Somatostatin-Dopamine Chimeric Molecules in Neuroendocrine Neoplasms
Abstract
1. Introduction
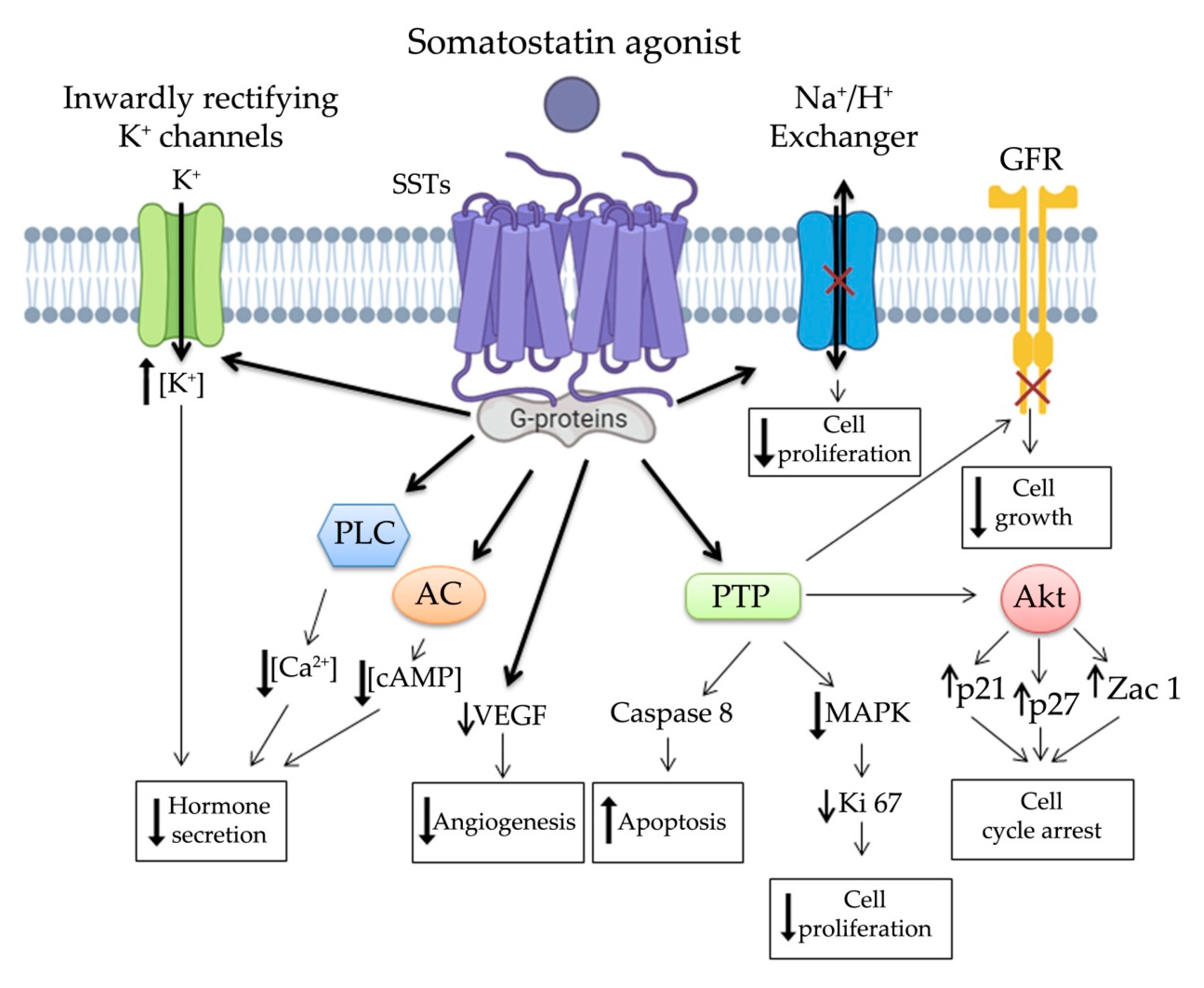
2. Somatostatin System in NENs
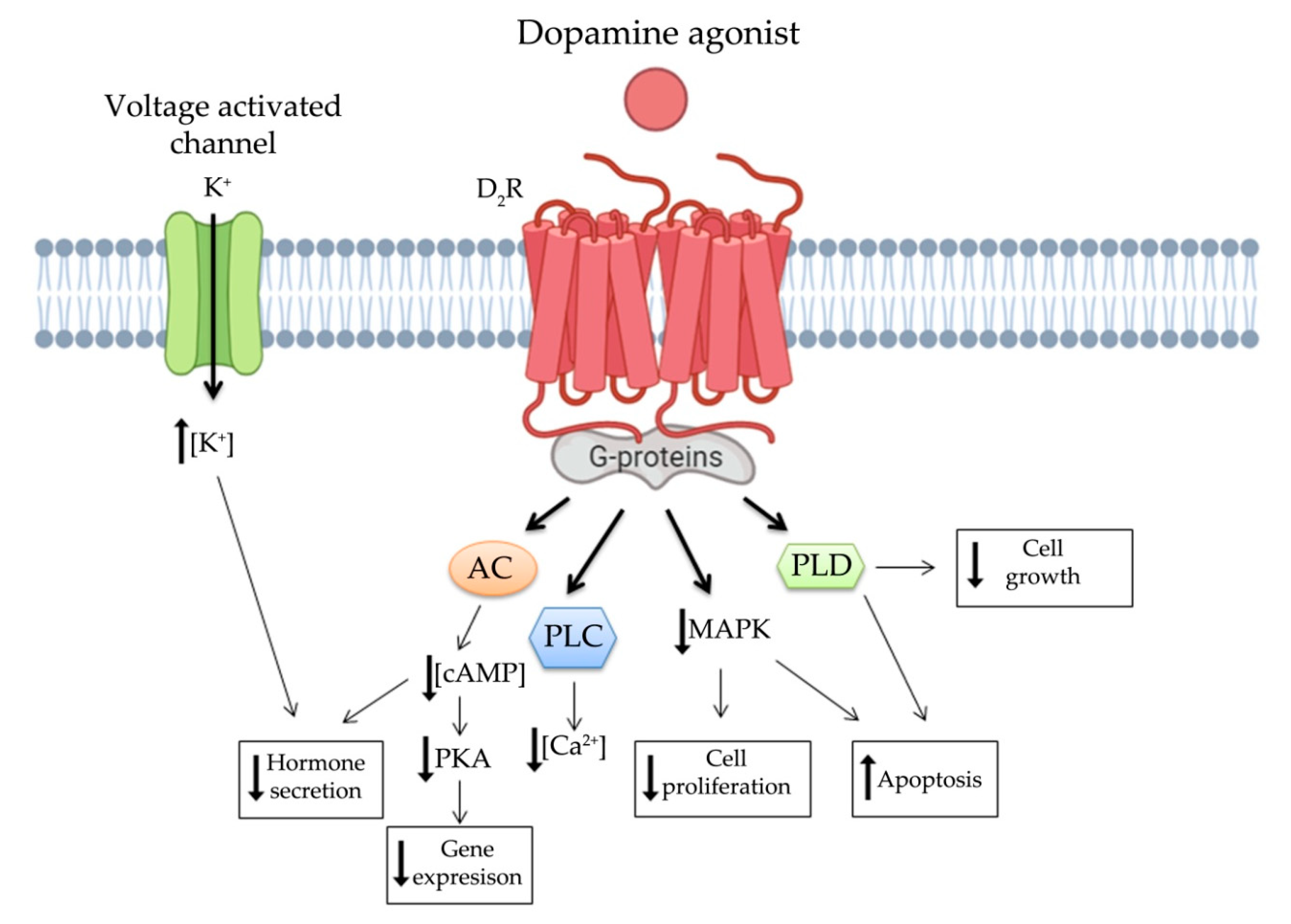
3. Dopamine System in NENs
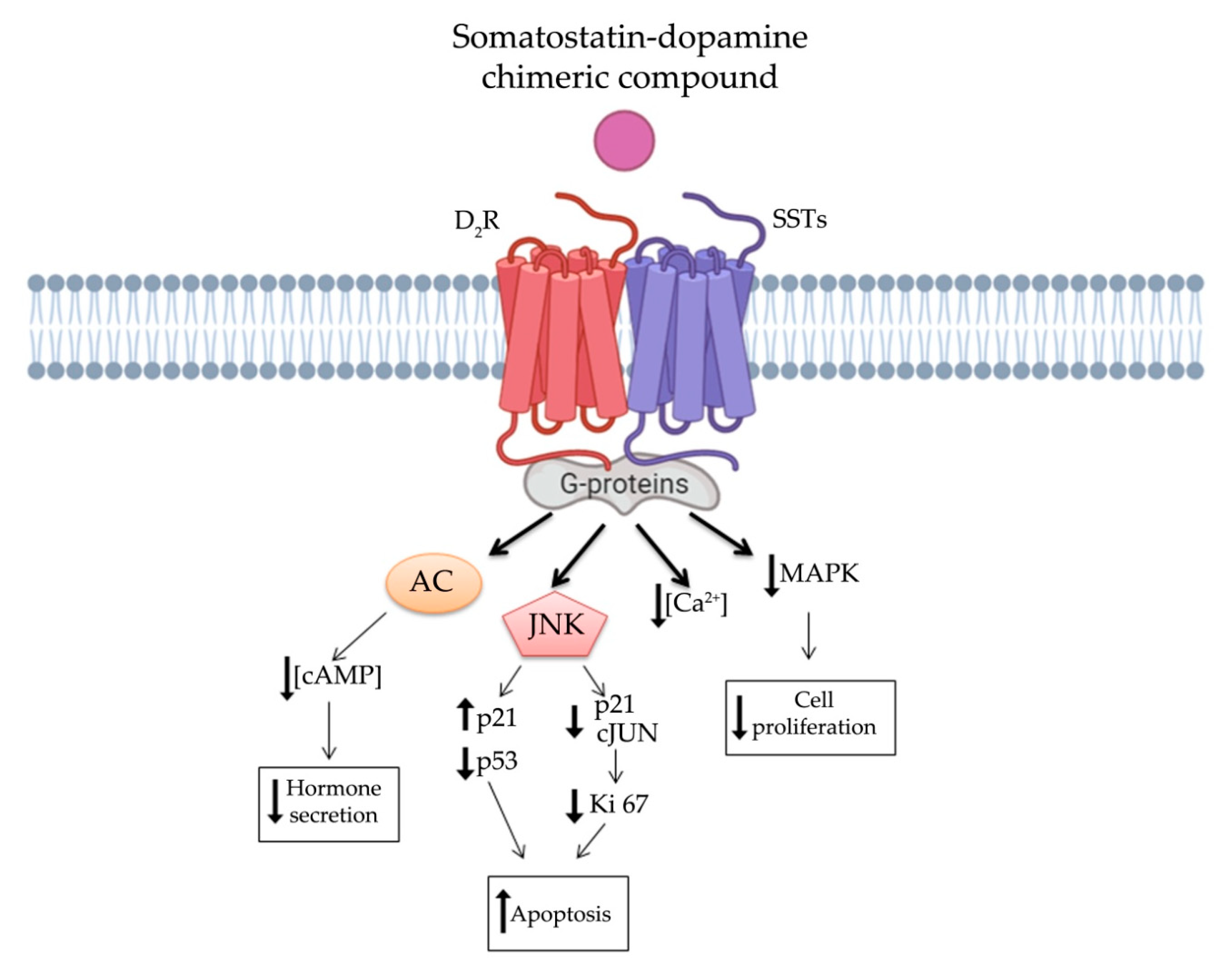
4. Somatostatin and Dopamine Receptors Interaction in NENs
5. Somatostatin-Dopamine Chimeric Compounds
5.1. First Generation Chimeras: Advantages and Limitations
5.2. Second Generation Chimeras: Advantages and Limitations
| Compound | Binding Affinity [125,127,134,137] | NEN Model | Anti-Tumor Activity | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|
| SST1 | SST2 | SST3 | SST4 | SST5 | D2R | ||||
| BIM-23A387 | 293 | 0.1 | 77.4 | ND | >1000 | 25 | Human and rat pituitary adenoma primary cells | ↓GH | [126] |
| Primary cultures of acromegaly | ↓GH | [11,127,128] | |||||||
| BIM-23A760 | 853 | 0.03 | 52 | 1000 | 3.1 | 15 | Primary cultures of acromegaly | ↓GH | [127,128] |
| Primary cultures of NFPA | ↓proliferation | [123,136,137] | |||||||
| Primary cultures of resistant prolactinoma | ↓PRL | [130,134] | |||||||
| Primary cultures of TSH-oma | ↓TSH | [131] | |||||||
| GEP NEN cell lines (BON-1 and QGP1) | ↓IGF-2 | [133] | |||||||
| Acromegaly patients (phase IIa study) | ↓GH serum levels | [140,141] | |||||||
| Broncopulmonary cell lines (NCI-H720 and NCI-H727) | ↓proliferation | [138] | |||||||
| In vivo NFPA mouse model | ↓proliferation | [139] | |||||||
| BIM-23A761 | 602 | 0.128 | 196 | >1000 | 8.7 | 25 | GEP NEN cell line (KRJ-I cell) | ↓proliferation | [138] |
| Primary cultures of acromegaly | ↓GH | [127] | |||||||
| BIM-23A758 | 549 * | 0.2 * | >1000 * | >1000 * | 43 * | 14 * | Human pancreatic NEN cells (BON-1) | ↑apoptosis, ↓Akt | [135] |
| Human midgut carcinoid cell (GOT-1) | ↑apoptosis, ↓Akt | [135] | |||||||
| Bronchopulmonary cell line (NCI-H727) | ↑apoptosis, ↓Akt | [135] | |||||||
| Primary cultures of acromegaly | ↓GH | [127] | |||||||
| BIM-23A781 | 93 | 0.8 | 11 | 42 | 4.5 | 29 | Primary cultures of acromegaly | ↓GH | [128] |
| Primary cultures of GEP NEN | ↓CgA | [132] | |||||||
| TBR-065 (BIM-23B065) | ND | 0.03 | ND | ND | 0.5 | 27.2 | Primary cultures of acromegaly | ↓cell viability, ↑apoptosis, ↓GH | [142,143,146] |
| Primary cultures of corticotropinoma | ↓cell viability, ↑apoptosis, ↓ATCH | [143] | |||||||
| Primary cultures of NFPA | ↑apoptosis, ↓CgA | [143] | |||||||
| AtT-20 cell line | ↑apoptosis | [143] | |||||||
| 3D cell models of QPG-1 and BON-1 cell | ↓ CgA, ↓Serotonin | [144] | |||||||
| MTC cell line (TT and MZ-CRC-1) | ↓cell viability, ↑apoptosis, ↓migration, ↓calcitonin | [145] | |||||||
| In vivo rat model | ↓GH | [147] | |||||||
| Healthy male volunteers (Phase I study) | ↓GH, ↓PRL and ↓IGF-I serum levels | [148,149] | |||||||
6. Conclusions
Funding
Conflicts of Interest
References
- DeLellis, R.A. The Neuroendocrine System and Its Tumors an Overview. Rev. Am. J. Clin. Pathol. 2001, 115 (Suppl. S5–S16). [Google Scholar] [CrossRef]
- Alexandraki, K.I.; Tsoli, M.; Kyriakopoulos, G.; Angelousi, A.; Nikolopoulos, G.; Kolomodi, D.; Kaltsas, G.A. Current concepts in the diagnosis and management of neuroendocrine neoplasms of unknown primary origin. Minerva Endocrinol. 2019, 44, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Tamagno, G.; Bennett, A.; Ivanovski, I. Lights and darks of neuroendocrine tumors of the appendix. Minerva Endocrinol. 2020, 45, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Rinke, A.; Wittenberg, M.; Schade-Brittinger, C.; Aminossadati, B.; Ronicke, E.; Gress, T.M.; Müller, H.H.; Arnold, R.; PROMID Study Group. Placebo-Controlled, Double-Blind, Prospective, Randomized Study on the Effect of Octreotide LAR in the Control of Tumor Growth in Patients with Metastatic Neuroendocrine Midgut Tumors (PROMID): Results of Long-Term Survival. Neuroendocrinology 2017, 104, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Caplin, M.E.; Pavel, M.; Ćwikła, J.B.; Phan, A.T.; Raderer, M.; Sedláčková, E.; Cadiot, G.; Wolin, E.M.; Capdevila, J.; Wall, L.; et al. CLARINET Investigators. Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: The CLARINET open-label extension study. Endocr. Relat. Cancer 2016, 23, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Mazziotti, G.; Mosca, A.; Frara, S.; Vitale, G.; Giustina, A. Somatostatin analogs in the treatment of neuroendocrine tumors: Current and emerging aspects. Expert Opin. Pharmacother. 2017, 18, 1679–1689. [Google Scholar] [CrossRef]
- Peillon, F.; Cesselin, F.; Bression, D.; Zygelman, N.; Brandi, A.M.; Nousbaum, A.; Mauborgne, A. In Vitro Effect of Dopamine and L-Dopa on Prolactin and Growth Hormone Release from Human Pituitary Adenomas. Clin. Endocrinol. Metab. 1979, 49, 737–741. [Google Scholar] [CrossRef]
- Ferrari, C.; Paracchi, A.; Romano, C.; Gerevini, G.; Boghen, M.; Barreca, A.; Fortini, P.; Dubini, A. Long-lasting lowering of serum growth hormone and prolactin levels by single and repetitive cabergoline administration in dopamine-responsive acromegalic patients. Clinical. Trial. Clin. Endocrinol. 1988, 29, 467–476. [Google Scholar] [CrossRef]
