Cutaneous Manifestations in Biological-Treated Inflammatory Bowel Disease Patients: A Narrative Review
Abstract
1. Introduction
2. Pathogenetic Insights
3. Infusion and Injection-Site Reactions
Management
4. Eczema
Management
5. Cutaneous Infections
Management
6. Psoriasiform Reactions
Management
7. Cutaneous Malignancies
Management
8. Lupus-Like Syndrome
Management
9. Vasculitis (Incl Erythema Elevatum Diutinum)
Management
10. Lichenoid Reactions
Management
11. Granulomatous Reactions
Management
12. Alopecia Areata/Totalis and Vitiligo
Management
13. Erythema Multiforme–Stevens Johnson Syndrome–Toxic Epidermal Necrolysis
Management
14. Dermatomyositis
Management
15. Diagnosis
16. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chateau, T.; Bonovas, S.; Le Berre, C.; Mathieu, N.; Danese, S.; Peyrin-Biroulet, L. Vedolizumab Treatment in Extra-Intestinal Manifestations in Inflammatory Bowel Disease: A Systematic Review. J. Crohns Colitis 2019, 13, 1569–1577. [Google Scholar] [CrossRef] [PubMed]
- Severs, M.; van Erp, S.J.H.; van der Valk, M.E.; Mangen, M.J.J.; Fidder, H.H.; van der Have, M.; van Bodegraven, A.A.; de Jong, D.J.; van der Woude, C.J.; Romberg-Camps, M.J.L.; et al. Smoking Is Associated With Extra-Intestinal Manifestations in Inflammatory Bowel Disease. J. Crohns Colitis 2016, 10, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.L.; Chandra, S.; Shih, D.Q. Skin Manifestations of Inflammatory Bowel Disease. Front. Physiol. 2012, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Greuter, T.; Navarini, A.; Vavricka, S.R. Skin Manifestations of Inflammatory Bowel Disease. Clin. Rev. Allergy Immunol. 2017, 53, 413–427. [Google Scholar] [CrossRef]
- Harbord, M.; Annese, V.; Vavricka, S.R.; Allez, M.; Barreiro-de Acosta, M.; Boberg, K.M.; Burisch, J.; De Vos, M.; De Vries, A.-M.; Dick, A.D.; et al. The First European Evidence-Based Consensus on Extra-Intestinal Manifestations in Inflammatory Bowel Disease. J. Crohns Colitis 2016, 10, 239–254. [Google Scholar] [CrossRef]
- Sridhar, S.; Maltz, R.M.; Boyle, B.; Kim, S.C. Dermatological Manifestations in Pediatric Patients with Inflammatory Bowel Diseases on Anti-TNF Therapy. Inflamm. Bowel Dis. 2018, 24, 2086–2092. [Google Scholar] [CrossRef]
- Cossio, M.-L.; Genois, A.; Jantchou, P.; Hatami, A.; Deslandres, C.; McCuaig, C. Skin Manifestations in Pediatric Patients Treated With a TNF-Alpha Inhibitor for Inflammatory Bowel Disease: A Retrospective Study [Formula: See Text]. J. Cutan Med. Surg. 2020, 24, 333–339. [Google Scholar] [CrossRef]
- Bremmer, M.; Deng, A.; Gaspari, A.A. A Mechanism-Based Classification of Dermatologic Reactions to Biologic Agents Used in the Treatment of Cutaneous Disease: Part 1. Dermatitis 2009, 20, 182–192. [Google Scholar] [CrossRef]
- Moustou, A.-E.; Matekovits, A.; Dessinioti, C.; Antoniou, C.; Sfikakis, P.P.; Stratigos, A.J. Cutaneous Side Effects of Anti-Tumor Necrosis Factor Biologic Therapy: A Clinical Review. J. Am. Acad. Dermatol. 2009, 61, 486–504. [Google Scholar] [CrossRef]
- Hindryckx, P.; Novak, G.; Costanzo, A.; Danese, S. Disease-Related and Drug-Induced Skin Manifestations in Inflammatory Bowel Disease. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 203–214. [Google Scholar] [CrossRef]
