360° Ab-Interno Schlemm’s Canal Viscodilation with OMNI Viscosurgical Systems for Open-Angle Glaucoma—Midterm Results
Abstract
:1. Introduction
2. Materials and Methods
2.1. Main Outcome Measures
2.2. Statistical Methods
2.3. Surgical Technique
3. Results
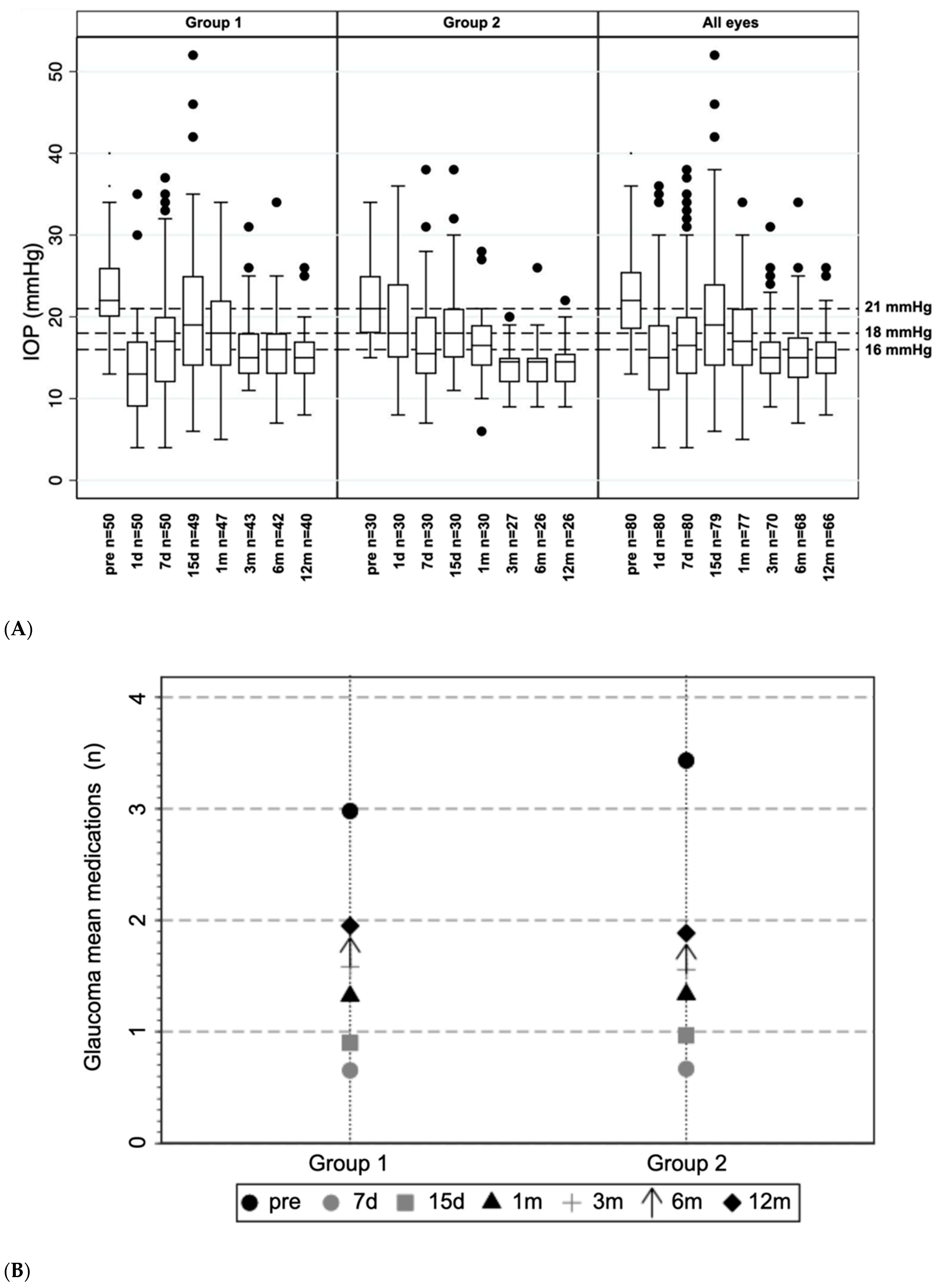
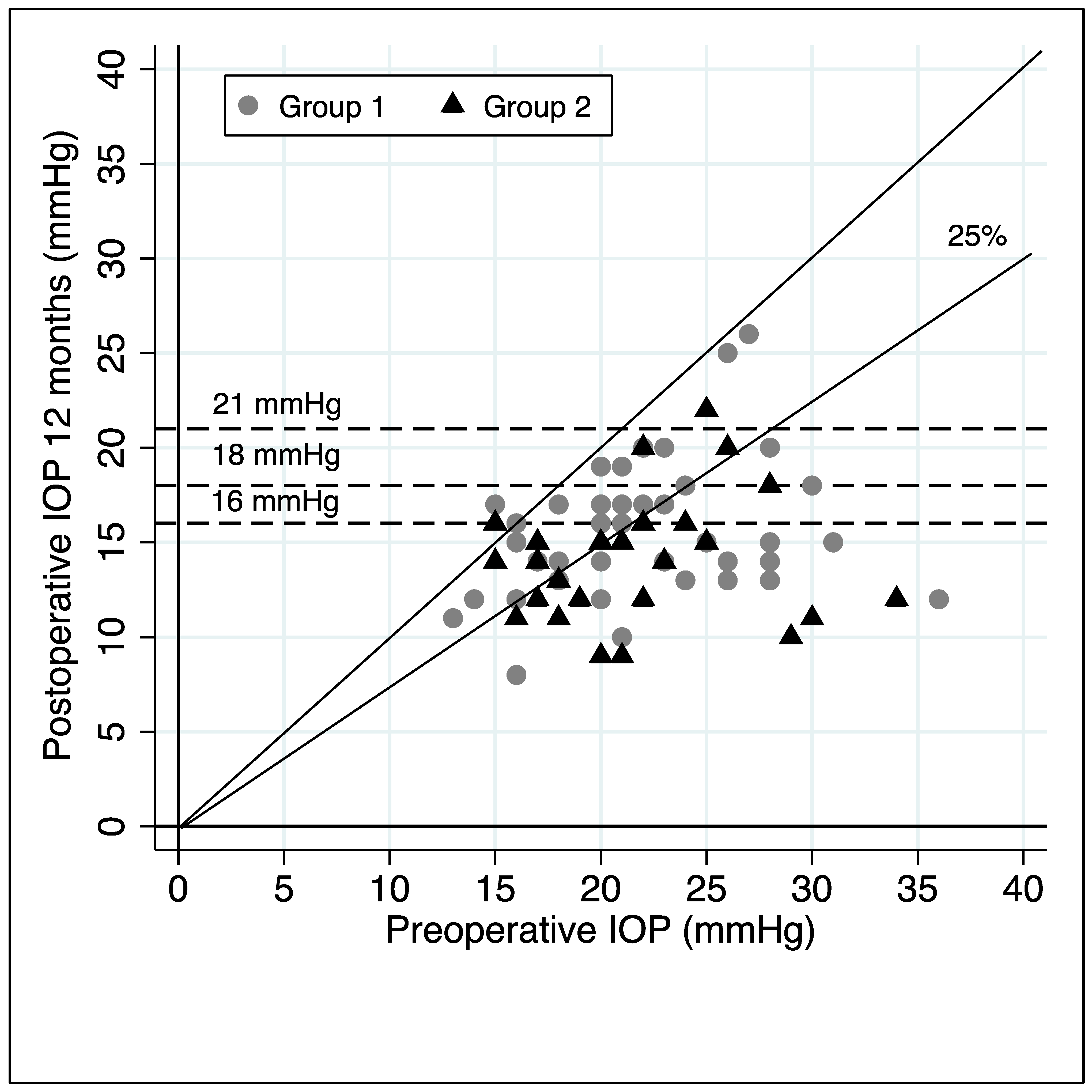
3.1. Differences in Intraocular Pressure and Antiglaucoma Medication Used
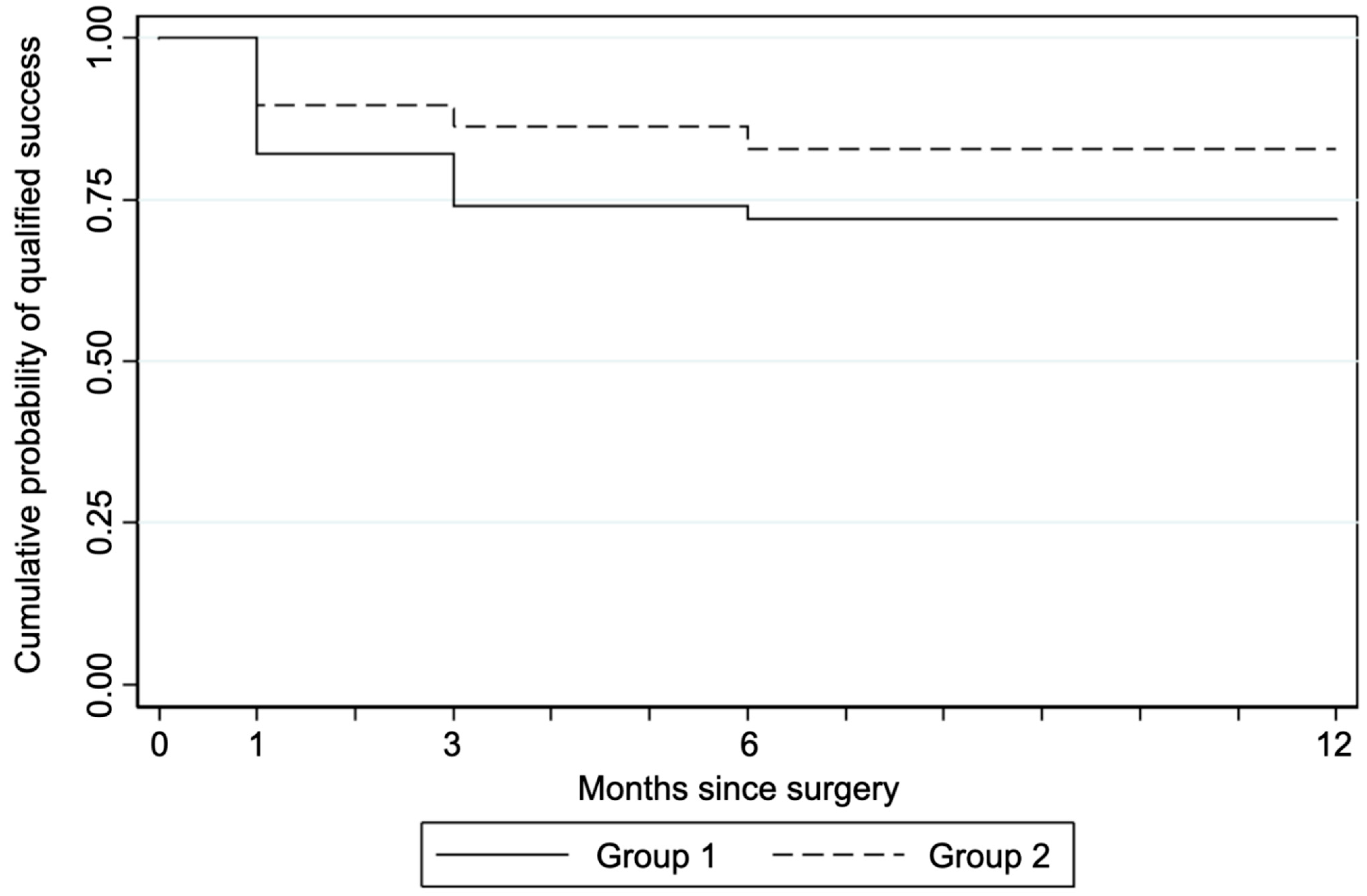
3.2. Success
3.3. Safety
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.H.; Rabiolo, A.; Morales, E.; Yu, F.; Afifi, A.A.; Nouri-Mahdavi, K.; Caprioli, J. Risk factors for fast visual field progression in glaucoma. Am. J. Ophthalmol. 2019, 207, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Kass, M.A.; Heuer, D.K.; Higginbotham, E.J.; Johnson, C.A.; Keltner, J.L.; Miller, J.P.; Parrish, R.K., II; Wilson, M.R.; Gordon, M.O. The Ocular Hypertension Treatment Study: A randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch. Ophthalmol. 2002, 120, 701–713. [Google Scholar] [CrossRef]
- The Advanced Glaucoma Intervention study (AGIS): 7. the relationship between control of intraocular pressure and visual field deterioration. Am. J. Ophthalmol. 2000, 130, 429–440. [CrossRef]
- Newman-Casey, P.A.; Robin, A.L.; Blachley, T.; Farris, K.; Heisler, M.; Resnicow, K.; Lee, P.P. The Most Common Barriers to Glaucoma Medication Adherence: A Cross-Sectional Survey. Ophthalmology 2015, 122, 1308–1316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dreer, L.E.; Girkin, C.; Mansberger, S.L. Determinants of medication adherence to topical glaucoma therapy. J. Glaucoma 2012, 21, 234–240. [Google Scholar] [CrossRef]