- Günther, T.; Tulipano, G.; Dournaud, P.; Bousquet, C.; Csaba, Z.; Kreienkamp, H.J.; Lupp, A.; Korbonits, M.; Castaño, J.P.; Wester, H.J.; et al. International Union of Basic and Clinical Pharmacology. CV. Somatostatin Receptors: Structure, Function, Ligands, and New Nomenclature. Pharmacol. Rev. 2018, 70, 763–835. [Google Scholar] [CrossRef]
- Rocheville, M.; Lange, D.C.; Kumar, U.; Patel, S.C.; Patel, R.C.; Patel, Y.C. Receptors for dopamine and somatostatin: Formation of heterooligomers with enhanced functional activity. Science 2000, 288, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Saveanu, A.; Lavaque, E.; Gunz, G.; Barlier, A.; Kim, S.; Taylor, J.E.; Culler, M.D.; Enjalbert, A.; Jaquet, P. Demonstration of Enhanced Potency of a Chimeric Somatostatin-Dopamine Molecule, BIM-23A387, in Suppressing Growth Hormone and Prolactin Secretion from Human Pituitary Somatotroph Adenoma Cells. J. Clin. Endocrinol. Metab. 2002, 87, 554. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, D. Somatostatin Receptors in the lUPHAR Compendium of Receptor Characterization and Classification; Nightingale Press: Royston, UK, 2000; pp. 354–364. [Google Scholar]
- Puig-Domingo, M.; Marazuela, M. Precision Medicine in the Treatment of Acromegaly. Minerva Endocrinol. 2019, 44, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Fayos, A.C.; García-Martínez, A.; Herrera-Martínez, A.D.; Jiménez-Vacas, J.M.; Vázquez-Borrego, M.C.; Castaño, J.P.; Picó, A.; Gahete, M.D.; Luque, R.M. Molecular Determinants of the Response to Medical Treatment of Growth Hormone Secreting Pituitary Neuroendocrine Tumors. Minerva Endocrinol. 2019, 44, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Molè, D.; Gentilin, E.; Ibañez-Costa, A.; Gagliano, T.; Gahete, M.D.; Tagliati, F.; Rossi, R.; Pelizzo, M.R.; Pansini, G.; Luque, R.M.; et al. The expression of the truncated isoform of somatostatin receptor subtype 5 associates with aggressiveness in medullary thyroid carcinoma cells. Endocrine 2015, 50, 442–452. [Google Scholar] [CrossRef]
- Luque, R.M.; Ibáñez-Costa, A.; Neto, L.V.; Taboada, G.F.; Hormaechea-Agulla, D.; Kasuki, L.; Venegas-Moreno, E.; Moreno-Carazo, A.; Gálvez, M.Á.; Soto-Moreno, A.; et al. Truncated somatostatin receptor variant sst5TMD4 confers aggressive features (proliferation, invasion and reduced octreotide response) to somatotropinomas. Cancer Lett. 2015, 359, 299–306. [Google Scholar] [CrossRef]
- Durán-Prado, M.; Saveanu, A.; Luque, R.M.; Gahete, M.D.; Gracia-Navarro, F.; Jaquet, P.; Dufour, H.; Malagón, M.M.; Culler, M.D.; Barlier, A.; et al. A potential inhibitory role for the new truncated variant of somatostatin receptor 5, sst5TMD4, in pituitary adenomas poorly responsive to somatostatin analogs. J. Clin. Endocrinol. Metab. 2010, 95, 2497–2502. [Google Scholar] [CrossRef]
- Sampedro-Núñez, M.; Luque, R.M.; Ramos-Levi, A.M.; Gahete, M.D.; Serrano-Somavilla, A.; Villa-Osaba, A.; Adrados, M.; Ibáñez-Costa, A.; Martín-Pérez, E.; Culler, M.D.; et al. Presence of sst5TMD4, a Truncated Splice Variant of the Somatostatin Receptor Subtype 5, Is Associated to Features of Increased Aggressiveness in Pancreatic Neuroendocrine Tumors. Oncotarget 2016, 7, 6593–6608. [Google Scholar] [CrossRef]
- Martinez, V. Somatostatin. In Handbook of Biologically Active Peptides: Gastrointestinal Peptides, Copyright © 2021; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 1320–1329, Section XIII, Chapter 180. [Google Scholar]
- Schonbrunn, A. Selective agonism in somatostatin receptor signaling and regulation. Mol. Cell Endocrinol. 2008, 286, 35–39. [Google Scholar] [CrossRef][Green Version]
- Siehler, S.; Hoyer, D. Characterisation of human recombinant somatostatin receptors. 2. Modulation of GTPgammaS binding. Naunyn Schmiedebergs Arch. Pharmacol. 1999, 360, 500–509. [Google Scholar] [CrossRef]
- Liu, Q.; Cescato, R.; Dewi, D.A.; Rivier, J.; Reubi, J.C.; Schonbrunn, A. Receptor signaling and endocytosis are differentially regulated by somatostatin analogs. Mol. Pharmacol. 2005, 68, 90–101. [Google Scholar] [CrossRef]
- Florio, T.; Rim, C.; Hershberger, R.E.; Loda, M.; Stork, P.J. The Somatostatin Receptor SSTR1 Is Coupled to Phosphotyrosine Phosphatase Activity in CHO-K1 Cells. Mol. Endocrinol. 1994, 8, 1289–1297. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tsuzaki, S.; Moses, A. Somatostatin Inhibits Deoxyribonucleic Acid Synthesis Induced by Both Thyrotropin and Insulin-Like Growth factor-I in FRTL5 Cells. Endocrinology 1990, 126, 3131–3138. [Google Scholar] [CrossRef] [PubMed]
- Florio, T.; Scorizello, A.; Fattore, M.; D’Alto, V.; Salzano, S.; Rossi, G.; Berlingieri, M.T.; Fusco, A.; Schettini, G. Somatostatin Inhibits PC Cl3 Thyroid Cell Proliferation through the Modulation of Phosphotyrosine Activity. Impairment of the Somatostatinergic Effects by Stable Expression of E1A. J. Biol. Chem. 1996, 271, 6129–6136. [Google Scholar] [CrossRef] [PubMed]
- Reardon, D.B.; Wood, S.L.; Brautigan, D.L.; Bell, G.I.; Dent, P.; Sturgill, T.W. Activation of a Protein Tyrosine Phosphatase and Inactivation of Raf-1 by Somatostatin. Biochem. J. 1996, 314, 401–404. [Google Scholar] [CrossRef]
