Potential Add-On Effects of Manual Therapy Techniques in Migraine Patients: A Randomised Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Randomisation, Blinding and Masking
2.4. Assessments
2.5. Intervention Procedures
2.6. Statistics
2.7. Sample Size Calculation
3. Results
3.1. Participants
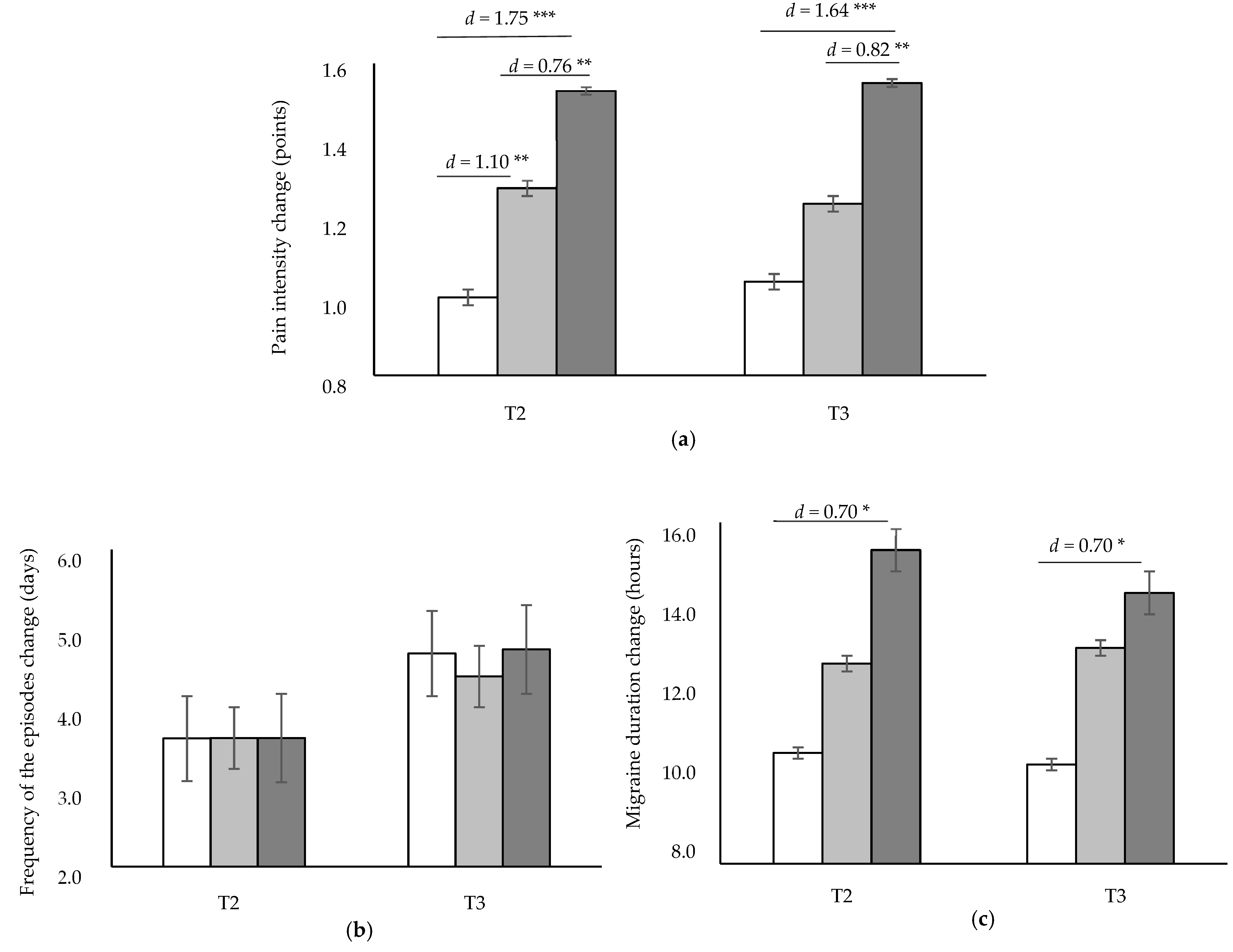
3.2. Effectiveness of the Interventions on Pain Intensity, Frequency of the Episodes and Migraine Duration
3.3. Effectiveness of the Interventions on Migraine Disability and Impact
3.4. Effectiveness of the Interventions on Depression and Anxiety Levels
3.5. Effectiveness of Interventions on Patient Global Impression of Change
3.6. Subgroup Analysis by Diagnosis
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef] [Green Version]
- Steiner, T.J.; Stovner, L.J.; Vos, T.; Jensen, R.; Katsarava, Z. Migraine is first cause of disability in under 50s: Will health politicians now take notice? J. Headache Pain 2018, 19, 17. [Google Scholar] [CrossRef] [PubMed]
- Louter, M.A.; Pijpers, J.A.; Wardenaar, K.J.; van Zwet, E.W.; van Hemert, A.M.; Zitman, F.G.; Ferrari, M.; Penninx, B.; Terwindt, G. Symptom dimensions of affective disorders in migraine patients. J. Psychosom. Res. 2015, 79, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Kong, F.; Buse, D.C. Predictors of episodic migraine transformation to chronic migraine: A systematic review and meta-analysis of observational cohort studies. Cephalalgia 2020, 40, 503–516. [Google Scholar] [CrossRef]
- Buse, D.C.; Greisman, J.D.; Baigi, K.; Lipton, R.B. Migraine Progression: A Systematic Review. Headache 2019, 59, 306–338. [Google Scholar] [CrossRef]
- May, A.; Schulte, L.H. Chronic migraine: Risk factors, mechanisms and treatment. Nat. Rev. Neurol. 2016, 12, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Peres, M.F.P.; Mercante, J.P.P.; Tobo, P.R.; Kamei, H.; Bigal, M.E. Anxiety and depression symptoms and migraine: A symptom-based approach research. J. Headache Pain 2017, 18, 37. [Google Scholar] [CrossRef] [Green Version]
- Puledda, F.; Shields, K. Non-Pharmacological Approaches for Migraine. Neurotherapeutics 2018, 15, 336–345. [Google Scholar] [CrossRef] [Green Version]
- Moore, C.S.; Sibbritt, D.W.; Adams, J. A critical review of manual therapy use for headache disorders: Prevalence, profiles, motivations, communication and self-reported effectiveness. BMC Neurol. 2017, 17, 61. [Google Scholar] [CrossRef] [Green Version]
- Bialosky, J.E.; Bishop, M.D.; Price, D.D.; Robinson, M.E.; George, S.Z. The mechanisms of manual therapy in the treatment of musculoskeletal pain: A comprehensive model. Man Ther. 2009, 14, 531–538. [Google Scholar] [CrossRef] [Green Version]
