Towards a Reliable and Rapid Automated Grading System in Facial Palsy Patients: Facial Palsy Surgery Meets Computer Science
Abstract
:1. Introduction
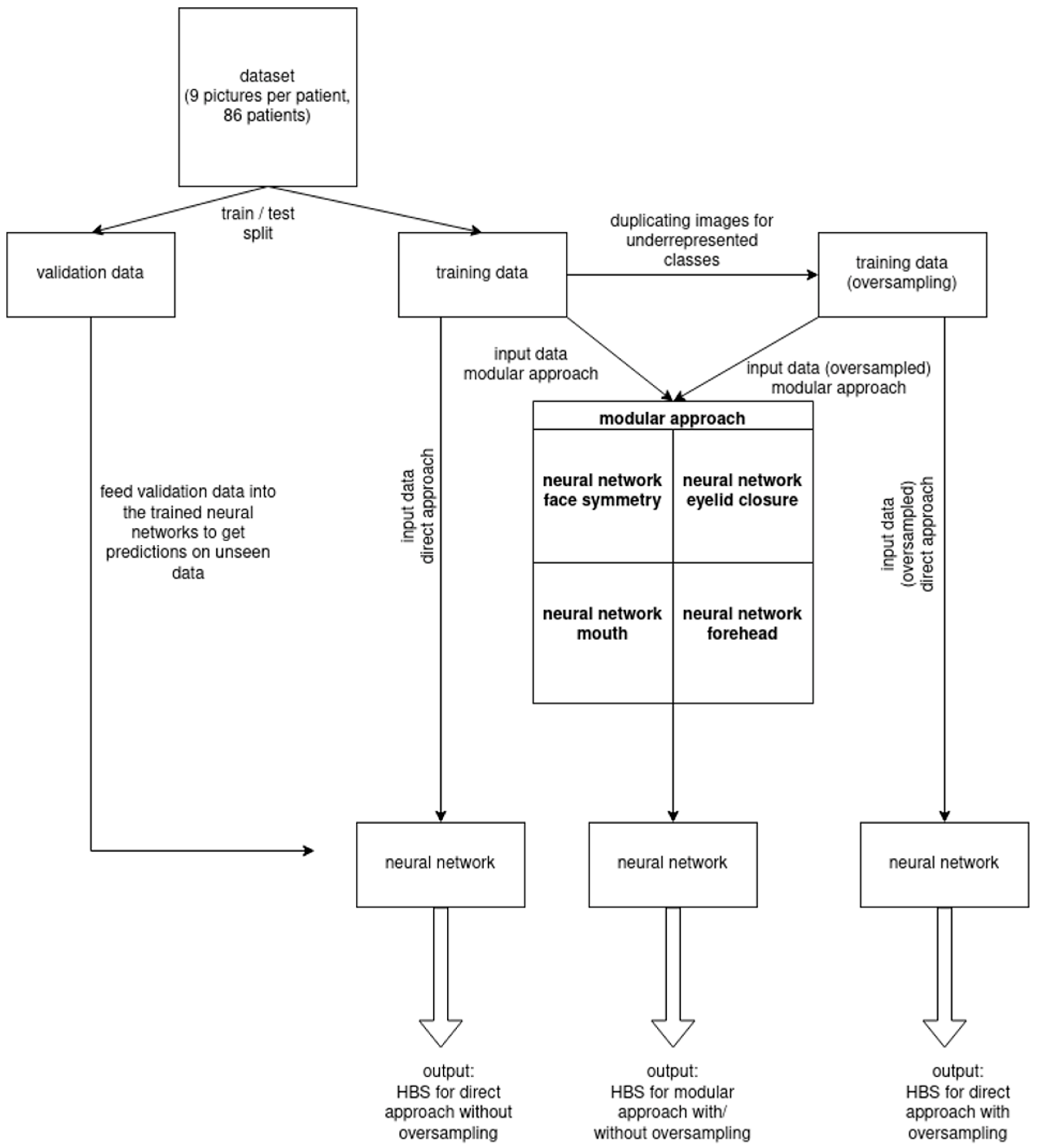
2. Materials and Methods
3. Results
3.1. Direct Classification Approach Yielded Significantly Enhanced Outcomes
3.2. Early Fusion Showed Refined Results
3.3. Oversampling Technique Did Not Influence Classification Performance of the Neural Networks
3.4. Combination of Early Fusion and Direct Form Yielded Optimized Classification Scores
3.5. External Databases and Recently Used- (LRU-) Caches Accelerated Runtimes
3.6. Direct Classification Approach Yielded Enhanced Outcomes
3.7. System Evaluation with F1-Score and Accuracy
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Steinhäuser, J.; Volk, G.F.; Thielker, J.; Geitner, M.; Kuttenreich, A.-M.; Klingner, C.M.; Dobel, C.; Guntinas-Lichius, O. Multidisciplinary Care of Patients with Facial Palsy: Treatment of 1220 Patients in a German Facial Nerve Center. JCM 2022, 11, 427. [Google Scholar] [CrossRef] [PubMed]
- Agostini, F. Idiopathic Facial Palsy: Umbrella Review of Systematic Reviews and Meta-Analyses. J. Biol. Regul. Homeost. Agents 2020, 34, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Rosson, G.; Redett, R. Facial Palsy: Anatomy, Etiology, Grading, and Surgical Treatment. J. Reconstr. Microsurg. 2008, 24, 379–389. [Google Scholar] [CrossRef]
- Heckmann, J.G.; Urban, P.P.; Pitz, S.; Guntinas-Lichius, O.; Gágyor, I. The Diagnosis and Treatment of Idiopathic Facial Paresis (Bell’s Palsy). Dtsch. Ärzteblatt Int. 2019, 41, 692. [Google Scholar] [CrossRef] [PubMed]
- Gilden, D.H. Bell’s Palsy. N. Engl. J. Med. 2004, 351, 1323–1331. [Google Scholar] [CrossRef]
- Morales, D.R.; Donnan, P.T.; Daly, F.; Staa, T.V.; Sullivan, F.M. Impact of Clinical Trial Findings on Bell’s Palsy Management in General Practice in the UK 2001–2012: Interrupted Time Series Regression Analysis. BMJ Open 2013, 3, e003121. [Google Scholar] [CrossRef]
- Pouwels, S.; Sanches, E.E.; Chaiet, S.R.; de Jongh, F.W.; Beurskens, C.H.G.; Monstrey, S.J.; Luijmes, R.E.; Siemann, I.; Ramnarain, D.; Marres, H.A.M.; et al. Association between Duration of Peripheral Facial Palsy, Severity, and Age of the Patient, and Psychological Distress. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 3048–3054. [Google Scholar] [CrossRef]
- Plumbaum, K.; Volk, G.F.; Boeger, D.; Buentzel, J.; Esser, D.; Steinbrecher, A.; Hoffmann, K.; Jecker, P.; Mueller, A.; Radtke, G.; et al. Inpatient Treatment of Patients with Acute Idiopathic Peripheral Facial Palsy: A Population-Based Healthcare Research Study. Clin. Otolaryngol. 2017, 42, 1267–1274. [Google Scholar] [CrossRef]
- Finsterer, J. Management of Peripheral Facial Nerve Palsy. Eur. Arch. Otorhinolaryngol. 2008, 265, 743–752. [Google Scholar] [CrossRef]