- Pan, M.G.; Florio, T.; Stork, P.J. G-protein activation of a hormone-stimulated phosphatase in human tumor-cells. Science 1992, 256, 1215–1217. [Google Scholar] [CrossRef]
- Cheung, N.W.; Boyages, S.C. Somatostatin-14 and its analog octreotide exert a cytostatic effect on GH3 rat pituitary tumor cell proliferation via a transient G0/ G1 cell cycle block. Endocrinology 1995, 136, 4174–4181. [Google Scholar] [CrossRef]
- Guillermet-Guibert, J.; Saint-Laurent, N.; Davenne, L.; Rochaix, P.; Cuvillier, O.; Culler, M.D.; Pradayrol, L.; Buscail, L.; Susini, C.; Bousquet, C. Novel synergistic mechanism for sst2 somatostatin and TNFalpha receptors to induce apoptosis: Crosstalk between NF-kappaB and JNK pathways. Cell Death Differ. 2007, 14, 197–208. [Google Scholar] [CrossRef]
- Theodoropoulou, M.; Zhang, J.; Laupheimer, S.; Paez-Pereda, M.; Erneux, C.; Florio, T.; Pagotto, U.; Stalla, G.K. Octreotide, a somatostatin analogue, mediates its antiproliferative action in pituitary tumor cells by altering phosphatidylinositol 3-kinase signaling and inducing Zac1 expression. Cancer Res. 2006, 66, 1576–1582. [Google Scholar] [CrossRef]
- Lopez, F.; Estève, J.P.; Buscail, L.; Delesque, N.; Saint-Laurent, N.; Théveniau, M.; Nahmias, C.; Vaysse, N.; Susini, C. The tyrosine phosphatase SHP-1 associates with the sst2 somatostatin receptor and is an essential component of sst2-mediated inhibitory growth signaling. J. Biol. Chem. 1997, 272, 24448–24454. [Google Scholar] [CrossRef]
- Sharma, K.; Srikant, C.B. Induction of wild-type p53, Bax, and acidic endonuclease during somatostatin-signaled apoptosis in MCF-7 human breast cancer cells. Int. J. Cancer 1998, 76, 259–266. [Google Scholar] [CrossRef]
- Liu, D.; Martino, G.; Thangaraju, M.; Sharma, M.; Halwani, F.; Shen, S.H.; Patel, Y.C.; Srikant, C.B. Caspase-8-mediated intracellular acidification precedes mitochondrial dysfunction in somatostatin-induced apoptosis. J. Biol. Chem. 2000, 275, 9244–9250. [Google Scholar] [CrossRef] [PubMed]
- Thangaraju, M.; Sharma, K.; Leber, B.; Andrews, D.W.; Shen, S.H.; Srikant, C.B. Regulation of acidification and apoptosis by SHP-1 and Bcl-2. J. Biol. Chem. 1999, 274, 29549–29557. [Google Scholar] [CrossRef] [PubMed]
- Teijeiro, R.; Rios, R.; Costoya, J.A.; Castro, R.; Bello, J.L.; Devesa, J.; Arce, V.M. Activation of human somatostatin receptor 2 promotes apoptosis through a mechanism that is independent from induction of p53. Cell Physiol. Biochem. 2002, 12, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Florio, T. Molecular mechanisms of the antiproliferative activity of somatostatin receptors (SSTRs) in neuroendocrine tumors. Front. Biosci. 2008, 13, 822–840. [Google Scholar] [CrossRef][Green Version]
- Ambler, G.R.; Butler, A.A.; Padmanabhan, J.; Breier, B.H.; Gluckman, P.D. The effects of octreotide on GH receptor and IGF-I expression in the GH-deficient rat. J. Endocrinol. 1996, 149, 223–231. [Google Scholar] [CrossRef]
- Häyry, P.; Räisänen, A.; Ustinov, J.; Mennander, A.; Paavonen, T. Somatostatin analog lanreotide inhibits myocyte replication and several growth factors in allograft arteriosclerosis. FASEB J. 1993, 7, 1055–1060. [Google Scholar] [CrossRef]
- Trouillas, J.; Vasiljevic, A.; Lapoirie, M.; Chinezu, L.; Jouanneau, E.; Raverot, G. Pathological Markers of Somatotroph Pituitary Neuroendocrine Tumors Predicting the Response to Medical Treatment. Minerva Endocrinol. 2019, 44, 129–136. [Google Scholar] [CrossRef]
- Shimon, I.; Taylor, J.E.; Dong, J.Z.; Bitonte, R.A.; Kim, S.; Morgan, B.; Coy, D.H.; Culler, M.D.; Melmed, S. Somatostatin receptor subtype specificity in human fetal pituitary cultures. Differential role of SSTR2 and SSTR5 for growth hormone, thyroid-stimulating hormone, and prolactin regulation. J. Clin. Investig. 1997, 99, 789–798. [Google Scholar] [CrossRef]
- Murray, R.D.; Kim, K.; Ren, S.G.; Chelly, M.; Umehara, Y.; Melmed, S. Central and peripheral actions of somatostatin on the growth hormone-IGF-I axis. J. Clin. Invest. 2004, 114, 349–356. [Google Scholar] [CrossRef]
- Chowers, Y.; Cahalon, L.; Lahav, M.; Schor, H.; Tal, R.; Bar-Meir, S.; Levite, M. Somatostatin through its specific receptor inhibits spontaneous and TNF-alpha- and bacteria-induced IL-8 and IL-1 beta secretion from intestinal epithelial cells. J. Immunol. 2000, 165, 2955–2961. [Google Scholar] [CrossRef]
- Grimaldi, M.; Florio, T.; Schettini, G. Somatostatin inhibits interleukin 6 release from rat cortical type I astrocytes via the inhibition of adenylyl cyclase. Biochem. Biophys. Res. Commun. 1997, 235, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Florio, T.; Morini, M.; Villa, V.; Arena, S.; Corsaro, A.; Thellung, S.; Culler, M.D.; Pfeffer, U.; Noonan, D.M.; Schettini, G.; et al. Somatostatin Inhibits Tumor Angiogenesis and Growth via Somatostatin receptor-3-mediated Regulation of Endothelial Nitric Oxide Synthase and Mitogen-Activated Protein Kinase Activities. Endocrinology 2003, 144, 1574–1584. [Google Scholar] [CrossRef] [PubMed]
- Theodoropoulou, M.; Stalla, G.K. Somatostatin receptors: From signaling to clinical practice. Front. Neuroendocrinol. 2013, 34, 228–252. [Google Scholar] [CrossRef] [PubMed]
