Efficacy of Home-Based Transcranial Direct Current Stimulation on Experimental Pain Sensitivity in Older Adults with Knee Osteoarthritis: A Randomized, Sham-Controlled Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Interventions and Procedures
2.4. Outcomes
2.4.1. Clinical Pain Intensity
2.4.2. Experimental Pain Sensitivity
2.4.3. Sample Size
2.4.4. Statistical Analysis
3. Results
3.1. Participants
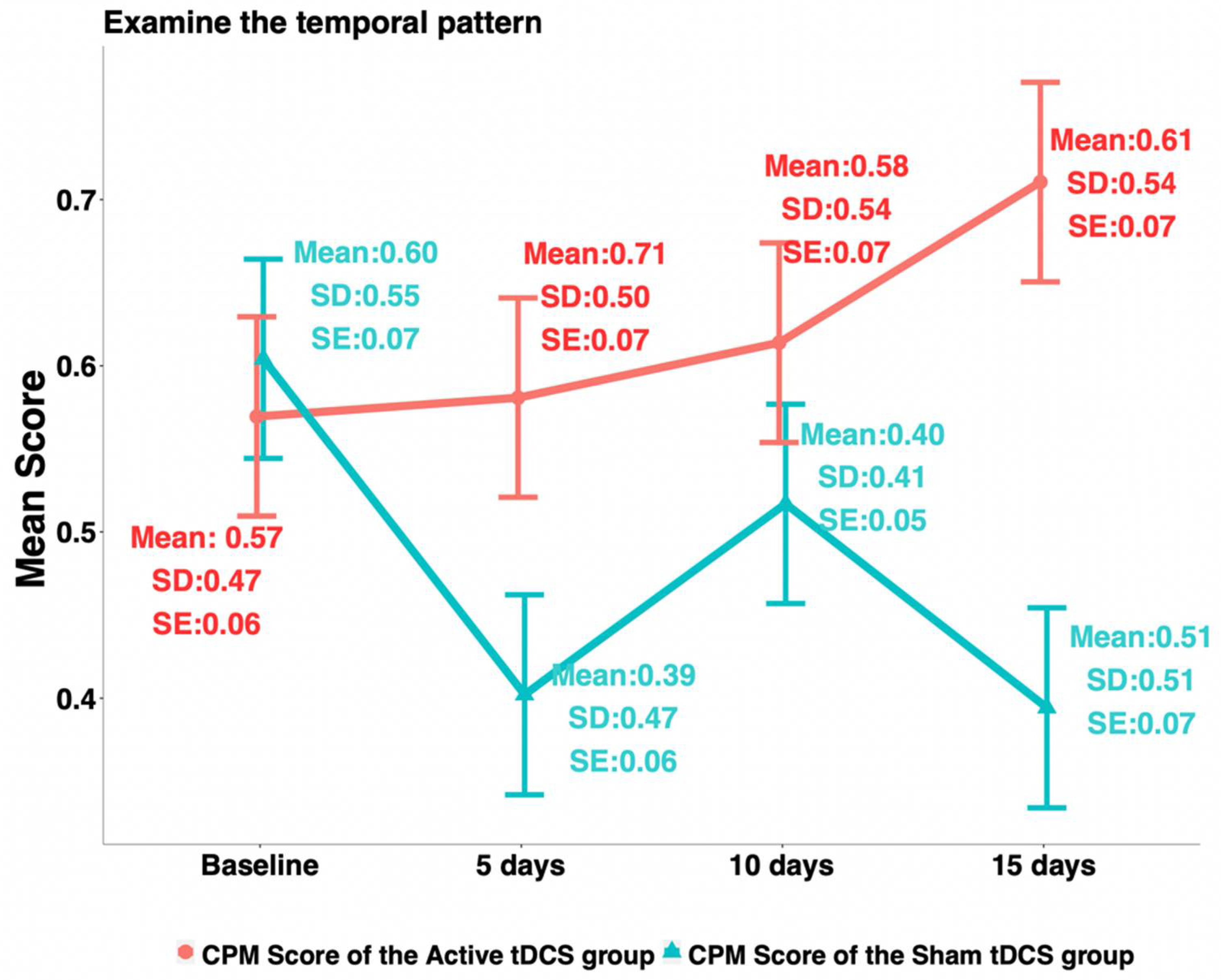
3.2. Experimental Pain Sensitivity
3.3. Relationship between Clinical Pain Intensity and Experimental Pain Sensitivity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- O’Neill, T.W.; Felson, D.T. Mechanisms of Osteoarthritis (OA) Pain. Curr. Osteoporos. Rep. 2018, 16, 611–616. [Google Scholar] [CrossRef]
- Barbour, K.E.; Boring, M.; Helmick, C.G.; Murphy, L.B.; Qin, J. Prevalence of Severe Joint Pain Among Adults with Doctor-Diagnosed Arthritis—United States, 2002–2014. MMWR Morb. Mortal. Wkly. Rep. 2016, 65, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Wylde, V.; Beswick, A.; Bruce, J.; Blom, A.; Howells, N.; Gooberman-Hill, R. Chronic pain after total knee arthroplasty. EFORT Open Rev. 2018, 3, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Soni, A.; Wanigasekera, V.; Mezue, M.; Cooper, C.; Javaid, M.; Price, A.J.; Tracey, I. Central Sensitization in Knee Osteoarthritis: Relating Presurgical Brainstem Neuroimaging and PainDETECT-Based Patient Stratification to Arthroplasty Outcome. Arthritis Rheumatol. 2019, 71, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Fingleton, C.; Smart, K.; Moloney, N.; Fullen, B.M.; Doody, C. Pain sensitization in people with knee osteoarthritis: A systematic review and meta-analysis. Osteoarthr. Cartil. 2015, 23, 1043–1056. [Google Scholar] [CrossRef]
- Curatolo, M. Reflections on Pain Sensitivity Washington, DC: International Association for the Study of Pain. Available online: https://www.iasp-pain.org/publications/relief-news/article/pain-sensitivity/ (accessed on 10 July 2022).