- Toulgoat, F.; Sarrazin, J.L.; Benoudiba, F.; Pereon, Y.; Auffray-Calvier, E.; Daumas-Duport, B.; Lintia-Gaultier, A.; Desal, H.A. Facial Nerve: From Anatomy to Pathology. Diagn. Interv. Imaging. 2013, 94, 1033–1042. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.Y.; Oh, D.J.; Park, B.; Choi, H.G. Bell’s palsy and obesity, alcohol consumption and smoking: A nested case-control study using a national health screening cohort. Sci. Rep. 2020, 10, 4248. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xu, L.; Luo, T.; Wu, F.; Zhao, B.; Li, X. The Etiology of Bell’s Palsy: A Review. J. Neurol. 2020, 267, 1896–1905. [Google Scholar] [CrossRef] [PubMed]
- Peitersen, E. Bell’s Palsy: The Spontaneous Course of 2,500 Peripheral Facial Nerve Palsies of Different Etiologies. Acta Otolaryngol Suppl 2002, 122, 4–30. [Google Scholar] [CrossRef]
- Vakharia, K.; Vakharia, K. Bell’s Palsy. Facial Plast. Surg. Clin. N. Am. 2016, 24, 1–10. [Google Scholar] [CrossRef]
- Kosins, A.M.; Hurvitz, K.A.; Evans, G.R.; Wirth, G.A. Facial Paralysis for the Plastic Surgeon. Can. J. Plast. Surg. 2007, 15, 77–82. [Google Scholar] [CrossRef]
- Bradbury, E.T.; Simons, W.; Sanders, R. Psychological and Social Factors in Reconstructive Surgery for Hemi-Facial Palsy. J. Plast. Reconstr. Aesthetic Surg. 2006, 59, 272–278. [Google Scholar] [CrossRef]
- Fu, L.; Patel, B.C. Lagophthalmos. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Jowett, N. A General Approach to Facial Palsy. Otolaryngol. Clin. N. Am. 2018, 51, 1019–1031. [Google Scholar] [CrossRef]
- Yanagihara, N.; Honda, N. Edematous Swelling of the Facial Nerve in Bell’s Palsy. Acta Oto-Laryngol. 2000, 120, 667–671. [Google Scholar] [CrossRef]
- Sullivan, F.M.; Swan, I.R.C.; Donnan, P.T.; Morrison, J.M.; Smith, B.H.; McKinstry, B.; Davenport, R.J.; Vale, L.D.; Clarkson, J.E.; Hammersley, V.; et al. Early Treatment with Prednisolone or Acyclovir in Bell’s Palsy. N. Engl. J. Med. 2007, 357, 1598–1607. [Google Scholar] [CrossRef]
- Prud’hon, S.; Kubis, N. La paralysie faciale périphérique a frigore. Rev. Médecine Interne 2019, 40, 28–37. [Google Scholar] [CrossRef]
- Benaim, J.L.; Amar, O.; Alliez, A.; Bertrand, B. Apport de l’électroneuromyographie dans les paralysies faciales périphériques. Ann. Chir. Plast. Esthétique 2015, 60, 374–376. [Google Scholar] [CrossRef]
- Guarin, D.L.; Yunusova, Y.; Taati, B.; Dusseldorp, J.R.; Mohan, S.; Tavares, J.; van Veen, M.M.; Fortier, E.; Hadlock, T.A.; Jowett, N. Toward an Automatic System for Computer-Aided Assessment in Facial Palsy. Facial Plast. Surg. Aesthetic Med. 2020, 22, 42–49. [Google Scholar] [CrossRef] [PubMed]