- Gedde, S.J.; Singh, K.; Schiffman, J.C.; Feuer, W.J. Tube versus Trabeculectomy Study Group. The Tube Versus Trabeculectomy Study: Interpretation of results and application to clinical practice. Curr. Opin. Ophthalmol. 2012, 23, 118–126. [Google Scholar] [CrossRef]
- Gedde, S.J.; Herndon, L.W.; Brandt, J.D.; Budenz, D.L.; Feuer, W.J.; Schiffman, J.C. Surgical complications in the tube versus trabeculectomy study during the first year of follow-up. Am. J. Ophthalmol. 2007, 143, 23–31. [Google Scholar] [CrossRef]
- Saheb, H.; Ahmed, I.I. Micro-invasive glaucoma surgery: Current perspectives and future directions. Curr. Opin. Ophthalmol. 2012, 23, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Brusini, P.; Filacorda, S. Enhanced glaucoma staging system (GSS 2) for classifying functional damage in glaucoma. J. Glaucoma 2006, 15, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Klink, T.; Sauer, J.; Körber, N.J.; Grehn, F.; Much, M.M.; Thederan, L.; Matlach, J.; Salgado, J.P. Quality of life following glaucoma surgery: Canaloplasty versus trabeculectomy. Clin. Ophthalmol. 2015, 9, 7–16. [Google Scholar] [PubMed] [Green Version]
- Battista, S.A.; Lu, Z.; Hofmann, S.; Freddo, T.; Overby, D.R.; Gong, H. Reduction of the available area for aqueous humor outflow and increase in meshwork herniations into collector channels following acute IOP elevation in bovine eyes. Investig. Ophthalmol. Vis. Sci. 2008, 49, 5346–5352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grieshaber, M.C.; Pienaar, A.; Olivier, J.; Stegmann, R. Comparing two tensioning suture sizes for 360 degrees viscocanalostomy (canaloplasty): A randomised controlled trial. Eye 2010, 24, 1220–1226. [Google Scholar] [CrossRef]