- Haller, H.; Lauche, R.; Sundberg, T.; Dobos, G.; Cramer, H. Craniosacral therapy for chronic pain: A systematic review and meta-analysis of randomized controlled trials. BMC Musculoskelet. Disord. 2019, 21, 1. [Google Scholar] [CrossRef] [PubMed]
- Cutler, M.; Holland, B.; Stupski, B.; Gamber, R.; Smith, M. Cranial manipulation can alter sleep latency and sympathetic nerve activity in humans: A pilot study. J. Altern. Complement. Med. 2005, 11, 103–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miana, L.; Bastos, V.H.D.V.; Machado, S.; Arias-Carrión, O.; Nardi, A.E.; Almeida, L.; Ribeiro, P.; Machado, D.; King, H.; Silva, J.G. Changes in alpha band activity associated with application of the compression of fourth ventricular (CV-4) osteopathic procedure: A qEEG pilot study. J. Bodyw. Mov. Ther. 2013, 17, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Espí-López, G.V.; Ruescas-Nicolau, M.A.; Nova-Redondo, C.; Benítez-Martínez, J.C.; Dugailly, P.M.; Falla, D. Effect of Soft Tissue Techniques on Headache Impact, Disability, and Quality of Life in Migraine Sufferers: A Pilot Study. J. Altern. Complement. Med. 2018, 24, 1099–1107. [Google Scholar] [CrossRef]
- Arnadottir, T.S.; Sigurdardottir, A.K. Is craniosacral therapy effective for migraine? Tested with HIT-6 Questionnaire. Complement. Ther. Clin. Pract. 2013, 19, 11–14. [Google Scholar] [CrossRef]
- Muñoz-Gómez, E.; Inglés, M.; Aguilar-Rodríguez, M.; Mollà-Casanova, S.; Sempere-Rubio, N.; Serra-Añó, P.; Espí-López, G.V. Effect of a Craniosacral Therapy Protocol in People with Migraine: A Randomized Controlled Trial. J. Clin. Med. 2022, 11, 759. [Google Scholar] [CrossRef]
- Voogt, L.; de Vries, J.; Meeus, M.; Struyf, F.; Meuffels, D.; Nijs, J. Analgesic effects of manual therapy in patients with musculoskeletal pain: A systematic review. Man Ther. 2015, 20, 250–256. [Google Scholar] [CrossRef] [Green Version]
- Wirth, B.; Gassner, A.; de Bruin, E.D.; Axén, I.; Swanenburg, J.; Humphreys, B.K.; Schweinhardt, P. Neurophysiological Effects of High Velocity and Low Amplitude Spinal Manipulation in Symptomatic and Asymptomatic Humans: A Systematic Literature Review. Spine (Phila Pa 1976) 2019, 44, E914–E926. [Google Scholar] [CrossRef]
- Chaibi, A.; Benth, J.Š.; Tuchin, P.J.; Russell, M.B. Chiropractic spinal manipulative therapy for migraine: A three-armed, single-blinded, placebo, randomized controlled trial. Eur. J. Neurol. 2017, 24, 143–153. [Google Scholar] [CrossRef] [Green Version]
- Muñoz-Gómez, E.; Inglés, M.; Serra-Añó, P.; Espí-López, G.V. Effectiveness of a manual therapy protocol based on articulatory techniques in migraine patients. A randomized controlled trial. Musculoskelet. Sci. Pract. 2021, 54, 102386. [Google Scholar] [CrossRef]