- House, J.W.; Brackmann, D.E. Facial Nerve Grading System. Otolaryngol. Head Neck Surg. 1985, 93, 146–147. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Lee, H.Y. Acute Peripheral Facial Palsy: Recent Guidelines and a Systematic Review of the Literature. J. Korean Med. Sci. 2020, 35, e245. [Google Scholar] [CrossRef] [PubMed]
- Banks, C.A.; Bhama, P.K.; Park, J.; Hadlock, C.R.; Hadlock, T.A. Clinician-Graded Electronic Facial Paralysis Assessment: The EFACE. Plast. Reconstr. Surg. 2015, 136, 223e–230e. [Google Scholar] [CrossRef] [PubMed]
- Greene, J.J.; Tavares, J.; Guarin, D.L.; Hadlock, T. Clinician and Automated Assessments of Facial Function Following Eyelid Weight Placement. JAMA Facial Plast. Surg. 2019, 21, 387–392. [Google Scholar] [CrossRef]
- Ahrens, A.; Skarada, D.; Wallace, M.; Cheung, J.Y.; Neely, J.G. Rapid Simultaneous Comparison System for Subjective Grading Scales Grading Scales for Facial Paralysis. Am. J. Otol. 1999, 20, 667–671. [Google Scholar]
- Cabrol, C.; Elarouti, L.; Montava, A.-L.; Jarze, S.; Mancini, J.; Lavieille, J.-P.; Barry, P.; Montava, M. Sunnybrook Facial Grading System: Intra-Rater and Inter-Rater Variabilities. Otol. Neurotol. 2021, 42, 1089–1094. [Google Scholar] [CrossRef]
- Mandlik, V.; Ruewe, M.; Engelmann, S.; Geis, S.; Taeger, C.; Kehrer, M.; Tamm, E.R.; Bleys, R.; Prantl, L.; Kehrer, A. Significance of the Marginal Mandibular Branch in Relation to Facial Palsy Reconstruction: Assessment of Microanatomy and Macroanatomy Including Axonal Load in 96 Facial Halves. Ann. Plast. Surg. 2019, 83, e43–e49. [Google Scholar] [CrossRef]
- Kehrer, A.; Engelmann, S.; Ruewe, M.; Geis, S.; Taeger, C.; Kehrer, M.; Prantl, L.; Tamm, E.; Bleys, R.R.L.A.W.; Mandlik, V. Anatomical Study of the Zygomatic and Buccal Branches of the Facial Nerve: Application to Facial Reanimation Procedures. Clin. Anat. 2019, 32, 480–488. [Google Scholar] [CrossRef]
- Zabojova, J.; Thrikutam, N.; Tolley, P.; Perez, J.; Rozen, S.M.; Rodriguez-Lorenzo, A. Relational Anatomy of the Mimetic Muscles and Its Implications on Free Functional Muscle Inset in Facial Reanimation. Ann. Plast. Surg. 2018, 81, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Baugh, R.F.; Basura, G.J.; Ishii, L.E.; Schwartz, S.R.; Drumheller, C.M.; Burkholder, R.; Deckard, N.A.; Dawson, C.; Driscoll, C.; Gillespie, M.B.; et al. Clinical Practice Guideline: Bell’s Palsy. Otolaryngol. Head Neck Surg. 2013, 149, S1–S27. [Google Scholar] [CrossRef]
- Salinas, R.A.; Alvarez, G.; Daly, F.; Ferreira, J. Corticosteroids for Bell’s Palsy (Idiopathic Facial Paralysis). Cochrane Database Syst. Rev. 2010, 7, CD001942. [Google Scholar]
