Feasibility of Combining Transcranial Direct Current Stimulation and Active Fully Embodied Virtual Reality for Visual Height Intolerance: A Double-Blind Randomized Controlled Study
Abstract
:1. Introduction
2. Material and Methods
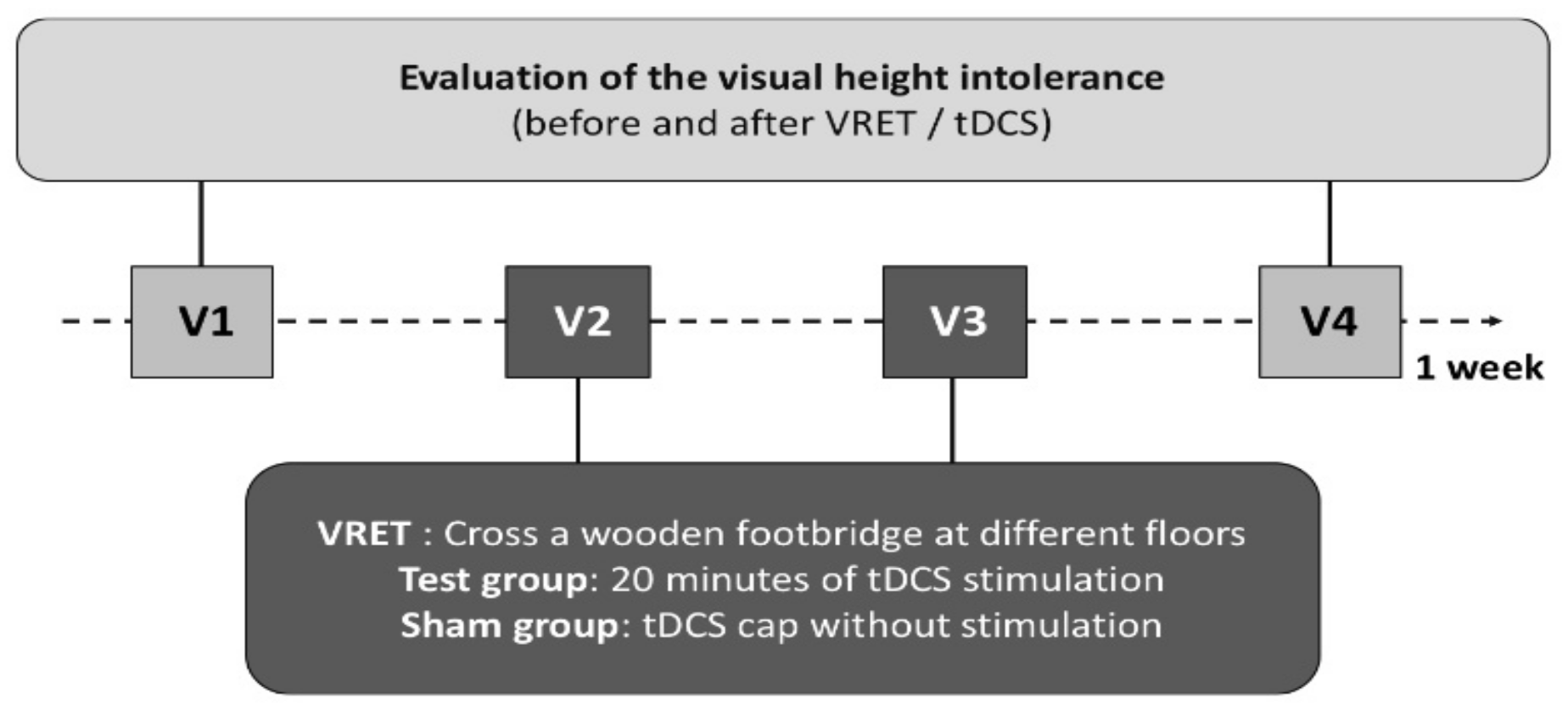
2.1. Design
- Two assessment sessions measuring SUD during a virtual reality task lasting 10 min to measure participants’ fear of height at baseline (V1) and after VRET (V4).
- Two exposure sessions (V2 and V3), during which participants were exposed to height (VRET) for 20 min while being stimulated online (active tDCS) or not (sham tDCS). Two sessions is the minimal number of sessions needed to observe VRET efficacy in the literature. We chose that number since the aim of research regarding potentiating the VRET effect with other interventions is to achieve a stronger and quicker effect [20].