- Hukovic, N.; Panetta, R.; Kumar, U.; Patel, Y.C. Agonist-dependent regulation of cloned human somatostatin receptor types 1-5 (hSSTR1-5): Subtype selective internalization or upregulation. Endocrinology 1996, 137, 4046–4049. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Csaba, Z.; Peineau, S.; Dournaud, P. Molecular mechanisms of somatostatin receptor trafficking. J. Mol. Endocrinol. 2012, 48, R1–R12. [Google Scholar] [CrossRef] [PubMed]
- Hipkin, R.W.; Friedman, J.; Clark, R.B.; Eppler, C.M.; Schonbrunn, A. Agonist-induced desensitization, internalization, and phosphorylation of the sst2A somatostatin receptor. J. Biol. Chem. 1997, 272, 13869–13876. [Google Scholar] [CrossRef]
- Patel, Y.C.; Wheatley, T. In Vivo and In Vitro Plasma Disappearance and Metabolism of somatostatin-28 and somatostatin-14 in the Rat. Endocrinology 1983, 112, 220–225. [Google Scholar] [CrossRef]
- Bauer, W.; Briner, U.; Doepfner, W.; Haller, R.; Huguenin, R.; Marbach, P.; Petcher, T.J.; Pless, J. SMS 201-995: A Very Potent and Selective Octapeptide Analogue of Somatostatin with Prolonged Action. Life Sci. 1982, 31, 1133–1140. [Google Scholar] [CrossRef]
- Taylor, J.E.; Bogden, A.E.; Moreau, J.P.; Coy, D.H. In Vitro and In Vivo Inhibition of Human Small Cell Lung Carcinoma (NCI-H69) Growth by a Somatostatin Analogue. Biochem. Biophys. Res. Commun. 1988, 153, 81–86. [Google Scholar] [CrossRef]
- Vitale, G.; Tagliaferri, P.; Caraglia, M.; Rampone, E.; Ciccarelli, A.; Bianco, A.R.; Abbruzzese, A.; Lupoli, G. Slow Release Lanreotide in Combination with interferon-alpha2b in the Treatment of Symptomatic Advanced Medullary Thyroid Carcinoma. J. Clin. Endocrinol. Metab. 2000, 85, 983–988. [Google Scholar] [CrossRef]
- Lupoli, G.; Cascone, E.; Arlotta, F.; Vitale, G.; Celentano, L.; Salvatore, M.; Lombardi, G. Treatment of Advanced Medullary Thyroid Carcinoma with a Combination of Recombinant Interferon alpha-2b and Octreotide. Cancer 1996, 78, 1114–1118. [Google Scholar] [CrossRef]
- Hofland, J.; Herrera-Martínez, A.D.; Zandee, W.T.; de Herder, W.W. Management of carcinoid syndrome: A systematic review and meta-analysis. Endocr. Relat. Cancer 2019, 26, R145–R156. [Google Scholar] [CrossRef] [PubMed]
- Al-Efraij, K.; Aljama, M.A.; Kennecke, H.F. Association of Dose Escalation of Octreotide Long-Acting Release on Clinical Symptoms and Tumor Markers and Response among Patients with Neuroendocrine Tumors. Cancer Med. 2015, 4, 864–870. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, D.; Ducreux, M.; Bommelaer, G.; Wemeau, J.L.; Bouché, O.; Catus, F.; Blumberg, J.; Ruszniewski, P. Treatment of carcinoid syndrome: A prospective crossover evaluation of lanreotide versus octreotide in terms of efficacy, patient acceptability, and tolerance. Cancer 2000, 88, 770–776. [Google Scholar] [CrossRef]
- Cella, D.; Beaumont, J.L.; Hudgens, S.; Marteau, F.; Feuilly, M.; Houchard, A.; Lapuerta, P.; Ramage, J.; Pavel, M.; Hörsch, D.; et al. Relationship Between Symptoms and Health-related Quality-of-life Benefits in Patients with Carcinoid Syndrome: Post Hoc Analyses from TELESTAR. Clin. Ther. 2018, 40, 2006–2020. [Google Scholar] [CrossRef] [PubMed]
- Tuvia, S.; Salama, P.; Weinstein, I.; Marom, K.; Neumark, E.; Arama, M.L.; Mishli, N.; Levy, S.; Lapidot, T.; Kadoshi, R.; et al. Octreolin™, a safe oral alternative for parenteral octreotide treatment. In Proceedings of the Fifth International Congress of the GRS and the IGF Society, New York, NY, USA, 3–7 October 2010. [Google Scholar]
- Bruns, C.; Lewis, I.; Briner, U.; Meno-Tetang, G.; Weckbecker, G. SOM230: A Novel Somatostatin Peptidomimetic with Broad Somatotropin Release Inhibiting Factor (SRIF) Receptor Binding and a Unique Antisecretory Profile. Eur. J. Endocrinol. 2002, 146, 707–716. [Google Scholar] [CrossRef]
- Schmid, H.A. Pasireotide (SOM230): Development, Mechanism of Action and Potential Applications. Mol. Cell Endocrinol. 2008, 286, 69–74. [Google Scholar] [CrossRef]
- Murray, R.D.; Kim, K.; Ren, S.G.; Lewis, I.; Weckbecker, G.; Bruns, C.; Melmed, S. The novel somatostatin ligand (SOM230) Regulates Human and Rat Anterior Pituitary Hormone Secretion. J. Clin. Endocrinol. Metab. 2004, 89, 3027–3032. [Google Scholar] [CrossRef][Green Version]
- Van der Hoek, J.; de Herder, W.W.; Feelders, R.A.; van der Lely, A.J.; Uitterlinden, P.; Boerlin, V.; Bruns, C.; Poon, K.W.; Lewis, I.; Weckbecker, G.; et al. A Single-Dose Comparison of the Acute Effects between the New Somatostatin Analog SOM230 and Octreotide in Acromegalic Patients. J. Clin. Endocrinol. Metab. 2004, 89, 638–645. [Google Scholar] [CrossRef]
- Onofri, C.; Theodoropoulou, M.; Losa, M.; Uhl, E.; Lange, M.; Arzt, E.; Stalla, G.K.; Renner, U. Localization of Vascular Endothelial Growth Factor (VEGF) Receptors in Normal and Adenomatous Pituitaries: Detection of a Non-Endothelial Function of VEGF in Pituitary Tumours. J. Endocrinol. 2006, 191, 249–261. [Google Scholar] [CrossRef]