- Krasnov, M.M. Sinusotomy. Trans. Am. Acad. Ophthalmol. Otolaryngol. 1972, 76, 368–374. [Google Scholar]
- Stegmann, R.; Pienaar, A.; Miller, D. Viscocanalostomy for open-angle glaucoma in Black African patients. J. Cataract Refract. Surg. 1999, 25, 316–322. [Google Scholar] [CrossRef]
- Lewis, R.A.; von Wolff, K.; Tetz, M.; Korber, N.; Kearney, J.R.; Shingleton, B.; Samuelson, T.W. Canaloplasty: Circumferential viscodilation and tensioning of Schlemm’s canal using a flexible microcatheter for the treatment of open-angle glaucoma in adults. Interim clinical study analysis. J. Catataract Refract. Surg. 2007, 33, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.A.; von Wolff, K.; Tetz, M.; Koerber, N.; Kearney, J.R.; Shingleton, B.J.; Samuelson, T.W. Canaloplasty: Three-year results of circumferential viscodilation and tensioning of Schlemm canal using a microcatheter to treat open-angle glaucoma. J. Cataract Refract. Surg. 2011, 37, 682–690. [Google Scholar] [CrossRef]
- Bull, H.; von Wolff, K.; Körber, N.; Tetz, M. Three-year canaloplasty outcomes for the treatment of open-angle glaucoma: European study results. Graefes Arch. Clin. Exp. Ophthalmol. 2011, 245, 1537–1545. [Google Scholar] [CrossRef]
- Brusini, P. Canaloplasty in open-angle glaucoma surgery: A four-year follow-up. Sci. World J. 2014, 2014, 469609. [Google Scholar] [CrossRef] [Green Version]
- Kerr, N.M.; Wang, J.; Barton, K. Minimally invasive glaucoma surgery as primary stand-alone surgery for glaucoma. Clin. Exp. Ophthalmol. 2017, 45, 393–400. [Google Scholar] [CrossRef]
