Long-Term Experience with Balloon Dilation for Short Bulbar and Membranous Urethral Strictures: Establishing a Baseline in the Active Drug Treatment Era
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Complications
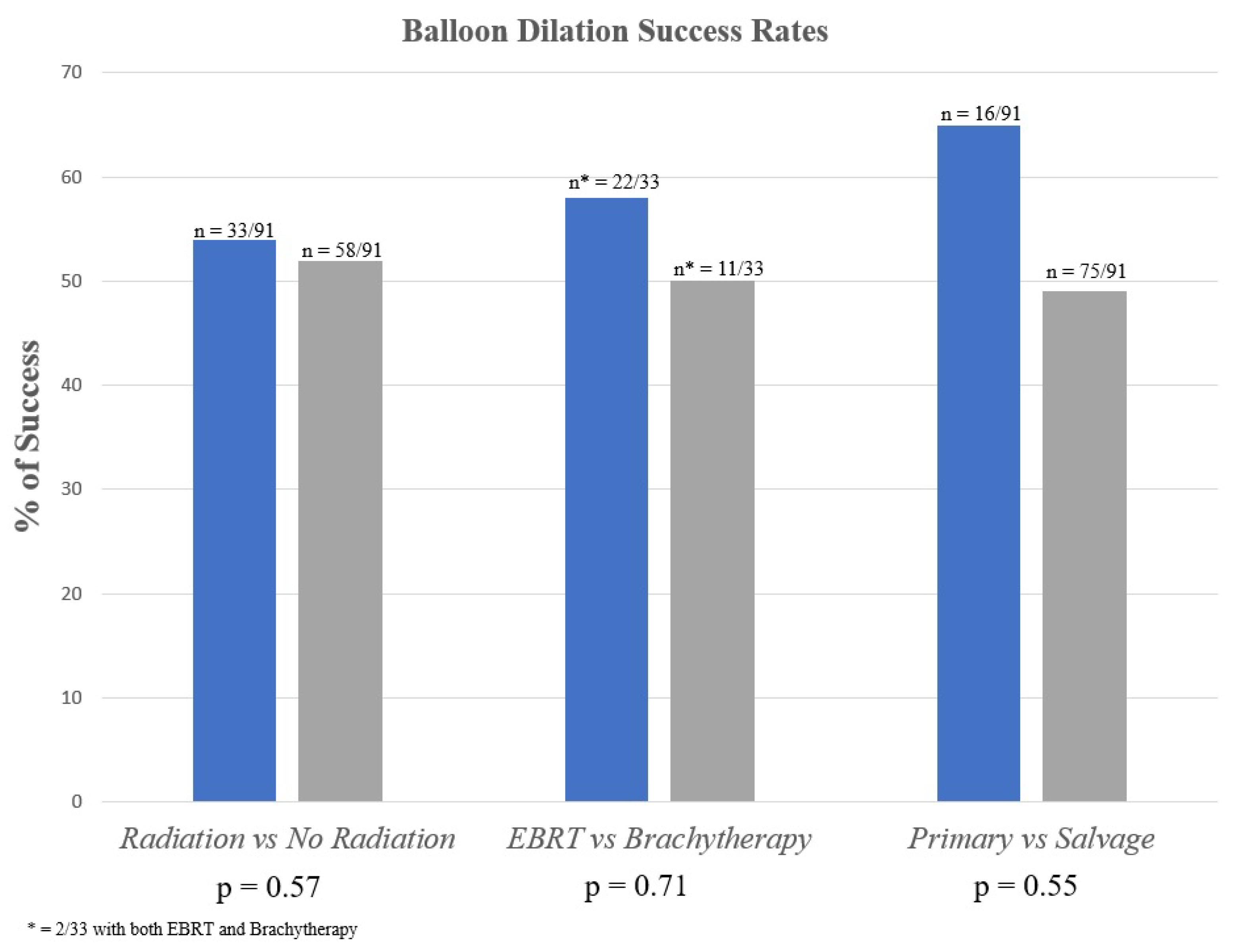
3.2. Irradiated vs. Non-Irradiated Patients
3.3. Primary vs. Salvage Patients
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Verla, W.; Oosterlinck, W.; Spinoit, A.F.; Waterloos, M. A Comprehensive Review Emphasizing Anatomy, Etiology, Diagnosis, and Treatment of Male Urethral Stricture Disease. Biomed Res. Int. 2019, 2019, 9046430. [Google Scholar] [CrossRef] [PubMed]
- Santucci, R.A.; Joyce, G.F.; Wise, M. Male urethral stricture disease. J. Urol. 2007, 177, 1667–1674. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.; Blakely, S.; Nikolavsky, D. Advancements in transurethral management of urethral stricture disease. Curr. Opin. Urol. 2021, 31, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.S.; Hofer, M.D.; Oberlin, D.T.; Milose, J.; Flury, S.C.; Morey, A.F.; Gonzalez, C.M. Practice Patterns in the Treatment of Urethral Stricture Among American Urologists: A Paradigm Change? Urology 2015, 86, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Hudak, S.J.; Atkinson, T.H.; Morey, A.F. Repeat transurethral manipulation of bulbar urethral strictures is associated with increased stricture complexity and prolonged disease duration. J. Urol. 2012, 187, 1691–1695. [Google Scholar] [CrossRef] [PubMed]
- Wessells, H.; Angermeier, K.W.; Elliott, S.; Gonzalez, C.M.; Kodama, R.; Peterson, A.C.; Reston, J.; Rourke, K.; Stoffel, J.; Vanni, A.J.; et al. Male Urethral Stricture: American Urological Association Guideline. J. Urol. 2017, 197, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Virasoro, R.; Delong, J.M.; Mann, R.A.; Estrella, R.E.; Pichardo, M.; Lay, R.R.; Espino, G.; Roth, J.D.; Elliott, S.P. A drug-coated balloon treatment for urethral stricture disease: Interim results from the ROBUST I study. Can. Urol. Assoc. J. 2020, 14, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.A.; Virasoro, R.; DeLong, J.M.; Estrella, R.E.; Pichardo, M.; Lay, R.R.; Espino, G.; Roth, J.D.; Elliott, S.P. A drug-coated balloon treatment for urethral stricture disease: Two-year results from the ROBUST I Study. Can. Urol. Assoc. J. 2021, 15, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.A.; Rozanski, A.T.; Shakir, N.A.; Viers, B.R.; Ward, E.E.; Bergeson, R.L.; Morey, A.F. Balloon dilation performs poorly as a salvage management strategy for recurrent bulbar urethral strictures following failed urethroplasty. Transl. Androl. Urol. 2020, 9, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Gelman, J.; Liss, M.A.; Cinman, N.M. Direct vision balloon dilation for the management of urethral strictures. J. Endourol. 