Unraveling Bicuspid Aortic Valve Enigmas by Multimodality Imaging: Clinical Implications
Abstract
:1. Introduction
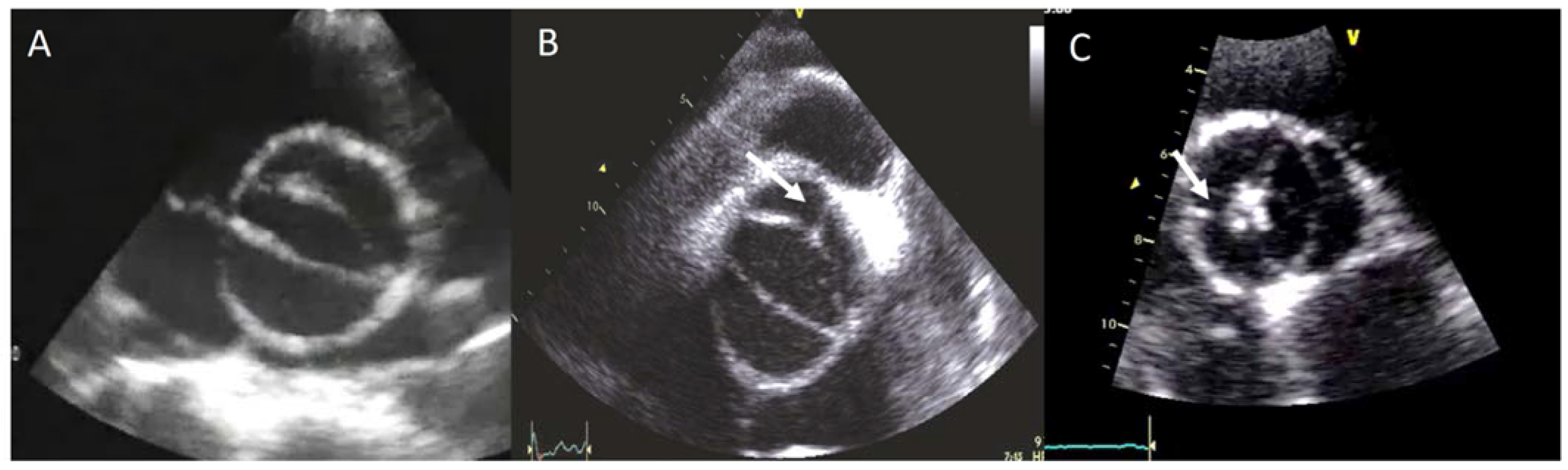
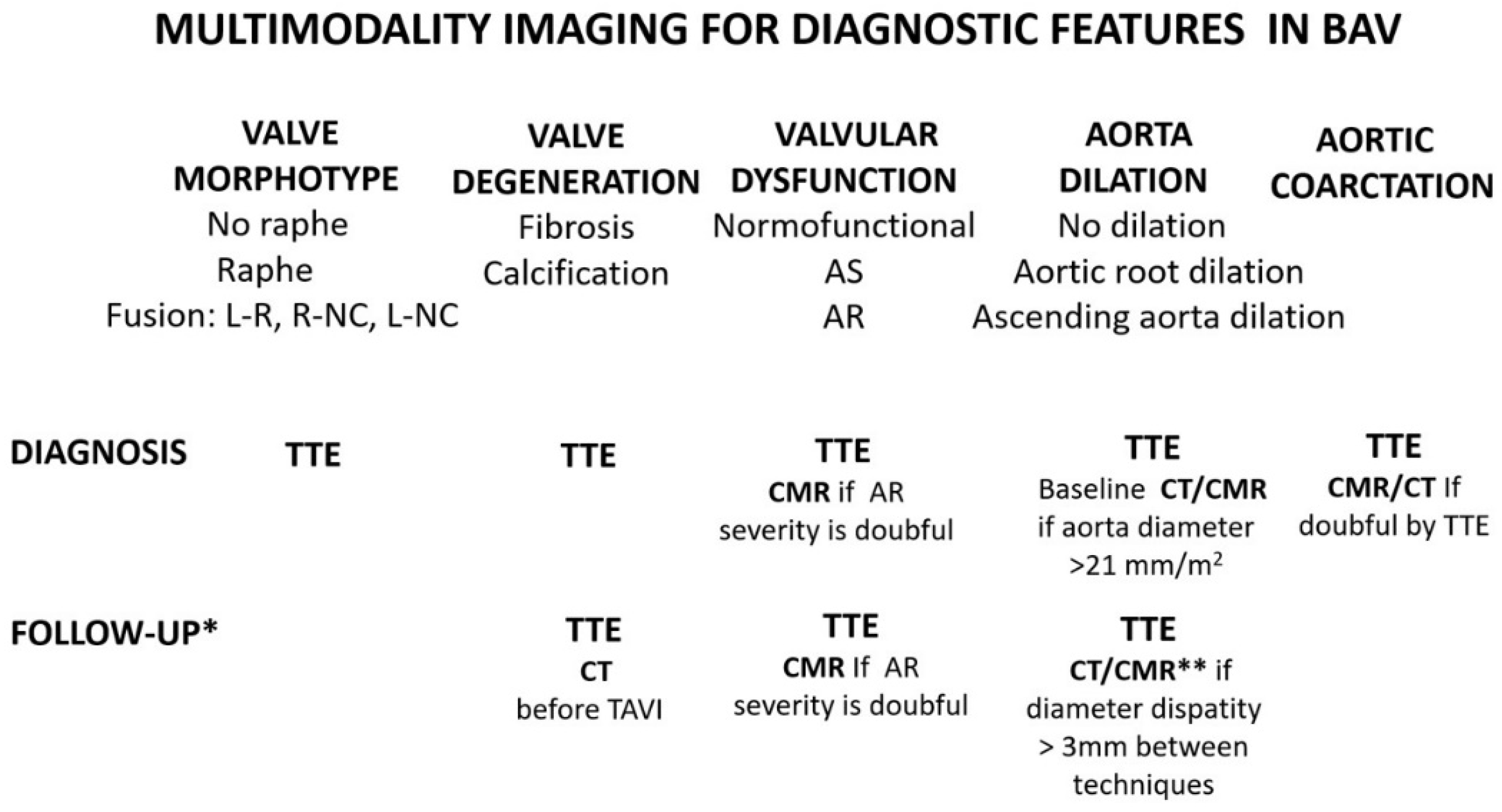
2. Diagnosis and BAV Phenotype
3. Familial Screening
4. Valvular Dysfunction
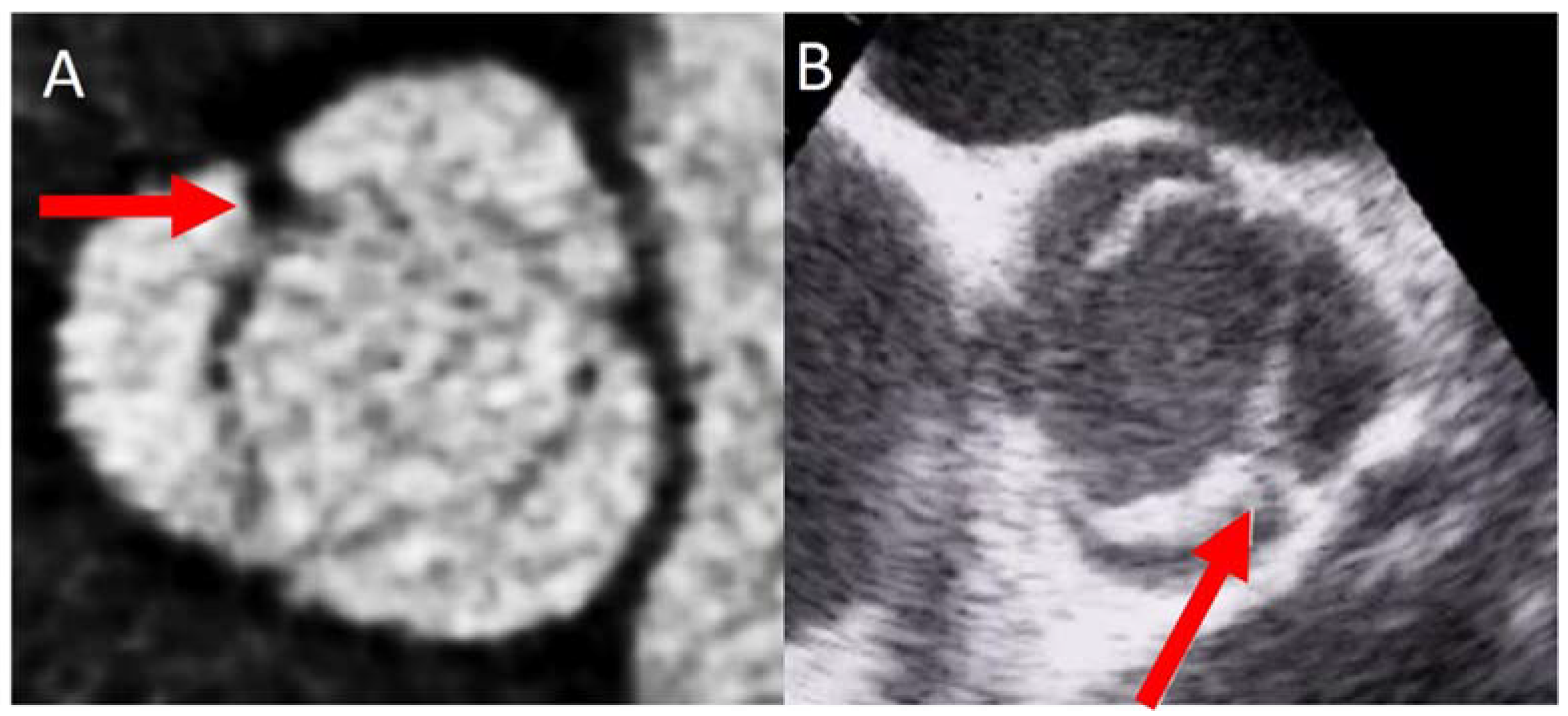
4.1. Valve Calcification
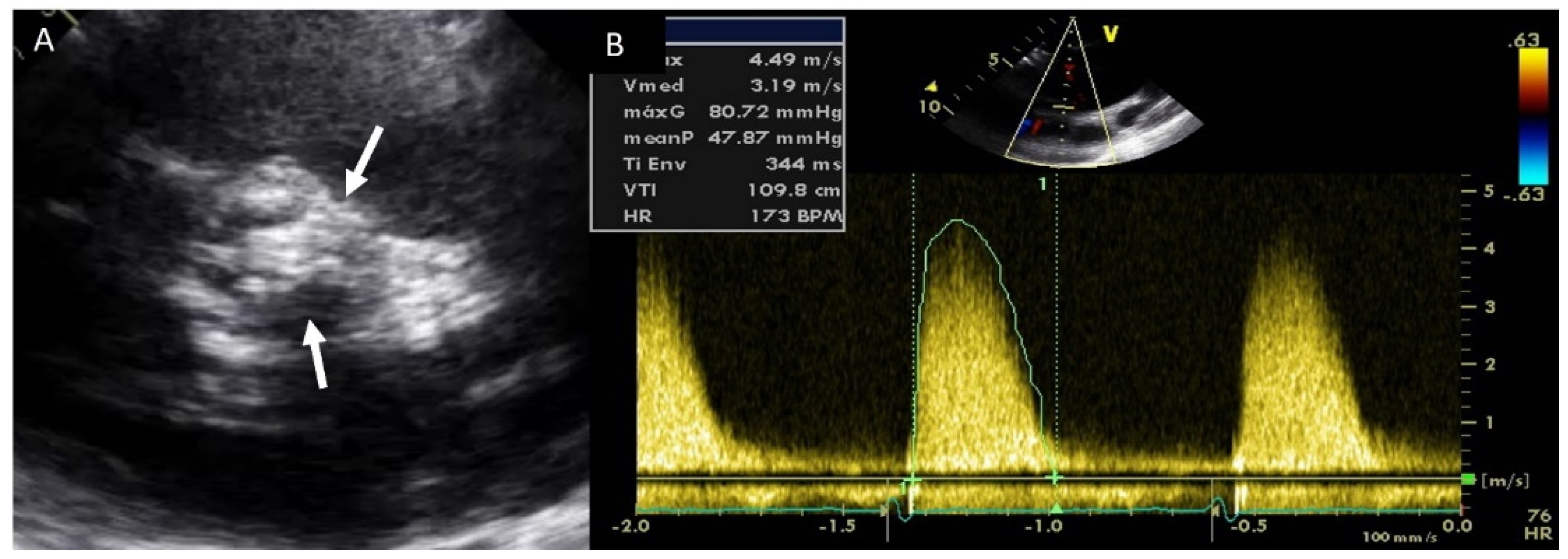
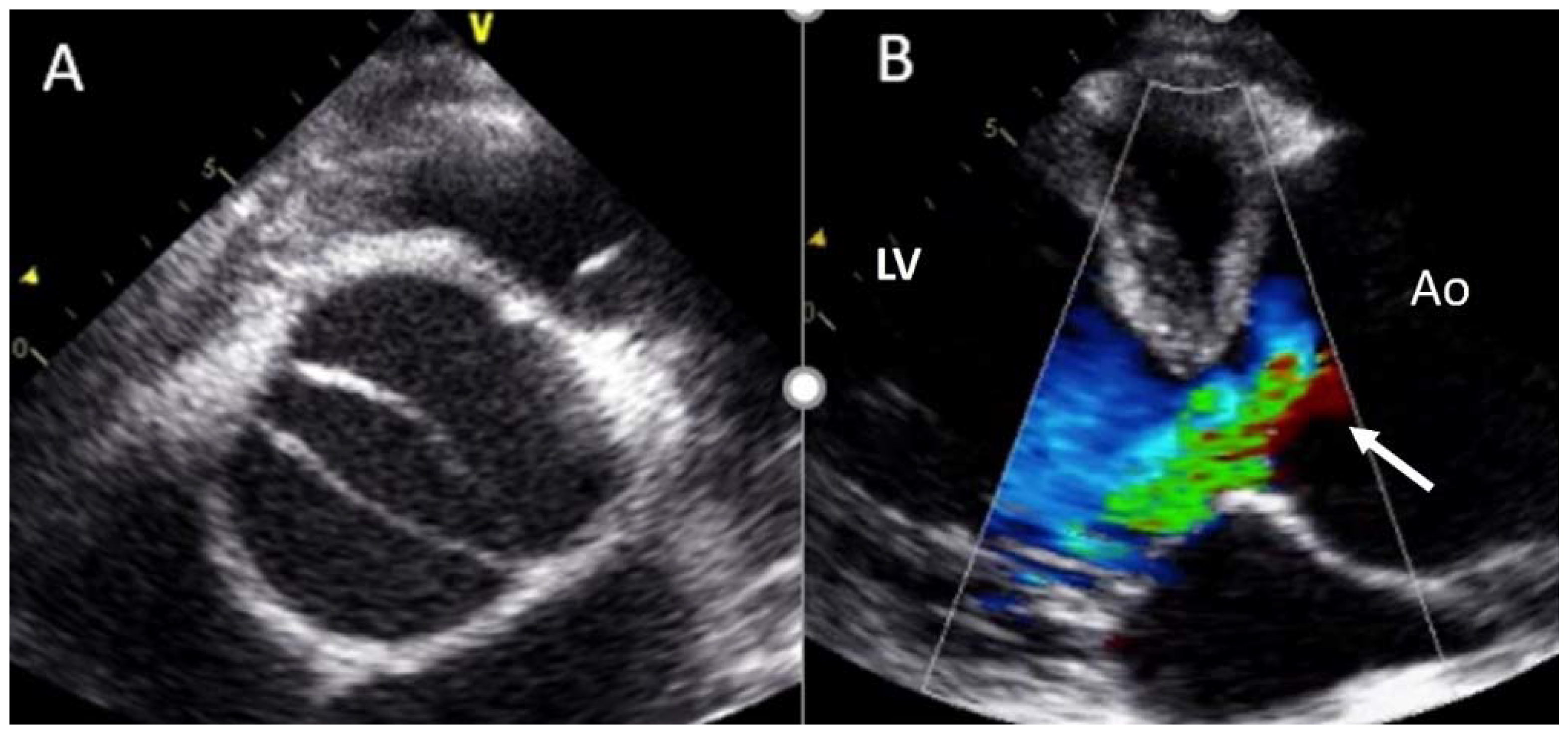
4.2. Aortic Stenosis