- Cheifetz, A.; Smedley, M.; Martin, S.; Reiter, M.; Leone, G.; Mayer, L.; Plevy, S. The Incidence and Management of Infusion Reactions to Infliximab: A Large Center Experience. Am. J. Gastroenterol. 2003, 98, 1315–1324. [Google Scholar] [CrossRef]
- Nigam, G.B.; Bhandare, A.P.; Antoniou, G.A.; Limdi, J.K. Systematic Review and Meta-Analysis of Dermatological Reactions in Patients with Inflammatory Bowel Disease Treated with Anti-Tumour Necrosis Factor Therapy. Eur. J. Gastroenterol. Hepatol. 2021, 33, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Nyboe Andersen, N.; Pasternak, B.; Basit, S.; Andersson, M.; Svanström, H.; Caspersen, S.; Munkholm, P.; Hviid, A.; Jess, T. Association between Tumor Necrosis Factor-α Antagonists and Risk of Cancer in Patients with Inflammatory Bowel Disease. JAMA 2014, 311, 2406–2413. [Google Scholar] [CrossRef]
- Long, M.D.; Martin, C.F.; Pipkin, C.A.; Herfarth, H.H.; Sandler, R.S.; Kappelman, M.D. Risk of Melanoma and Nonmelanoma Skin Cancer among Patients with Inflammatory Bowel Disease. Gastroenterology 2012, 143, 390–399.e1. [Google Scholar] [CrossRef]
- Scharl, S.; Barthel, C.; Rossel, J.-B.; Biedermann, L.; Misselwitz, B.; Schoepfer, A.M.; Straumann, A.; Vavricka, S.R.; Rogler, G.; Scharl, M.; et al. Malignancies in Inflammatory Bowel Disease: Frequency, Incidence and Risk Factors-Results from the Swiss IBD Cohort Study. Am. J. Gastroenterol. 2019, 114, 116–126. [Google Scholar] [CrossRef]
- De Bandt, M.; Sibilia, J.; Le Loët, X.; Prouzeau, S.; Fautrel, B.; Marcelli, C.; Boucquillard, E.; Siame, J.L.; Mariette, X. Club Rhumatismes et Inflammation Systemic Lupus Erythematosus Induced by Anti-Tumour Necrosis Factor Alpha Therapy: A French National Survey. Arthritis Res. Ther. 2005, 7, R545–R551. [Google Scholar] [CrossRef]
- Picardo, S.; So, K.; Venugopal, K. Anti-TNF-Induced Lupus in Patients with Inflammatory Bowel Disease. JGH Open 2020, 4, 507–510. [Google Scholar] [CrossRef]
- Almoallim, H.; Al-Ghamdi, Y.; Almaghrabi, H.; Alyasi, O. Anti-Tumor Necrosis Factor-α Induced Systemic Lupus Erythematosus. Open Rheumatol. J. 2012, 6, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Pichler, W.J. Adverse Side-Effects to Biological Agents. Allergy 2006, 61, 912–920. [Google Scholar] [CrossRef]
- Baert, F.; Noman, M.; Vermeire, S.; Van Assche, G.; D’ Haens, G.; Carbonez, A.; Rutgeerts, P. Influence of Immunogenicity on the Long-Term Efficacy of Infliximab in Crohn’s Disease. N. Engl. J. Med. 2003, 348, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, P.E.; van der Heijde, D.M.; St Clair, E.W.; Furst, D.E.; Breedveld, F.C.; Kalden, J.R.; Smolen, J.S.; Weisman, M.; Emery, P.; Feldmann, M.; et al. Infliximab and Methotrexate in the Treatment of Rheumatoid Arthritis. Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group. N. Engl. J. Med. 2000, 343, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Garcovich, S.; De Simone, C.; Genovese, G.; Berti, E.; Cugno, M.; Marzano, A.V. Paradoxical Skin Reactions to Biologics in Patients With Rheumatologic Disorders. Front. Pharmacol. 2019, 10, 282. [Google Scholar] [CrossRef] [PubMed]