2011, 25, 1249–1251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, S.-C.; Wu, H.-Y.; Wang, W.; Xu, L.-W.; Ding, G.-Q.; Zhang, Z.-G.; Li, G.-H. High-pressure balloon dilation for male anterior urethral stricture: Single-center experience. J. Zhejiang Univ. Sci. B 2016, 17, 722–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammed, N.; Kestin, L.; Ghilezan, M.; Krauss, D.; Vicini, F.; Brabbins, D.; Gustafson, G.; Ye, H.; Martinez, A. Comparison of acute and late toxicities for three modern high-dose radiation treatment techniques for localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Hindson, B.R.; Millar, J.L.; Matheson, B. Urethral strictures following high-dose-rate brachytherapy for prostate cancer: Analysis of risk factors. Brachytherapy 2013, 12, 50–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, L.; Williams, S.G.; Tai, K.H.; Foroudi, F.; Cleeve, L.; Duchesne, G. Urethral stricture following high dose rate brachytherapy for prostate cancer. Radiother. Oncol. 2009, 91, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Hampson, L.A.; McAninch, J.W.; Breyer, B.N. Male urethral strictures and their management. Nat. Rev. Urol 2014, 11, 43–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erickson, B.A.; Elliott, S.P.; Voelzke, B.B.; Myers, J.B.; Broghammer, J.A.; Smith, T.G.; McClung, C.D.; Alsikafi, N.F.; Brant, W.O. Multi-institutional 1-year bulbar urethroplasty outcomes using a standardized prospective cystoscopic follow-up protocol. Urology 2014, 84, 213–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baradaran, N.; Fergus, K.B.; Moses, R.A.; Patel, D.P.; Gaither, T.W.; Voelzke, B.B.; Smith, T.G.; Erickson, B.A.; Elliott, S.P.; Alsikafi, N.F.; et al. Clinical significance of cystoscopic urethral stricture recurrence after anterior urethroplasty: A multi-institution analysis from Trauma and Urologic Reconstructive Network of Surgeons (TURNS). World J. Urol. 2019, 37, 2763–2768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Clinical Factor | Successful Patients n = 45/91 (49%) | Failed Patients n = 46/91 (51%) | p-Value |
|---|---|---|---|
| CAD | 14 | 7 | 0.07 |
| DM | 8 | 12 | 0.34 |
| HTN | 22 | 29 | 0.17 |
| HLD | 23 | 19 | 0.35 |
| COPD | 2 | 0 | 0.24 |
| CHF | 2 | 2 | 1 |
| Smoking History | 23 | 19 | 0.35 |
| Malignancy History | 18 | 19 | 0.99 |
| History of Trauma | 5 | 7 | 0.26 |
| Age at Surgery Mean (SD) | 60 (17) | 62 (12) | 0.26 |
| BMI Mean (SD) | 30 (6) | 31 (8) | 0.15 |
| Stricture Location (n) | - | - | - |
| Bulbar | 36/45 (80%) | 39 (85%) | 0.55 |