- Linder, T.E.; Abdelkafy, W.; Cavero-Vanek, S. The Management of Peripheral Facial Nerve Palsy: “Paresis” Versus “Paralysis” and Sources of Ambiguity in Study Designs. Otol. Neurotol. 2010, 31, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Madhok, V.B.; Gagyor, I.; Daly, F.; Somasundara, D.; Sullivan, M.; Gammie, F.; Sullivan, F. Corticosteroids for Bell’s Palsy (Idiopathic Facial Paralysis). Cochrane Database Syst. Rev. 2016, 2016, CD001942. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, M.; Ito, T.; Furukawa, T.; Hirayama, A.; Kakehata, S. The Effect of Insulin-like Growth Factor 1 on the Recovery of Facial Nerve Function in a Guinea Pig Model of Facial Palsy. J. Physiol. Sci. 2020, 70, 28. [Google Scholar] [CrossRef]
- Miller, M.Q.; Hadlock, T.A. Beyond Botox: Contemporary Management of Nonflaccid Facial Palsy. Facial Plast. Surg. Aesthetic Med. 2020, 22, 65–70. [Google Scholar] [CrossRef]
- Labbé, D.; Bénichou, L.; Iodice, A.; Giot, J.-P. Signe du Depressor Anguli Oris (DAO) dans les parésies faciales. Comment le rechercher et libérer le sourire (note technique). Ann. Chir. Plast. Esthétique 2012, 57, 281–285. [Google Scholar] [CrossRef]
- Lafer, M.P.; March, T.M. Management of Long-Standing Flaccid Facial Palsy. Otolaryngol. Clin. N. Am. 2018, 51, 1141–1150. [Google Scholar] [CrossRef]
- Hontanilla, B.; Olivas-Menayo, J.; Marré, D.; Cabello, Á.; Aubá, C. Maximizing the Smile Symmetry in Facial Paralysis Reconstruction: An Algorithm Based on Twenty Years’ Experience. Facial Plast. Surg. 2021, 37, 360–369. [Google Scholar] [CrossRef]
- Garcia, R.M.; Hadlock, T.A.; Klebuc, M.J.; Simpson, R.L.; Zenn, M.R.; Marcus, J.R. Contemporary Solutions for the Treatment of Facial Nerve Paralysis. Plast. Reconstr. Surg. 2015, 135, 1025e–1046e. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Jeong, S.S.; Oh, T.S. Masseter Nerve-Based Facial Palsy Reconstruction. Arch. Craniofac. Surg. 2020, 21, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Jowett, N.; Hadlock, T.A. Free Gracilis Transfer and Static Facial Suspension for Midfacial Reanimation in Long-Standing Flaccid Facial Palsy. Otolaryngol. Clin. N. Am. 2018, 51, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.S.; Kim, H.B.; Choi, J.W.; Jeong, W.S. Facial Reanimation with Masseter Nerve–Innervated Free Gracilis Muscle Transfer in Established Facial Palsy Patients. Arch. Plast. Surg. 2019, 46, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Royall, D. Mobile Health: We’ve Got an App for That! Can. J. Diet. Pract. Res. 2014, 75, 4. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, B.; Bagala, M.; Creighton, A.; Leavey, T.; Nicholls, S.; Wood, C.; Longman, J.; Barker, J.; Pit, S. Mobile Phone Applications and Their Use in the Self-Management of Type 2 Diabetes Mellitus: A Qualitative Study among App Users and Non-App Users. Diabetol. Metab. Syndr. 2019, 11, 84. [Google Scholar] [CrossRef] [PubMed]
- Postel-Vinay, N.; Steichen, O.; Pébelier, E.; Persu, A.; Berra, E.; Bobrie, G.; Savard, S.; Nogueria, J.; Azizi, M. Home Blood Pressure Monitoring and E-Health: Investigation of Patients’ Experience with the Hy-Result System. Blood Press. Monit. 2020, 25, 155–161. [Google Scholar] [CrossRef]