2.2. Participants
2.3. Experimental Setup
2.3.1. VR Device
2.3.2. VR Environment
Assessment Sessions (V1 and V4)
Exposure Sessions (V2 and V3)
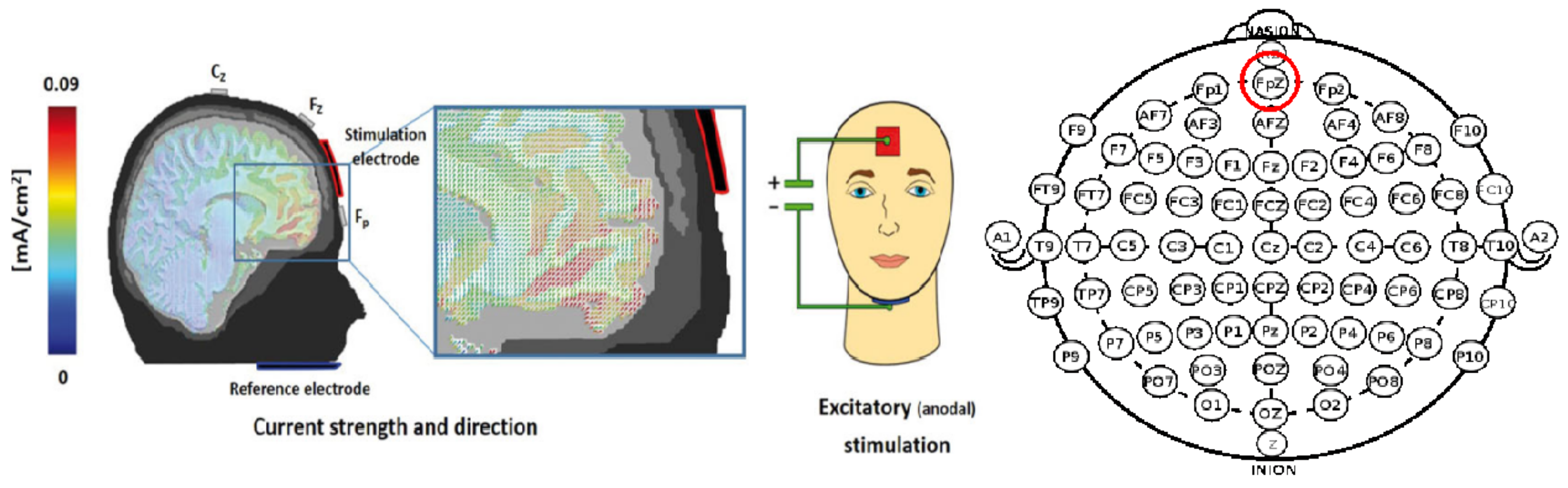
2.3.3. tDCS Device and Protocol
2.4. Measures
2.5. Ethic
2.6. Statistical Analysis
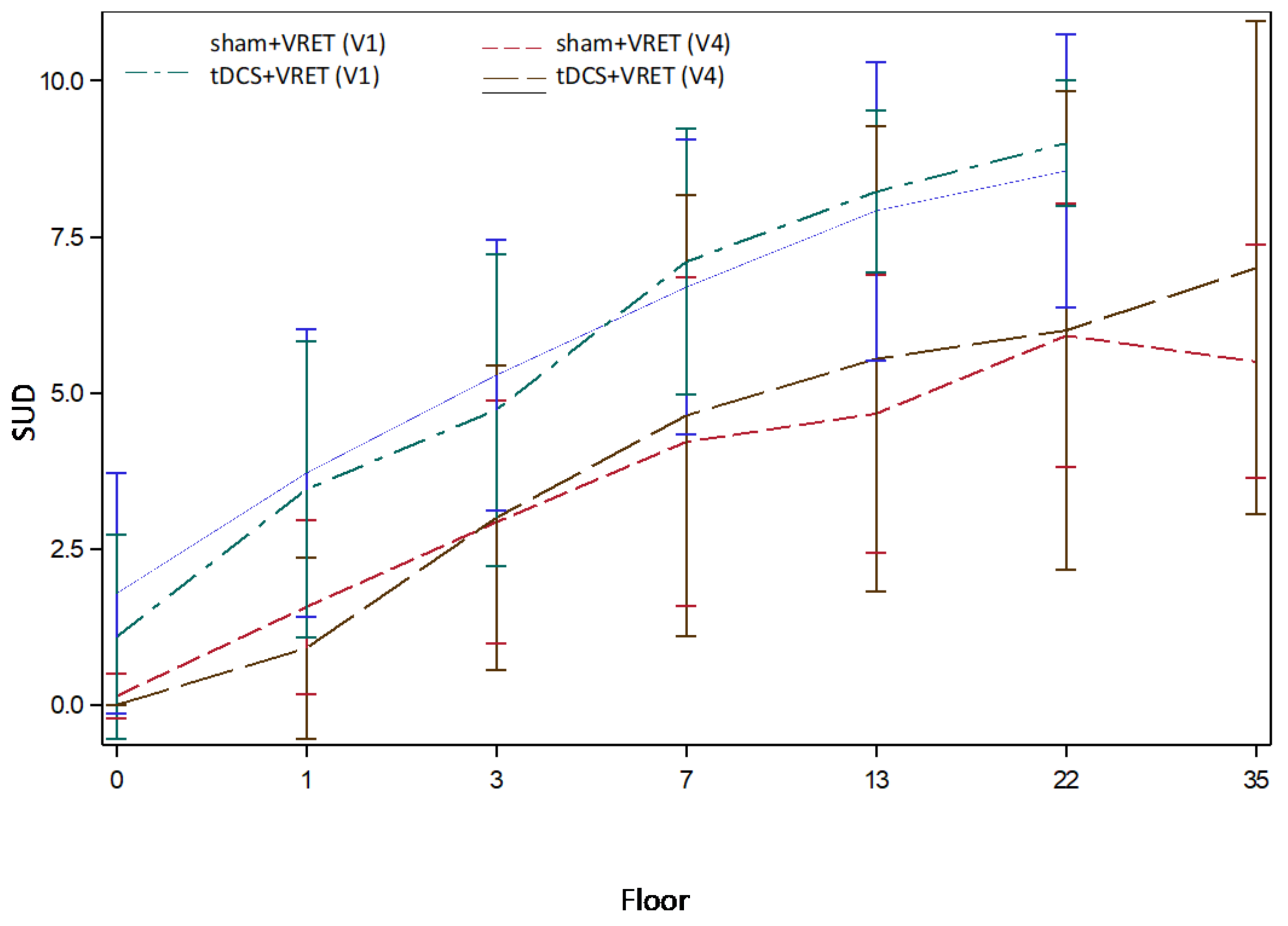
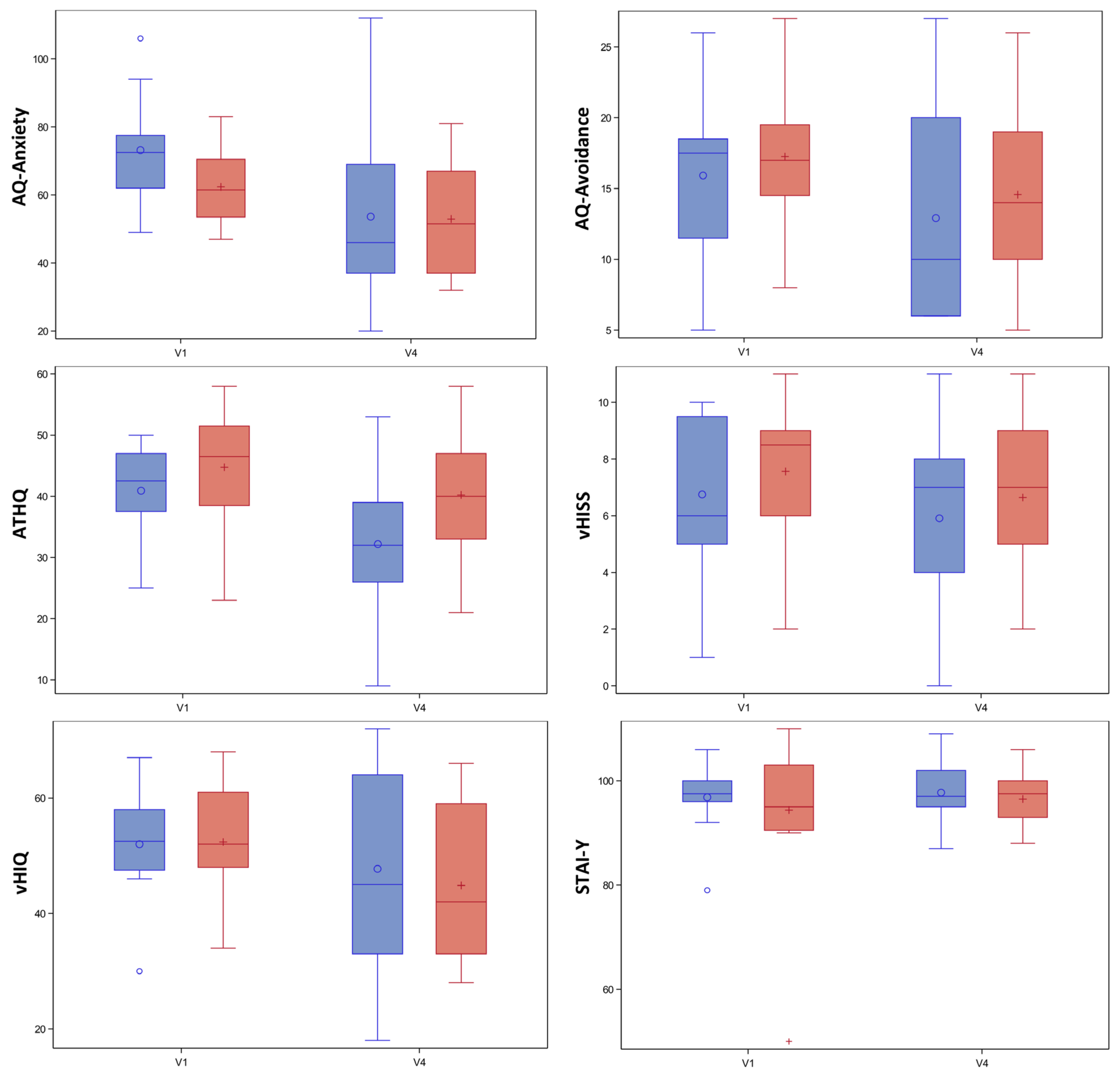
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bandelow, B.; Michaelis, S. Epidemiology of anxiety disorders in the 21st century. Dialogues Clin. Neurosci. 2015, 17, 327–335. [Google Scholar]
- Kyu, H.H.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1859–1922. [Google Scholar] [CrossRef] [Green Version]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); American Psychiatric Pub.: Washington, DC, USA, 2013. [Google Scholar]
- Golden, R.N. Making advances where it matters: Improving outcomes in mood and anxiety disorders. CNS Spectr. 2004, 9, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Sartori, S.B.; Singewald, N. Novel pharmacological targets in drug development for the treatment of anxiety and anxiety-related disorders. Pharmacol. Ther. 2019, 204, 107402. [Google Scholar] [CrossRef] [PubMed]
- Bystritsky, A. Treatment-resistant anxiety disorders. Mol. Psychiatry 2006, 11, 805–814. [Google Scholar] [CrossRef] [Green Version]
- Schottenbauer, M.A.; Glass, C.R.; Arnkoff, D.B.; Tendick, V.; Gray, S.H. Nonresponse and dropout rates in outcome studies on PTSD: Review and methodological considerations. Psychiatry 2008, 71, 134–168. [Google Scholar] [CrossRef] [PubMed]
- Lurie, I.; Levine, S.Z. Meta-analysis of dropout rates in SSRIs versus placebo in randomized clinical trials of PTSD. J. Nerv. Ment. Dis. 2010, 198, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Baxter, A.J.; Vos, T.; Scott, K.M.; Ferrari, A.J.; Whiteford, H.A. The global burden of anxiety disorders in 2010. Psychol. Med. 2014, 44, 2363–2374. [Google Scholar] [CrossRef]
- Wittchen, H.U.; Jacobi, F.; Rehm, J.; Gustavsson, A.; Svensson, M.; Jönsson, B.; Olesen, J.; Allgulander, C.; Alonso, J.; Faravelli, C.; et al. The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur. Neuropsychopharmacol. 2011, 21, 655–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapfhammer, H.-P.; Huppert, D.; Grill, E.; Fitz, W.; Brandt, T. Visual height intolerance and acrophobia: Clinical characteristics and comorbidity patterns. Eur. Arch. Psychiatry Clin. Neurosci. 2015, 265, 375–385. [Google Scholar] [CrossRef]
- Huppert, D.; Grill, E.; Brandt, T. A New Questionnaire for Estimating the Severity of Visual Height Intolerance and Acrophobia by a Metric Interval Scale. Front. Neurol. 2017, 8, 211. [Google Scholar] [CrossRef] [Green Version]
- McNally, R.J. Mechanisms of exposure therapy: How neuroscience can improve psychological treatments for anxiety disorders. Clin. Psychol. Rev. 2007, 27, 750–759. [Google Scholar] [CrossRef]
- Myers, K.M.; Davis, M. Behavioral and neural analysis of extinction. Neuron 2002, 36, 567–584. [Google Scholar] [CrossRef] [Green Version]
- Vervliet, B.; Craske, M.G.; Hermans, D. Fear extinction and relapse: State of the art. Annu. Rev. Clin. Psychol. 2013, 9, 215–248. [Google Scholar] [CrossRef]
