Relationship between Cardiopulmonary, Mitochondrial and Autonomic Nervous System Function Improvement after an Individualised Activity Programme upon Chronic Fatigue Syndrome Patients
Abstract
1. Introduction
2. Materials and Methods
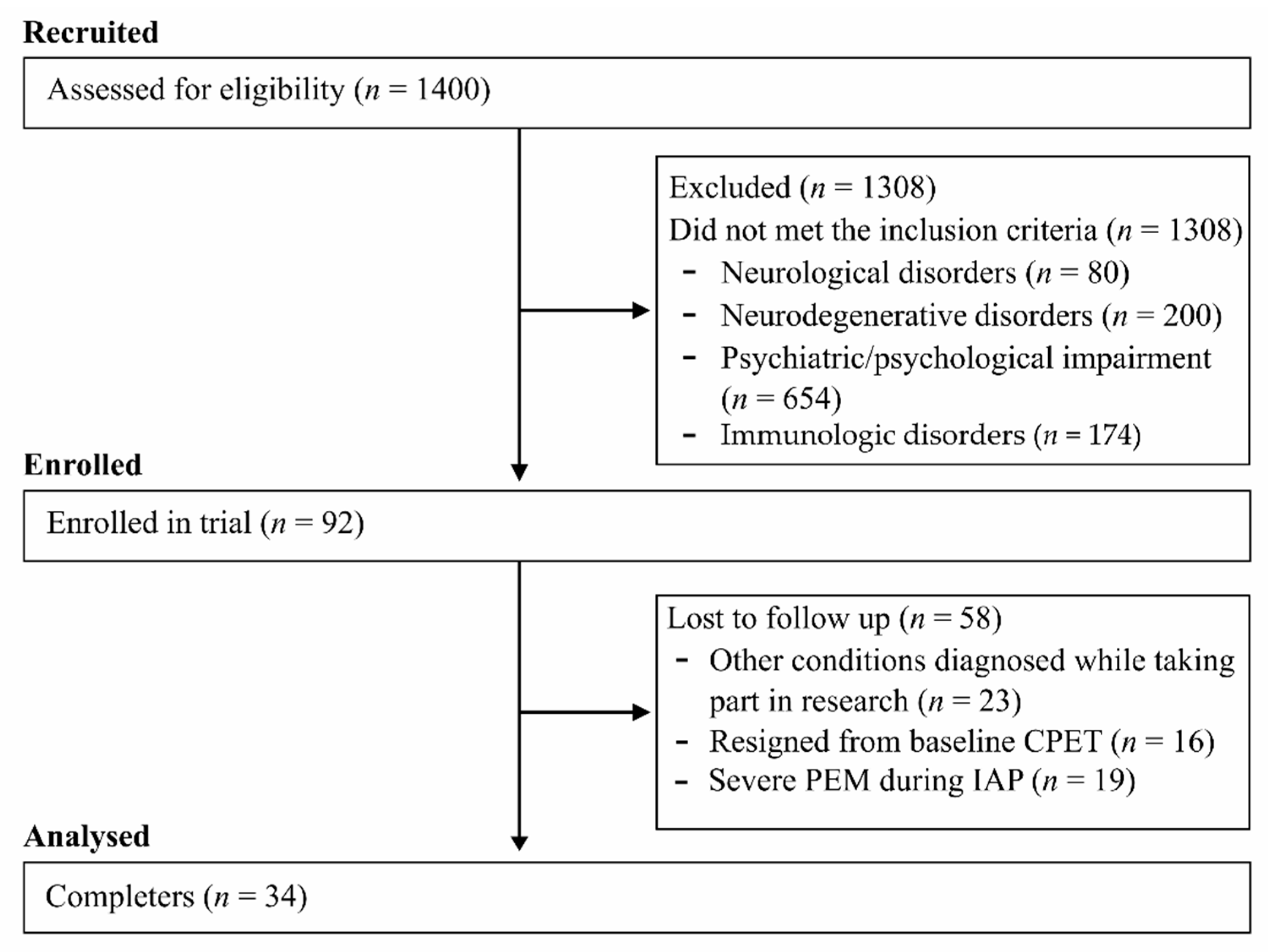
2.1. Recruitment and Eligibility
Anxiety and Depression
2.2. Outcome Measures
2.2.1. Body Composition Analysis
2.2.2. Fatigue
2.2.3. Autonomic Nervous System (ANS) Functioning
2.2.4. Mitochondrial Function
2.2.5. Cardiorespiratory Function
2.3. Intervention
Individualised Activity Programme (IAP)
2.4. Statistical Methods
3. Results
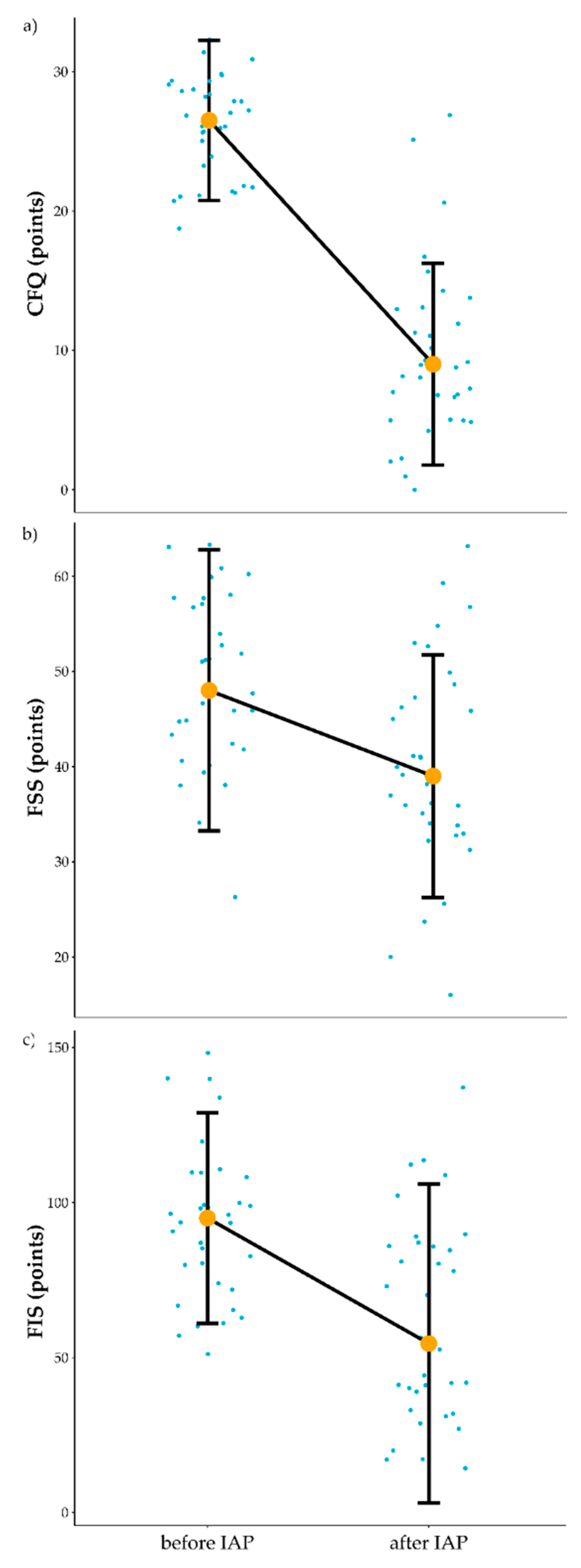
3.1. Influence of IAP on Fatigue
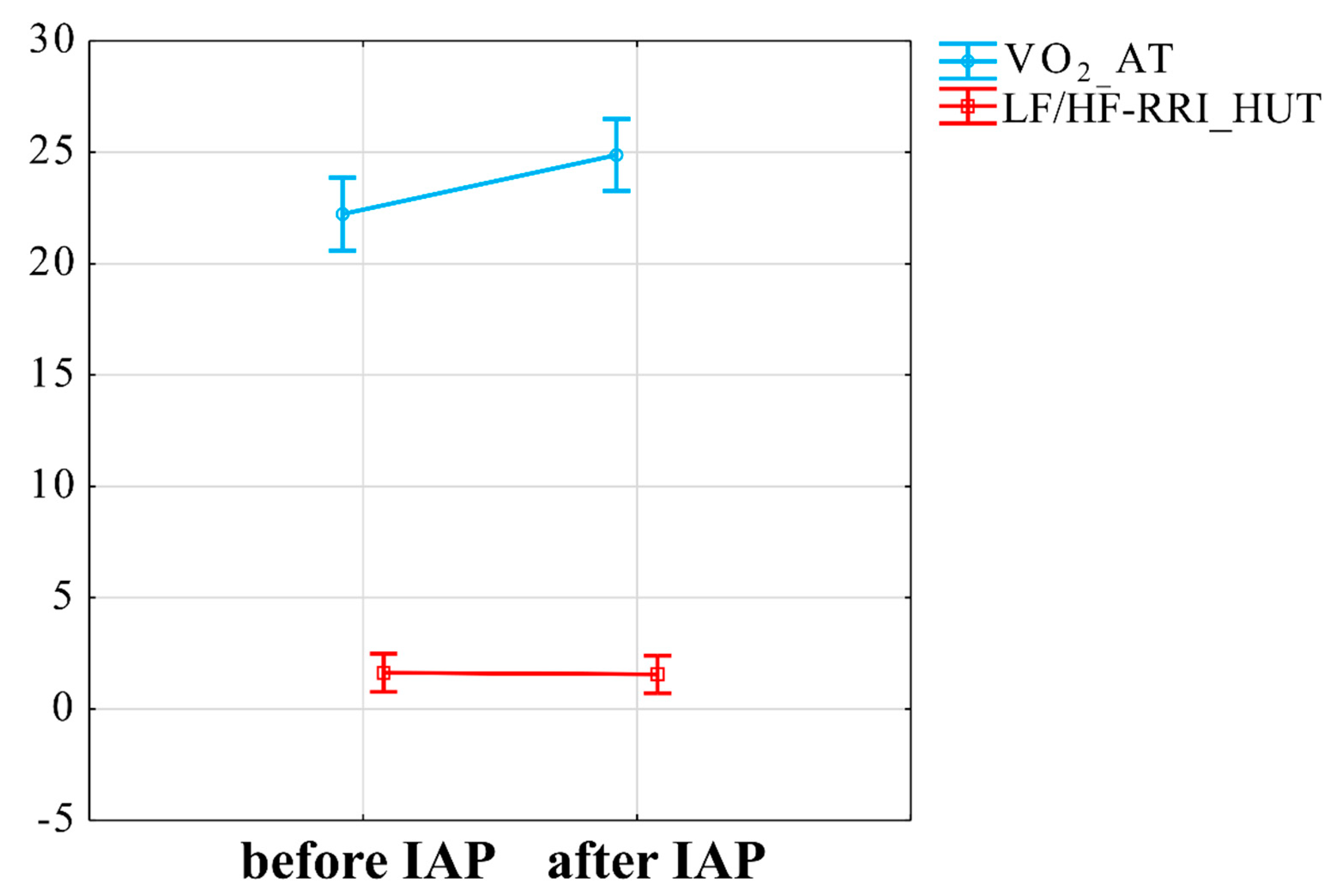
3.2. Influence of IAP on Cardiorespiratory Function
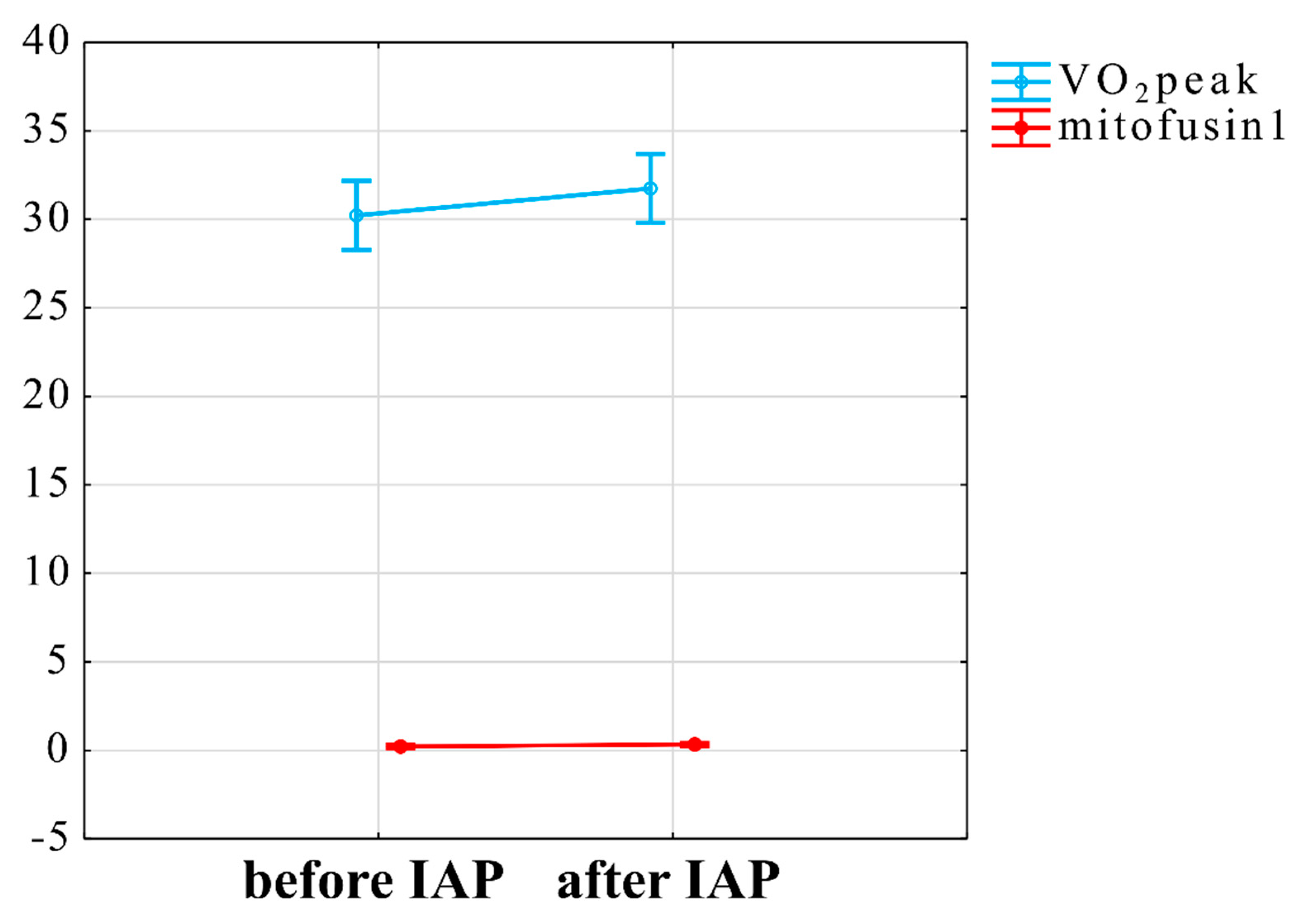
3.3. Influence of IAP on Mitochondrial Function
3.4. Interaction between VO2peak Improvement, Mitochondrial Plasma Markers and ANS Changes
4. Discussion
4.1. Influence of IAP on Self-Reported Fatigue and Peak Oxygen Uptake
4.2. Relationship of Peak Oxygen Uptake Improvement and Mitofusin1 Level
4.3. The Relationship between of Submax VO2 Improvement and ANS Responsiveness
4.4. Study Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments

Conflicts of Interest
References
- Prins, J.B.; Bleijenberg, G.; van der Meer, J.W.M. Chronic fatigue syndrome—Authors’ reply. Lancet 2006, 367, 1575. [Google Scholar] [CrossRef]
- Jason, L.A.; McManimen, S.; Sunnquist, M.; Brown, A.; Newton, J.L.; Strand, E.B. Examining the Institute of Medicine’s Recommendations Regarding Chronic Fatigue Syndrome: Clinical Versus Research Criteria. J. Neurol. Psychol. 2015, 2015, 441577253. [Google Scholar]
- Komaroff, A.L. Advances in understanding the pathophysiology of chronic fatigue syndrome. JAMA 2019, 322, 499–500. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.J.; Bahl, J.S.; Buckley, J.D.; Thomson, R.L.; Davison, K. Evidence of altered cardiac autonomic regulation in myalgic encephalomyelitis/chronic fatigue syndrome: A systematic review and meta-analysis. Medicine 2019, 98, e1760. [Google Scholar] [CrossRef]