- Martorella, G.; Mathis, K.; Miao, H.; Wang, D.; Park, L.; Ahn, H. Self-administered Transcranial Direct Current Stimulation for Pain in Older Adults with Knee Osteoarthritis: A Randomized Controlled Trial. Brain Stimul. 2022, 15, 902–909. [Google Scholar] [CrossRef]
- Ahn, H.; Woods, A.J.; Kunik, M.E.; Bhattacharjee, A.; Chen, Z.; Choi, E.; Fillingim, R.B. Efficacy of transcranial direct current stimulation over primary motor cortex (anode) and contralateral supraorbital area (cathode) on clinical pain severity and mobility performance in persons with knee osteoarthritis: An experimenter- and participant-blinded, randomized, sham-controlled pilot clinical study. Brain Stimul. 2017, 10, 902–909. [Google Scholar]
- Bartholomew, C.; Lack, S.; Neal, B. Altered pain processing and sensitisation is evident in adults with patellofemoral pain: A systematic review including meta-analysis and meta-regression. Scand. J. Pain 2019, 20, 11–27. [Google Scholar] [CrossRef]
- Antal, A.; Terney, D.; Kuhnl, S.; Paulus, W. Anodal transcranial direct current stimulation of the motor cortex ameliorates chronic pain and reduces short intracortical inhibition. J. Pain Symptom Manag. 2010, 39, 890–903. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Cohen, L.G.; Wassermann, E.M.; Priori, A.; Lang, N.; Antal, A.; Paulus, W.; Hummel, F.; Boggio, P.S.; Fregni, F.; et al. Transcranial direct current stimulation: State of the art 2008. Brain Stimul. 2008, 1, 206–223. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; Antal, A.; Ayache, S.S.; Benninger, D.H.; Brunelin, J.; Cogiamanian, F.; Cotelli, M.; De Ridder, D.; Ferrucci, R.; Langguth, B.; et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol. 2017, 128, 56–92. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.; Suchting, R.; Woods, A.J.; Miao, H.; Green, C.; Cho, R.Y.; Choi, E.; Fillingim, R. Bayesian analysis of the effect of transcranial direct current stimulation on experimental pain sensitivity in older adults with knee osteoarthritis: Randomized sham-controlled pilot clinical study. J. Pain Res. 2018, 11, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Suchting, R.; Kapoor, S.; Mathis, K.B.; Ahn, H. Changes in Experimental Pain Sensitivity from Using Home-Based Remotely Supervised Transcranial Direct Current Stimulation in Older Adults with Knee Osteoarthritis. Pain Med. 2020, 21, 2676–2683. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.; Zhong, C.; Miao, H.; Chaoul, A.; Park, L.; Yen, I.H.; Vila, M.A.; Sorkpor, S.; Abdi, S. Efficacy of combining home-based transcranial direct current stimulation with mindfulness-based meditation for pain in older adults with knee osteoarthritis: A randomized controlled pilot study. J. Clin. Neurosci. 2019, 70, 140–145. [Google Scholar] [CrossRef]
- Altman, R.; Asch, E.; Bloch, D.; Bole, G.; Borenstein, D.; Brandt, K.; Christy, W.; Cooke, T.D. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 1986, 29, 1039–1049. [Google Scholar] [CrossRef]
- Edwards, R.R.; Fillingim, R.B.; Ness, T.J. Age-related differences in endogenous pain modulation: A comparison of diffuse noxious inhibitory controls in healthy older and younger adults. Pain 2003, 101, 155–165. [Google Scholar] [CrossRef]
- Fregni, F.; Gimenes, R.; Valle, A.C.; Ferreira, M.J.L.; Rocha, R.R.; Natalle, L.; Bravo, R.; Rigonatti, S.P.; Freedman, S.D.; Nitsche, M.A.; et al. A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum. 2006, 54, 3988–3998. [Google Scholar] [CrossRef]