- Davids, A.M.; Pahlitzsch, M.; Boeker, A.; Winterhalter, S.; Maier-Wenzel, A.K.; Klamann, M. Ab interno canaloplasty (ABiC)—12-month results of a new minimally invasive glaucoma surgery (MIGS). Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 1947–1953. [Google Scholar] [CrossRef]
- Cagini, C.; Peruzzi, C.; Fiore, T.; Spadea, L.; Lippera, M.; Lippera, S. Canaloplasty: Current value in the management of glaucoma. J. Ophthalmol. 2016, 2016, 7080475. [Google Scholar] [CrossRef] [Green Version]
- Hughes, T.; Traynor, M. Clinical results of ab interno canaloplasty in patients with open-angle glaucoma. Clin. Ophthalmol. 2020, 14, 3641–3650. [Google Scholar] [PubMed]
- Körber, N. Ab interno canaloplasty for the treatment of glaucoma: A case series study. Spektrum Augenheilkd 2018, 32, 223–227. [Google Scholar] [CrossRef] [Green Version]
- Kazerounian, S.; Zimbelmann, M.; Lörtscher, M.; Hommayda, S.; Tsirkinidou, I.; Müller, M. Canaloplasty ab interno (AbiC)—2-Year-Results of a Novel Minimally Invasive Glaucoma Surgery (MIGS) Technique. Klin. Monbl. Augenheilkd. 2021, 238, 1113–1119. [Google Scholar] [CrossRef]
- Ondrejka, S.; Körber, N. 360 ab-interno Schlemm’s canal viscodilation in primary open-angle glaucoma. Clin. Ophthalmol. 2019, 13, 1235–1246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vold, S.D.; Williamson, B.K.; Hirsch, L.; Aminlari, A.E.; Cho, A.S.; Nelson, C.; Dickerson, J.E., Jr. Canaloplasty and Trabeculotomy with the OMNI System in Pseudophakic Patients with Open-Angle Glaucoma: The ROMEO Study. Ophthalmol. Glaucoma 2020, 4, 173–181. [Google Scholar] [CrossRef]