- Fortin, J.; Klinger, T.; Wagner, C.; Sterner, H.; Madritsch, C.; Grullenberger, R. The Task Force Monitor—A Non-invasive Beat-to-beat Monitor for Hemodynamic and Autonomic Function of the Human Body. In Proceedings of the 20th Annual International Conference of the IEEE; Engineering in Medicine and Biology Society: Austin, TX, USA, 1988. [Google Scholar]
- Fortin, J.; Marte, W.; Grullenberger, R. Continuous non-invasive blood pressure monitoring using concentrically interlocking control loops. Comput. Biol. Med. 2006, 36, 941–957. [Google Scholar] [CrossRef] [PubMed]
- Laborde, S.; Mosley, E.; Thayer, J.F. Heart rate variability and cardiac vagal tone in psychophysiological research–recommendations for experiment planning, data analysis, and data reporting. Front Psychol. 2017, 8, 213. [Google Scholar] [CrossRef]
- Perna, G.; Riva, A.; Defillo, A.; Sangiorgio, E.; Nobile, M.; Caldirola, D. Heart rate variability: Can it serve as a marker of mental health resilience? Special Section on “Translational and Neuroscience Studies in Affective Disorders” Section Editor, Maria Nobile MD, PhD. J. Affect. Disord. 2020, 263, 754–761. [Google Scholar] [CrossRef]
- Naschitz, J.E.; Rosner, I.; Rozenbaum, M.; Gaitini, L.; Bistritzki, I.; Zuckerman, E.; Sabo, E.; Yeshurun, D. The capnography head-up tilt test for evaluation of chronic fatigue syndrome. Semin. Arthritis Rheum. 2000, 30, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Cheshire, W.P.; Goldstein, D.S. Autonomic uprising: The tilt table test in autonomic medicine. Clin. Auton. Res. 2019, 29, 215–230. [Google Scholar] [CrossRef]
- Raczak, G.; Danilowicz-Szymanowicz, L.; Kobuszewska-Chwirot, M.; Ratkowski, W.; Figura-Chmielewska, M.; Szwoch, M. Long-term exercise training improves autonomic nervous system profile in professional runners. Kardiol. Pol. 2006, 64, 135. [Google Scholar]
- Soltani, M.; Baluchi, M.J.; Boullosa, D.; Daraei, A.; Govindasamy, K.; Dehbaghi, K.M.; Mollabashi, S.S.; Doyle–Baker, P.K.; Basati, G.; Saeidi, A.; et al. Endurance training intensity has greater effects than volume on heart rate variability and arterial stiffness adaptations in sedentary adult men: A Randomized Controlled Trial. Res. Square 2021. [Google Scholar] [CrossRef]
- Esco, M.R.; Flatt, A.A.; Nakamura, F.Y. Initial weekly HRV response is related to the prospective change in VO2max in female soccer players. Int. J. Sports Med. 2016, 37, 436–441. [Google Scholar] [CrossRef]
