---|
| Mitochondrial disease in adults | Clinical aspects and diagnosis | [2] |
| Mitochondrial energy generation disorders: genes, mechanisms, and clues to pathology. | Genetic discovery and functional characterization | [3] |
| Mitochondrial Disease Genetics Update Recent insights into the Molecular Diagnosis and Expanding Phenotype of Primary Mitochondrial Disease. | Update of novel mitochondrial disease genes and pathogenic variants | [4] |
| Mitochondrial disease in adults: what’s old and what’s new? | Disease mechanism and clinical aspects in adults | [5] |
| Mutations causing mitochondrial disease: What is new and what challenges remain? | Advances in mitochondrial genetics | [6] |
| Human diseases associated with defects in assembly of OXPHOS complexes. | Factors involved in assembly human OXPHOS complex | [7] |
| Complex I deficiency: clinical features, biochemistry and molecular genetics. | Advances in the structure, function and assembly of complex I | [8] |
| The genetics and pathology of mitochondrial disease. | Genetic discovery and advances in mitochondrial pathology | [9] |
| Nuclear gene mutations as the cause of mitochondrial complex III deficiency | Discuss the nuclear-encoded proteins in which mutations have been found to be associated to CIII deficiency | [10] |
| Cytochrome c oxidase deficiency | Genetic etiology and clinical manifestations in COX deficiency | [11] |
| Mitochondrial disease in children. | Clinical aspects and diagnosis | [12] |
| Mitochondrial DNA depletion syndromes: review and updates of genetic basis, manifestations, and therapeutic options. | Genetic basis, clinical manifestation, and therapeutic options | [13] |
| Clinical and genetic spectrum of mitochondrial neurogastrointestinal encephalomyopathy | Symptomatology, diagnostic procedures, and hurdles, in vitro and in vivo models, experimental therapies | [14] |
| Mitochondrial dynamics: overview of molecular mechanisms | Overview of the molecular mechanisms that govern mitochondrial fission and fusion in mammals. | [15] |
| Nuclear genes involved in mitochondrial diseases caused by instability of mitochondrial DNA | Overview of nuclear genes involved in mitochondrial diseases | [16] |
| Genes | Type of Article | Type of Mutation | Patients N/Age at the Time of the Examination | Clinical Presentation | References |
|---|---|---|---|---|---|
| MT-ND1 | CR | m.4087A>G (pThr261Ala) in homoplasmy | 1 pt/41 yo | Ptosis, ophthalmoparesis, weakness, rabdomyolisis after suxamethonium injection | [31] |
| MT-ND1 | CR | m.3365T>C (pLeu20Pro) | F/28 yo | Exercise intolerance, fatigue, weakness. Metabolic acidosis, high serum lactate concentration | [32] |
| MT-ND1 | CR | m.4175G>A (pTrp290 *) | NA/22 yo | Exercise intolerance, exertion-related muscle weakness and pain. Ptosis. High serum lactate concentration | [32] |
| MT-ND1 | CR | m.3902_3908invACCTTGC | M/43 yo | Exercise intolerance and myalgia. Weakness. | [36] |
| MT-ND2 | CR | m.4831G>A (pGly121Asp) | M/21 yo | Fatigability, muscle weakness. High serum lactate concentration. | [33] |
| MT-ND2 | CR | m.5133_5134del | M/28 yo | Exercise intolerance. High serum lactate concentration | [20] |
| MT-ND4 | CR | m.11832G>A (pTrp358 *) | M/38 yo | Exercise intolerance, fatigue, myalgia. | [34] |
| MT-ND5 | CR | m.13271T>C (p. Leu312Pro) | F/27 yo | Exercise intolerance. High serum lactate concentration. | [35] |
| MT-ND6 | CR | m.14512_14513del p.(Met54Serfs *7) | M/27 yo | Exercise intolerance, muscle weakness, ptosis, intermittent diplopia. | [38] |