- Vinny, P.; Takkar, A.; Lal, V.; Padma, M.; Sylaja, P.; Narasimhan, L.; Dwivedi, S.; Nair, P.; Iype, T.; Gupta, A.; et al. Mobile Application as a Complementary Tool for Differential Diagnosis in Neuro-Ophthalmology: A Multicenter Cross-Sectional Study. Indian J. Ophthalmol. 2021, 69, 1491. [Google Scholar] [CrossRef]
- Györi, E.; Mayrhofer, M.; Schwaiger, B.M.; Pona, I.; Tzou, C.H. Functional Results after Facial Reanimation in Iatrogenic Facial Palsy. Microsurgery 2020, 40, 145–153. [Google Scholar] [CrossRef]
- Bianchi, B.; Ferri, A.; Sesenna, E. Facial Reanimation after Nerve Sacrifice in the Treatment of Head and Neck Cancer. Curr. Opin. Otolaryngol. Head Neck Surg. 2012, 20, 114–119. [Google Scholar] [CrossRef]
- Humphrey, C.; Kriet, J. Nerve Repair and Cable Grafting for Facial Paralysis. Facial Plast. Surg. 2008, 24, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Vásquez, L.M.; Medel, R. Lagophthalmos after Facial Palsy: Current Therapeutic Options. Ophthalmic Res. 2014, 52, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Grixti, A.; Sadri, M.; Edgar, J.; Datta, A.V. Common Ocular Surface Disorders in Patients in Intensive Care Units. Ocul. Surf. 2012, 10, 26–42. [Google Scholar] [CrossRef] [PubMed]
- Nemet, A.Y.; Vinker, S. Considerations and Complications after Bells’ Palsy. J. Clin. Neurosci. 2015, 22, 1949–1953. [Google Scholar] [CrossRef]
- Riches, N.; Panagioti, M.; Alam, R.; Cheraghi-Sohi, S.; Campbell, S.; Esmail, A.; Bower, P. The Effectiveness of Electronic Differential Diagnoses (DDX) Generators: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0148991. [Google Scholar] [CrossRef]
- Szczepura, A.; Holliday, N.; Neville, C.; Johnson, K.; Khan, A.J.K.; Oxford, S.W.; Nduka, C. Raising the Digital Profile of Facial Palsy: National Surveys of Patients’ and Clinicians’ Experiences of Changing UK Treatment Pathways and Views on the Future Role of Digital Technology. J. Med. Internet Res. 2020, 22, e20406. [Google Scholar] [CrossRef]
- Petrides, G.; Clark, J.R.; Low, H.; Lovell, N.; Eviston, T.J. Three-Dimensional Scanners for Soft-Tissue Facial Assessment in Clinical Practice. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 605–614. [Google Scholar] [CrossRef]
- Taeger, J.; Bischoff, S.; Hagen, R.; Rak, K. Utilization of Smartphone Depth Mapping Cameras for App-Based Grading of Facial Movement Disorders: Development and Feasibility Study. JMIR Mhealth Uhealth 2021, 9, e19346. [Google Scholar] [CrossRef]
- Miller, M.Q.; Hadlock, T.A.; Fortier, E.; Guarin, D.L. The Auto-EFACE: Machine Learning–Enhanced Program Yields Automated Facial Palsy Assessment Tool. Plast. Reconstr. Surg. 2021, 147, 467–474. [Google Scholar] [CrossRef]