- Bevilaqua-Grossi, D.; Gonçalves, M.C.; Carvalho, G.F.; Florencio, L.L.; Dach, F.; Speciali, J.G.; Bigal, M.E.; Chaves, T.C. Additional Effects of a Physical Therapy Protocol on Headache Frequency, Pressure Pain Threshold, and Improvement Perception in Patients With Migraine and Associated Neck Pain: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2016, 97, 866–874. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.; Geroin, C.; Valè, N.; Marchioretto, F.; Turrina, A.; Dimitrova, E.; Tamburin, S.; Serina, A.; Castellazzi, P.; Meschieri, A.; et al. Does myofascial and trigger point treatment reduce pain and analgesic intake in patients undergoing onabotulinumtoxin A injection due to chronic intractable migraine? Eur. J. Phys. Rehabil. Med. 2018, 54, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Voigt, K.; Liebnitzky, J.; Burmeister, U.; Sihvonen-Riemenschneider, H.; Beck, M.; Voigt, R.; Bergmann, A. Efficacy of osteopathic manipulative treatment of female patients with migraine: Results of a randomized controlled trial. J. Altern. Complement. Med. 2011, 17, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, G.F.; Schwarz, A.; Szikszay, T.M.; Adamczyk, W.M.; Bevilaqua-Grossi, D.; Luedtke, K. Physical therapy and migraine: Musculoskeletal and balance dysfunctions and their relevance for clinical practice. Braz. J. Phys. Ther. 2020, 24, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Luedtke, K.; Starke, W.; May, A. Musculoskeletal dysfunction in migraine patients. Cephalalgia 2018, 38, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Bishop, M.; Mintken, P.; Bialosky, J.; Cleland, J. Patient Expectations of Benefit From Interventions for Neck Pain and Resulting Influence on Outcomes. J. Orthop. Sports Phys. Ther. 2013, 43, 457–465. [Google Scholar] [CrossRef] [Green Version]
- Bishop, M.D.; Bialosky, J.E.; Cleland, J.A. Patient expectations of benefit from common interventions for low back pain and effects on outcome: Secondary analysis of a clinical trial of manual therapy interventions. J Man Manip Ther. 2011, 19, 20–25. [Google Scholar] [CrossRef] [Green Version]
- Bishop, M.D.; Mintken, P.; Bialosky, J.E.; Cleland, J.A. Factors shaping expectations for complete relief from symptoms during rehabilitation for patients with spine pain. Physiother. Theory Pract. 2019, 35, 70–79. [Google Scholar] [CrossRef]
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018, 38, 1–211. [CrossRef]
- Stewart, W.; Lipton, R.; White, J.; Dowson, A.; Kolodner, K.; Liberman, J.; Sawyer, J. An international study to assess reliability of the Migraine Disability Assessment (MIDAS) score. Neurology 1999, 22, 988–994. [Google Scholar] [CrossRef]
- Kelly, A. The minimum clinically significant difference in visual analogue scale pain score does not differ with severity of pain. Emerg. Med. J. 2001, 18, 205–207. [Google Scholar] [CrossRef] [Green Version]
- Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Diener, H.C.; Lipton, R.B.; Aurora, S.K.; Nolan, M.E.; Silberstein, S.D. Assessing clinically meaningful treatment effects in controlled trials: Chronic migraine as an example. J. Pain 2015, 16, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, G.F.; Luedtke, K.; Braun, T. Minimal important change and responsiveness of the Migraine Disability Assessment Score (MIDAS) questionnaire. J. Headache Pain 2021, 22, 126. [Google Scholar] [CrossRef] [PubMed]
- Stewart, W.; Lipton, R.; Dowson, A.; Sawyer, J. Development and testing of the migraine disability assessment (MIDAS) questionnaire to assess headache-related disability. Neurology 2001, 56 (Suppl. 1), S20–S28. [Google Scholar] [CrossRef] [PubMed]
- Kawata, A.; Coeytaux, R.; DeVellis, R.; Kahn, K. Psychometric properties of the HIT-6 among patients in a headache-specialty practice. Headache 2005, 45, 638–643. [Google Scholar] [CrossRef]
- Coeytaux, R.R.; Kaufman, J.S.; Chao, R.; Mann, J.D.; DeVellis, R.F. Four methods of estimating the minimal important difference score were compared to establish a clinically significant change in Headache Impact Test. J. Clin. Epidemiol. 2006, 59, 374–380. [Google Scholar] [CrossRef]
- Rendas, R.; Yang, M.; Varon, S.; DeGryse, R.; Kosinski, M. Validation of the Headache Impact Test (HIT-6) in patients with chronic migraine. Health Qual. Life Outcomes 2014, 12, 117. [Google Scholar] [CrossRef] [Green Version]
- Beck, A.; Steer, R.; Brown, G. Manual for the Beck Depression Inventory-II; Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Dworkin, R.H.; Turk, D.C.; Wyrwich, K.W.; Beaton, D.; Cleeland, C.S.; Farrar, J.T.; Haythornthwaite, J.A.; Jensen, M.P.; Kerns, R.D.; Ader, D.N.; et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J. Pain 2008, 9, 105–121. [Google Scholar] [CrossRef]
- Wang, Y.P.; Gorenstein, C. Psychometric properties of the Beck Depression Inventory-II: A comprehensive review. Braz. J. Psychiatry 2013, 35, 416–431. [Google Scholar] [CrossRef] [Green Version]
- Spielberger, C.; Gorsuch, R.; Lushene, R.; Vagg, P.; Jacobs, G. Manual for State-Trait Anxiety Inventory; Consulting Psychologist Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Taghizadeh, N.; Tremblay, A.; Cressman, S.; Peacock, S.; McWilliams, A.M.; MacEachern, P.; Johnston, M.R.; Goffin, J.; Goss, G.; Nicholas, G.; et al. Health-related quality of life and anxiety in the PAN-CAN lung cancer screening cohort. BMJ Open 2019, 9, e024719. [Google Scholar] [CrossRef]
- Guy, W. ECDEU Assessment Manual for Psychopharmacology; U.S. Dept. of Health, Education, and Welfare, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute of Mental Health, Psychopharmacology Research Branch, Division of Extramural Research Programs: Rockville, MD, USA, 1976.