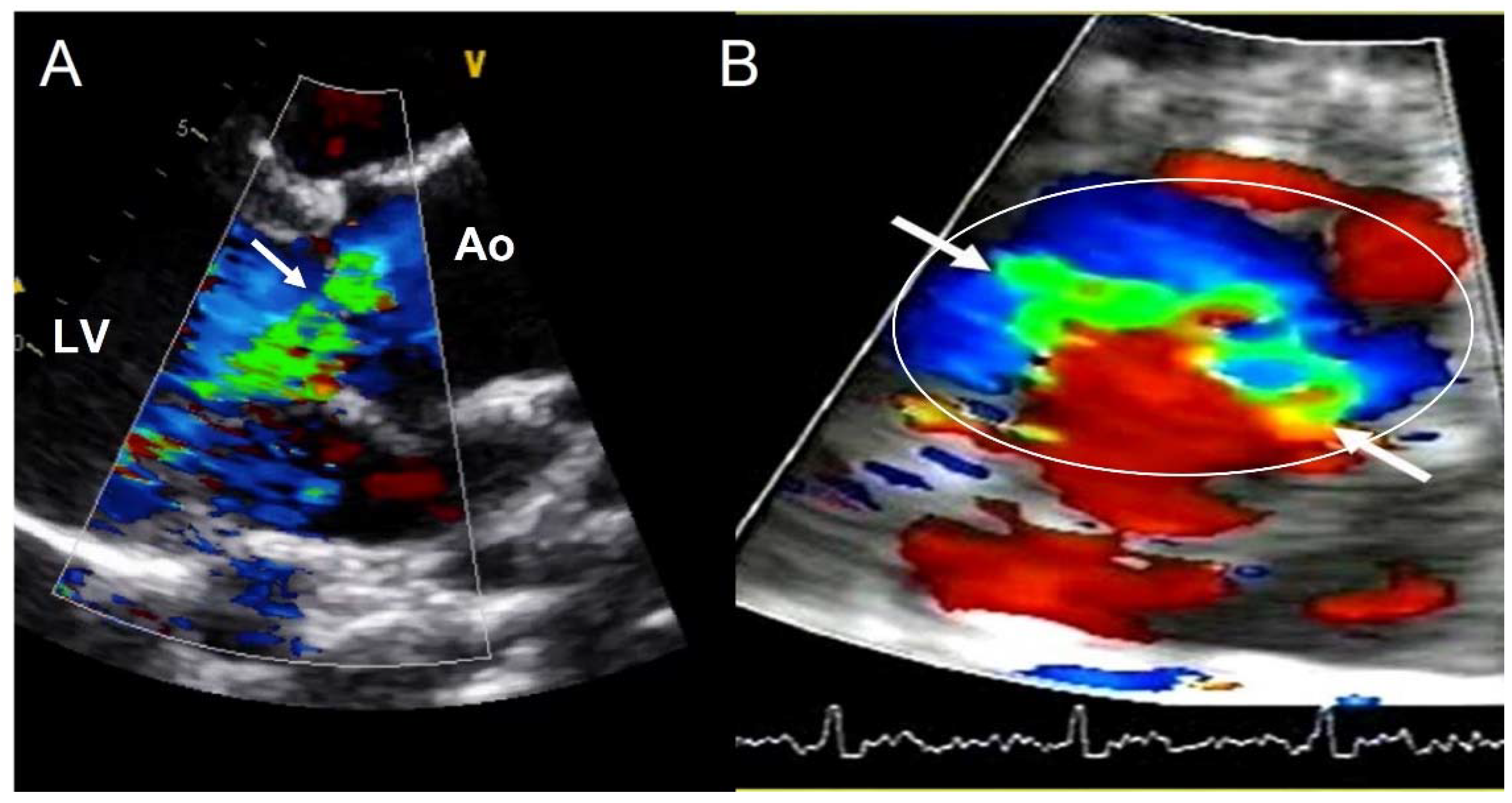
4.3. Aortic Regurgitation
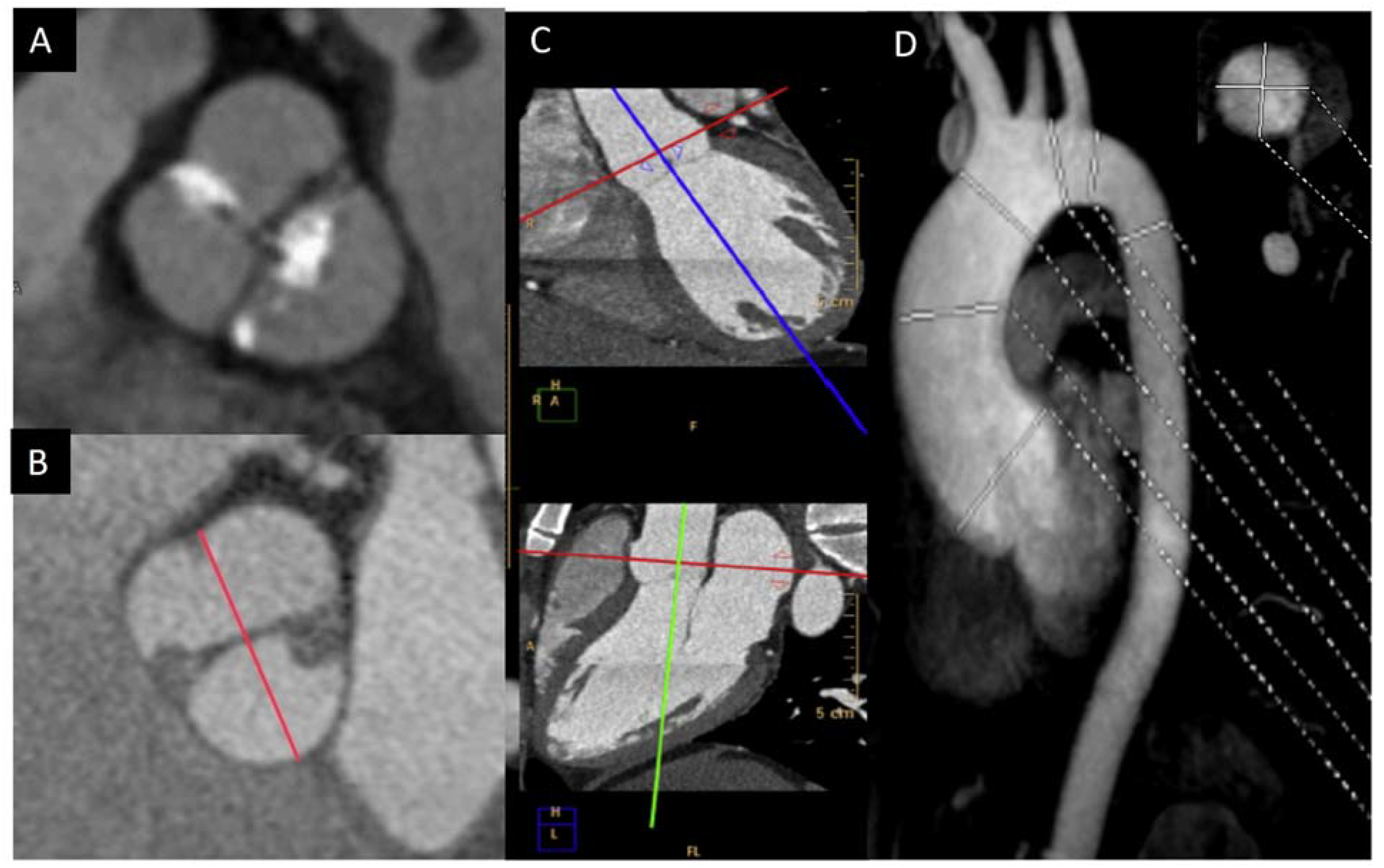
5. Aorta Involvement
6. Imaging Predictors
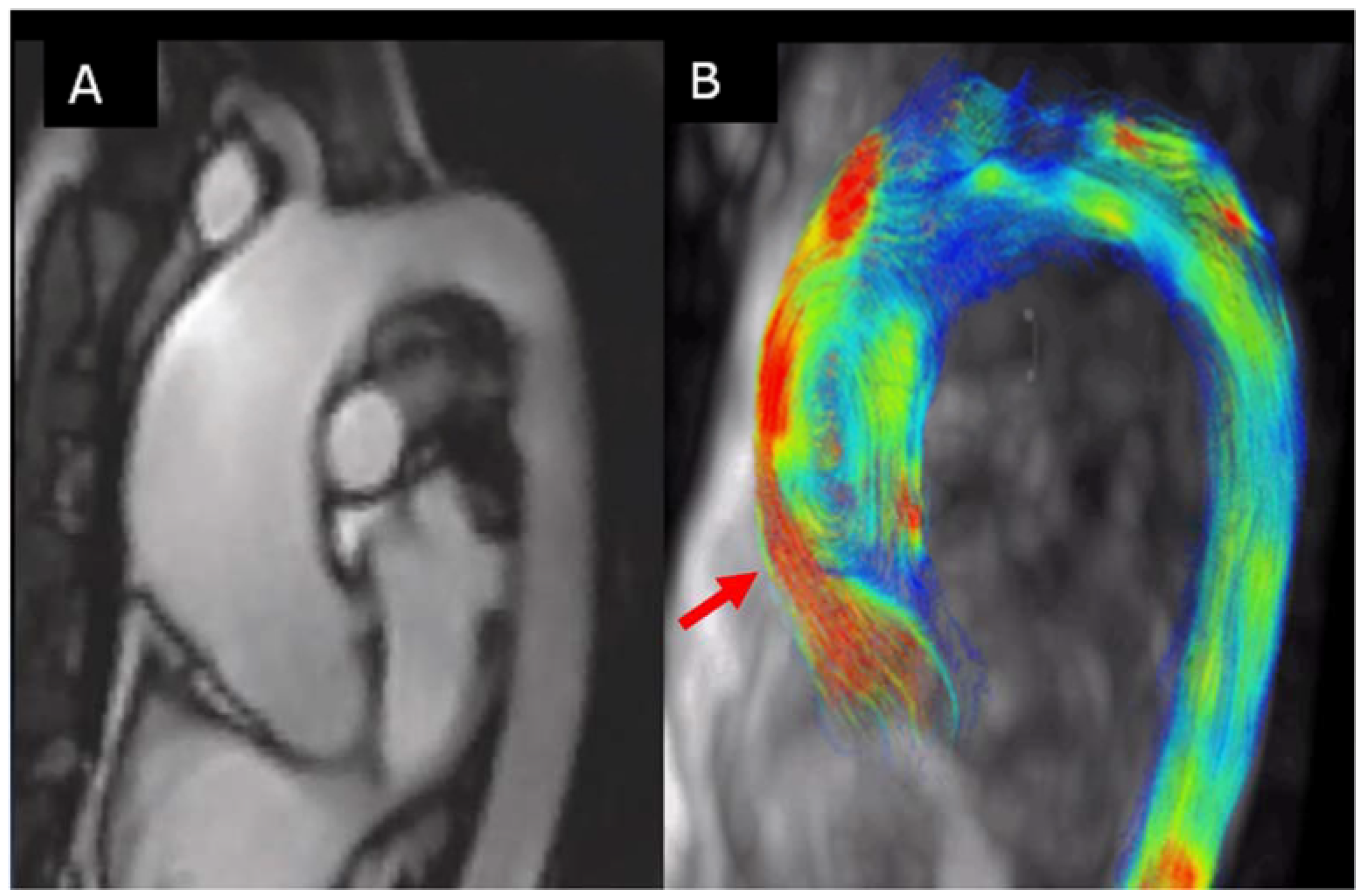
6.1. Aorta Biomechanics Predictor Parameters
6.2. Aortic Flow Hemodynamic Parameters
7. Imaging Surveillance
8. Complications: Surgical and TAVI Indications
8.1. Infective Endocarditis
8.2. Aortic Dissection
8.3. Aortic Stenosis: Surgical and TAVR Indications
8.4. Aortic Regurgitation: Valve Repair and Replacement
8.5. Ascending Aorta Surgery
9. Critical Knowledge Gaps
10. Highlights
- -
- Multimodality imaging is the cornerstone of the diagnosis, follow-up, and surgical treatment indication in BAV patients.
- -
- Although TTE is the technique of choice to identify valve morphotype, valve dysfunction, and ascending aorta dilation, CT and CMR improve the accuracy of aorta size measurement.
- -
- A major benefit of CT is its superior spatial resolution, by which the aortic valve can be evaluated for morphology and calcification, ascending aorta size accuracy, and reproducibility measured and coronary arteries assessed.
- -
- CMR offers the unique opportunity to transition from anatomic to dynamic imaging of the ascending aorta by assessing its functional properties and blood flow patterns. 4D-flow CMR may provide new parameters to predict the rate of progressive aorta enlargement.