- van der Laken, C.J.; Voskuyl, A.E.; Roos, J.C.; Stigter van Walsum, M.; de Groot, E.R.; Wolbink, G.; Dijkmans, B.A.C.; Aarden, L.A. Imaging and Serum Analysis of Immune Complex Formation of Radiolabelled Infliximab and Anti-Infliximab in Responders and Non-Responders to Therapy for Rheumatoid Arthritis. Ann. Rheum. Dis. 2007, 66, 253–256. [Google Scholar] [CrossRef]
- Garcia-Romo, G.S.; Caielli, S.; Vega, B.; Connolly, J.; Allantaz, F.; Xu, Z.; Punaro, M.; Baisch, J.; Guiducci, C.; Coffman, R.L.; et al. Netting Neutrophils Are Major Inducers of Type I IFN Production in Pediatric Systemic Lupus Erythematosus. Sci. Transl. Med. 2011, 3, 73ra20. [Google Scholar] [CrossRef]
- Palucka, A.K.; Blanck, J.-P.; Bennett, L.; Pascual, V.; Banchereau, J. Cross-Regulation of TNF and IFN-Alpha in Autoimmune Diseases. Proc. Natl. Acad. Sci. USA 2005, 102, 3372–3377. [Google Scholar] [CrossRef]
- Checkley, L.A.; Kristofek, L.; Kile, S.; Bolgar, W. Incidence and Management of Infusion Reactions to Infliximab in an Alternate Care Setting. Dig. Dis. Sci. 2019, 64, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Kerbleski, J.F.; Gottlieb, A.B. Dermatological Complications and Safety of Anti-TNF Treatments. Gut 2009, 58, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Umetsu, R.; Kato, Y.; Hane, Y.; Sasaoka, S.; Motooka, Y.; Hatahira, H.; Abe, J.; Fukuda, A.; Naganuma, M.; et al. Age-Related Trends in Injection Site Reaction Incidence Induced by the Tumor Necrosis Factor-α (TNF-α) Inhibitors Etanercept and Adalimumab: The Food and Drug Administration Adverse Event Reporting System, 2004–2015. Int. J. Med. Sci. 2017, 14, 102–109. [Google Scholar] [CrossRef]
- Thomaidou, E.; Ramot, Y. Injection Site Reactions with the Use of Biological Agents. Dermatol. Ther. 2019, 32, e12817. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Baert, F.; Danese, S.; Krznarić, Ž.; Kobayashi, T.; Yao, X.; Chen, J.; Rosario, M.; Bhatia, S.; Kisfalvi, K.; et al. Efficacy and Safety of Vedolizumab Subcutaneous Formulation in a Randomized Trial of Patients With Ulcerative Colitis. Gastroenterology 2020, 158, 562–572.e12. [Google Scholar] [CrossRef]
- Fumery, M.; Tilmant, M.; Yzet, C.; Brazier, F.; Loreau, J.; Turpin, J.; Le Mouel, J.P.; Goeb, V.; Nguyen-Khac, E.; Singh, S.; et al. Premedication as Primary Prophylaxis Does Not Influence the Risk of Acute Infliximab Infusion Reactions in Immune-Mediated Inflammatory Diseases: A Systematic Review and Meta-Analysis. Dig. Liver Dis. 2019, 51, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Sator, P. Safety and Tolerability of Adalimumab for the Treatment of Psoriasis: A Review Summarizing 15 Years of Real-Life Experience. Ther. Adv. Chronic Dis. 2018, 9, 147–158. [Google Scholar] [CrossRef]
- Nakamura, M.; Lee, K.; Singh, R.; Zhu, T.H.; Farahnik, B.; Abrouk, M.; Koo, J.; Bhutani, T. Eczema as an Adverse Effect of Anti-TNFα Therapy in Psoriasis and Other Th1-Mediated Diseases: A Review. J. Dermatolog. Treat. 2017, 28, 237–241. [Google Scholar] [CrossRef]
- Segaert, S.; Hermans, C. Clinical Signs, Pathophysiology and Management of Cutaneous Side Effects of Anti-Tumor Necrosis Factor Agents. Am. J. Clin. Dermatol. 2017, 18, 771–787. [Google Scholar] [CrossRef]
- Flendrie, M.; Vissers, W.H.P.M.; Creemers, M.C.W.; de Jong, E.M.G.J.; van de Kerkhof, P.C.M.; van Riel, P.L.C.M. Dermatological Conditions during TNF-Alpha-Blocking Therapy in Patients with Rheumatoid Arthritis: A Prospective Study. Arthritis Res. Ther. 2005, 7, R666–R676. [Google Scholar] [CrossRef]