- Vidal, S.; Oliveira, M.C.; Kovacs, K.; Scheithauer, B.W.; Lloyd, R. Immunolocalization of Vascular Endothelial Growth Factor in the GH3 Cell Line. Cell Tissue Res. 2000, 300, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.; Blanchard, M.P.; Albertelli, M.; Barbieri, F.; Brue, T.; Niccoli, P.; Delpero, J.R.; Monges, G.; Garcia, S.; Ferone, D.; et al. Pasireotide and octreotide antiproliferative effects and sst2 trafficking in human pancreatic neuroendocrine tumor cultures. Endocr. Relat. Cancer 2014, 21, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Zatelli, M.C.; Piccin, D.; Vignali, C.; Tagliati, F.; Ambrosio, M.R.; Bondanelli, M.; Cimino, V.; Bianchi, A.; Schmid, H.A.; Scanarini, M.; et al. Pasireotide, a Multiple Somatostatin Receptor Subtypes Ligand, Reduces Cell Viability in Non-Functioning Pituitary Adenomas by Inhibiting Vascular Endothelial Growth Factor Secretion. Endocr. Relat. Cancer 2007, 14, 91–102. [Google Scholar] [CrossRef]
- Batista, D.L.; Zhang, X.; Gejman, R.; Ansell, P.J.; Zhou, Y.; Johnson, S.A.; Swearingen, B.; Hedley-Whyte, E.T.; Stratakis, C.A.; Klibanski, A. The Effects of SOM230 on Cell Proliferation and Adrenocorticotropin Secretion in Human Corticotroph Pituitary Adenomas. J. Clin. Endocrinol. Metab. 2006, 91, 4482–4488. [Google Scholar] [CrossRef] [PubMed]
- Stueven, A.K.; Kayser, A.; Wetz, C.; Amthauer, H.; Wree, A.; Tacke, F.; Wiedenmann, B.; Roderburg, C.; Jann, H. Somatostatin Analogues in the Treatment of Neuroendocrine Tumors: Past, Present and Future. Int. J. Mol. Sci. 2019, 20, 3049. [Google Scholar] [CrossRef]
- Cives, M.; Kunz, P.L.; Morse, B.; Coppola, D.; Schell, M.J.; Campos, T.; Nguyen, P.T.; Nandoskar, P.; Khandelwal, V.; Strosberg, J.R. Phase II clinical trial of pasireotide long-acting repeatable in patients with metastatic neuroendocrine tumors. Endocr. Relat. Cancer 2015, 22, 1–9. [Google Scholar] [CrossRef]
- Wolin, E.M.; Jarzab, B.; Eriksson, B.; Walter, T.; Toumpanakis, C.; Morse, M.A.; Tomassetti, P.; Weber, M.M.; Fogelman, D.R.; Ramage, J.; et al. Phase III study of pasireotide long-acting release. Drug Des. Devel. Ther. 2015, 9, 5075–5086. [Google Scholar] [CrossRef]
- Vitale, G.; Dicitore, A.; Sciammarella, C.; Di Molfetta, S.; Rubino, M.; Faggiano, A.; Colao, A. Pasireotide in the treatment of neuroendocrine tumors: A review of the literature. Endocr. Relat. Cancer 2018, 25, R351–R364. [Google Scholar] [CrossRef]
- Afargan, M.; Janson, E.T.; Gelerman, G.; Rosenfeld, R.; Ziv, O.; Karpov, O.; Wolf, A.; Bracha, M.; Shohat, D.; Liapakis, G.; et al. Novel Long-Acting Somatostatin Analog with Endocrine Selectivity: Potent Suppression of Growth Hormone but Not of Insulin. Endocrinology 2001, 142, 477–486. [Google Scholar] [CrossRef]
- Gazal, S.; Gelerman, G.; Ziv, O.; Karpov, O.; Litman, P.; Bracha, M.; Afargan, M.; Gilon, C. Human Somatostatin Receptor Specificity of Backbone-Cyclic Analogues Containing Novel Sulfur Building Units. J. Med. Chem. 2002, 45, 1665–1671. [Google Scholar] [CrossRef]
- Shimon, I.; Rubinek, T.; Hadani, M.; Alhadef, N. PTR-3173 (Somatoprim), a Novel Somatostatin Analog with Affinity for Somatostatin Receptors 2, 4 and 5 Is a Potent Inhibitor of Human GH Secretion. J. Endocrinol. Investig. 2004, 27, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Plöckinger, U.; Hoffmann, U.; Geese, M.; Lupp, A.; Buchfelder, M.; Flitsch, J.; Vajkoczy, P.; Jakob, W.; Saeger, W.; Schulz, S.; et al. DG3173 (Somatoprim), a Unique Somatostatin Receptor Subtypes 2-, 4- And 5-selective Analogue, Effectively Reduces GH Secretion in Human GH-secreting Pituitary Adenomas Even in Octreotide Non-Responsive Tumours. Eur. J. Endocrinol. 2012, 166, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Pivonello, R.; Ferone, D.; Lombardi, G.; Colao, A.; Lamberts, S.; Hofland, L.J. Novel insights in dopamine receptor physiology. Eur. J. Endocrinol. 2007, 156, S13–S21. [Google Scholar] [CrossRef] [PubMed]
- Missale, C.; Nash, S.R.; Robinson, S.W.; Jaber, M.; Caron, M.G. Dopamine Receptors: From Structure to Function. Physiol. Rev. 1998, 78, 189–225. [Google Scholar] [CrossRef]
- Ben-Shlomo, A.; Liu, N.A.; Melmed, S. Somatostatin and dopamine receptor regulation of pituitary somatotroph adenomas. Pituitary 2017, 20, 93–99. [Google Scholar] [CrossRef]
- Senogles, S.E. D2S dopamine receptor mediates phospholipase D and antiproliferation. Mol. Cell Endocrinol. 2003, 2209, 61–69. [Google Scholar] [CrossRef]
- De Camilli, P.; Macconi, D.; Spada, A. Dopamine inhibits adenylate cyclase in human prolactin-secreting pituitary adenomas. Nature 1979, 278, 252–254. [Google Scholar] [CrossRef]
- Bevan, J.S.; Webster, J.; Burke, C.W.; Scanlon, M.F. Dopamine Agonists and Pituitary Tumor Shrinkage. Endocr. Rev. 1992, 13, 220–240. [Google Scholar] [CrossRef]
- Choi, E.Y.; Jeong, D.; Park, K.W.; Baik, J.H. G protein-mediated mitogen-activated protein kinase activation by two dopamine D2 receptors. Biochem. Biophys. Res. Commun. 1999, 256, 33–40. [Google Scholar] [CrossRef]
- Pathak, R.D.; Tran, T.H.; Burshell, A.L. A case of dopamine agonists inhibiting pancreatic polypeptide secretion from an islet cell tumor. J. Clin. Endocrinol. Metab. 2004, 89, 581–584. [Google Scholar] [CrossRef][Green Version]