- -
- Therefore, a multimodality approach may offer a comprehensive assessment of morphology, risk stratification, and therapy guidance of BAV disease.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Michelena, H.I.; Prakash, S.K.; Corte, A.D.; Bissell, M.M.; Anavekar, N.; Mathieu, P.; Bosse, Y.; Limongelli, G.; Bossone, E.; Benson, D.W.; et al. Bicuspid aortic valve identifying knowledge gaps and rising to the challenge from the international bicuspid aortic valve consortium (BAVCON). Circulation 2014, 129, 2691–2704. [Google Scholar] [CrossRef]
- Michelena, H.I.; Della Corte, A.; Prakash, S.K.; Milewicz, D.M.; Evangelista, A.; Enriquez-Sarano, M. Bicuspid aortic valve aortopathy in adults: Incidence, etiology, and clinical significance. Int. J. Cardiol. 2015, 201, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Michelena, H.I.; Khanna, A.D.; Mahoney, D.; Margaryan, E.; Topilsky, Y.; Suri, R.M.; Eidem, B.; Edwards, W.D.; Sundt, T.M., 3rd; Enriquez-Sarano, M. Incidence of aortic complications in patients with bicuspid aortic valves. JAMA 2011, 306, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Michelena, H.I.; Katan, O.; Suri, R.M.; Baddour, L.M.; Enriquez-Sarano, M. Incidence of Infective Endocarditis in Patients With Bicuspid Aortic Valves in the Community. Mayo Clin. Proc. 2016, 91, 122–123. [Google Scholar] [CrossRef] [PubMed]
- Girdauskas, E.; Borger, M.A.; Secknus, M.-A.; Girdauskas, G.; Kuntze, T. Is aortopathy in bicuspid aortic valve disease a congenital defect or a result of abnormal hemodynamics? A critical reappraisal of a one-sided argument. Eur. J. Cardio-Thoracic Surg. Off. J. Eur. Assoc. Cardio-Thoracic Surg. 2011, 39, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Barker, A.J.; Markl, M.; Bürk, J.; Lorenz, R.; Bock, J.; Bauer, S.; Schulz-Menger, J.; von Knobelsdorff-Brenkenhoff, F. Bicuspid aortic valve is associated with altered wall shear stress in the ascending aorta. Circ. Cardiovasc. Imaging 2012, 5, 457–466. [Google Scholar] [CrossRef]
- Hope, M.D.; Hope, T.A.; Meadows, A.K.; Ordovas, K.G.; Urbania, T.H.; Alley, M.T.; Higgins, C.B. Bicuspid aortic valve: Four-dimensional MR evaluation of ascending aortic systolic flow patterns. Radiology 2010, 255, 53–61. [Google Scholar] [CrossRef]
- Tadros, T.M.; Klein, M.D.; Shapira, O.M. Ascending aortic dilatation associated with bicuspid aortic valve: Pathophysiology, molecular biology, and clinical implications. Circulation 2009, 119, 880–890. [Google Scholar] [CrossRef]
- Schaefer, B.M.; Lewin, M.B.; Stout, K.K.; Gill, E.; Prueitt, A.; Byers, P.H.; Otto, C.M. The bicuspid aortic valve: An integrated phenotypic classification of leaflet morphology and aortic root shape. Heart 2008, 94, 1634–1638. [Google Scholar] [CrossRef]
- Murphy, I.G.; Collins, J.; Powell, A.; Markl, M.; McCarthy, P.; Malaisrie, S.C.; Carr, J.C.; Barker, A.J. Comprehensive 4-stage categorization of bicuspid aortic valve leaflet morphology by cardiac MRI in 386 patients. Int. J. Cardiovasc. Imaging 2017, 33, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, R.O.J.; Tajik, A.J.; Edwards, W.D.; Reeder, G.S.; Shub, C.; Seward, J.B. Accuracy of 2-dimensional echocardiographic diagnosis of congenitally bicuspid aortic valve: Echocardiographic-anatomic correlation in 115 patients. Am. J. Cardiol. 1983, 51, 1469–1473. [Google Scholar] [CrossRef]
- Sun, B.J.; Lee, S.; Jang, J.Y.; Kwon, O.; Bae, J.S.; Lee, J.H.; Kim, D.-H.; Jung, S.-H.; Song, J.-M.; Kang, D.-H.; et al. Performance of a Simplified Dichotomous Phenotypic Classification of Bicuspid Aortic Valve to Predict Type of Valvulopathy and Combined Aortopathy. J. Am. Soc. Echocardiogr. Off. Publ. Am. Soc. Echocardiogr. 2017, 30, 1152–1161. [Google Scholar] [CrossRef]
- Jilaihawi, H.; Chen, M.; Webb, J.; Himbert, D.; Ruiz, C.E.; Rodés-Cabau, J.; Pache, G.; Colombo, A.; Nickenig, G.; Lee, M.; et al. A Bicuspid Aortic Valve Imaging Classification for the TAVR Era. JACC Cardiovasc. Imaging 2016, 9, 1145–1158. [Google Scholar] [CrossRef] [PubMed]
- Sievers, H.-H.; Schmidtke, C. A classification system for the bicuspid aortic valve from 304 surgical specimens. J. Thorac. Cardiovasc. Surg. 2007, 133, 1226–1233. [Google Scholar] [CrossRef]
- Sabet, H.Y.; Edwards, W.D.; Tazelaar, H.D.; Daly, R.C. Congenitally bicuspid aortic valves: A surgical pathology study of 542 cases (1991 through 1996) and a literature review of 2,715 additional cases. Mayo Clin. Proc. 1999, 74, 14–26. [Google Scholar] [CrossRef]
- Angelini, A.; Ho, S.Y.; Anderson, R.H.; Devine, W.A.; Zuberbuhler, J.R.; Becker, A.E.; Davies, M.J. The morphology of the normal aortic valve as compared with the aortic valve having two leaflets. J. Thorac. Cardiovasc. Surg. 1989, 98, 362–367. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Song, J.-K.; Song, J.-M.; Kang, D.-H.; Yun, S.-C.; Kang, D.-W.; Kwon, S.U.; Kim, J.S. Association between anatomic features of atrial septal abnormalities obtained by omni-plane transesophageal echocardiography and stroke recurrence in cryptogenic stroke patients with patent foramen ovale. Am. J. Cardiol. 2010, 106, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Michelena, H.I.; Della Corte, A.; Evangelista, A.; Maleszewski, J.J.; Edwards, W.D.; Roman, M.J.; Devereux, R.B.; Fernández, B.; Asch, F.M.; Barker, A.J.; et al. Summary: International Consensus Statement on Nomenclature and Classification of the Congenital Bicuspid Aortic Valve and Its Aortopathy, for Clinical, Surgical, Interventional and Research Purposes. Ann. Thorac. Surg. 2021, 112, 1005–1022. [Google Scholar] [CrossRef]
- Michelena, H.I.; Della Corte, A.; Evangelista, A.; Maleszewski, J.J.; Enriquez-Sarano, M.; Bax, J.J.; Otto, C.M.; Schäfers, H.-J. Speaking a common language: Introduction to a standard terminology for the bicuspid aortic valve and its aortopathy. Prog. Cardiovasc. Dis. 2020, 63, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Michelena, H.I.; Chandrasekaran, K.; Topilsky, Y.; Messika-Zeitoun, D.; Della Corte, A.; Evangelista, A.; Schäfers, H.-J.; Enriquez-Sarano, M. The Bicuspid Aortic Valve Condition: The Critical Role of Echocardiography and the Case for a Standard Nomenclature Consensus. Prog. Cardiovasc. Dis. 2018, 61, 404–415. [Google Scholar] [CrossRef] [PubMed]