- Lynch, J.P.; Ferreira, M.A.; Phipps, S. Th2/Th17 Reciprocal Regulation: Twists and Turns in the Complexity of Asthma Phenotypes. Ann. Transl. Med. 2016, 4, S59. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Foulkes, A.C.; Mason, K.; Smith, C.H.; Griffiths, C.E.M.; Warren, R.B. Phenotypic Switch to Eczema in Patients Receiving Biologics for Plaque Psoriasis: A Systematic Review. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 1440–1448. [Google Scholar] [CrossRef]
- Ramos, J.M.; García-Sepulcre, M.F.; Rodríguez, J.C.; Padilla, S.; Gutiérrez, F. Mycobacterium Marinum Infection Complicated by Anti-Tumour Necrosis Factor Therapy. J. Med. Microbiol. 2010, 59, 617–621. [Google Scholar] [CrossRef]
- Nguyen, E.D.; Gabel, C.K.; Kroshinsky, D. Assessing the Incidence of Skin and Soft Tissue Infection in Patients on Biologics. J. Am. Acad. Dermatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wintzell, V.; Svanström, H.; Melbye, M.; Jess, T.; Olén, O.; Ludvigsson, J.F.; Pasternak, B. Use of Tumour Necrosis Factor-α Inhibitors and the Risk of Serious Infection in Paediatric Inflammatory Bowel Disease in Denmark: A Nationwide Cohort Study. Lancet Gastroenterol. Hepatol. 2019, 4, 845–853. [Google Scholar] [CrossRef]
- Ham, M.; Cullen, G.; Cheifetz, A.S. Varicella Zoster Virus Infection in Patients with Inflammatory Bowel Disease. Gastroenterol. Hepatol. (N Y) 2013, 9, 56–58. [Google Scholar]
- Nyboe Andersen, N.; Pasternak, B.; Friis-Møller, N.; Andersson, M.; Jess, T. Association between Tumour Necrosis Factor-α Inhibitors and Risk of Serious Infections in People with Inflammatory Bowel Disease: Nationwide Danish Cohort Study. BMJ 2015, 350, h2809. [Google Scholar] [CrossRef]
- Fréling, E.; Baumann, C.; Cuny, J.-F.; Bigard, M.-A.; Schmutz, J.-L.; Barbaud, A.; Peyrin-Biroulet, L. Cumulative Incidence of, Risk Factors for, and Outcome of Dermatological Complications of Anti-TNF Therapy in Inflammatory Bowel Disease: A 14-Year Experience. Am. J. Gastroenterol. 2015, 110, 1186–1196. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, D.; Guidi, L.; Ferraro, P.M.; Marzo, M.; Felice, C.; Celleno, L.; Landi, R.; Andrisani, G.; Pizzolante, F.; De Vitis, I.; et al. Paradoxical Psoriasis in a Large Cohort of Patients with Inflammatory Bowel Disease Receiving Treatment with Anti-TNF Alpha: 5-Year Follow-up Study. Aliment. Pharmacol. Ther. 2015, 42, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Cleynen, I.; Van Moerkercke, W.; Billiet, T.; Vandecandelaere, P.; Vande Casteele, N.; Breynaert, C.; Ballet, V.; Ferrante, M.; Noman, M.; Assche, G.V.; et al. Characteristics of Skin Lesions Associated With Anti-Tumor Necrosis Factor Therapy in Patients With Inflammatory Bowel Disease: A Cohort Study. Ann. Intern. Med. 2016, 164, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Richette, P.; Viguier, M.; Bachelez, H.; Bardin, T. Psoriasis Induced by Anti-Tumor Necrosis Factor Therapy: A Class Effect? J. Rheumatol. 2007, 34, 438–439. [Google Scholar] [PubMed]