- Sebastiao, E.; Hubbard, E.A.; Klaren, R.E.; Pilutti, L.A.; Motl, R.W. Fitness and its association with fatigue in persons with multiple sclerosis. Scand. J. Med. Sci. Sports 2017, 27, 1776–1784. [Google Scholar] [CrossRef]
- Franklin, J.D.; Atkinson, G.; Atkinson, J.M.; Batterham, A. Peak oxygen uptake in chronic fatigue syndrome/myalgic encephalomyelitis: A meta analysis. Int. J. Sports Med. 2019, 40, 77–87. [Google Scholar] [CrossRef]
- Myhill, S.; Booth, N.E.; McLaren-Howard, J. Chronic fatigue syndrome and mitochondrial dysfunction. Int. J. Clin. Exp. Med. 2009, 2, 1. [Google Scholar]
- Behan, W.M.H.; More, I.A.R.; Behan, P.O. Mitochondrial abnormalities in the postviral fatigue syndrome. Acta Neuropathol. 1991, 83, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Byrne, E.; Trounce, I.; Dennett, X. Chronic relapsing myalgia (postviral): Clinical, histological and biochemical studies. Aust. N. Z. J. Med. 1985, 15, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Vecchiet, L.; Montanari, G.; Pizzigallo, E.; Iezzi, S.; de Bigontina, P.; Dragani, L.; Vecchiet, J.; Giamberardino, M.A. Sensory characterization of somatic parietal tissues in humans with chronic fatigue syndrome. Neurosci. Lett. 1996, 208, 117–120. [Google Scholar] [CrossRef]
- Bell, M.B.; Bush, Z.; McGinnis, G.R.; Rowe, G.C. Adult skeletal muscle deletion of Mitofusin 1 and 2 impedes exercise performance and training capacity. J. Appl. Physiol. 2019, 126, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Cartoni, R.; Léger, B.; Hock, M.B.; Praz, M.; Crettenand, A.; Pich, S.; Ziltener, J.L.; Luthi, F.; Dériaz, O.; Zorzano, A.; et al. Mitofusins 1/2 and ERRalpha expression are increased in human skeletal muscle after physical exercise. J. Physiol. 2005, 567, 349–358. [Google Scholar] [CrossRef]
- Lebeau, J.; Saunders, J.M.; Moraes, V.W.; Madhavan, A.; Madrazo, N.; Anthony, M.C.; Wiseman, R.L. The PERK arm of the unfolded protein response regulates mitochondrial morphology during acute endoplasmic reticulum stress. Cell Rep. 2018, 22, 2827–2836. [Google Scholar] [CrossRef]