- Sperling, J.S.; Lubat, E. Forme fruste or “Incomplete” bicuspid aortic valves with very small raphes: The prevalence of bicuspid valve and its significance may be underestimated. Int. J. Cardiol. 2015, 184, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Guala, A.; Rodriguez-Palomares, J.; Galian-Gay, L.; Teixido-Tura, G.; Johnson, K.M.; Wieben, O.; Sao Aviles, A.; Evangelista, A. Partial Aortic Valve Leaflet Fusion Is Related to Deleterious Alteration of Proximal Aorta Hemodynamics. Circulation 2019, 139, 2707–2709. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, T.G.; Mwangi, I.; Horgan, S.J.; Cradock, A.; Fitzpatrick, P.; Murray, J.G. Steady-state free-precession (SSFP) cine MRI in distinguishing normal and bicuspid aortic valves. J. Magn. Reson. Imaging 2008, 28, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Clavel, M.-A.; Pibarot, P.; Messika-Zeitoun, D.; Capoulade, R.; Malouf, J.; Aggarval, S.; Araoz, P.A.; Michelena, H.I.; Cueff, C.; Larose, E.; et al. Impact of aortic valve calcification, as measured by MDCT, on survival in patients with aortic stenosis: Results of an international registry study. J. Am. Coll. Cardiol. 2014, 64, 1202–1213. [Google Scholar] [CrossRef]
- Dux-Santoy, L.; Rodríguez-Palomares, J.F.; Teixidó-Turà, G.; Ruiz-Muñoz, A.; Casas, G.; Valente, F.; Servato, M.L.; Galian-Gay, L.; Gutiérrez, L.; González-Alujas, T.; et al. Registration-based semi-automatic assessment of aortic diameter growth rate from contrast-enhanced computed tomography outperforms manual quantification. Eur. Radiol. 2021. [Google Scholar] [CrossRef]
- Goldstein, S.A.; Evangelista, A.; Abbara, S.; Arai, A.; Asch, F.M.; Badano, L.P.; Bolen, M.A.; Connolly, H.M.; Cuellar-Calabria, H.; Czerny, M.; et al. Multimodality imaging of diseases of the thoracic aorta in adults: From the American Society of Echocardiography and the European Association of Cardiovascular Imaging: Endorsed by the Society of Cardiovascular Computed Tomography and Society for Cardiova. J. Am. Soc. Echocardiogr. 2015, 28, 119–182. [Google Scholar] [CrossRef]
- Baumgartner, H.; Falk, V.; Bax, J.J.; De Bonis, M.; Hamm, C.; Holm, P.J.; Iung, B.; Lancellotti, P.; Lansac, E.; Rodriguez Muñoz, D.; et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2017, 38, 2739–2791. [Google Scholar] [CrossRef]
- Huntington, K.; Hunter, A.G.; Chan, K.L. A prospective study to assess the frequency of familial clustering of congenital bicuspid aortic valve. J. Am. Coll. Cardiol. 1997, 30, 1809–1812. [Google Scholar] [CrossRef]
- Robledo-Carmona, J.; Rodríguez-Bailón, I.; Carrasco-Chinchilla, F.; Fernández, B.; Jiménez-Navarro, M.; Porras-Martin, C.; Montiel-Trujillo, A.; García-Pinilla, J.M.; Such-Martínez, M.; De Teresa-Galván, E. Hereditary patterns of bicuspid aortic valve in a hundred families. Int. J. Cardiol. 2013, 168, 3443–3449. [Google Scholar] [CrossRef]
- Biner, S.; Rafique, A.M.; Ray, I.; Cuk, O.; Siegel, R.J.; Tolstrup, K. Aortopathy is prevalent in relatives of bicuspid aortic valve patients. J. Am. Coll. Cardiol. 2009, 53, 2288–2295. [Google Scholar] [CrossRef]
- Galian-Gay, L.; Carro Hevia, A.; Teixido-Turà, G.; Rodríguez Palomares, J.; Gutiérrez-Moreno, L.; Maldonado, G.; Gonzàlez-Alujas, M.T.; Sao-Aviles, A.; Gallego, P.; Calvo-Iglesias, F.; et al. Familial clustering of bicuspid aortic valve and its relationship with aortic dilation in first-degree relatives. Heart 2019, 105, 603–608. [Google Scholar] [CrossRef]
- Tzemos, N.; Therrien, J.; Yip, J.; Thanassoulis, G.; Tremblay, S.; Jamorski, M.T.; Webb, G.D.; Siu, S.C. Outcomes in adults with bicuspid aortic valves. JAMA—J. Am. Med. Assoc. 2008, 300, 1317–1325. [Google Scholar] [CrossRef]
- Masri, A.; Svensson, L.G.; Griffin, B.P.; Desai, M.Y. Contemporary natural history of bicuspid aortic valve disease: A systematic review. Heart 2017, 103, 1323–1330. [Google Scholar] [CrossRef]
- Kong, W.K.F.; Delgado, V.; Poh, K.K.; Regeer, M.V.; Ng, A.C.T.; McCormack, L.; Yeo, T.C.; Shanks, M.; Parent, S.; Enache, R.; et al. Prognostic Implications of Raphe in Bicuspid Aortic Valve Anatomy. JAMA Cardiol. 2017, 2, 285–292. [Google Scholar] [CrossRef]
- Evangelista, A.; Gallego, P.; Calvo-Iglesias, F.; Bermejo, J.; Robledo-Carmona, J.; Sanchez, V.; Saura, D.; Arnold, R.; Carro, A.; Maldonado, G.; et al. Anatomical and clinical predictors of valve dysfunction and aortic dilation in bicuspid aortic valve disease. Heart 2018, 104, 566–573. [Google Scholar] [CrossRef]
- Mathieu, P.; Bossé, Y.; Huggins, G.S.; Della Corte, A.; Pibarot, P.; Michelena, H.I.; Limongelli, G.; Boulanger, M.-C.; Evangelista, A.; Bédard, E.; et al. The pathology and pathobiology of bicuspid aortic valve: State of the art and novel research perspectives. J. Pathol. Clin. Res. 2015, 1, 195–206. [Google Scholar] [CrossRef]
- Yousry, M.; Rickenlund, A.; Petrini, J.; Jenner, J.; Liska, J.; Eriksson, P.; Franco-Cereceda, A.; Eriksson, M.J.; Caidahl, K. Aortic valve type and calcification as assessed by transthoracic and transoesophageal echocardiography. Clin. Physiol. Funct. Imaging 2015, 35, 306–313. [Google Scholar] [CrossRef]
- Choi, B.H.; Ko, S.M.; Shin, J.K.; Chee, H.K.; Kim, J.S.; Kim, J. Association between aortic valvular calcification and characteristics of the aortic valve in patients with bicuspid aortic valve stenosis. Acta Radiol. 2019, 60, 468–477. [Google Scholar] [CrossRef]
- Shen, M.; Tastet, L.; Capoulade, R.; Arsenault, M.; Bédard, É.; Clavel, M.-A.; Pibarot, P. Effect of bicuspid aortic valve phenotype on progression of aortic stenosis. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 727–734. [Google Scholar] [CrossRef]
- Yang, L.-T.; Boler, A.; Medina-Inojosa, J.R.; Scott, C.G.; Maurer, M.J.; Eleid, M.F.; Enriquez-Sarano, M.; Tribouilloy, C.; Michelena, H.I. Aortic Stenosis Progression, Cardiac Damage, and Survival: Comparison Between Bicuspid and Tricuspid Aortic Valves. JACC Cardiovasc. Imaging 2021, 14, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.K.F.; Regeer, M.V.; Ng, A.C.T.; McCormack, L.; Poh, K.K.; Yeo, T.C.; Shanks, M.; Parent, S.; Enache, R.; Popescu, B.A.; et al. Sex Differences in Phenotypes of Bicuspid Aortic Valve and Aortopathy: Insights from a Large Multicenter, International Registry. Circ. Cardiovasc. Imaging 2017, 10, e005155. [Google Scholar] [CrossRef]
- Michelena, H.I.; Suri, R.M.; Katan, O.; Eleid, M.F.; Clavel, M.-A.; Maurer, M.J.; Pellikka, P.A.; Mahoney, D.; Enriquez-Sarano, M. Sex Differences and Survival in Adults With Bicuspid Aortic Valves: Verification in 3 Contemporary Echocardiographic Cohorts. J. Am. Heart Assoc. 2016, 5, e004211. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-T.; Michelena, H.I.; Maleszewski, J.J.; Schaff, H.V.; Pellikka, P.A. Contemporary Etiologies, Mechanisms, and Surgical Approaches in Pure Native Aortic Regurgitation. Mayo Clin. Proc. 2019, 94, 1158–1170. [Google Scholar] [CrossRef]