- Sarkisian, S.R.; Mathews, B.; Ding, K.; Patel, A.; Nicek, Z. 360° ab-interno trabeculotomy in refractory primary open-angle glaucoma. Clin. Ophthalmol. 2019, 13, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Grabska-Liberek, I.; Duda, P.; Rogowska, M.; Majszyk-Ionescu, J.; Skowyra, A.; Koziorowska, A.; Kane, I.; Chmielewski, J. 12-month interim results of a prospective study of patients with mild to moderate open-angle glaucoma undergoing combined viscodilation of Schlemm’s canal and collector channels and 360° trabeculotomy as a standalone procedure or combined with cataract surgery. Eur. J. Ophthalmol. 2021. online ahead of print. [Google Scholar] [CrossRef]
- Brown, R.H.; Tsegaw, S.; Dhamdhere, K.; Lynch, M.G. Viscodilation of Schlemm’s canal and trabeculotomy combined with cataract surgery for reducing intraocular pressure in open-angle glaucoma. J. Cataract Refract. Surg. 2020, 46, 644–645. [Google Scholar] [CrossRef]
- Brusini, P.; Tosoni, C. Canaloplasty after failed trabeculectomy: A possible option. J. Glaucoma 2014, 23, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xin, C.; Han, Y.; Shi, Y.; Ziaei, S.; Wang, N. Intermediate outcomes of ab externo circumferential trabeculotomy and canaloplasty in POAG patients with prior incisional glaucoma surgery. BMC Ophthalmol. 2020, 20, 389. [Google Scholar] [CrossRef] [PubMed]
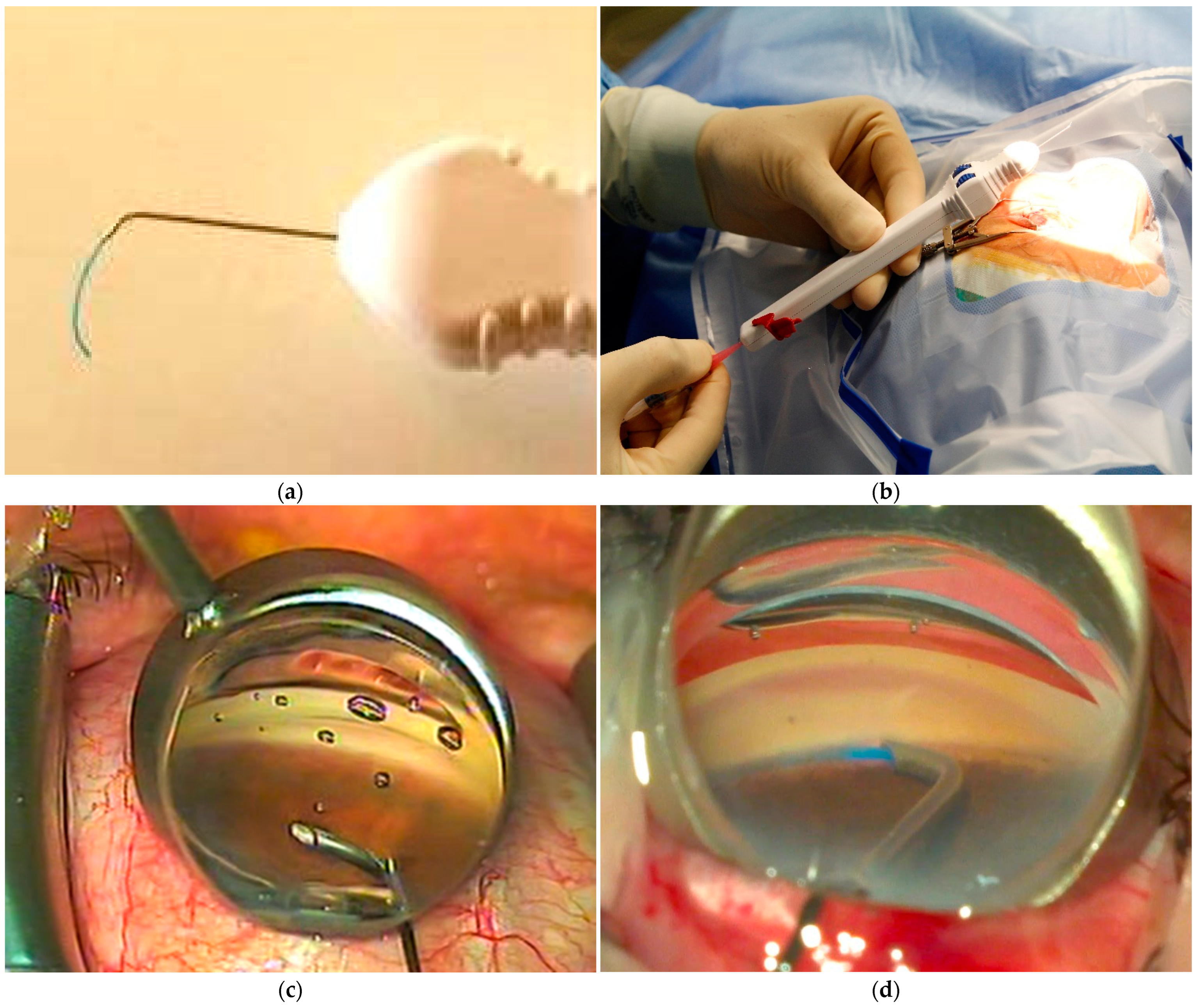
| All Eyes | Group 1 (SC Viscodilation) | Group 2 (SC Vscd + Phaco) | Test on Differences (Statistical Significance) | |
|---|---|---|---|---|
| Eyes, n | 80 | 50 | 30 | |
| Patients, n | 73 | 47 | 26 | |
| Age in years | ||||
| Mean Age ± SD (years) | 74.5 ± 7.5 | 74.2 ± 8.0 | 75.0 ± 6.6 | t test (p = 0.32) |
| Age Range (years) | 56–93 | 56–93 | 62–89 | |
| Gender, n (%) | ||||
| Female | 43 (58.9) | 26 (55.3) | 17 (65.4) | Chi2 test on gender distribution (p = 0.35) |