- Gandiga, P.C.; Hummel, F.C.; Cohen, L.G. Transcranial DC stimulation (tDCS): A tool for double-blind sham-controlled clinical studies in brain stimulation. Clin. Neurophysiol. 2006, 117, 845–850. [Google Scholar] [CrossRef]
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res. 2011, 63 (Suppl. S11), S240–S252. [Google Scholar] [CrossRef]
- Dworkin, R.H.; Turk, D.C.; McDermott, M.P.; Peirce-Sandner, S.; Burke, L.B.; Cowan, P.; Farrar, J.T.; Hertz, S.; Raja, S.N.; Rappaport, B.A.; et al. Interpreting the clinical importance of group differences in chronic pain clinical trials: IMMPACT recommendations. Pain 2009, 146, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Uddin, Z.; MacDermid, J.C. Quantitative Sensory Testing in Chronic Musculoskeletal Pain. Pain Med. 2016, 17, 1694–1703. [Google Scholar] [CrossRef] [Green Version]
- Weaver, K.R.; Griffioen, M.A.; Klinedinst, N.J.; Galik, E.; Duarte, A.C.; Colloca, L.; Resnick, B.; Dorsey, S.G.; Renn, C.L. Quantitative Sensory Testing Across Chronic Pain Conditions and Use in Special Populations. Front. Pain Res. 2021, 2, 779068. [Google Scholar] [CrossRef]
- Yarnitsky, D.; Arendt-Nielsen, L.; Bouhassira, D.; Edwards, R.R.; Fillingim, R.; Granot, M.; Hansson, P.; Lautenbacher, S.; Marchand, S.; Wilder-Smith, O. Recommendations on terminology and practice of psychophysical DNIC testing. Eur. J. Pain 2010, 14, 339. [Google Scholar] [CrossRef]
- Kennedy, D.L.; Kemp, H.I.; Ridout, D.; Yarnitsky, D.; Rice, A.S.C. Reliability of conditioned pain modulation: A systematic review. Pain 2016, 157, 2410–2419. [Google Scholar] [CrossRef] [PubMed]
- Carlesso, L.C.; Law, L.F.; Wang, N.; Nevitt, M.; Lewis, C.E.; Neogi, T.; Multicenter Osteoarthritis Study Group. Association of Pain Sensitization and Conditioned Pain Modulation to Pain Patterns in Knee Osteoarthritis. Arthritis Care Res. 2022, 74, 107–112. [Google Scholar] [CrossRef]
- Foucher, K.C.; Chmell, S.J.; Courtney, C.A. Duration of symptoms is associated with conditioned pain modulation and somatosensory measures in knee osteoarthritis. J. Orthop. Res. 2019, 37, 136–142. [Google Scholar] [CrossRef]
- Yarnitsky, D.; Bouhassira, D.; Drewes, A.; Fillingim, R.; Granot, M.; Hansson, P.; Landau, R.; Marchand, S.; Matre, D.; Nilsen, K.; et al. Recommendations on practice of conditioned pain modulation (CPM) testing. Eur. J. Pain 2015, 19, 805–806. [Google Scholar] [CrossRef]
- Tavares, D.R.B.; Okazaki, J.E.F.; Santana, M.V.D.A.; Pinto, A.C.P.N.; Tutiya, K.K.; Gazoni, F.M.; Pinto, C.B.; Santos, F.C.; Fregni, F.; Trevisani, V.F.M. Motor cortex transcranial direct current stimulation effects on knee osteoarthritis pain in elderly subjects with dysfunctional descending pain inhibitory system: A randomized controlled trial. Brain Stimul. 2021, 14, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Bartley, E.J.; King, C.D.; Sibille, K.T.; Cruz-Almeida, Y.; Riley, J.L., 3rd; Glover, T.L.; Goodin, B.R.; Sotolongo, A.S.; Herbert, M.S.; Bulls, H.W.; et al. Enhanced Pain Sensitivity Among Individuals with Symptomatic Knee Osteoarthritis: Potential Sex Differences in Central Sensitization. Arthritis Care Res. 2016, 68, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yao, J.; Zhang, W.; Chen, S.; Peng, W. Effects of transcranial direct current stimulation on experimental pain perception: A systematic review and meta-analysis. Clin. Neurophysiol. 2021, 132, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.B.; Teixeira Costa, B.; Duarte, D.; Fregni, F. Transcranial Direct Current Stimulation as a Therapeutic Tool for Chronic Pain. J. ECT 2018, 34, e36–e50. [Google Scholar] [CrossRef] [PubMed]