- Morina, N.; Ijntema, H.; Meyerbröker, K.; Emmelkamp, P.M.G. Can virtual reality exposure therapy gains be generalized to real-life? A meta-analysis of studies applying behavioral assessments. Behav. Res. Ther. 2015, 74, 18–24. [Google Scholar] [CrossRef]
- Opriş, D.; Pintea, S.; García-Palacios, A.; Botella, C.; Szamosközi, Ş.; David, D. Virtual reality exposure therapy in anxiety disorders: A quantitative meta-analysis. Depress. Anxiety 2012, 29, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Emmelkamp, P.M.G.; Krijn, M.; Hulsbosch, A.M.; de Vries, S.; Schuemie, M.J.; van der Mast, C. Virtual reality treatment versus exposure in vivo: A comparative evaluation in acrophobia. Behav. Res. Ther. 2002, 40, 509–516. [Google Scholar] [CrossRef]
- Boeldt, D.; McMahon, E.; McFaul, M.; Greenleaf, W. Using Virtual Reality Exposure Therapy to Enhance Treatment of Anxiety Disorders: Identifying Areas of Clinical Adoption and Potential Obstacles. Front. Psychiatry 2019, 10, 773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ressler, K.J.; Rothbaum, B.O.; Tannenbaum, L.; Anderson, P.; Graap, K.; Zimand, E.; Hodges, L.; Davis, M. Cognitive enhancers as adjuncts to psychotherapy: Use of D-cycloserine in phobic individuals to facilitate extinction of fear. Arch. Gen. Psychiatry 2004, 61, 1136–1144. [Google Scholar] [CrossRef]
- Robinson, O.J.; Pike, A.C.; Cornwell, B.; Grillon, C. The translational neural circuitry of anxiety. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1353–1360. [Google Scholar] [CrossRef] [Green Version]
- Kalisch, R.; Wiech, K.; Critchley, H.D.; Dolan, R.J. Levels of appraisal: A medial prefrontal role in high-level appraisal of emotional material. NeuroImage 2006, 30, 1458–1466. [Google Scholar] [CrossRef] [Green Version]
- Marin, M.-F.; Camprodon, J.A.; Dougherty, D.D.; Milad, M.R. Device-based brain stimulation to augment fear extinction: Implications for PTSD treatment and beyond. Depress. Anxiety 2014, 31, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Ochsner, K.N.; Silvers, J.A.; Buhle, J.T. Functional imaging studies of emotion regulation: A synthetic review and evolving model of the cognitive control of emotion. Ann. N. Y. Acad. Sci. 2012, 1251, E1–E24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marin, M.-F.; Zsido, R.G.; Song, H.; Lasko, N.B.; Killgore, W.D.S.; Rauch, S.L.; Simon, N.M.; Milad, M.R. Skin Conductance Responses and Neural Activations During Fear Conditioning and Extinction Recall Across Anxiety Disorders. JAMA Psychiatry 2017, 74, 622–631. [Google Scholar] [CrossRef]
- Aleman, A. Use of repetitive transcranial magnetic stimulation for treatment in psychiatry. Clin. Psychopharmacol. Neurosci. Off. Sci. J. Korean Coll. Neuropsychopharmacol. 2013, 11, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Tortella, G.; Casati, R.; Aparicio, L.V.M.; Mantovani, A.; Senço, N.; D’Urso, G.; Brunelin, J.; Guarienti, F.; Selingardi, P.M.L.; Muszkat, D.; et al. Transcranial direct current stimulation in psychiatric disorders. World J. Psychiatry 2015, 5, 88–102. [Google Scholar] [CrossRef]
- Bajbouj, M.; Padberg, F. A perfect match: Noninvasive brain stimulation and psychotherapy. Eur. Arch. Psychiatry Clin. Neurosci. 2014, 264 (Suppl. 1), S27–S33. [Google Scholar] [CrossRef]