- Perrot, S.; Lantéri-Minet, M. Patients’ Global Impression of Change in the management of peripheral neuropathic pain: Clinical relevance and correlations in daily practice. Eur. J. Pain 2019, 23, 1117–1128. [Google Scholar] [CrossRef] [PubMed]
- Kamper, S.J.; Maher, C.G.; Mackay, G. Global rating of change scales: A review of strengths and weaknesses and considerations for design. J. Man Manip. Ther. 2009, 17, 163–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Côté, P.; Kreitz, B.; Cassidy, J.; Thiel, H. The validity of the extension-rotation test as a clinical screening procedure before neck manipulation: A secondary analysis. J. Manip. Physiol. Ther. 1996, 19, 159–164. [Google Scholar]
- Chaibi, A.; Russell, M.B. A risk-benefit assessment strategy to exclude cervical artery dissection in spinal manual-therapy: A comprehensive review. Ann. Med. 2019, 51, 118–127. [Google Scholar] [CrossRef] [Green Version]
- Thalheimer, W.; Cook, S. How to calculate effect sizes from published research: A simplified methodology. Work-Learn. Res. 2002, 1, 1–9. [Google Scholar]
- Tassorelli, C.; Tramontano, M.; Berlangieri, M.; Schweiger, V.; D’Ippolito, M.; Palmerini, V.; Bonazza, S.; Rosa, R.; Cerbo, R.; Buzzi, M.G. Assessing and treating primary headaches and cranio-facial pain in patients undergoing rehabilitation for neurological diseases. J. Headache Pain 2017, 18, 99. [Google Scholar]
- Magnusson, J.E.; Becker, W.J. Migraine Frequency and Intensity: Relationship with Disability and Psychological Factors. Headache 2003, 43, 1049–1059. [Google Scholar] [CrossRef]
- Bialosky, J.E.; Beneciuk, J.M.; Bishop, M.D.; Coronado, R.A.; Penza, C.W.; Simon, C.B.; George, S.Z. Unraveling the Mechanisms of Manual Therapy: Modeling an Approach. J. Orthop. Sports Phys. Ther. 2018, 48, 8–18. [Google Scholar] [CrossRef]
- Ghanbari, A.; Askarzadeh, S.; Petramfar, P.; Mohamadi, M. Migraine responds better to a combination of medical therapy and trigger point management than routine medical therapy alone. NeuroRehabilitation 2015, 37, 157–163. [Google Scholar] [CrossRef]
- Lascurain-Aguirrebeña, I.; Newham, D.; Critchley, D.J. Mechanism of Action of Spinal Mobilizations: A Systematic Review. Spine 2016, 41, 159–172. [Google Scholar] [CrossRef]
- Luedtke, K.; Basener, A.; Bedei, S.; Castien, R.; Chaibi, A.; Falla, D.; Fernández-De-Las-Peñas, C.; Gustafsson, M.; Hall, T.; Jull, G.; et al. Outcome measures for assessing the effectiveness of non-pharmacological interventions in frequent episodic or chronic migraine: A Delphi study. BMJ Open 2020, 10, e029855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pradeep, R.; Nemichandra, S.C.; Harsha, S.; Radhika, K. Migraine Disability, Quality of Life, and Its Predictors. Ann. Neurosci. 2020, 27, 18–23. [Google Scholar]