- Leng, Z.G.; Lin, S.J.; Wu, Z.R.; Guo, Y.H.; Cai, L.; Shang, H.B.; Tang, H.; Xue, Y.J.; Lou, M.Q.; Zhao, W.; et al. Activation of DRD5 (dopamine receptor D5) inhibits tumor growth by autophagic cell death. Autophagy 2017, 13, 1404–1419. [Google Scholar] [CrossRef]
- Stefaneanu, L.; Kovacs, K.; Horvath, E.; Buchfelder, M.; Fahlbusch, R.; Lancranjan, L. Dopamine D2 receptor gene expression in human adenohypophysial adenomas. Endocrine 2001, 14, 329–336. [Google Scholar] [CrossRef]
- Pivonello, C.; Rousaki, P.; Negri, M.; Sarnataro, M.; Napolitano, M.; Marino, F.Z.; Patalano, R.; De Martino, M.C.; Sciammarella, C.; Faggiano, A.; et al. Effects of the single and combined treatment with dopamine agonist, somatostatin analog and mTOR inhibitors in a human lung carcinoidcell line: An in vitro study. Endocrine 2017, 56, 603–620. [Google Scholar] [CrossRef] [PubMed]
- Mattei, A.M.; Ferrari, C.; Baroldi, P.; Cavioni, V.; Paracchi, A.; Galparoli, C.; Romano, C.; Spellecchia, D.; Gerevini, G.; Crosignani, P.G. Prolactin-lowering Effect of Acute and Once Weekly Repetitive Oral Administration of Cabergoline at Two Dose Levels in Hyperprolactinemic Patients. J. Clin. Endocrinol. Metab. 1988, 66, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Verhelst, J.; Abs, R.; Maiter, D.; van den Bruel, A.; Vandeweghe, M.; Velkeniers, B.; Mockel, J.; Lamberigts, G.; Petrossians, P.; Coremans, P.; et al. Cabergoline in the treatment of hyperprolactinemia: A study in 455 patients. J. Clin. Endocrinol. Metab. 1999, 84, 2518–2522. [Google Scholar] [CrossRef] [PubMed]
- Colao, A.; Di Sarno, A.; Landi, M.L.; Scavuzzo, F.; Cappabianca, P.; Pivonello, R.; Volpe, R.; Di Salle, F.; Cirillo, S.; Annunziato, L.; et al. Macroprolactinoma shrinkage during cabergoline treatment is greater in naive patients than in patients pretreated with other dopamine agonists: A prospective study in 110 patients. J. Clin. Endocrinol. Metab. 2000, 85, 2247–2252. [Google Scholar] [CrossRef] [PubMed]
- Maiter, D. Management of Dopamine Agonist-Resistant Prolactinoma. Neuroendocrinology 2019, 109, 42–50. [Google Scholar] [CrossRef]
- Abs, J.R.; Verhelst, D.; Maiter, K.; Van Acker, F.; Nobels, J.L.; Coolens, C.; Beckers, M.e.A. Cabergoline in the Treatment of Acromegaly: A Study in 64 Patients. J. Clin. Endocrinol. Metab. 1998, 83, 374–378. [Google Scholar] [CrossRef]
- Kuhn, E.; Chanson, P. Cabergoline in acromegaly. Pituitary 2017, 20, 121–128. [Google Scholar] [CrossRef]
- Sandret, L.; Maison, P.; Chanson, P. Place of cabergoline in acromegaly: A meta-analysis. J. Clin. Endocrinol. Metab. 2011, 96, 1327–1335. [Google Scholar] [CrossRef]
- Gadelha, M.R.; Vieira Neto, L. Efficacy of medical treatment in Cushing’s disease: A systematic review. Clin. Endocrinol. 2014, 80, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, T.; Trantakis, C.; Biesold, M.; Prothmann, S.; Guenzel, S.; Schober, R.; Paschke, R. Minor tumour shrinkage in nonfunctioning pituitary adenomas by long-term treatment with the dopamine agonist cabergoline. Pituitary 2001, 4, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.C.; Naves, L.A.; Silva, A.O.; de Castro, L.F.; Casulari, L.A.; Azevedo, M.F. Short-term treatment with cabergoline can lead to tumor shrinkage in patients with nonfunctioning pituitary adenomas. Pituitary 2013, 16, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Batista, R.L.; Musolino, N.; Cescato, V.; da Silva, G.O.; Medeiros, R.; Herkenhoff, C.; Trarbach, E.B.; Cunha-Neto, M.B. Nonfunctioning Pituitary Adenoma: A Single-Center, Open-Label, 2-Year Randomized Clinical Trial. Am. J. Clin. Oncol. 2019, 42, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Kwekkeboom, D.J.; Hofland, L.J.; van Koetsveld, P.M.; Singh, R.; van den Berge, J.H.; Lamberts, S.W. Bromocriptine increasingly suppresses the in vitro gonadotropin and alpha-subunit release from pituitary adenomas during long term culture. J. Clin. Endocrinol. Metab. 1990, 71, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Kwekkeboom, D.J.; Lamberts, S.W. Long-term treatment with the dopamine agonist CV 205-502 of patients with a clinically non-functioning, gonadotroph, or alpha-subunit secreting pituitary adenoma. Clin. Endocrinol. 1992, 36, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Farrell, W.E.; Clark, A.J.; Stewart, M.F.; Crosby, S.R.; White, A. Bromocriptine inhibits pro-opiomelanocortin mRNA and ACTH precursor secretion in small cell lung cancer cell lines. J. Clin. Investig. 1992, 90, 705–710. [Google Scholar] [CrossRef]
- Reith, P.; Monnot, E.A.; Bathija, P.J. Prolonged suppression of a corticotropin-producing bronchial Carcinoid by Oral Bromocriptine. Arch. Intern. Med. 1987, 147, 989–991. [Google Scholar] [CrossRef]
- Pivonello, R.; Ceresol, E.; Albertelli, M.; Faggiano, A.; Torre, G.; De Martino, M.C.; Lombardi, G.; Minuto, F.; Colao, A.; Ferone, D. Expression of somatostatin and dopamine receptors and effect of chimeric somatostatin–dopamine molecules on cell proliferation in pancreatic neuroendocrine tumors. In Proceedings of the 5th Annual Conference, European Neuroendocrine Tumour Society, Paris, France, 6–8 March2008. [Google Scholar]