- Vicario, C.M.; Salehinejad, M.A.; Felmingham, K.; Martino, G.; Nitsche, M.A. A systematic review on the therapeutic effectiveness of non-invasive brain stimulation for the treatment of anxiety disorders. Neurosci. Biobehav. Rev. 2019, 96, 219–231. [Google Scholar] [CrossRef]
- Herrmann, M.J.; Katzorke, A.; Busch, Y.; Gromer, D.; Polak, T.; Pauli, P.; Deckert, J. Medial prefrontal cortex stimulation accelerates therapy response of exposure therapy in acrophobia. Brain Stimul. 2017, 10, 291–297. [Google Scholar] [CrossRef]
- Brunoni, A.R.; Amadera, J.; Berbel, B.; Volz, M.S.; Rizzerio, B.G.; Fregni, F. A systematic review on reporting and assessment of adverse effects associated with transcranial direct current stimulation. Int. J. Neuropsychopharmacol. 2011, 14, 1133–1145. [Google Scholar] [CrossRef]
- Bikson, M.; Grossman, P.; Thomas, C.; Zannou, A.L.; Jiang, J.; Adnan, T.; Mourdoukoutas, A.P.; Kronberg, G.; Truong, D.; Boggio, P.; et al. Safety of Transcranial Direct Current Stimulation: Evidence Based Update 2016. Brain Stimul. 2016, 9, 641–661. [Google Scholar] [CrossRef] [Green Version]
- Nikolin, S.; Huggins, C.; Martin, D.; Alonzo, A.; Loo, C.K. Safety of repeated sessions of transcranial direct current stimulation: A systematic review. Brain Stimul. 2018, 11, 278–288. [Google Scholar] [CrossRef]
- Abend, R.; Jalon, I.; Gurevitch, G.; Sar-El, R.; Shechner, T.; Pine, D.S.; Hendler, T.; Bar-Haim, Y. Modulation of fear extinction processes using transcranial electrical stimulation. Transl. Psychiatry 2016, 6, e913. [Google Scholar] [CrossRef] [PubMed]
- Dittert, N.; Hüttner, S.; Polak, T.; Herrmann, M.J. Augmentation of Fear Extinction by Transcranial Direct Current Stimulation (tDCS). Front. Behav. Neurosci. 2018, 12, 76. [Google Scholar] [CrossRef] [Green Version]
- Van’t Wout, M.; Shea, M.T.; Larson, V.C.; Greenberg, B.D.; Philip, N.S. Combined transcranial direct current stimulation with virtual reality exposure for posttraumatic stress disorder: Feasibility and pilot results. Brain Stimulation 2019, 12, 41–43. [Google Scholar] [CrossRef]
- Sauvaget, A.; Tostivint, A.; Etcheverrigaray, F.; Pichot, A.; Dert, C.; Schirr-Bonnais, S.; Clouet, J.; Sellal, O.; Mauduit, N.; Leux, C.; et al. Hospital production cost of transcranial direct current stimulation (tDCS) in the treatment of depression. Neurophysiol. Clin. 2019, 49, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Jamil, A.; Batsikadze, G.; Kuo, H.-I.; Labruna, L.; Hasan, A.; Paulus, W.; Nitsche, M.A. Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation. J. Physiol. 2017, 595, 1273–1288. [Google Scholar] [CrossRef]
- Dagar, S.; Chowdhury, S.R.; Bapi, R.S.; Dutta, A.; Roy, D. Near-Infrared Spectroscopy—Electroencephalography-Based Brain-State-Dependent Electrotherapy: A Computational Approach Based on Excitation-Inhibition Balance Hypothesis. Front. Neurol. 2016, 7, 123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Raedt, R.; Vanderhasselt, M.-A.; Baeken, C. Neurostimulation as an intervention for treatment resistant depression: From research on mechanisms towards targeted neurocognitive strategies. Clin. Psychol. Rev. 2015, 41, 61–69. [Google Scholar] [CrossRef] [Green Version]
- Miniussi, C.; Harris, J.A.; Ruzzoli, M. Modelling non-invasive brain stimulation in cognitive neuroscience. Neurosci. Biobehav. Rev. 2013, 37, 1702–1712. [Google Scholar] [CrossRef] [Green Version]