- Tondera, D.; Grandemange, S.; Jourdain, A.; Karbowski, M.; Mattenberger, Y.; Herzig, S.; Da Cruz, S.; Clerc, P.; Raschke, I.; Merkwirth, C.; et al. SLP-2 is required for stress-induced mitochondrial hyperfusion. EMBO J. 2009, 28, 1589–1600. [Google Scholar] [CrossRef]
- Sgarbi, G.; Matarrese, P.; Pinti, M.; Lanzarini, C.; Ascione, B.; Gibellini, L.; Dika, E.; Patrizi, A.; Tommasino, C.; Capri, M.; et al. Mitochondria hyperfusion and elevated autophagic activity are key mechanisms for cellular bioenergetic preservation in centenarians. Aging 2014, 6, 296–310. [Google Scholar] [CrossRef]
- Rossignol, R.; Gilkerson, R.; Aggeler, R.; Yamagata, K.; Remington, S.J.; Capaldi, R.A. Energy substrate modulates mitochondrial structure and oxidative capacity in cancer cells. Cancer Res. 2004, 64, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Chakrabarti, O. Mitochondrial hyperfusion: A friend or a foe. Biochem. Soc. Trans. 2020, 48, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.C.; Wilson, R.J.; Yan, Z. Molecular mechanisms for mitochondrial adaptation to exercise training in skeletal muscle. FASEB J. 2016, 30, 13–22. [Google Scholar] [CrossRef]
- Cvejic, E.; Lloyd, A.R.; Vollmer-Conna, U. Neurocognitive improvements after best-practice intervention for chronic fatigue syndrome: Preliminary evidence of divergence between objective indices and subjective perceptions. Compr. Psychiatry 2016, 66, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Castell, B.D.; Kazantzis, N.; Moss-Morris, R.E. Cognitive behavioral therapy and graded exercise for chronic fatigue syndrome: A meta-analysis. Clin. Psychol. Sci. Pract. 2011, 18, 311–324. [Google Scholar] [CrossRef]
- Larun, L.; Brurberg, K.G.; Odgaard-Jensen, J.; Price, J.R. Exercise therapy for chronic fatigue syndrome. Cochrane Database Syst. Rev. 2016, 25, CD003200. [Google Scholar] [CrossRef]
- Wilshire, C.E.; Kindlon, T.; Courtney, R.; Matthees, A.; Tuller, D.; Geraghty, K.; Levin, B. Rethinking the treatment of chronic fatigue syndrome—A reanalysis and evaluation of findings from a recent major trial of graded exercise and CBT. BMC Psychol. 2018, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- McPhee, G. Cognitive behaviour therapy and objective assessments in chronic fatigue syndrome. J. Health Psychol. 