- Myerson, S.G.; d’Arcy, J.; Mohiaddin, R.; Greenwood, J.P.; Karamitsos, T.D.; Francis, J.M.; Banning, A.P.; Christiansen, J.P.; Neubauer, S. Aortic regurgitation quantification using cardiovascular magnetic resonance: Association with clinical outcome. Circulation 2012, 126, 1452–1460. [Google Scholar] [CrossRef]
- Kammerlander, A.A.; Wiesinger, M.; Duca, F.; Aschauer, S.; Binder, C.; Zotter Tufaro, C.; Nitsche, C.; Badre-Eslam, R.; Schönbauer, R.; Bartko, P.; et al. Diagnostic and Prognostic Utility of Cardiac Magnetic Resonance Imaging in Aortic Regurgitation. JACC Cardiovasc. Imaging 2019, 12, 1474–1483. [Google Scholar] [CrossRef]
- Spampinato, R.A.; Jahnke, C.; Paetsch, I.; Hilbert, S.; Löbe, S.; Lindemann, F.; Strotdrees, E.; Hindricks, G.; Borger, M.A. Grading of aortic regurgitation by cardiovascular magnetic resonance and pulsed Doppler of the left subclavian artery: Harmonizing grading scales between imaging modalities. Int. J. Cardiovasc. Imaging 2020, 36, 1517–1526. [Google Scholar] [CrossRef]
- Rodriguez-Palomares, J.F.; Teixido-Tura, G.; Galuppo, V.; Cuellar, H.; Laynez, A.; Gutierrez, L.; Gonzalez-Alujas, M.T.; Garcia-Dorado, D.; Evangelista, A. Multimodality Assessment of Ascending Aortic Diameters: Comparison of Different Measurement Methods. J. Am. Soc. Echocardiogr. 2016, 29, 819–826.e4. [Google Scholar] [CrossRef] [PubMed]
- Vis, J.C.; Rodriguez-Palomares, J.F.; Teixido-Tura, G.; Galian-Gay, L.; Granato, C.; Guala, A.; Sao-Aviles, A.; Gutierrez, L.; Gonzalez-Alujas, T.; Garcia-Dorado, D.; et al. Implications of Asymmetry and Valvular Morphotype on Echocardiographic Measurements of the Aortic Root in Bicuspid Aortic Valve. J. Am. Soc. Echocardiogr. 2019, 32, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Girdauskas, E.; Rouman, M.; Disha, K.; Fey, B.; Dubslaff, G.; von Kodolitsch, Y.; Reichenspurner, H.; Borger, M.A.; Kuntze, T. Morphologic and Functional Markers of Aortopathy in Patients With Bicuspid Aortic Valve Insufficiency Versus Stenosis. Ann. Thorac. Surg. 2017, 103, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Beppu, S.; Suzuki, S.; Matsuda, H.; Ohmori, F.; Nagata, S.; Miyatake, K. Rapidity of progression of aortic stenosis in patients with congenital bicuspid aortic valves. Am. J. Cardiol. 1993, 71, 322–327. [Google Scholar] [CrossRef]
- Michelena, H.I.; Desjardins, V.A.; Avierinos, J.-F.; Russo, A.; Nkomo, V.T.; Sundt, T.M.; Pellikka, P.A.; Tajik, A.J.; Enriquez-Sarano, M. Natural history of asymptomatic patients with normally functioning or minimally dysfunctional bicuspid aortic valve in the community. Circulation 2008, 117, 2776–2784. [Google Scholar] [CrossRef]
- Lee, S.Y.; Shim, C.Y.; Kim, D.; Cho, I.; Hong, G.-R.; Ha, J.-W.; Chung, N. Factors Determining Aortic Valve Dysfunction in Korean Subjects With a Bicuspid Aortic Valve. Am. J. Cardiol. 2017, 119, 2049–2055. [Google Scholar] [CrossRef]
- Detaint, D.; Michelena, H.I.; Nkomo, V.T.; Vahanian, A.; Jondeau, G.; Sarano, M.E. Aortic dilatation patterns and rates in adults with bicuspid aortic valves: A comparative study with Marfan syndrome and degenerative aortopathy. Heart 2014, 100, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Della Corte, A.; Bancone, C.; Buonocore, M.; Dialetto, G.; Covino, F.E.; Manduca, S.; Scognamiglio, G.; D’Oria, V.; De Feo, M. Pattern of ascending aortic dimensions predicts the growth rate of the aorta in patients with bicuspid aortic valve. JACC Cardiovasc. Imaging 2013, 6, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Thanassoulis, G.; Yip, J.W.L.; Filion, K.; Jamorski, M.; Webb, G.; Siu, S.C.; Therrien, J. Retrospective study to identify predictors of the presence and rapid progression of aortic dilatation in patients with bicuspid aortic valves. Nat. Clin. Pract. Cardiovasc. Med. 2008, 5, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Guala, A.; Rodriguez-Palomares, J.; Dux-Santoy, L.; Teixido-Tura, G.; Maldonado, G.; Galian, L.; Huguet, M.; Valente, F.; Gutierrez, L.; Gonzalez-Alujas, T.; et al. Influence of Aortic Dilation on the Regional Aortic Stiffness of Bicuspid Aortic Valve Assessed by 4-Dimensional Flow Cardiac Magnetic Resonance: Comparison With Marfan Syndrome and Degenerative Aortic Aneurysm. JACC Cardiovasc. Imaging 2019, 12, 1020–1029. [Google Scholar] [CrossRef]
- Nistri, S.; Grande-Allen, J.; Noale, M.; Basso, C.; Siviero, P.; Maggi, S.; Crepaldi, G.; Thiene, G. Aortic elasticity and size in bicuspid aortic valve syndrome. Eur. Heart J. 2008, 29, 472–479. [Google Scholar] [CrossRef]
- Huang, F.Q.; Le Tan, J. Pattern of aortic dilatation in different bicuspid aortic valve phenotypes and its association with aortic valvular dysfunction and elasticity. Heart. Lung Circ. 2014, 23, 32–38. [Google Scholar] [CrossRef]
- Goudot, G.; Mirault, T.; Rossi, A.; Zarka, S.; Albuisson, J.; Achouh, P.; Pernot, M.; Messas, E. Segmental aortic stiffness in patients with bicuspid aortic valve compared with first-degree relatives. Heart 2019, 105, 130–136. [Google Scholar] [CrossRef]
- Rodriguez-Palomares, J.F.; Dux-Santoy, L.; Guala, A.; Kale, R.; Maldonado, G.; Teixido-Tura, G.; Galian, L.; Huguet, M.; Valente, F.; Gutierrez, L.; et al. Aortic flow patterns and wall shear stress maps by 4D-flow cardiovascular magnetic resonance in the assessment of aortic dilatation in bicuspid aortic valve disease. J. Cardiovasc. Magn. Reson. 2018, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Bissell, M.M.; Hess, A.T.; Biasiolli, L.; Glaze, S.J.; Loudon, M.; Pitcher, A.; Davis, A.; Prendergast, B.; Markl, M.; Barker, A.J.; et al. Aortic dilation in bicuspid aortic valve disease: Flow pattern is a major contributor and differs with valve fusion type. Circ. Cardiovasc. Imaging 2013, 6, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Dux-Santoy, L.; Guala, A.; Teixidó-Turà, G.; Ruiz-Muñoz, A.; Maldonado, G.; Villalva, N.; Galian, L.; Valente, F.; Gutiérrez, L.; González-Alujas, T.; et al. Increased rotational flow in the proximal aortic arch is associated with its dilation in bicuspid aortic valve disease. Eur. Heart J. Cardiovasc. Imaging 2019, 20, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Mahadevia, R.; Barker, A.J.; Schnell, S.; Entezari, P.; Kansal, P.; Fedak, P.W.M.; Malaisrie, S.C.; McCarthy, P.; Collins, J.; Carr, J.; et al. Bicuspid aortic cusp fusion morphology alters aortic three-dimensional outflow patterns, wall shear stress, and expression of aortopathy. Circulation 2014, 129, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Guzzardi, D.G.; Barker, A.J.; van Ooij, P.; Malaisrie, S.C.; Puthumana, J.J.; Belke, D.D.; Mewhort, H.E.M.; Svystonyuk, D.A.; Kang, S.; Verma, S.; et al. Valve-Related Hemodynamics Mediate Human Bicuspid Aortopathy: Insights From Wall Shear Stress Mapping. J. Am. Coll. Cardiol. 2015, 66, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Guala, A.; Dux-Santoy, L.; Teixido-Tura, G.; Johnson, K.M.; Evangelista, A.; Rodríguez-Palomares, J. Raphe in bicuspid aortic valve without significant aortic valve disease is unrelated to aortic hemodynamics and stiffness. Rev. Esp. Cardiol. (Engl. Ed.) 2020, 73, 89–90. [Google Scholar] [CrossRef]