- Junghofer, M.; Winker, C.; Rehbein, M.A.; Sabatinelli, D. Noninvasive Stimulation of the Ventromedial Prefrontal Cortex Enhances Pleasant Scene Processing. Cereb. Cortex 2017, 27, 3449–3456. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, S.K.; Prasanna, S.S. Virtual Reality and Noninvasive Brain Stimulation in Stroke: How Effective Is Their Combination for Upper Limb Motor Improvement?—A Meta-Analysis. PM&R 2018, 10, 1261–1270. [Google Scholar] [CrossRef]
- Beeli, G.; Casutt, G.; Baumgartner, T.; Jäncke, L. Modulating presence and impulsiveness by external stimulation of the brain. Behav. Brain Funct. 2008, 4, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, D.C. Comparison of self-report and overt-behavioral procedures for assessing acrophobia. Behav. Ther. 1977, 8, 17–23. [Google Scholar] [CrossRef]
- Wolpe, J. 77ie Practice of Behavior Therapy; Pergamon Press: New York, NY, USA, 1969. [Google Scholar]
- Roesmann, K.; Kroker, T.; Hein, S.; Rehbein, M.; Winker, C.; Leehr, E.J.; Klucken, T.; Junghöfer, M. Transcranial Direct Current Stimulation of the Ventromedial Prefrontal Cortex Modulates Perceptual and Neural Patterns of Fear Generalization. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, in press. [Google Scholar] [CrossRef]
- Abelson, J.L.; Curtis, G.C. Cardiac and neuroendocrine responses to exposure therapy in height phobics: Desynchrony within the “physiological response system”. Behav. Res. Ther. 1989, 27, 561–567. [Google Scholar] [CrossRef] [Green Version]
- Steinman, S.A.; Teachman, B.A. Cognitive Processing and Acrophobia: Validating the Heights Interpretation Questionnaire. J. Anxiety Disord. 2011, 25, 896–902. [Google Scholar] [CrossRef] [Green Version]
- Spielberger, C.D.; Sydeman, S.J. State-trait anxiety inventory and state-trait anger expression inventory. In The Use of Psychological Testing for Treatment Planning and Outcome Assessment; Maruish, M.E., Ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1994. [Google Scholar]
- Guy, W. ECDEU Assessment Manual for Psychopharmacology; US Department of Health, Education, and Welfare, Public Health Service, Psychopharmacology Research Branch, Division of Extramural Research Programs: Rockville, MD, USA, 1976. [Google Scholar]
- Schubert, T.; Friedmann, F.; Regenbrecht, H. The experience of presence: Factor analytic insights. Presence Teleoperators Virtual Environ. 2001, 10, 266–281. [Google Scholar] [CrossRef]
- Kennedy, R.S.; Lane, N.E.; Berbaum, K.S.; Lilienthal, M.G. Simulator sickness questionnaire: An enhanced method for quantifying simulator sickness. Int. J. Aviat. Psychol. 1993, 3, 203–220. [Google Scholar] [CrossRef]
- Rabinak, C.A.; Angstadt, M.; Lyons, M.; Mori, S.; Milad, M.R.; Liberzon, I.; Phan, K.L. Cannabinoid modulation of prefrontal-limbic activation during fear extinction learning and recall in humans. Neurobiol. Learn. Mem. 2014, 113, 125–134. [Google Scholar] [CrossRef] [Green Version]
- Coelho, C.M.; Santos, J.A.; Silva, C.; Wallis, G.; Tichon, J.; Hine, T.J. The role of self-motion in acrophobia treatment. Cyberpsychol. Behav. Impact Internet Multimed. Virtual Real. Behav. Soc. 2008, 11, 723–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coelho, C.M.; Silva, C.F.; Santos, J.A.; Tichon, J.; Wallis, G. Contrasting the effectiveness and efficiency of virtual reality and real environments in the treatment of acrophobia. PsychNology J. 2008, 6, 203–216. [Google Scholar]