- Banks, C.A.; Jowett, N.; Azizzadeh, A.; Beurskens, C.; Bhama, P.; Borschel, G.; Coombs, C.; Coulson, S.; Croxon, G.; Diels, J.; et al. Worldwide Testing of the eFACE Facial Nerve Clinician-Graded Scale. Plast. Reconstr. Surg. 2017, 139, 491e–498e. [Google Scholar] [CrossRef]
- Dusseldorp, J.R.; van Veen, M.M.; Mohan, S.; Hadlock, T.A. Outcome Tracking in Facial Palsy. Otolaryngol. Clin. N. Am. 2018, 51, 1033–1050. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.N.; Susarla, S.M.; Hohman, M.H.; Henstrom, D.K.; Cheney, M.L.; Hadlock, T.A. A Comparison of Facial Nerve Grading Systems. Ann. Plast. Surg. 2013, 70, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Fuchigami, T.; Nakamura, K.; Kondo, E.; Sato, G.; Kitamura, Y.; Takeda, N. New Method to Evaluate Sequelae of Static Facial Asymmetry in Patients with Facial Palsy Using Three-Dimensional Scanning Analysis. Auris Nasus Larynx 2022, 49, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Horta, R.; Nascimento, R.; Gerós, A.; Aguiar, P.; Silva, A.; Amarante, J. A Novel System for Assessing Facial Muscle Movements: The Facegram 3D. Surg. Innov. 2018, 25, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Wu, J.; Zhong, W.; Wei, M.; Tong, J.; Yu, H.; Wang, L. Automatic Facial Paralysis Assessment via Computational Image Analysis. J. Healthc. Eng. 2020, 2020, 2398542. [Google Scholar] [CrossRef] [PubMed]
- Haase, D.; Minnigerode, L.; Volk, G.F.; Denzler, J.; Guntinas-Lichius, O. Automated and Objective Action Coding of Facial Expressions in Patients with Acute Facial Palsy. Eur. Arch. Otorhinolaryngol. 2015, 272, 1259–1267. [Google Scholar] [CrossRef]
- Meier-Gallati, V.; Scriba, H. Objective Assessment of the Reliability of the House–Brackmann and Fisch Grading of Synkinesis. Eur. Arch. Otorhinolaryngol. 2017, 274, 4217–4223. [Google Scholar] [CrossRef]
- Stoeckli, S.J.; Scriba, H.; Pollak, A.; Linder, T.E. Correlation of an Objective, Computer-Based System (OSCAR) with Subjective Evaluation Methods in the Assessment of Facial Motion in Patients with Bell’s Palsy. Otol. Neurotol. 2002, 23, S71–S72. [Google Scholar] [CrossRef]
- Tzou, C.-H.J.; Pona, I.; Placheta, E.; Hold, A.; Michaelidou, M.; Artner, N.; Kropatsch, W.; Gerber, H.; Frey, M. Evolution of the 3-Dimensional Video System for Facial Motion Analysis: Ten Years’ Experiences and Recent Developments. Ann. Plast. Surg. 2012, 69, 173–185. [Google Scholar] [CrossRef]
- Mothes, O.; Modersohn, L.; Volk, G.F.; Klingner, C.; Witte, O.W.; Schlattmann, P.; Denzler, J.; Guntinas-Lichius, O. Automated Objective and Marker-Free Facial Grading Using Photographs of Patients with Facial Palsy. Eur. Arch. Otorhinolaryngol. 2019, 276, 3335–3343. [Google Scholar] [CrossRef]