| Male | 30 (41.1) | 21 (44.7) | 9 (34.6) | |
| Preoperative IOP, mean ± SD | 22.5 ± 5.3 | 23.0 ± 5.7 | 21.5 ± 4.7 | t test (p = 0.11) |
| Preoperative medications, mean n ± SD | 3.2 ± 1.0 | 3.0 ± 1.1 | 3.4 ± 0.8 | t test (p = 0.03) |
| Preoperative BCVA, mean ± SD | 7.5 ± 2.5 | 7.9 ± 2.4 | 6.8 ± 2.1 | t test (p = 0.02) |
| Glaucoma diagnosis, n (%) | ||||
| Primary open angle glaucoma | 51 (63.8) | 37 (74.0) | 14 (46.7) | Chi2 test (p = 0.01) |
| Pseudoexfoliative glaucoma | 27 (33.7) | 12 (24.0) | 15 (50.0) | Chi2 test (p = 0.01) |
| Pigmentary dispersion glaucoma | 2 (2.5) | 1 (2.0) | 1 (3.3) | Chi2 test (p = 0.71) |
| Baseline lens status, n (%) | ||||
| Pseudophakic | 44 (55.0) | 44 (88.0) | 0 (0) | Chi2 test (p = 0.00) |
| Phakic | 36 (45.0) | 6 (12.0) | 30 (100) | Chi2 test (p = 0.00) |
| Previous surgery, n (%) | - | - | - | |
| Cataract | 44 (55.0) | 44 (88.0) | 0 (0.0) | Chi2 test (p = 0.00) |
| Laser trabeculoplasty | 15 (18.8) | 10 (20.0) | 5 (16.7) | Chi2 test (p = 0.71) |
| Deep sclerectomy | 10 (12.5) | 10 (20.0) | 0 (0.0) | Chi2 test (p = 0.01) |
| Trabeculectomy | 3 (3.8) | 3 (6.0) | 0 (0.0) | Chi2 test (p = 0.01) |
| BL | 7 Days | 15 Days | 1 Month | 3 Months | 6 Months | 12 Months | ||
|---|---|---|---|---|---|---|---|---|
| All eyes | Eyes, n | 80 | 80 | 79 | 77 | 70 | 68 | 66 |
| Mean IOP, mmHg ± SD | 22.5 ± 5.3 | 17.8 ± 7.5 | 20.5 ± 8.4 | 17.8 ± 5.5 | 15.6 ± 4.0 | 15.3 ± 4.6 | 15.0 ± 3.6 | |
| Mean IOP reduction from BL, mmHg (%) | 4.7 (18%) | 1.8 (5.3%) | 4.5 (16.6%) | 6.5 (26.9%) | 6.8 (28.4%) | 6.9 (29.0%) | ||
| p-Value | (p < 0.001) | (p = 0.035) | p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | ||