- Ezzedine, K.; Visseaux, L.; Cadiot, G.; Brixi, H.; Bernard, P.; Reguiai, Z. Ustekinumab for Skin Reactions Associated with Anti-Tumor Necrosis Factor-α Agents in Patients with Inflammatory Bowel Diseases: A Single-Center Retrospective Study. J. Dermatol. 2019, 46, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Pijls, P.A.R.R.; Gilissen, L.P.L. Vedolizumab Is an Effective Alternative in Inflammatory Bowel Disease Patients with Anti-TNF-Alpha Therapy-Induced Dermatological Side Effects. Dig. Liver Dis. 2016, 48, 1391–1393. [Google Scholar] [CrossRef]
- Anderson, A.; Ferris, L.K.; Click, B.; Ramos-Rivers, C.; Koutroubakis, I.E.; Hashash, J.G.; Dunn, M.; Barrie, A.; Schwartz, M.; Regueiro, M.; et al. Low Rates of Dermatologic Care and Skin Cancer Screening Among Inflammatory Bowel Disease Patients. Dig. Dis. Sci. 2018, 63, 2729–2739. [Google Scholar] [CrossRef]
- Greuter, T.; Vavricka, S.; König, A.O.; Beaugerie, L.; Scharl, M. Swiss IBDnet, an official working group of the Swiss Society of Gastroenterology Malignancies in Inflammatory Bowel Disease. Digestion 2020, 101 (Suppl. 1), 136–145. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Feagan, B.G.; Danese, S.; O’Brien, C.D.; Ott, E.; Marano, C.; Baker, T.; Zhou, Y.; Volger, S.; Tikhonov, I.; et al. Safety of Ustekinumab in Inflammatory Bowel Disease: Pooled Safety Analysis of Results from Phase 2/3 Studies. Inflamm. Bowel Dis. 2020. [Google Scholar] [CrossRef]
- Pudipeddi, A.; Kariyawasam, V.; Haifer, C.; Baraty, B.; Paramsothy, S.; Leong, R.W. Safety of Drugs Used for the Treatment of Crohn’s Disease. Expert Opin. Drug Saf. 2019, 18, 357–367. [Google Scholar] [CrossRef]
- van den Heuvel, T.R.A.; Wintjens, D.S.J.; Jeuring, S.F.G.; Wassink, M.H.H.; Romberg-Camps, M.J.L.; Oostenbrug, L.E.; Sanduleanu, S.; Hameeteman, W.H.; Zeegers, M.P.; Masclee, A.A.; et al. Inflammatory Bowel Disease, Cancer and Medication: Cancer Risk in the Dutch Population-Based IBDSL Cohort. Int. J. Cancer 2016, 139, 1270–1280. [Google Scholar] [CrossRef]
- Farraye, F.A.; Melmed, G.Y.; Lichtenstein, G.R.; Kane, S.V. ACG Clinical Guideline: Preventive Care in Inflammatory Bowel Disease. Am. J. Gastroenterol. 2017, 112, 241–258. [Google Scholar] [CrossRef]
- De Bandt, M. Anti-TNF-Alpha-Induced Lupus. Arthritis Res. Ther. 2019, 21, 235. [Google Scholar] [CrossRef]
- Bolton, C.; Chen, Y.; Hawthorne, R.; Schepel, I.R.M.; Harriss, E.; Hofmann, S.C.; Ellis, S.; Clarke, A.; Wace, H.; Martin, B.; et al. Systematic Review: Monoclonal Antibody-Induced Subacute Cutaneous Lupus Erythematosus. Drugs R D 2020, 20, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Tierney, E.; Kirthi, S.; Ramsay, B.; Ahmad, K. Ustekinumab-Induced Subacute Cutaneous Lupus. JAAD Case Rep. 2019, 5, 271–273. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goh, S.W.; Jamil, A.; Nor, N.M.; Cader, R.A.; Shaharir, S.S. Exacerbation of Systemic Lupus Erythematosus in a Patient with Concomitant Chronic Plaque Psoriasis Treated with Ustekinumab. Indian J. Dermatol. Venereol. Leprol. 2020, 86, 68–70. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y. State-of-the-Art Treatment of Systemic Lupus Erythematosus. Int. J. Rheum Dis. 2020, 23, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Bolton, C.; Mouyis, K.; Bhamra, K.; Steuer, A. The First Case of Natalizumab-Induced Subacute Cutaenous Lupus Erythematosus. Rheumatology (Oxf.) 2020, 59, 1446–1447. [Google Scholar] [CrossRef] [PubMed]