- Kim, H.; Kim, S.; Kim, Y.; Park, K. A Smartphone-Based Automatic Diagnosis System for Facial Nerve Palsy. Sensors 2015, 15, 26756–26768. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Soraghan, J.J.; O’Reilly, B.F.; Xing, D. Quantitative Analysis of Facial Paralysis Using Local Binary Patterns in Biomedical Videos. IEEE Trans. Biomed. Eng. 2009, 56, 1864–1870. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sajid, M.; Shafique, T.; Baig, M.; Riaz, I.; Amin, S.; Manzoor, S. Automatic Grading of Palsy Using Asymmetrical Facial Features: A Study Complemented by New Solutions. Symmetry 2018, 10, 242. [Google Scholar] [CrossRef]
- Brown, S.; Isaacson, B.; Kutz, W.; Barnett, S.; Rozen, S.M. Facial Nerve Trauma: Clinical Evaluation and Management Strategies. Plast. Reconstr. Surg. 2019, 143, 1498–1512. [Google Scholar] [CrossRef]
| (A) | Processing Method | Oversampling | F1-Score | Accuracy |
|---|---|---|---|---|
| Module form | sequential | no | 0.355 | 0.621 |
| yes | 0.330 | 0.600 | ||
| Early Fusion | no | 0.980 | 0.990 | |
| yes | 0.967 | 0.983 | ||
| Late Fusion | no | 0.817 | 0.900 | |
| yes | 0.808 | 0.895 |
| (B) | Processing Method | Oversampling | F1-Score | Accuracy |
|---|---|---|---|---|
| Direct form | sequential | no | 0.884 | 0.942 |
| yes | 0.914 | 0.968 | ||
| Early Fusion | no | 1.000 | 1.000 | |
| yes | 1.000 | 1.000 | ||
| Late Fusion | no | 0.895 | 0.964 | |
| yes | 0.927 | 0.963 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knoedler, L.; Baecher, H.; Kauke-Navarro, M.; Prantl, L.; Machens, H.-G.; Scheuermann, P.; Palm, C.; Baumann, R.; Kehrer, A.; Panayi, A.C.; et al. Towards a Reliable and Rapid Automated Grading System in Facial Palsy Patients: Facial Palsy Surgery Meets Computer Science. J. Clin. Med. 2022, 11, 4998. https://doi.org/10.3390/jcm11174998
Knoedler L, Baecher H, Kauke-Navarro M, Prantl L, Machens H-G, Scheuermann P, Palm C, Baumann R, Kehrer A, Panayi AC, et al. Towards a Reliable and Rapid Automated Grading System in Facial Palsy Patients: Facial Palsy Surgery Meets Computer Science. Journal of Clinical Medicine. 2022; 11(17):4998. https://doi.org/10.3390/jcm11174998
Chicago/Turabian StyleKnoedler, Leonard, Helena Baecher, Martin Kauke-Navarro, Lukas Prantl, Hans-Günther Machens, Philipp Scheuermann, Christoph Palm, Raphael Baumann, Andreas Kehrer, Adriana C. Panayi, and et al. 2022. "Towards a Reliable and Rapid Automated Grading System in Facial Palsy Patients: Facial Palsy Surgery Meets Computer Science" Journal of Clinical Medicine 11, no. 17: 4998. https://doi.org/10.3390/jcm11174998
APA StyleKnoedler, L., Baecher, H., Kauke-Navarro, M., Prantl, L., Machens, H.-G., Scheuermann, P., Palm, C., Baumann, R., Kehrer, A., Panayi, A. C., & Knoedler, S. (2022). Towards a Reliable and Rapid Automated Grading System in Facial Palsy Patients: Facial Palsy Surgery Meets Computer Science. Journal of Clinical Medicine, 11(17), 4998. https://doi.org/10.3390/jcm11174998