- Oldrati, V.; Colombo, B.; Antonietti, A. Combination of a short cognitive training and tDCS to enhance visuospatial skills: A comparison between online and offline neuromodulation. Brain Res. 2018, 1678, 32–39. [Google Scholar] [CrossRef] [PubMed]
- D’Urso, G.; Mantovani, A.; Patti, S.; Toscano, E.; de Bartolomeis, A. Transcranial Direct Current Stimulation in Obsessive-Compulsive Disorder, Posttraumatic Stress Disorder, and Anxiety Disorders. J. ECT 2018, 34, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Slater, M.; Lotto, B.; Arnold, M.M.; Sánchez-Vives, M.V. How we experience immersive virtual environments: The concept of presence and its measurement. Anu. Psicol. 2009, 40, 193–210. [Google Scholar]
- Diemer, J.; Alpers, G.W.; Peperkorn, H.M.; Shiban, Y.; Mühlberger, A. The impact of perception and presence on emotional reactions: A review of research in virtual reality. Front. Psychol. 2015, 6, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumgartner, T.; Speck, D.; Wettstein, D.; Masnari, O.; Beeli, G.; Jäncke, L. Feeling present in arousing virtual reality worlds: Prefrontal brain regions differentially orchestrate presence experience in adults and children. Front. Hum. Neurosci. 2008, 2, 8. [Google Scholar] [CrossRef] [Green Version]
- Bjekic, J.; Zivanovic, M.; Filipović, S. Transcranial Direct Current Stimulation (tDCS) for Memory Enhancement. J. Vis. Exp. 2021, 2021, e62681. [Google Scholar] [CrossRef]
- Minarik, T.; Berger, B.; Althaus, L.; Bader, V.; Biebl, B.; Brotzeller, F.; Fusban, T.; Hegemann, J.; Jesteadt, L.; Kalweit, L.; et al. The Importance of Sample Size for Reproducibility of tDCS Effects. Front. Hum. Neurosci. 2016, 10, 453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berlim, M.T.; Van den Eynde, F.; Daskalakis, Z.J. Clinical utility of transcranial direct current stimulation (tDCS) for treating major depression: A systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. J. Psychiatr. Res. 2013, 47, 1–7. [Google Scholar] [CrossRef]
- Shiozawa, P.; Fregni, F.; Benseñor, I.M.; Lotufo, P.A.; Berlim, M.T.; Daskalakis, J.Z.; Cordeiro, Q.; Brunoni, A.R. Transcranial direct current stimulation for major depression: An updated systematic review and meta-analysis. Int. J. Neuropsychopharmacol. 2014, 17, 1443–1452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nitsche, M.A.; Doemkes, S.; Karaköse, T.; Antal, A.; Liebetanz, D.; Lang, N.; Tergau, F.; Paulus, W. Shaping the effects of transcranial direct current stimulation of the human motor cortex. J. Neurophysiol. 2007, 97, 3109–3117. [Google Scholar] [CrossRef] [Green Version]
- Marković, V.; Vicario, C.M.; Yavari, F.; Salehinejad, M.A.; Nitsche, M.A. A Systematic Review on the Effect of Transcranial Direct Current and Magnetic Stimulation on Fear Memory and Extinction. Front. Hum. Neurosci. 2021, 15, 655947. [Google Scholar] [CrossRef]
- Chen, Y.; Lu, X.; Yu, P.; Zeng, L.; Ye, H.; Shi, Q.; Guo, W. Modulating the Activity of the VMPFC with tDCS Alters the Social Framing Effect. Front. Behav. Neurosci. 2021, 15, 677006. [Google Scholar] [CrossRef]