- Alexandra, J.; Akash, G.; Heitham, A.-B. P-016 Vedolizumab and Drug-Induced Lupus: An Unexpected Phenomenon. Inflamm. Bowel Dis. 2017, 23, S10–S11. [Google Scholar] [CrossRef]
- Cohen, R.D.; Bhayat, F.; Blake, A.; Travis, S. The Safety Profile of Vedolizumab in Ulcerative Colitis and Crohn’s Disease: 4 Years of Global Post-Marketing Data. J. Crohns Colitis 2020, 14, 192–204. [Google Scholar] [CrossRef]
- Saint Marcoux, B.; De Bandt, M. CRI (Club Rhumatismes et Inflammation) Vasculitides Induced by TNFalpha Antagonists: A Study in 39 Patients in France. Jt. Bone Spine 2006, 73, 710–713. [Google Scholar] [CrossRef]
- Mohan, N.; Edwards, E.T.; Cupps, T.R.; Slifman, N.; Lee, J.-H.; Siegel, J.N.; Braun, M.M. Leukocytoclastic Vasculitis Associated with Tumor Necrosis Factor-Alpha Blocking Agents. J. Rheumatol. 2004, 31, 1955–1958. [Google Scholar]
- Asarch, A.; Gottlieb, A.B.; Lee, J.; Masterpol, K.S.; Scheinman, P.L.; Stadecker, M.J.; Massarotti, E.M.; Bush, M.L. Lichen Planus-like Eruptions: An Emerging Side Effect of Tumor Necrosis Factor-Alpha Antagonists. J. Am. Acad. Dermatol. 2009, 61, 104–111. [Google Scholar] [CrossRef]
- Darrigade, A.S.; Goussot, J.-F.; Milpied, B.; Taieb, A.; Seneschal, J. Pigmented Lichenoid Drug Eruption: A New Clinical Presentation of Interface Dermatitis Induced by Anti-TNF Alpha Drugs. Eur. J. Dermatol. 2016, 26, 633–634. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.; Basile, A.; Bair, B.; Fivenson, D. Lichenoid Reactions in Association with Tumor Necrosis Factor Alpha Inhibitors. J. Clin. Aesthet Dermatol. 2015, 8, 45–49. [Google Scholar] [PubMed]
- Dodiuk-Gad, R.P.; Shear, N.H. Granulomatous Drug Eruptions. Dermatol. Clin. 2015, 33, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Deng, A.; Harvey, V.; Sina, B.; Strobel, D.; Badros, A.; Junkins-Hopkins, J.M.; Samuels, A.; Oghilikhan, M.; Gaspari, A. Interstitial Granulomatous Dermatitis Associated with the Use of Tumor Necrosis Factor Alpha Inhibitors. Arch. Dermatol. 2006, 142, 198–202. [Google Scholar] [CrossRef]
- Cassone, G.; Citriniti, G.; Sebastiani, M.; Manfredi, A.; Cesinaro, A.; Ferri, C. Reactive Granulomatous Dermatitis during Anti-TNF Therapy: A Case Report and Review of the Literature. Clin. Med. Rev. Case Rep. 2017, 4, 172. [Google Scholar]
- Ribeiro, L.B.P.; Rego, J.C.G.; Estrada, B.D.; Bastos, P.R.; Piñeiro Maceira, J.M.; Sodré, C.T. Alopecia Secondary to Anti-Tumor Necrosis Factor-Alpha Therapy. An. Bras. Dermatol. 2015, 90, 232–235. [Google Scholar] [CrossRef]
- Toda-Brito, H.; Lopes, L.; Soares-Almeida, L.; Filipe, P. Adalimumab-Induced Psoriatic Alopecia/Alopecia Areata-like Reaction in a Patient with Crohn’s Disease. Dermatol. Online J. 2015, 21, 6. [Google Scholar]
- Le Bidre, E.; Chaby, G.; Martin, L.; Perrussel, M.; Sassolas, B.; Sigal, M.-L.; Kaassis, C.; Lespessailles, E.; Nseir, A.; Estève, E. Alopecia areata during anti-TNF alpha therapy: Nine cases. Ann. Dermatol. Venereol. 2011, 138, 285–293. [Google Scholar] [CrossRef]
- Bae, J.M.; Kim, M.; Lee, H.H.; Kim, K.-J.; Shin, H.; Ju, H.J.; Kim, G.M.; Park, C.J.; Park, H.J. Increased Risk of Vitiligo Following Anti-Tumor Necrosis Factor Therapy: A 10-Year Population-Based Cohort Study. J. Investig. Dermatol. 2018, 138, 768–774. [Google Scholar] [CrossRef]