| Membranous | 9/45 (20%) | 7 (15%) | |
| Prior Management (n) | - | - | - |
| Prior Endoscopic Management * | 23/45 (51%) | 27/46 (59%) | 0.47 |
| Prior Urethroplasty | 22/45 (49%) | 29/46 (63%) | 0.17 |
| Timing from Latest Treatment to Balloon: Median Months | 9.5 (4–29) | 6 (4–22) | 0.27 |
| Irradiated Patients n = 33/91 (36%) | Non-Irradiated Patients n = 58/91 (64%) | p-Value | |
|---|---|---|---|
| Follow-up Cystoscopy Findings | n = 16/33 (48%) | n = 27/58 (47%) | - |
| >16Fr | 8 | 11 | 0.86 |
| <16Fr | 8 | 16 | |
| Symptom/Cystoscopic Concordance | - | - | - |
| No Sx and >16Fr | 3 | 7 | 0.7 |
| No Sx and <16Fr | 0 | 3 | 0.28 |
| Sx and >16Fr | 5 | 4 | 0.26 |
| Sx and <16Fr | 8 | 13 | 0.91 |
| Failed Patients | n = 15/33 (45%) | n = 28/58 (48%) | 0.57 |
| Median (IQR) Time to Failure in Months | 7 (3–13) | 5 (3–13) | 0.33 |
| Management Post-Balloon Failure | n = 15/33 (45%) | n = 27/58 (47%) | - |
| Catheter Dilation | 0 | 4 | 0.28 |
| DVIU | 2 | 1 | 0.25 |
| Repeat Balloon Dilation | 5 | 3 | 0.09 |
| Suprapubic Tube | 6 | 2 | 0.01 * |
| Urethroplasty | 1 | 16 | 0.003 * |
| Perineal Urethrostomy | 0 | 0 | 1 |
| Urinary Diversion | 1 | 1 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beeder, L.A.; Cook, G.S.; Nealon, S.W.; Badkhshan, S.; Sanders, S.C.; Perito, D.P.; Hudak, S.J.; Morey, A.F. Long-Term Experience with Balloon Dilation for Short Bulbar and Membranous Urethral Strictures: Establishing a Baseline in the Active Drug Treatment Era. J. Clin. Med. 2022, 11, 3095. https://doi.org/10.3390/jcm11113095
Beeder LA, Cook GS, Nealon SW, Badkhshan S, Sanders SC, Perito DP, Hudak SJ, Morey AF. Long-Term Experience with Balloon Dilation for Short Bulbar and Membranous Urethral Strictures: Establishing a Baseline in the Active Drug Treatment Era. Journal of Clinical Medicine. 2022; 11(11):3095. https://doi.org/10.3390/jcm11113095
Chicago/Turabian StyleBeeder, Lauren A., Grayden S. Cook, Samantha W. Nealon, Shervin Badkhshan, Sarah C. Sanders, Dylan P. Perito, Steven J. Hudak, and Allen F. Morey. 2022. "Long-Term Experience with Balloon Dilation for Short Bulbar and Membranous Urethral Strictures: Establishing a Baseline in the Active Drug Treatment Era" Journal of Clinical Medicine 11, no. 11: 3095. https://doi.org/10.3390/jcm11113095
APA StyleBeeder, L. A., Cook, G. S., Nealon, S. W., Badkhshan, S., Sanders, S. C., Perito, D. P., Hudak, S. J., & Morey, A. F. (2022). Long-Term Experience with Balloon Dilation for Short Bulbar and Membranous Urethral Strictures: Establishing a Baseline in the Active Drug Treatment Era. Journal of Clinical Medicine, 11(11), 3095. https://doi.org/10.3390/jcm11113095