2017, 22, 1181–1186. [Google Scholar] [CrossRef]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.C.; Dobbins, J.G.; Komaroff, A. The chronic fatigue syndrome: A comprehensive approach to its definition and study. Ann. Intern Med. 1994, 121, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Zalewski, P.; Kujawski, S.; Tudorowska, M.; Morten, K.; Tafil-Klawe, M.; Klawe, J.J.; Strong, J.; Estévez-López, F.; Murovska, M.; Newton, J.L. The Impact of a Structured Exercise Programme upon Cognitive Function in Chronic Fatigue Syndrome Patients. Brain Sci. 2019, 10, 4. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Ball, R.; Ranieri, W. Comparison of Beck Depression Inventories -IA and -II in psychiatric outpatients. J. Pers. Assess. 1996, 67, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Morriss, R.; Wearden, A.; Mullis, R. Exploring the validity of the Chalder Fatigue scale in chronic fatigue syndrome. J. Psychosom. Res. 1998, 45, 411–417. [Google Scholar] [CrossRef]
- Valko, P.O.; Bassetti, C.L.; Bloch, K.E.; Held, U.; Baumann, C.R. Validation of the fatigue severity scale in a Swiss cohort. Sleep 2008, 31, 1601–1607. [Google Scholar] [CrossRef] [PubMed]
- Frith, J.; Newton, J. Fatigue impact scale. Occup. Med. 2010, 60, 159. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, A.M.; Mainardi, L.T.; Meloni, C.; Chierchiu, S.; Cerutti, S. Continuous monitoring of the sympatho-vagal balance through spectral analysis. IEEE Eng. Med. Biol. Mag. 1997, 16, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Kenny, R.A.; O’Shea, D.; Parry, S.W. The Newcastle protocols for head-up tilt table testing in the diagnosis of vasovagal syncope, carotid sinus hypersensitivity, and related disorders. Heart 2000, 83, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Estévez, M.; Machado, C.; Leisman, G.; Estévez-Hernández, T.; Arias-Morales, A.; Machado, A.; Montes-Brown, J. Spectral analysis of heart rate variability. Int. J. Disabil. Hum. Dev. 2016, 15, 5–17. [Google Scholar] [CrossRef]
- Froelicher, V.F.; Thompson, A.; Noguera, I.; Davis, G.; Stewart, A.J.; Triebwasser, J.H. Prediction of Maximal Oxygen Consumption: Comparison of the Bruce and Balke treadmill protocols. Chest 1975, 68, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Linda, S.; Thompson, W.R.; Gordon, N.F.; Pescatello, L.S. (Eds.) ACSM’s Guidelines for Exercise Testing and Prescription; Wolters Kluwer Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2010; Volume 8, pp. 35–96. [Google Scholar]