| TMEM126B | CR | c.635G>T (p.Gly212Val) and c.401delA (pAsn134Ilefs *2) | 2F and 4M/ 21–36 yo | Exercise intolerance, muscle weakness. High serum lactate concentration | [39] |
| TMEM126B | CR | c.635G>T (p.Gly212Val) and c.397G>A (p.Asp133Asn) (2/3); c.635G>T (pGly212Val) and c.208C>T (p.Gln70 *) | 1F and 2M/ 22–38 yo | Proximal muscle weakness. Exercise induced myalgia. High serum lactate concentration | [40] |
| ACAD9 | CS | 42 missense mutations, | 41F and 29M/22 days—44 yo | Exercise intolerance, muscle weakness, cardiomyopathy. Metabolic acidosis. High serum lactate concentration | [41] |
| 1 frame shift, 1 nonsense, | |||||
| 7 splice sites and 1 initiation codon | |||||
| MT-CYB | CR | m.15084G>A (pTrp113 *); | 2F and 3M/ 32—52 yo | Exercise intolerance, muscle weakness. High serum lactate concentration. Exercise-induced myoglobinuria. | [34] |
| m.15168G>A (pTrp141 *); | |||||
| m.15723G>A (pTrp326 *); | |||||
| m.14846G>A (pGly34Ser); | |||||
| m.15498_15521del | |||||
| MT-CYB | CR | m.15615G>A (pGly290Asp) | M/29 yo | Exercise intolerance | [42] |
| MT-CYB | CR | m.15242G>A (pGly166 *) | F/34 yo | Exercise intolerance. High serum lactate concentration; encephalopathy with seizures and hallucinations. | [43] |
| MT-CYB | CR | m.1517G>A (pGly142 *) | F/40 yo | Exercise intolerance, muscle weakness, fatigue and myalgia | [44] |
| MT-CYB | CR | m.15800C>T (pGln352 *) | F/24 yo | Exercise intolerance, fatigue. | [45] |
| MT-CO1 | CR | m.6708G>A (predicting the loss of the last 245 amino acids of 514 in COX II) | F/30 yo | Exercise intolerance. Muscle weakness and fatigue, myalgia. Exercise-induced myoglobinuria. | [46] |
| MT-CO2 | CR | m7671T>A p(Met29Lys) | M/14 yo | Muscle weakness and fatigue. High serum lactate concentration. | [47] |
| MT-CO2 | CR | m8088delT (pLeu168 *) | F/16 yo | Exercise intolerance, muscle weakness. High serum lactate concentration. Anemia | [48] |
| MT-CO3 | CR | m9379G>A (pTrp58 *) | M/20 yo | Exercise intolerance. Muscle weakness, hypotonia and scapular winging; The symptoms spontaneously regressed through the years. | [49] |
| COX6A2 | CR | c.117C>A (p.Ser39Arg) and c.127T>C (p.Cys43Arg) | M/9 yo | Muscle weakness, hypotonia, facial weakness, high arched palate since infancy | [50] |
| COX10 | CR | c.1007A>T (p.Asp336Val) and c.1015C>T (p.Arg339Trp) | F/37 yo | Failure to thrive. Exercise intolerance, muscle weakness and fatigue, renal Fanconi syndrome. Metabolic acidosis. | [51] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arena, I.G.; Pugliese, A.; Volta, S.; Toscano, A.; Musumeci, O. Molecular Genetics Overview of Primary Mitochondrial Myopathies. J. Clin. Med. 2022, 11, 632. https://doi.org/10.3390/jcm11030632
Arena IG, Pugliese A, Volta S, Toscano A, Musumeci O. Molecular Genetics Overview of Primary Mitochondrial Myopathies. Journal of Clinical Medicine. 2022; 11(3):632. https://doi.org/10.3390/jcm11030632
Chicago/Turabian StyleArena, Ignazio Giuseppe, Alessia Pugliese, Sara Volta, Antonio Toscano, and Olimpia Musumeci. 2022. "Molecular Genetics Overview of Primary Mitochondrial Myopathies" Journal of Clinical Medicine 11, no. 3: 632. https://doi.org/10.3390/jcm11030632
APA StyleArena, I. G., Pugliese, A., Volta, S., Toscano, A., & Musumeci, O. (2022). Molecular Genetics Overview of Primary Mitochondrial Myopathies. Journal of Clinical Medicine, 11(3), 632. https://doi.org/10.3390/jcm11030632