- Reubi, J.C.; Waser, B.; Schaer, J.C.; Laissue, J.A. Somatostatin receptor sst 1 -sst 5 expression in normal and neoplastic human tissues using receptor autoradiography with subtype-selective ligands. Eur. J. Nucl. Med. 2001, 28, 836–846. [Google Scholar] [CrossRef]
- Vitale, G.; Pivonello, R.; Ferone, D.; De Martino, M.C.; Auriemma, R.S.; Caraglia, M.; Abbruzzese, A.; Lombardi, G.; Colao, A. The role of somatostatin receptors in the medical treatment of acromegaly. Dig. Liver Dis. 1995, 36 (Suppl. S1), S55–S59. [Google Scholar] [CrossRef]
- Miller, G.M.; Alexander, J.M.; Bikkal, H.A.; Katznelson, L.; Zervas, N.T.; Klibanski, A. Somatostatin receptor subtype gene expression in pituitary adenomas. J. Clin. Endocrinol. Metab. 1995, 80, 1386–1392. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hofland, L.J. Somatostatin and somatostatin receptors in Cushing’s disease. Mol. Cell Endocrinol. 2008, 286, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Reubi, J.C.; Waser, B. Concomitant expression of several peptide receptors in neuroendocrine tumours: Molecular basis for in vivo multireceptor tumour targeting. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 781–793. [Google Scholar] [CrossRef] [PubMed]
- Pivonello, R.; Ferone, D.; de Herder, W.W.; Kros, J.M.; De Caro, M.L.; Arvigo, M.; Annunziato, L.; Lombardi, G.; Colao, A.; Hofland, L.J.; et al. Dopamine receptor expression and function in corticotroph pituitary tumors. J. Clin. Endocrinol. Metab. 2004, 89, 2452–2462. [Google Scholar] [CrossRef]
- Ferone, D.; de Herder, W.W.; Pivonello, R.; Kros, J.M.; van Koetsveld, P.M.; de Jong, T.; Minuto, F.; Colao, A.; Lamberts, S.W.; Hofland, L.J. Correlation of in vitro and in vivo somatotropic adenoma responsiveness to somatostatin analogs and dopamine agonists with immunohistochemical evaluation of somatostatin and dopamine receptors and electron microscopy. J. Clin. Endocrinol. Metabol. 2008, 93, 1412–1417. [Google Scholar] [CrossRef]
- Saveanu, A.; Jaquet, P.; Brue, T.; Barlier, A. Relevance of coexpression of somatostatin and dopamine D2 receptors in pituitary adenomas. Mol. Cell Endocrinol. 2008, 286, 206–213. [Google Scholar] [CrossRef]
- Diakatou, E.; Kaltsas, G.; Tzivras, M.; Kanakis, G.; Papaliodi, E.; Kontogeorgos, G. Somatostatin and dopamine receptor profile of gastroenteropancreatic neuroendocrine tumors: An immunohistochemical study. Endocr. Pathol. 2011, 22, 24–30. [Google Scholar] [CrossRef]
- Grossrubatscher, E.; Veronese, S.; Ciaramella, P.D.; Pugliese, R.; Boniardi, M.; De Carlis, L.; Torre, M.; Ravini, M.; Gambacorta, M.; Loli, P. High expression of dopamine receptor subtype 2 in a large series of neuroendocrine tumors. Cancer Biol. Ther. 2008, 7, 1970–1978. [Google Scholar] [CrossRef][Green Version]
- Lemmer, K.; Ahnert-Hilger, G.; Höpfner, M.; Hoegerle, S.; Faiss, S.; Grabowski, P.; Jockers-Scherübl, M.; Riecken, E.O.; Zeitz, M.; Scherübl, H. Expression of dopamine receptors and transporter in neuroendocrine gastrointestinal tumor cells. Life Sci. 2002, 71, 667–678. [Google Scholar] [CrossRef]
- O’Toole, D.; Saveanu, A.; Couvelard, A.; Gunz, G.; Enjalbert, A.; Jaquet, P.; Ruszniewski, P.; Barlier, A. The analysis of quantitative expression of somatostatin and dopamine receptors in gastro-entero-pancreatic tumours opens new therapeutic strategies. Eur. J. Endocrinol. 2006, 155, 849–857. [Google Scholar] [CrossRef]
- Srirajaskanthan, R.; Watkins, J.; Marelli, L.; Khan, K.; Caplin, M.E. Expression of Somatostatin and Dopamine 2 Receptors in Neuroendocrine Tumours and the Potential Role for New Biotherapies. Neuroendocrinology 2009, 89, 308–314n. [Google Scholar] [CrossRef] [PubMed]
- Ferone, D.; Arvigo, M.; Semino, C.; Jaquet, P.; Saveanu, A.; Taylor, J.E.; Moreau, J.P.; Culler, M.D.; Albertelli, M.; Minuto, F.; et al. Somatostatin and Dopamine Receptor Expression in Lung Carcinoma Cells and Effects of Chimeric Somatostatin-Dopamine Molecules on Cell Proliferation. Am. J. Physiol. Endocrinol. Metab. 2005, 289, E1044–E1050. [Google Scholar] [CrossRef] [PubMed]
- Kidd, M.; Modlin, I.M.; Pfragner, R.; Eick, G.N.; Champaneria, M.C.; Chan, A.K.; Camp, R.L.; Mane, S.M. Small bowel carcinoid (enterochromaffin cell) neoplasia exhibits transforming growth factor-beta1-mediated regulatory abnormalities including up-regulation of C-Myc and MTA1. Cancer 2007, 109, 2420–2431. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, C.; Feelders, R.A.; Waaijers, A.M.; van Koetsveld, P.M.; Sprij-Mooij, D.M.; Lamberts, S.W.; Hofland, L.J. Differential regulation of human dopamine D2 and somatostatin receptor subtype expression by glucocorticoids in vitro. J. Mol. Endocrinol. 2009, 42, 47–56. [Google Scholar] [CrossRef]
- Baragli, A.; Alturaihi, H.; Watt, H.L.; Abdallah, A.; Kumar, U. Heterooligomerization of human dopamine receptor 2 and somatostatin receptor 2: Co-immunoprecipitation and fluorescence resonance energy transfer analysis. Cell Signal. 2007, 19, 2304–2316. [Google Scholar] [CrossRef]
- Gatto, F.; Hofland, L.J. The role of somatostatin and dopamine D2 receptors in endocrine tumors. Rev. Endocr. Relat. Cancer 2011, 18, R233–R251. [Google Scholar] [CrossRef]