- Cerritelli, F.; Ginevri, L.; Messi, G.; Caprari, E.; Di Vincenzo, M.; Renzetti, C.; Cozzolino, V.; Barlafante, G.; Foschi, N.; Provinciali, L. Clinical effectiveness of osteopathic treatment in chronic migraine: 3-Armed randomized controlled trial. Complement. Ther. Med. 2015, 23, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Wanderley, D.; Valença, M.M.; de Souza Costa Neto, J.J.; Martins, J.V.; Raposo, M.C.F.; de Oliveira, D.A. Contract-relax technique compared to static stretching in treating migraine in women: A randomized pilot trial. J. Bodyw. Mov. Ther. 2019, 24, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Tuchin, P.J.; Pollard, H.; Bonello, R. A randomized controlled trial of chiropractic spinal manipulative therapy for migraine. J. Manip. Physiol. Ther. 2000, 23, 91–95. [Google Scholar] [CrossRef]
- Rist, P.M.; Hernandez, A.; Bernstein, C.; Kowalski, M.; Osypiuk, K.; Vining, R.; Long, C.R.; Goertz, C.; Song, R.; Wayne, P.M. The Impact of Spinal Manipulation on Migraine Pain and Disability: A Systematic Review and Meta-Analysis. Headache 2019, 59, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Espí-López, G.; López-Bueno, L.; Vicente-Herrero, M.; Martínez-Arnau, F. Efficacy of manual therapy on anxiety and depression in patients with tension-type headache. A randomized controlled clinical trial. Int. J. Ost. Med. 2016, 22, 11–20. [Google Scholar] [CrossRef]
- Diener, H.C.; Tassorelli, C.; Dodick, D.W.; Silberstein, S.D.; Lipton, R.B.; Ashina, M.; Becker, W.J.; Ferrari, M.D.; Goadsby, P.J.; Pozo-Rosich, P.; et al. Guidelines of the International Headache Society for controlled trials of acute treatment of migraine attacks in adults: Fourth edition. Cephalalgia 2019, 39, 687–710. [Google Scholar] [CrossRef] [Green Version]
- Beier, D.; Callesen, H.E.; Carlsen, L.N.; Birkefoss, K.; Tómasdóttir, H.; Wűrtzen, H.; Høst, C.V.; Hansen, J.M. Manual joint mobilisation techniques, supervised physical activity, psychological treatment, acupuncture and patient education in migraine treatment. A systematic review and meta-analysis. Cephalalgia 2021, 22, 3331024211034489. [Google Scholar] [CrossRef]

| STG n = 25 | AG n = 25 | STAG n = 25 | Total n = 75 | |
|---|---|---|---|---|
| Medication | ||||
| Preventive | 2 (8%) | 8 (32%) | 9 (36%) | 19 (25.33%) |
| Symptomatic | 23 (92%) | 16 (64%) | 16 (64%) | 55 (73.33%) |
| None | 0 (0%) | 1 (4%) | 0 (0%) | 1 (1.33%) |
| Migraine duration (hours) | 24.24 (24.62) | 23.20 (20.28) | 24.88 (15.73) | 24.11 (20.26) |
| Pain intensity (MIDAS) | 7.28 (1.09) | 7.40 (1.13) | 7.56 (1.10) | 7.41 (1.10) |
| Frequency of occurrence (MIDAS) | 25.08 (11.02) | 24.84 (12.26) | 24.48 (11.41) | 24.8 (11.42) |
| Migraine Disability (MIDAS) | 35.76 (21.06) | 34.64 (20.23) | 35.72 (15.84) | 35.37 (18.93) |
| Migraine Impact (HIT-6) | 63.44 (4.34) | 64.44 (6.49) | 64.00 (4.7) | 63.96 (5.21) |
| Depression (BDI-II) | 7.68 (6.96) | 8.64 (7.92) | 8.00 (5.21) | 8.11 (6.71) |
| State-Anxiety (STAI-state) | 40.84 (13.38) | 43.76 (13.94) | 43.76 (12.32) | 42.79 (13.12) |
| Trait-Anxiety (STAI-trait) | 38.64 (12.46) | 40.32 (12.85) | 40.00 (12.57) | 39.65 (12.48) |
| Anxiety (STAI total) | 39.74 (12.44) | 42.04 (13.13) | 41.88 (11.85) | 41.22 (12.36) |