- Schneider, D.A.; Phillips, S.E.; Stoffolano, S.H. The simplified V-slope method of detecting the gas exchange threshold. Med. Sci. Sports Exerc. 1993, 25, 1180–1184. [Google Scholar] [CrossRef] [PubMed]
- Field, A. Discovering Statistics using SPSS; Sage Publications: London, UK, 2009; 550p. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://ggplot2.tidyverse.org (accessed on 9 October 2019).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Kujawski, S.; Cossington, J.; Słomko, J.; Dawes, H.; Strong, J.W.; Estevez-Lopez, F.; Murovska, M.; Newton, J.L.; Hodges, L.; Zalewski, P. Prediction of Discontinuation of Structured Exercise Programme in Chronic Fatigue Syndrome Patients. J. Clin. Med. 2020, 9, 3436. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, M.; Goldsmith, K.A.; Johnson, A.L.; Chalder, T.; Walker, J.; White, P.D. Rehabilitative treatments for chronic fatigue syndrome: Long-term follow-up from the PACE trial. Lancet Psychiatry 2015, 2, 1067–1074. [Google Scholar] [CrossRef]
- White, P.D.; Goldsmith, K.A.; Johnson, A.L.; Potts, L.; Walwyn, R.; DeCesare, J.C.; Baber, H.L.; Burgess, M.; Clark, L.V.; Cox, D.L.; et al. Comparison of adaptive pacing therapy, cognitive behaviour therapy, graded exercise therapy, and specialist medical care for chronic fatigue syndrome (PACE): A randomised trial. Lancet 2011, 377, 823–836. [Google Scholar] [CrossRef]
- Clark, L.V.; Pesola, F.; Thomas, J.M.; Vergara-Williamson, M.; Beynon, M.; White, P.D. Guided graded exercise self-help plus specialist medical care versus specialist medical care alone for chronic fatigue syndrome (GETSET): A pragmatic randomised controlled trial. Lancet 2017, 390, 363–373. [Google Scholar] [CrossRef]
- Wilshire, C.E.; McPhee, G.; Science for MECFQ. Submission to the public review on common data elements for ME/CFS: Problems with the Chalder Fatigue Questionnaire. Science for ME. 2018. Available online: https://huisartsvink.files.wordpress.com/2018/08/wilshire-mcphee-cfq-cde-critique-for-s4me-final.pdf (accessed on 26 March 2021).