- Tauber, M.; Beneton, N.; Reygagne, P.; Bachelez, H.; Viguier, M. Alopecia Areata Developing during Ustekinumab Therapy: Report of Two Cases. Eur. J. Dermatol. 2013, 23, 912–913. [Google Scholar] [CrossRef] [PubMed]
- Verros, C.; Rallis, E.; Crowe, M. Letter: Alopecia Areata during Ustekinumab Administration: Co-Existence or an Adverse Reaction? Dermatol. Online J. 2012, 18, 14. [Google Scholar]
- Guttman-Yassky, E.; Ungar, B.; Noda, S.; Suprun, M.; Shroff, A.; Dutt, R.; Khattri, S.; Min, M.; Mansouri, Y.; Zheng, X.; et al. Extensive Alopecia Areata Is Reversed by IL-12/IL-23p40 Cytokine Antagonism. J. Allergy Clin. Immunol. 2016, 137, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Anthony, N.; Bourneau-Martin, D.; Ghamrawi, S.; Lagarce, L.; Babin, M.; Briet, M. Drug-Induced Vitiligo: A Case/Non-Case Study in Vigibase®, the WHO Pharmacovigilance Database. Fundam Clin. Pharmacol. 2020, 34, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Elkady, A.; Bonomo, L.; Amir, Y.; Vekaria, A.S.; Guttman-Yassky, E. Effective Use of Ustekinumab in a Patient with Concomitant Psoriasis, Vitiligo, and Alopecia Areata. JAAD Case Rep. 2017, 3, 477–479. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Borrás-Blasco, J.; Navarro-Ruiz, A.; Borrás, C.; Casterá, E. Adverse Cutaneous Reactions Induced by TNF-Alpha Antagonist Therapy. South Med. J. 2009, 102, 1133–1140. [Google Scholar] [CrossRef]
- Ahdout, J.; Haley, J.C.; Chiu, M.W. Erythema Multiforme during Anti-Tumor Necrosis Factor Treatment for Plaque Psoriasis. J. Am. Acad. Dermatol. 2010, 62, 874–879. [Google Scholar] [CrossRef]
- Hasegawa, A.; Abe, R. Recent Advances in Managing and Understanding Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis. F1000Res 2020, 9. [Google Scholar] [CrossRef]
- Roselli, J.; Innocenti, T.; Lynch, E.N.; Parisio, L.; Apolito, P.; Mello, T.; Macrì, G.; Milla, M.; Biagini, M.R.; Tarocchi, M.; et al. Stevens-Johnson Syndrome and Herpes Simplex Type 1 Infection during Adalimumab Therapy for Crohn’s Disease. Case Rep. Gastrointest Med. 2020, 2020, 3875024. [Google Scholar] [CrossRef]
- Tseng, C.-C.; Chang, S.-J.; Liao, W.-T.; Chan, Y.-T.; Tsai, W.-C.; Ou, T.-T.; Wu, C.-C.; Sung, W.-Y.; Hsieh, M.-C.; Yen, J.-H. Increased Cumulative Incidence of Dermatomyositis in Ulcerative Colitis: A Nationwide Cohort Study. Sci. Rep. 2016, 6, 28175. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-H.; Chang, C.; Lian, Z.-X. Polymyositis and Dermatomyositis—Challenges in Diagnosis and Management. J. Transl. Autoimmun. 2019, 2, 100018. [Google Scholar] [CrossRef] [PubMed]
- Campanilho-Marques, R.; Deakin, C.T.; Simou, S.; Papadopoulou, C.; Wedderburn, L.R.; Pilkington, C.A. Juvenile Dermatomyositis Research Group (JDRG) Retrospective Analysis of Infliximab and Adalimumab Treatment in a Large Cohort of Juvenile Dermatomyositis Patients. Arthritis Res. Ther. 2020, 22, 79. [Google Scholar] [CrossRef]
- Brunasso, A.M.G.; Aberer, W.; Massone, C. New Onset of Dermatomyositis/Polymyositis during Anti-TNF-α Therapies: A Systematic Literature Review. Sci. World J. 2014, 2014, 179180. [Google Scholar] [CrossRef] [PubMed]