| Change T2–T1 | Change T3–T1 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| STG | AG | STAG | STG vs. STAG | AG vs. STAG | STG vs. AG | STG | AG | STAG | STG vs. STAG | AG vs. STAG | STG vs. AG | |
| Migraine Disability (MIDAS) | 6.00 (3.28) | 4.48 (2.71) | 5.68 (2.93) | d = −0.10 | d = 0.43 | d = −0.51 | 12.00 (8.58) | 10.36 (7.11) | 12.76 (6.97) | d = 0.10 | d = 0.34 | d = −0.21 |
| Migraine Impact (HIT-6) | 7.75 (7.96) | 8.48 (4.02) | 9.68 (4.83) | d = 0.30 | d = 0.27 | d = 0.12 | 6.71 (5.83) | 7.56 (4.44) | 8.80 (5.39) | d = 0.37 | d = 0.25 | d = 0.17 |
| Depression (BDI-II) | 2.38 (9.61) | 1.88 (3.60) | 2.52 (3.51) | d = 0.02 | d = 0.18 | d = −0.08 | 1.75 (4.67) | 0.83 (4.64) | 1.52 (4.00) | d = −0.05 | d = 0.16 | d = −0.20 |
| State-Anxiety (STAI-state) | 5.23 (16.44) | 2.25 (10.33) | 5.36 (5.50) | d = 0.01 | d = 0.39 | d = −0.22 | 2.77 (7.98) | 3.67 (9.36) | 5.16 (5.91) | d = 0.34 | d = 0.20 | d = 0.10 |
| Trait-Anxiety (STAI-trait) | 5.19 (12.13) | 4.16 (5.19) | 4.40 (8.71) | d = −0.08 | d = 0.03 | d = −0.12 | 5.19 (6.47) | 4.56 (6.25) | 4.16 (8.48) | d = −0.14 | d = −0.05 | d = −0.10 |
| Anxiety (STAI total) | 4.13 (16.09) | 3.06 (6.90) | 4.88 (6.41) | d = 0.07 | d = 0.27 | d = −0.09 | 3.87 (5.67) | 3.77 (5.98) | 4.66 (6.68) | d = 0.13 | d = 0.14 | d = −0.02 |
| T2 | T3 | |||||
|---|---|---|---|---|---|---|
| STG | AG | STAG | STG | AG | STAG | |
| Patient global impression of change (PGICS) | ||||||
| No change | 7 (28) | 3 (12) | 1 (4) | 8 (32) | 6 (24) | 1 (4) |
| Small improvement | 5 (20) | 9 (36) | 4 (16) | 14 (56) | 14 (56) | 12 (48) |
| Moderate improvement | 11 (44) | 12 (48) | 18 (72) | 3 (12) ** | 5 (20) * | 12 (48) |
| Large improvement | 2 (8) | 1 (4) | 2 (8) | 0 (0) | 0 (0) | 0 (0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Gómez, E.; Serra-Añó, P.; Mollà-Casanova, S.; Sempere-Rubio, N.; Aguilar-Rodríguez, M.; Espí-López, G.V.; Inglés, M. Potential Add-On Effects of Manual Therapy Techniques in Migraine Patients: A Randomised Controlled Trial. J. Clin. Med. 2022, 11, 4686. https://doi.org/10.3390/jcm11164686
Muñoz-Gómez E, Serra-Añó P, Mollà-Casanova S, Sempere-Rubio N, Aguilar-Rodríguez M, Espí-López GV, Inglés M. Potential Add-On Effects of Manual Therapy Techniques in Migraine Patients: A Randomised Controlled Trial. Journal of Clinical Medicine. 2022; 11(16):4686. https://doi.org/10.3390/jcm11164686
Chicago/Turabian StyleMuñoz-Gómez, Elena, Pilar Serra-Añó, Sara Mollà-Casanova, Núria Sempere-Rubio, Marta Aguilar-Rodríguez, Gemma V. Espí-López, and Marta Inglés. 2022. "Potential Add-On Effects of Manual Therapy Techniques in Migraine Patients: A Randomised Controlled Trial" Journal of Clinical Medicine 11, no. 16: 4686. https://doi.org/10.3390/jcm11164686
APA StyleMuñoz-Gómez, E., Serra-Añó, P., Mollà-Casanova, S., Sempere-Rubio, N., Aguilar-Rodríguez, M., Espí-López, G. V., & Inglés, M. (2022). Potential Add-On Effects of Manual Therapy Techniques in Migraine Patients: A Randomised Controlled Trial. Journal of Clinical Medicine, 11(16), 4686. https://doi.org/10.3390/jcm11164686