- Jason, L.A.; Evans, M.; Brown, M.; Porter, N.; Brown, A.; Hunnell, J.; Anderson, V.; Lerch, A. Fatigue scales and chronic fatigue syndrome: Issues of sensitivity and specificity. Disabil. Stud. Q. Winter 2011, 31, 1375. [Google Scholar] [CrossRef]
- Al Amir Dache, Z.A.A.; Otandault, A.; Tanos, R.; Pastor, B.; Meddeb, R.; Sanchez, C.; Arena, G.; Lasorsa, L.; Bennett, A.; Grange, T.; et al. Blood contains circulating cell-free respiratory competent mitochondria. FASEB J. 2020, 34, 3616–3630. [Google Scholar] [CrossRef] [PubMed]
- Puhm, F.; Afonyushkin, T.; Resch, U.; Obermayer, G.; Rohde, M.; Penz, T.; Schuster, M.; Wagner, G.; Rendeiro, A.F.; Melki, I.; et al. Mitochondria Are a Subset of Extracellular Vesicles Released by Activated Monocytes and Induce Type I IFN and TNF Responses in Endothelial Cells. Circ. Res. 2019, 125, 43–52. [Google Scholar] [CrossRef]
- Zachari, M.; Ktistakis, N.T. Mammalian Mitophagosome Formation: A Focus on the Early Signals and Steps. Front. Cell Dev. Biol. 2020, 8, 171. [Google Scholar] [CrossRef]
- Bhattacharyya, K.; Mukherjee, S. Fluorescent Metal Nano-Clusters as Next Generation Fluorescent Probes for Cell Imaging and Drug Delivery. Bull. Chem. Soc. Jpn. 2018, 91, 447–454. [Google Scholar] [CrossRef]
- Bernimoulin, M.; Waters, E.K.; Foy, M.; Steele, B.M.; Sullivan, M.; Falet, H.; Walsh, M.T.; Barteneva, N.; Geng, J.-G.; Hartwig, J.H.; et al. Differential stimulation of monocytic cells results in distinct populations of microparticles. J. Thromb. Haemost. 2009, 7, 1019–1028. [Google Scholar] [CrossRef]
- Sugiura, A.; McLelland, G.; Fon, E.A.; McBride, H.M. A new pathway for mitochondrial quality control: Mitochondrial-derived vesicles. EMBO J. 2014, 33, 2142–2156. [Google Scholar] [CrossRef]
- Westermann, B. Mitochondrial fusion and fission in cell life and death. Nat. Rev. Mol. Cell Biol. 2010, 11, 872–884. [Google Scholar] [CrossRef]
- Eisner, V.; Lenaers, G.; Hajnóczky, G. Mitochondrial fusion is frequent in skeletal muscle and supports excitation–contraction coupling. J. Cell Biol. 2014, 205, 179–195. [Google Scholar] [CrossRef]
- Goncalves, I.O.; Passos, E.; Diogo, C.V.; Rocha-Rodrigues, S.; Santos-Alves, E.; Oliveira, P.J.; Ascensão, A.; Magalhaes, J. Exercise mitigates mitochondrial permeability transition pore and quality control mechanisms alterations in nonalcoholic steatohepatitis. Appl. Physiol. Nutr. Metab. 2016, 41, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.G.R.; Lally, J.; Holloway, G.P.; Heigenhauser, G.J.F.; Bonen, A.; Spriet, L.L. Repeated transient mRNA bursts precede increases in transcriptional and mitochondrial proteins during training in human skeletal muscle. J. Physiol. 2010, 588, 4795–4810. [Google Scholar] [CrossRef] [PubMed]
- Flockhart, M.; Nilsson, L.C.; Tais, S.; Ekblom, B.; Apró, W.; Larsen, F.J. Excessive exercise training causes mitochondrial functional impairment and decreases glucose tolerance in healthy volunteers. Cell Metab. 2021. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Myalgic Encephalomyelitis (or Encephalopathy)/Chronic Fatigue Syndrome: Diagnosis and Management. Draft Guidance Consultation. 2020. Available online: https://www.nice.org.uk/guidance/indevelopment/gid-ng10091 (accessed on 26 March 2021).
- Davenport, T.E.; Lehnen, M.; Stevens, S.R.; Vanness, J.M.; Stevens, J.; Snell, C.R. Chronotropic Intolerance: An Overlooked Determinant of Symptoms and Activity Limitation in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome? Front. Pediatr. 2019, 7, 82. [Google Scholar] [CrossRef]
- Hodges, L.; Nielsen, T.; Cochrane, D.; Baken, D. The physiological time line of post-exertional malaise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Transl. Sports Med. 2020, 3, 243–249. [Google Scholar] [CrossRef]
- Holtzman, C.S.; Bhatia, S.; Cotler, J.; Jason, L.A. Assessment of Post-Exertional Malaise (PEM) in Patients with Myalgic Encephalomyelitis (ME) and Chronic Fatigue Syndrome (CFS): A Patient-Driven Survey. Diagnostics 2019, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterwijck, J.; Marušič, U.; De Wandele, I.; Paul, L.; Meeus, M.; Moorkens, G.; Lambrecht, L.; Danneels, L.; Nijs, J. The Role of Autonomic Function in Exercise-induced Endogenous Analgesia: A Case-control Study in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome and Healthy People. Pain Physician 2017, 20, E389–E399. [Google Scholar]
- Buchheit, M.; Gindre, C. Cardiac parasympathetic regulation: Respective associations with cardiorespiratory fitness and training load. Am. J. Physiol. Circ. Physiol. 2006, 291, H451–H458. [Google Scholar] [CrossRef] [PubMed]