- Guala, A.; Dux-Santoy, L.; Teixido-Tura, G.; Ruiz-Muñoz, A.; Laura Galian-Gay, M.L.; Servato, F.V.; Laura Gutiérrez, T.G.-A.; Johnson, K.M. Wall shear stress predicts aortic dilation in patients with bicuspid aortic valve. JACC Cardiovasc. Imaging 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Borger, M.A.; Fedak, P.W.M.; Stephens, E.H.; Gleason, T.G.; Girdauskas, E.; Ikonomidis, J.S.; Khoynezhad, A.; Siu, S.C.; Verma, S.; Hope, M.D.; et al. The American Association for Thoracic Surgery consensus guidelines on bicuspid aortic valve-related aortopathy: Executive summary. J. Thorac. Cardiovasc. Surg. 2018, 156, 473–480. [Google Scholar] [CrossRef]
- Oliver, J.M.; Alonso-Gonzalez, R.; Gonzalez, A.E.; Gallego, P.; Sanchez-Recalde, A.; Cuesta, E.; Aroca, A.; Lopez-Sendon, J.L. Risk of aortic root or ascending aorta complications in patients with bicuspid aortic valve with and without coarctation of the aorta. Am. J. Cardiol. 2009, 104, 1001–1006. [Google Scholar] [CrossRef]
- Otto, C.M.; Nishimura, R.A.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P.; Gentile, F.; Jneid, H.; Krieger, E.V.; Mack, M.; McLeod, C.; et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2021, 77, e25–e197. [Google Scholar] [CrossRef] [PubMed]
- Michelena, H.I.; Margaryan, E.; Miller, F.A.; Eleid, M.; Maalouf, J.; Suri, R.; Messika-Zeitoun, D.; Pellikka, P.A.; Enriquez-Sarano, M. Inconsistent echocardiographic grading of aortic stenosis: Is the left ventricular outflow tract important? Heart 2013, 99, 921–931. [Google Scholar] [CrossRef]
- Vahanian, A.; Beyersdorf, F.; Praz, F.; Milojevic, M.; Baldus, S.; Bauersachs, J.; Capodanno, D.; Conradi, L.; De Bonis, M.; De Paulis, R.; et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2021. [Google Scholar] [CrossRef] [PubMed]
- Holmes, D.R.J.; Nishimura, R.A.; Grover, F.L.; Brindis, R.G.; Carroll, J.D.; Edwards, F.H.; Peterson, E.D.; Rumsfeld, J.S.; Shahian, D.M.; Thourani, V.H.; et al. Annual Outcomes With Transcatheter Valve Therapy: From the STS/ACC TVT Registry. Ann. Thorac. Surg. 2016, 101, 789–800. [Google Scholar] [CrossRef]
- Yoon, S.-H.; Lefèvre, T.; Ahn, J.-M.; Perlman, G.Y.; Dvir, D.; Latib, A.; Barbanti, M.; Deuschl, F.; De Backer, O.; Blanke, P.; et al. Transcatheter Aortic Valve Replacement With Early- and New-Generation Devices in Bicuspid Aortic Valve Stenosis. J. Am. Coll. Cardiol. 2016, 68, 1195–1205. [Google Scholar] [CrossRef]
- Forrest, J.K.; Ramlawi, B.; Deeb, G.M.; Zahr, F.; Song, H.K.; Kleiman, N.S.; Chetcuti, S.J.; Michelena, H.I.; Mangi, A.A.; Skiles, J.A.; et al. Transcatheter Aortic Valve Replacement in Low-risk Patients With Bicuspid Aortic Valve Stenosis. JAMA Cardiol. 2021, 6, 50–57. [Google Scholar] [CrossRef]
- Ehrlich, T.; de Kerchove, L.; Vojacek, J.; Boodhwani, M.; El-Hamamsy, I.; De Paulis, R.; Lansac, E.; Bavaria, J.E.; El Khoury, G.; Schäfers, H.-J. State-of-the art bicuspid aortic valve repair in 2020. Prog. Cardiovasc. Dis. 2020, 63, 457–464. [Google Scholar] [CrossRef]
- Hagendorff, A.; Evangelista, A.; Fehske, W.; Schäfers, H.-J. Improvement in the Assessment of Aortic Valve and Aortic Aneurysm Repair by 3-Dimensional Echocardiography. JACC Cardiovasc. Imaging 2019, 12, 2225–2244. [Google Scholar] [CrossRef]
- Zeeshan, A.; Idrees, J.J.; Johnston, D.R.; Rajeswaran, J.; Roselli, E.E.; Soltesz, E.G.; Gillinov, A.M.; Griffin, B.; Grimm, R.; Hammer, D.F.; et al. Durability of Aortic Valve Cusp Repair With and Without Annular Support. Ann. Thorac. Surg. 2018, 105, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Lansac, E.; de Kerchove, L. Aortic valve repair techniques: State of the art. Eur. J. Cardio-Thoracic Surg. Off. J. Eur. Assoc. Cardio-Thoracic Surg. 2018, 53, 1101–1107. [Google Scholar] [CrossRef]
- Della Corte, A.; Body, S.C.; Booher, A.M.; Schaefers, H.-J.; Milewski, R.K.; Michelena, H.I.; Evangelista, A.; Pibarot, P.; Mathieu, P.; Limongelli, G.; et al. Surgical treatment of bicuspid aortic valve disease: Knowledge gaps and research perspectives. J. Thorac. Cardiovasc. Surg. 2014, 147, 1749–1757.e1. [Google Scholar] [CrossRef] [PubMed]
- Schneider, U.; Feldner, S.K.; Hofmann, C.; Schöpe, J.; Wagenpfeil, S.; Giebels, C.; Schäfers, H.-J. Two decades of experience with root remodeling and valve repair for bicuspid aortic valves. J. Thorac. Cardiovasc. Surg. 2017, 153, S65–S71. [Google Scholar] [CrossRef] [PubMed]
| TTE | CT | MRI | |
|---|---|---|---|
| VALVE MORPHOTYPE | +++ | ++ | +++ |
| RAPHE | +++ | +++ | ++ |
| VALVE DEGENERATION | ++ | +++ | ++ |
| AORTIC STENOSIS | ++ | ++ | + |
| AORTIC REGURGITATION | ++ | - | +++ |
| AORTIC ROOT DILATION | + | +++ | +++ |
| TUBULAR SEGMENT DILATION | + | ++ | +++ |
| AORTIC COARCTATION | + | +++ | +++ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evangelista Masip, A.; Galian-Gay, L.; Guala, A.; Lopez-Sainz, A.; Teixido-Turà, G.; Ruiz Muñoz, A.; Valente, F.; Gutierrez, L.; Fernandez-Galera, R.; Casas, G.; et al. Unraveling Bicuspid Aortic Valve Enigmas by Multimodality Imaging: Clinical Implications. J. Clin. Med. 2022, 11, 456. https://doi.org/10.3390/jcm11020456
Evangelista Masip A, Galian-Gay L, Guala A, Lopez-Sainz A, Teixido-Turà G, Ruiz Muñoz A, Valente F, Gutierrez L, Fernandez-Galera R, Casas G, et al. Unraveling Bicuspid Aortic Valve Enigmas by Multimodality Imaging: Clinical Implications. Journal of Clinical Medicine. 2022; 11(2):456. https://doi.org/10.3390/jcm11020456
Chicago/Turabian StyleEvangelista Masip, Arturo, Laura Galian-Gay, Andrea Guala, Angela Lopez-Sainz, Gisela Teixido-Turà, Aroa Ruiz Muñoz, Filipa Valente, Laura Gutierrez, Ruben Fernandez-Galera, Guillem Casas, and et al. 2022. "Unraveling Bicuspid Aortic Valve Enigmas by Multimodality Imaging: Clinical Implications" Journal of Clinical Medicine 11, no. 2: 456. https://doi.org/10.3390/jcm11020456
APA StyleEvangelista Masip, A., Galian-Gay, L., Guala, A., Lopez-Sainz, A., Teixido-Turà, G., Ruiz Muñoz, A., Valente, F., Gutierrez, L., Fernandez-Galera, R., Casas, G., Panaro, A., Marigliano, A., Huguet, M., González-Alujas, T., & Rodriguez-Palomares, J. (2022). Unraveling Bicuspid Aortic Valve Enigmas by Multimodality Imaging: Clinical Implications. Journal of Clinical Medicine, 11(2), 456. https://doi.org/10.3390/jcm11020456