- Pinal-Fernandez, I.; Kroodsma, C.T.; Mammen, A.L. Successful Treatment of Refractory Mechanic’s Hands with Ustekinumab in a Patient with the Antisynthetase Syndrome. Rheumatology (Oxf.) 2019, 58, 1307–1308. [Google Scholar] [CrossRef]
- Montoya, C.L.; Gonzalez, M.L.; Ospina, F.E.; Tobón, G.J. A Rare Case of Amyopathic Juvenile Dermatomyositis Associated With Psoriasis Successfully Treated With Ustekinumab. J. Clin. Rheumatol. 2017, 23, 129–130. [Google Scholar] [CrossRef]
- Hu, J.Z.; Billings, S.D.; Yan, D.; Fernandez, A.P. Histologic Comparison of Tumor Necrosis Factor-α Inhibitor-Induced Psoriasis and Psoriasis Vulgaris. J. Am. Acad. Dermatol. 2020, 83, 71–77. [Google Scholar] [CrossRef]
- Laga, A.C.; Vleugels, R.A.; Qureshi, A.A.; Velazquez, E.F. Histopathologic Spectrum of Psoriasiform Skin Reactions Associated with Tumor Necrosis Factor-α Inhibitor Therapy. A Study of 16 Biopsies. Am. J. Dermatopathol. 2010, 32, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Murphy, E.C.; Zahn, J.; Friedman, A.J. Is TNF-α Inhibitor-Induced Psoriasiform Dermatitis Always Psoriasiform? J. Drugs Dermatol. 2020, 19, 1009–1010. [Google Scholar] [PubMed]
- Succaria, F.; Bhawan, J. Cutaneous Side-Effects of Biologics in Immune-Mediated Disorders: A Histopathological Perspective. J. Dermatol. 2017, 44, 243–250. [Google Scholar] [CrossRef] [PubMed]
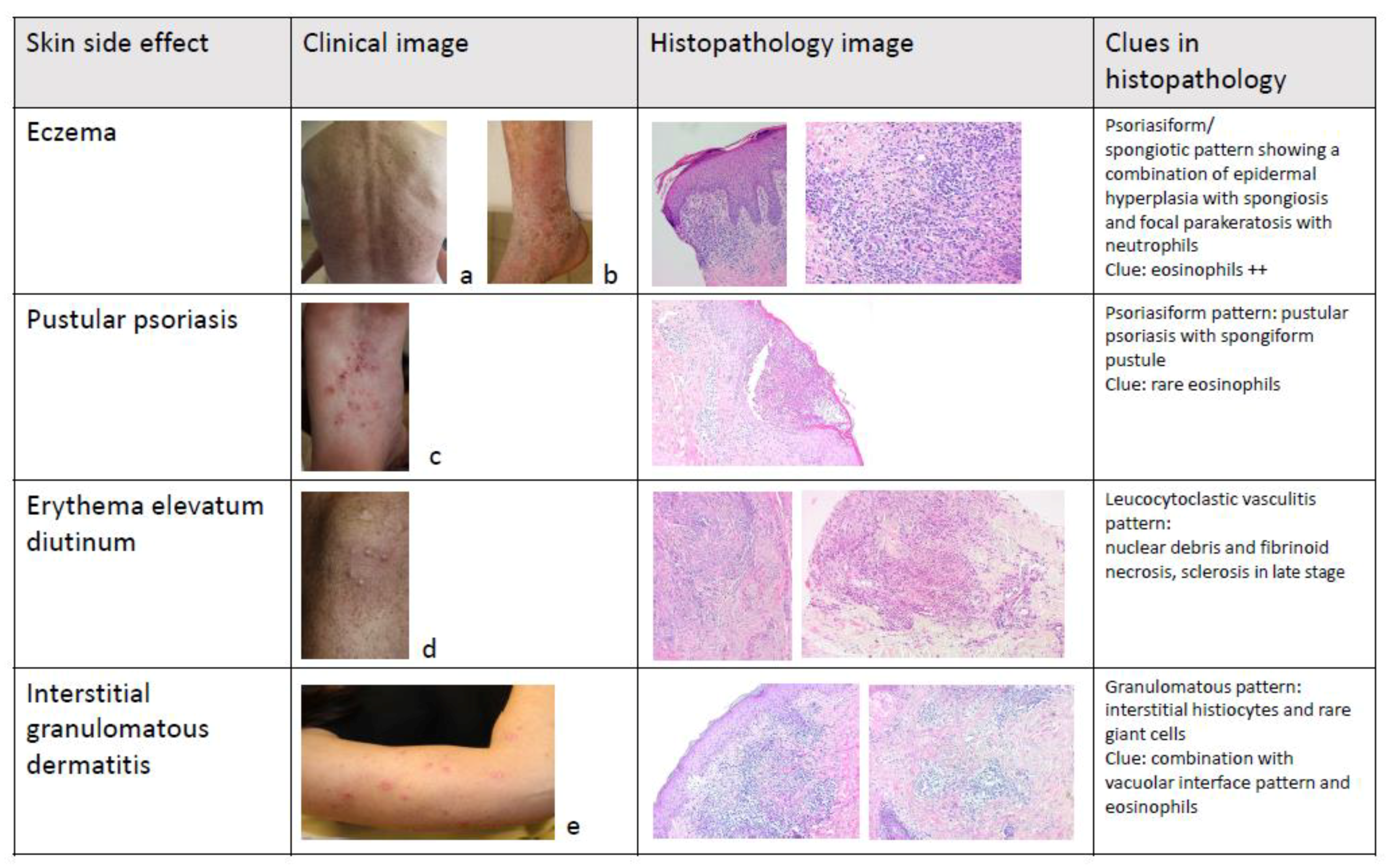
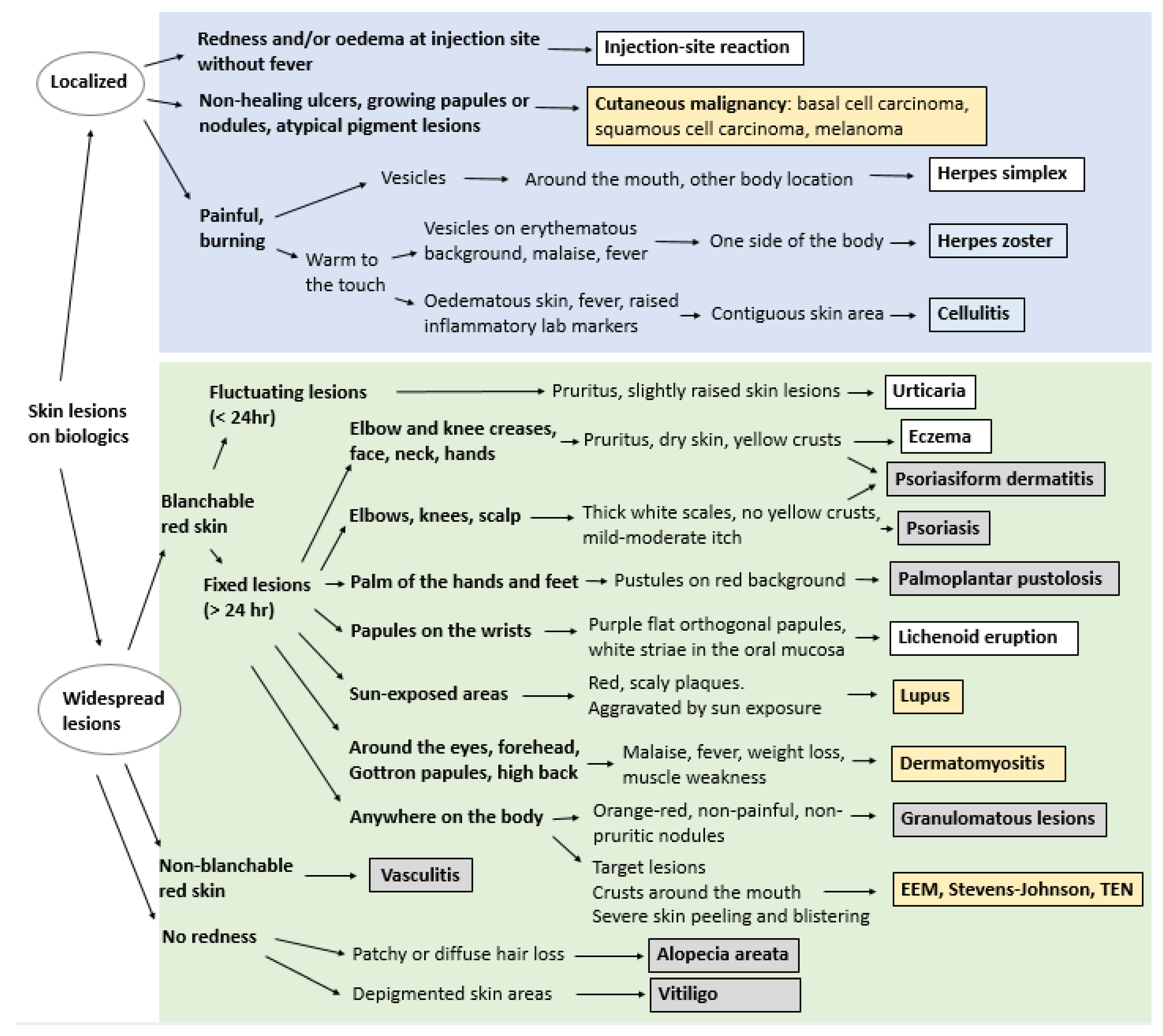
| Skin-Related Adverse Events on An-ti-TNF-α (Adalimumab, Certolizumab, Infliximab) | Incidence | Strength of Association |
|---|---|---|
| 1. Infusion reactions and injection site reactions | <5% and 10%, respectively [11] | Very strong |
| 2. Xerosis and eczema | 3–9% [12] | Moderate |
| 3. Cutaneous infections | 5–11% [12] | Strong |
| 4. Psoriasiform reactions | 4–7% [12] | Strong |
| 5. Cutaneous malignancies | 0.3–1.4% [13,14,15] | Mild |
| 6. Lupus-like syndrome | <1% [16] | Strong |
| 7. Vasculitis | <0.5% [17,18] | Strong |
| 8. Lichenoid drug reaction | Cases | Moderate |
| 9. Granulomatous reactions | Cases | Moderate |
| 10. Alopecia areata/totalis and/or vitiligo | Cases | Moderate |
| 11. Erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis | Cases | Strong |
| 12. Dermatomyositis | Cases | Strong |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lambert, J.L.W.; De Schepper, S.; Speeckaert, R. Cutaneous Manifestations in Biological-Treated Inflammatory Bowel Disease Patients: A Narrative Review. J. Clin. Med. 2021, 10, 1040. https://doi.org/10.3390/jcm10051040
Lambert JLW, De Schepper S, Speeckaert R. Cutaneous Manifestations in Biological-Treated Inflammatory Bowel Disease Patients: A Narrative Review. Journal of Clinical Medicine. 2021; 10(5):1040. https://doi.org/10.3390/jcm10051040
Chicago/Turabian StyleLambert, Jo L. W., Sofie De Schepper, and Reinhart Speeckaert. 2021. "Cutaneous Manifestations in Biological-Treated Inflammatory Bowel Disease Patients: A Narrative Review" Journal of Clinical Medicine 10, no. 5: 1040. https://doi.org/10.3390/jcm10051040
APA StyleLambert, J. L. W., De Schepper, S., & Speeckaert, R. (2021). Cutaneous Manifestations in Biological-Treated Inflammatory Bowel Disease Patients: A Narrative Review. Journal of Clinical Medicine, 10(5), 1040. https://doi.org/10.3390/jcm10051040