- Brietzke, A.P.; Zortea, M.; Carvalho, F.; Sanches, P.R.; Silva, D.P.J.; Torres, I.L.D.S.; Fregni, F.; Caumo, W. Large Treatment Effect with Extended Home-Based Transcranial Direct Current Stimulation over Dorsolateral Prefrontal Cortex in Fibromyalgia: A Proof of Concept Sham-Randomized Clinical Study. J. Pain 2020, 21, 212–224. [Google Scholar] [CrossRef]
- Sorkpor, S.; Galle, K.; Teixeira, A.; Colpo, G.; Ahn BJackson, N.; Miao, H.; Ahn, H. The Relationship Between Plasma BDNF and Pain in Older Adults with Knee Osteoarthritis. Biol. Res. Nurs. 2022, 23, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Suchting, R.; Teixeira, A.L.; Ahn, B.; Colpo, G.D.; Park, J.; Ahn, H. Changes in Brain-derived Neurotrophic Factor from Active and Sham Transcranial Direct Current Stimulation in Older Adults with Knee Osteoarthritis. Clin. J. Pain 2021, 37, 898–903. [Google Scholar] [CrossRef]
- Pacheco-Barrios, K.; Cardenas-Rojas, A.; Thibaut, A.; Costa, B.; Ferreira, I.; Caumo, W.; Fregni, F. Methods and strategies of tDCS for the treatment of pain: Current status and future directions. Expert Rev. Med. Devices 2020, 17, 879–898. [Google Scholar] [CrossRef]
| Variable | Sham tDCS (n = 60) | Active tDCS (n = 60) | p-Value |
|---|---|---|---|
| HPTh, M(SD) | 40.17 (3.42) | 39.40 (3.30) | 0.12 |
| HPTo, M(SD) | 45.06 (3.17) | 44.85 (2.74) | 0.51 |
| PPT, M(SD) | 2.57 (1.17) | 2.40 (1.02) | 0.57 |
| CPM, M(SD) | 0.60 (0.55) | 0.57 (0.47) | 0.58 |
| Variable | Sham Group (n = 60) | Active Group (n = 60) | Effect Size (d) | p-Value |
|---|---|---|---|---|
| HPTh Change | −1.133.08 | 1.133.43 | 0.69 | <0.01 |
| HPTo Change | −0.102.39 | 0.842.10 | 0.42 | 0.03 |
| PPT Change | −0.300.70 | 0.560.52 | 1.39 | <0.0001 |
| CPM Change | −0.210.65 | 0.140.67 | 0.53 | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martorella, G.; Mathis, K.; Miao, H.; Wang, D.; Park, L.; Ahn, H. Efficacy of Home-Based Transcranial Direct Current Stimulation on Experimental Pain Sensitivity in Older Adults with Knee Osteoarthritis: A Randomized, Sham-Controlled Clinical Trial. J. Clin. Med. 2022, 11, 5209. https://doi.org/10.3390/jcm11175209
Martorella G, Mathis K, Miao H, Wang D, Park L, Ahn H. Efficacy of Home-Based Transcranial Direct Current Stimulation on Experimental Pain Sensitivity in Older Adults with Knee Osteoarthritis: A Randomized, Sham-Controlled Clinical Trial. Journal of Clinical Medicine. 2022; 11(17):5209. https://doi.org/10.3390/jcm11175209
Chicago/Turabian StyleMartorella, Geraldine, Kenneth Mathis, Hongyu Miao, Duo Wang, Lindsey Park, and Hyochol Ahn. 2022. "Efficacy of Home-Based Transcranial Direct Current Stimulation on Experimental Pain Sensitivity in Older Adults with Knee Osteoarthritis: A Randomized, Sham-Controlled Clinical Trial" Journal of Clinical Medicine 11, no. 17: 5209. https://doi.org/10.3390/jcm11175209
APA StyleMartorella, G., Mathis, K., Miao, H., Wang, D., Park, L., & Ahn, H. (2022). Efficacy of Home-Based Transcranial Direct Current Stimulation on Experimental Pain Sensitivity in Older Adults with Knee Osteoarthritis: A Randomized, Sham-Controlled Clinical Trial. Journal of Clinical Medicine, 11(17), 5209. https://doi.org/10.3390/jcm11175209