| Mean MEDs, n ± SD | 3.2 ± 1.0 | 0.7 ± 1.2 | 0.9 ± 1.3 | 1.3 ± 1.4 | 1.6 ± 1.4 | 1.7 ± 1.4 | 1.9 ± 1.4 | |
| Mean MEDs reduction from BL, n (p-Value) | 2.5 (p < 0.001) | 2.2 (p < 0.001) | 1.8 (p < 0.001) | 1.6 (p < 0.001) | 1.5 (p < 0.001) | 1.2 (p < 0.001) | ||
| Group 1 | Eyes, n | 50 | 50 | 49 | 47 | 43 | 42 | 40 |
| Mean IOP, mmHg ± SD | 23.0 ± 5.7 | 17.8 ± 7.9 | 21.1 ± 9.5 | 18.6 ± 6.0 | 16.4 ± 4.4 | 15.8 ± 5.1 | 15.6 ± 3.6 | |
| Mean IOP reduction from BL, mmHg (%) | 5.2 (20.3%) | 1.6 (4.6%) | 4.2 (15.1%) | 5.9 (23.8%) | 6.5 (27.0%) | 6.5 (26.8%) | ||
| p-Value | p < 0.001 | p = 0.12 | p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | ||
| Mean MEDs, n ± SD | 3.0 ± 1.1 | 0.7 ± 1.2 | 0.9 ± 1.2 | 1.3 ± 1.4 | 1.6 ± 1.4 | 1.7 ± 1.4 | 2.0 ± 1.4 | |
| Mean MEDs reduction from BL, n (p-Value) | 2.3 (p < 0.001) | 2.1 (p < 0.001) | 1.7 (p < 0.001) | 1.4 (p < 0.001) | 1.2 (p < 0.001) | 1.0 (p < 0.001) | ||
| Group 2 | Eyes, n | 30 | 30 | 30 | 30 | 27 | 26 | 26 |
| Mean IOP, mmHg ± SD | 21.5 ± 4.7 | 17.6 ± 7.1 | 19.4 ± 6.3 | 16.6 ± 4.5 | 14.4 ± 2.9 | 14.5 ± 3.7 | 14.1 ± 3.3 | |
| Mean IOP reduction from BL, mmHg (%) | 3.9 (14.3%) | 2.1 (6.2%) | 4.9 (19.1%) | 7.4 (30.7%) | 7.2 (30.3%) | 7.6 (32.4%) | ||
| p-Value | p = 0.007 | p < 0.069 | p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | ||
| Mean MEDs, n ± SD | 3.4 ± 0.8 | 0.7 ± 1.1 | 1.0 ± 1.4 | 1.3 ± 1.3 | 1.6 ± 1.4 | 1.7 ± 1.3 | 1.9 ± 1.4 | |
| Mean MEDs reduction from BL, n (p-Value) | 2.8 (p < 0.001) | 2.5 (p < 0.001) | 2.1 (p < 0.001) | 1.9 (p < 0.001) | 1.8 (p < 0.001) | 1.6 (p < 0.001) | ||
| Success Rate (%) at 12 Months | ||||
|---|---|---|---|---|
| All Included Eyes | Group 1 | Group 2 | ||