- Kenney, W.L. Parasympathetic control of resting heart rate: Relationship to aerobic power. Med. Sci. Sports Exerc. 1985, 17, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Pichot, V.; Busso, T.; Roche, F.; Garet, M.; Costes, F.; Duverney, D.; Lacour, J.-R.; Barthélémy, J.-C. Autonomic adaptations to intensive and overload training periods: A laboratory study. Med. Sci. Sports Exerc. 2002, 34, 1660–1666. [Google Scholar] [CrossRef]
- Kouidi, E.; Haritonidis, K.; Koutlianos, N.; Deligiannis, A. Effects of athletic training on heart rate variability triangular index. Clin. Physiol. Funct. Imaging 2002, 22, 279–284. [Google Scholar] [CrossRef]
- Maciel, B.C.; Gallo, L.; Neto, J.A.M.; Filho, E.C.L.; Filho, J.T.; Manço, J.C. Parasympathetic contribution to bradycardia induced by endurance training in man. Cardiovasc. Res. 1985, 19, 642–648. [Google Scholar] [CrossRef]
- Shin, K.; Minamitani, H.; Onishi, S.; Yamazaki, H.; Lee, M. Autonomic differences between athletes and nonathletes: Spectral analysis approach. Med. Sci. Sports Exerc. 1997, 29, 1482–1490. [Google Scholar] [CrossRef]
- Hautala, A.J.; Mäkikallio, T.H.; Kiviniemi, A.; Laukkanen, R.T.; Nissilä, S.; Huikuri, H.V.; Tulppo, M.P. Cardiovascular autonomic function correlates with the response to aerobic training in healthy sedentary subjects. Am. J. Physiol. Circ. Physiol. 2003, 285, H1747–H1752. [Google Scholar] [CrossRef]
- Bouchard, C. Individual differences in the response to regular exercise. Int. J. Obes. Relat. Metab. Disord. 1995, 19 (Suppl. 4), S5–S8. [Google Scholar]
- Singh, J.P.; Larson, M.G.; O’Donnell, C.J.; Tsuji, H.; Evans, J.C.; Levy, D. Heritability of Heart Rate Variability: The Framingham Heart Study. Circulation 1999, 99, 2251–2254. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.P.; Larson, M.G.; O’Donnell, C.J.; Tsuji, H.; Corey, D.; Levy, D. Genome scan linkage results for heart rate variability (the Framingham Heart Study). Am. J. Cardiol. 2002, 90, 1290–1293. [Google Scholar] [CrossRef]
- Hosseini, S.; Vázquez-Villegas, P.; Rito-Palomares, M.; Martinez-Chapa, S.O. Advantages, Disadvantages and Modifications of Conventional ELISA. In Tunable Low-Power Low-Noise Amplifier for Healthcare Applications; Springer: Singapore, 2018; pp. 67–115. [Google Scholar]
- Gómez-Mora, E.; Carrillo, J.; Urrea, V.; Rigau, J.; Alegre, J.; Cabrera, C.; Oltra, E.; Castro-Marrero, J.; Blanco, J. Impact of Long-Term Cryopreservation on Blood Immune Cell Markers in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Implications for Biomarker Discovery. Front. Immunol. 2020, 11, 582330. [Google Scholar] [CrossRef] [PubMed]


| Variable (Unit) | Mean (SD) before IAP (n = 34) |
|---|---|
| Age (years) | 37.06 (7.9) |
| BMI (kg/m2) | 24.52 (3.2) |
| FFM (kg) | 54.45 (9.7) |
| Fat (%) | 25.04 (6.6) |
| HADS_A_(points) | 10.30 (3.8) |
| HADS_D_(points) | 8.76 (3.2) |
| BDI_(points) | 17.97 (9.1) |
| HR_(bpm) | 69.75 (7.9) |
| sBP_(mmHg) | 116.98 (12) |
| dBP_(mmHg) | 79.45 (10.8) |
| CI (l/min/m2)_ | 3.54 (0.9) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujawski, S.; Cossington, J.; Słomko, J.; Zawadka-Kunikowska, M.; Tafil-Klawe, M.; Klawe, J.J.; Buszko, K.; Jakovljevic, D.G.; Kozakiewicz, M.; Morten, K.J.; et al. Relationship between Cardiopulmonary, Mitochondrial and Autonomic Nervous System Function Improvement after an Individualised Activity Programme upon Chronic Fatigue Syndrome Patients. J. Clin. Med. 2021, 10, 1542. https://doi.org/10.3390/jcm10071542
Kujawski S, Cossington J, Słomko J, Zawadka-Kunikowska M, Tafil-Klawe M, Klawe JJ, Buszko K, Jakovljevic DG, Kozakiewicz M, Morten KJ, et al. Relationship between Cardiopulmonary, Mitochondrial and Autonomic Nervous System Function Improvement after an Individualised Activity Programme upon Chronic Fatigue Syndrome Patients. Journal of Clinical Medicine. 2021; 10(7):1542. https://doi.org/10.3390/jcm10071542
Chicago/Turabian StyleKujawski, Sławomir, Jo Cossington, Joanna Słomko, Monika Zawadka-Kunikowska, Małgorzata Tafil-Klawe, Jacek J. Klawe, Katarzyna Buszko, Djordje G. Jakovljevic, Mariusz Kozakiewicz, Karl J. Morten, and et al. 2021. "Relationship between Cardiopulmonary, Mitochondrial and Autonomic Nervous System Function Improvement after an Individualised Activity Programme upon Chronic Fatigue Syndrome Patients" Journal of Clinical Medicine 10, no. 7: 1542. https://doi.org/10.3390/jcm10071542
APA StyleKujawski, S., Cossington, J., Słomko, J., Zawadka-Kunikowska, M., Tafil-Klawe, M., Klawe, J. J., Buszko, K., Jakovljevic, D. G., Kozakiewicz, M., Morten, K. J., Dawes, H., Strong, J. W. L., Murovska, M., Van Oosterwijck, J., Estevez-Lopez, F., Newton, J. L., Hodges, L., Zalewski, P., & on behalf of the European Network on ME/CFS (EUROMENE). (2021). Relationship between Cardiopulmonary, Mitochondrial and Autonomic Nervous System Function Improvement after an Individualised Activity Programme upon Chronic Fatigue Syndrome Patients. Journal of Clinical Medicine, 10(7), 1542. https://doi.org/10.3390/jcm10071542