- Baeken, C.; Vanderhasselt, M.-A.; Remue, J.; Rossi, V.; Schiettecatte, J.; Anckaert, E.; De Raedt, R. One left dorsolateral prefrontal cortical HF-rTMS session attenuates HPA-system sensitivity to critical feedback in healthy females. Neuropsychologia 2014, 57, 112–121. [Google Scholar] [CrossRef] [Green Version]
- Pulopulos, M.M.; Schmausser, M.; De Smet, S.; Vanderhasselt, M.-A.; Baliyan, S.; Venero, C.; Baeken, C.; De Raedt, R. The effect of HF-rTMS over the left DLPFC on stress regulation as measured by cortisol and heart rate variability. Horm. Behav. 2020, 124, 104803. [Google Scholar] [CrossRef]
- Mehrsafar, A.H.; Rosa, M.A.S.; Zadeh, A.M.; Gazerani, P. A feasibility study of application and potential effects of a single session transcranial direct current stimulation (tDCS) on competitive anxiety, mood state, salivary levels of cortisol and alpha amylase in elite athletes under a real-world competition. Physiol. Behav. 2020, 227, 113173. [Google Scholar] [CrossRef]
- Carnevali, L.; Pattini, E.; Sgoifo, A.; Ottaviani, C. Effects of prefrontal transcranial direct current stimulation on autonomic and neuroendocrine responses to psychosocial stress in healthy humans. Stress 2020, 23, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Gianaros, P.J.; Wager, T.D. Brain-body pathways linking psychological stress and physical health. Curr. Dir. Psychol. Sci. 2015, 24, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Duran, N.L.; Mayer, S.E.; Abelson, J.L. Modeling neuroendocrine stress reactivity in salivary cortisol: Adjusting for peak latency variability. Stress 2014, 17, 285–295. [Google Scholar] [CrossRef] [PubMed]




| tDCS + VERT (n = 11) | Sham + VERT (n = 14) | Stat | p-Values | |
|---|---|---|---|---|
| Sex (male) | 2 (18.2%) | 5 (35.7%) | χ2 = 0.939 | 0.332 |
| Age | 37.27 ± 15.88 | 37.79 ± 17.68 | z = −0.164 | 0.869 |
| AQ anxiety | 74.00 ± 15.80 | 64.14 ± 10.41 | z = −1.591 | 0.112 |
| AQ avoidance | 15.73 ± 6.23 | 17.21 ± 2.94 | z = −0.852 | 0.394 |
| ATHQ | 41.36 ± 8.15 | 45.57 ± 9.80 | z = −1.372 | 0.170 |
| HIQ | 52.27 ± 9.80 | 52.79 ± 9.96 | z = −0.302 | 0.763 |
| STAI-Y-A | 48.64 ± 2.01 | 48.71 ± 4.68 | z = −0.277 | 0.782 |
| STAI-Y-B | 49.82 ± 3.09 | 49.07 ± 4.60 | z = −0.826 | 0.409 |
| STAI-Y total | 98.45 ± 3.64 | 97.79 ± 7.04 | z = −0.823 | 0.410 |
| vHISS | 6.82 ± 2.86 | 7.57 ± 2.85 | z = −0.635 | 0.526 |
| CGI Severity | 4.91 ± 0.70 | 4.79 ± 0.98 | z = −0.119 | 0.905 |
| SUD max | 8.55 ± 2.34 | 8.36 ± 2.27 | z = −0.173 | 0.863 |
| Mean (Sham Group) | 95% CI Lower | 95% CI Upper | Mean (Active Group) | 95% CI Lower | 95% CI Upper | Effect Size | |
|---|---|---|---|---|---|---|---|
| SSQ Scale at V2 SSQ scale at V3 | 8.642 5.000 | 6.658 3.323 | 10.628 6.677 | 11.364 7.909 | 5.368 1.246 | 17.359 14.572 | 0.363 0.358 |
| Scores | Mean (Sham Group) | 95% CI Lower | 95% CI Upper | Mean (Active Group) | 95% CI Lower | 95% CI Upper | Effect Size |
|---|---|---|---|---|---|---|---|
| SUD max | 6.2100 | 4.9800 | 7.4400 | 6.5500 | 4.1500 | 8.9500 | 0.1046 |
| AQ anxiety | 52.9300 | 44.4900 | 61.3700 | 53.6400 | 37.6800 | 69.6000 | 0.0326 |
| AQ avoidance | 14.5700 | 11.1700 | 17.9700 | 12.9100 | 8.1200 | 17.7000 | −0.2306 |
| ATHQ | 40.2100 | 34.4300 | 45.9900 | 32.1800 | 25.2500 | 39.1100 | −0.7193 |