| ≤16 mmHg | Complete | 14.1 | 14.0 | 14.3 |
| Qualified | 58.9 | 48.0 | 78.8 | |
| ≤18 mmHg | Complete | 15.4 | 14.0 | 17.9 |
| Qualified | 71.8 | 66.0 | 82.1 | |
| ≤21 mmHg | Complete | 17.9 | 18.0 | 17.9 |
| Qualified | 80.8 | 76.0 | 89.3 | |
| ≤16 mmHg and ≥25% IOP reduction | Complete | 10.3 | 10.0 | 10.7 |
| Qualified | 43.6 | 32.0 | 64.3 | |
| ≤18 mmHg and ≥25% IOP reduction | Complete | 11.5 | 10.0 | 14.0 |
| Qualified | 50.0 | 40.0 | 67.9 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toneatto, G.; Zeppieri, M.; Papa, V.; Rizzi, L.; Salati, C.; Gabai, A.; Brusini, P. 360° Ab-Interno Schlemm’s Canal Viscodilation with OMNI Viscosurgical Systems for Open-Angle Glaucoma—Midterm Results. J. Clin. Med. 2022, 11, 259. https://doi.org/10.3390/jcm11010259
Toneatto G, Zeppieri M, Papa V, Rizzi L, Salati C, Gabai A, Brusini P. 360° Ab-Interno Schlemm’s Canal Viscodilation with OMNI Viscosurgical Systems for Open-Angle Glaucoma—Midterm Results. Journal of Clinical Medicine. 2022; 11(1):259. https://doi.org/10.3390/jcm11010259
Chicago/Turabian StyleToneatto, Giacomo, Marco Zeppieri, Veronica Papa, Laura Rizzi, Carlo Salati, Andrea Gabai, and Paolo Brusini. 2022. "360° Ab-Interno Schlemm’s Canal Viscodilation with OMNI Viscosurgical Systems for Open-Angle Glaucoma—Midterm Results" Journal of Clinical Medicine 11, no. 1: 259. https://doi.org/10.3390/jcm11010259
APA StyleToneatto, G., Zeppieri, M., Papa, V., Rizzi, L., Salati, C., Gabai, A., & Brusini, P. (2022). 360° Ab-Interno Schlemm’s Canal Viscodilation with OMNI Viscosurgical Systems for Open-Angle Glaucoma—Midterm Results. Journal of Clinical Medicine, 11(1), 259. https://doi.org/10.3390/jcm11010259