| vHISS | 6.6400 | 5.2400 | 8.0400 | 5.9100 | 3.8700 | 7.9500 | −0.2410 |
| HIQ | 44.8600 | 37.3200 | 52.4000 | 47.7300 | 37.3200 | 58.1400 | 0.1820 |
| STAI | 96.5000 | 93.9776 | 99.0224 | 97.7273 | 94.2837 | 101.1713 | 0.2343 |
| CGI total | 15.3571 | 12.9703 | 17.7440 | 16.0909 | 11.6314 | 20.5505 | 0.1201 |
| IPQ | 6.5000 | 1.4649 | 11.535 | 10.2727 | 6.2177 | 14.3273 | 0.4610 |
| SSQ at V2 | 8.6429 | 6.6582 | 10.627 | 11.3636 | 5.3680 | 17.3592 | 0.3625 |
| SSQ at V3 | 5.0000 | 3.3227 | 6.6772 | 7.9090 | 1.2460 | 14.5721 | 0.3582 |
| Cortisol level after exposure | 0.9214 | 0.5945 | 1.2484 | 0.8636 | 0.5442 | 1.1830 | −0.1010 |
| Scores | Mean (Sham Group) | 95% CI Lower | 95% CI Upper | Mean (Active Group) | 95% CI Lower | 95% CI Upper | Effect Size | p-Value * |
|---|---|---|---|---|---|---|---|---|
| SUD max | −2.14 | −3.21 | −1.07 | −2.00 | −3.81 | −0.19 | 0.0549 | 0.484 |
| AQ anxiety | −11.21 | −18.16 | −4.26 | −20.36 | −34.83 | −5.89 | −0.4741 | 0.228 |
| AQ avoidance | −2.64 | −5.31 | 0.03 | −2.82 | −7.04 | 1.40 | −0.0296 | 0.913 |
| ATHQ | −5.36 | −9.10 | −1.62 | −9.18 | −17.62 | −0.74 | −0.3451 | 0.583 |
| vHISS | −0.93 | −1.46 | −0.40 | −0.91 | −2.12 | 0.30 | 0.0126 | 0.955 |
| HIQ total | −7.93 | −13.81 | −2.05 | −4.55 | −12.99 | 3.89 | 0.2685 | 0.459 |
| STAI total | −1.29 | −4.50 | 1.92 | −0.73 | −3.75 | 2.29 | 0.1014 | 0.978 |
| Cortisol level after exposure | −0.29 | −0.82 | 0.24 | 0.11 | 0.11 | 0.11 | 0.5657 | 0.131 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bulteau, S.; Laurin, A.; Bach-Ngohou, K.; Péré, M.; Vibet, M.-A.; Hardouin, J.-B.; Sebille, V.; Lagalice, L.; Faurel-Paul, É.; Acier, D.; et al. Feasibility of Combining Transcranial Direct Current Stimulation and Active Fully Embodied Virtual Reality for Visual Height Intolerance: A Double-Blind Randomized Controlled Study. J. Clin. Med. 2022, 11, 345. https://doi.org/10.3390/jcm11020345
Bulteau S, Laurin A, Bach-Ngohou K, Péré M, Vibet M-A, Hardouin J-B, Sebille V, Lagalice L, Faurel-Paul É, Acier D, et al. Feasibility of Combining Transcranial Direct Current Stimulation and Active Fully Embodied Virtual Reality for Visual Height Intolerance: A Double-Blind Randomized Controlled Study. Journal of Clinical Medicine. 2022; 11(2):345. https://doi.org/10.3390/jcm11020345
Chicago/Turabian StyleBulteau, Samuel, Andrew Laurin, Kalyane Bach-Ngohou, Morgane Péré, Marie-Anne Vibet, Jean-Benoit Hardouin, Véronique Sebille, Lydie Lagalice, Élodie Faurel-Paul, Didier Acier, and et al. 2022. "Feasibility of Combining Transcranial Direct Current Stimulation and Active Fully Embodied Virtual Reality for Visual Height Intolerance: A Double-Blind Randomized Controlled Study" Journal of Clinical Medicine 11, no. 2: 345. https://doi.org/10.3390/jcm11020345
APA StyleBulteau, S., Laurin, A., Bach-Ngohou, K., Péré, M., Vibet, M.-A., Hardouin, J.-B., Sebille, V., Lagalice, L., Faurel-Paul, É., Acier, D., Rabeyron, T., Riche, V.-P., Sauvaget, A., Melki, F., Vigier, T., Perreira Da Silva, M., Charlet, O., & Prié, Y. (2022). Feasibility of Combining Transcranial Direct Current Stimulation and Active Fully Embodied Virtual Reality for Visual Height Intolerance: A Double-Blind Randomized Controlled Study. Journal of Clinical Medicine, 11(2), 345. https://doi.org/10.3390/jcm11020345