- Lasolle, H.; Ferriere, A.; Vasiljevic, A.; Eimer, S.; Nunes, M.L.; Tabarin, A. Pasireotide-LAR in Acromegaly Patients Treated with a Combination Therapy: A Real-Life Study; Endocr Connect. Endocr. Connect. 2019, 8, 1383–1394. [Google Scholar] [CrossRef]
- Pivonello, R.; Ferone, D.; Lamberts, S.W.; Colao, A. Cabergoline plus Lanreotide for Ectopic Cushing’s Syndrome. N. Engl. J. Med. 2005, 352, 2457–2458. [Google Scholar] [CrossRef]
- Pivonello, R.; De Leo, M.; Cozzolino, A.; Colao, A. The Treatment of Cushing’s Disease. Endocr. Rev. 2015, 36, 385–486. [Google Scholar] [CrossRef]
- Sosa-Eroza, E.; Espinosa, E.; Ramírez-Rentería, C.; Mendoza, V.; Arreola, R.; Mercado, M. Treatment of Multiresistant Prolactinomas with a Combination of Cabergoline and Octreotide LAR. Endocrine 2018, 61, 343–348. [Google Scholar] [CrossRef]
- Hofland, L.J.; Vandamme, T.; Albertelli, M.; Ferone, D. Hormone and Receptor Candidates for Target and Biotherapy of Neuroendocrine Tumors. Front. Horm Res. 2015, 44, 216–238. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.G.; Kim, S.; Taylor, J.; Dong, J.; Moreau, J.P.; Culler, M.D.; Melmed, S. Suppression of rat and human growth hormone and prolactin secretion by a novel somatostatin/dopaminergic chimeric ligand. J. Clin. Endocrinol. Metab. 2003, 88, 5414–5421. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jaquet, P.; Gunz, G.; Saveanu, A.; Dufour, H.; Taylor, J.; Dong, J.; Kim, S.; Moreau, J.P.; Enjalbert, A.; Culler, M.D. Efficacy of Chimeric Molecules Directed Towards Multiple Somatostatin and Dopamine Receptors on Inhibition of GH and Prolactin Secretion From GH-secreting Pituitary Adenomas Classified as Partially Responsive to Somatostatin Analog Therapy. Eur. J. Endocrinol. 2005, 153, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Saveanu, A.; Gunz, G.; Guillen, S.; Dufour, H.; Culler, M.D.; Jaquet, P. Somatostatin and dopamine-somatostatin multiple ligands directed towards somatostatin and dopamine receptors in pituitary adenomas. Neuroendocrinology 2006, 283, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Florio, T.; Barbieri, F.; Spaziante, R.; Zona, G.; Hofland, L.J.; van Koetsveld, P.M.; Feelders, R.A.; Stalla, G.K.; Theodoropoulou, M.; Culler, M.D.; et al. Efficacy of a Dopamine-Somatostatin Chimeric Molecule, BIM-23A760, in the Control of Cell Growth from Primary Cultures of Human Non-Functioning Pituitary Adenomas: A Multi-Center Study. Multicent. Study Endocr. Relat. Cancer 2008, 15, 583–596. [Google Scholar] [CrossRef]
- Fusco, A.; Gunz, G.; Jaquet, P.; Dufour, H.; Germanetti, A.L.; Culler, M.D.; Barlier, A.; Saveanu, A. Somatostatinergic Ligands in Dopamine-Sensitive and -Resistant Prolactinomas. Eur. J. Endocrinol. 2008, 158, 595–603. [Google Scholar] [CrossRef]
- Gatto, F.; Barbieri, F.; Gatti, M.; Wurth, R.; Schulz, S.; Ravetti, J.L.; Zona, G.; Culler, M.D.; Saveanu, A.; Giusti, M.; et al. Balance Between Somatostatin and D2 Receptor Expression Drives TSH-secreting Adenoma Response to Somatostatin Analogues and Dopastatins. Clin. Endocrinol. 2012, 76, 407–414. [Google Scholar] [CrossRef]
- Couvelard, A.; Pélaprat, D.; Dokmak, S.; Sauvanet, A.; Voisin, T.; Couvineau, A.; Ruszniewski, P. Antisecretory Effects of Chimeric Somatostatin/Dopamine Receptor Ligands on Gastroenteropancreatic Neuroendocrine Tumors. Pancreas 2017, 46, 631–638. [Google Scholar] [CrossRef]
- Van Adrichem, R.C.; de Herder, W.W.; Kamp, K.; Brugts, M.P.; de Krijger, R.R.; Sprij-Mooij, D.M.; Lamberts, S.W.; van Koetsveld, P.M.; Janssen, J.A.; Hofland, L.J. Effects of Somatostatin Analogs and Dopamine Agonists on Insulin-Like Growth Factor 2-Induced Insulin Receptor Isoform A Activation by Gastroenteropancreatic Neuroendocrine Tumor Cells. Neuroendocrinology 2016, 103, 815–825. [Google Scholar] [CrossRef]
- Cuny, T.; Mohamed, A.; Graillon, T.; Roche, C.; Defilles, C.; Germanetti, A.L.; Couderc, B.; Figarella-Branger, D.; Enjalbert, A.; Barlier, A.; et al. Somatostatin Receptor sst2 Gene Transfer in Human Prolactinomas in Vitro: Impact on Sensitivity to Dopamine, Somatostatin and Dopastatin, in the Control of Prolactin Secretion. Mol. Cell Endocrinol. 2012, 355, 106–113. [Google Scholar] [CrossRef]
- Zitzmann, K.; Andersen, S.; Vlotides, G.; Spöttl, G.; Zhang, S.; Datta, R.; Culler, M.; Göke, B.; Auernhammer, C.J. The Novel Somatostatin Receptor 2/dopamine Type 2 Receptor Chimeric Compound BIM-23A758 Decreases the Viability of Human GOT1 Midgut Carcinoid Cells. Neuroendocrinology 2013, 98, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Gruszka, A.; Kunert-Radek, J.; Radek, A.; Pisarek, H.; Taylor, J.; Dong, J.Z.; Culler, M.D.; Pawlikowski, M. The Effect of Selective sst1, sst2, sst5 Somatostatin Receptors Agonists, a Somatostatin/Dopamine (SST/DA) Chimera and Bromocriptine on the “Clinically Non-Functioning” Pituitary Adenomas In Vitro. Life Sci. 2006, 78, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Peverelli, E.; Olgiati, L.; Locatelli, M.; Magni, P.; Fustini, M.F.; Frank, G.; Mantovani, G.; Beck-Peccoz, P.; Spada, A.; Lania, A. The Dopamine-Somatostatin Chimeric Compound BIM-23A760 Exerts Antiproliferative and Cytotoxic Effects in Human Non-Functioning Pituitary Tumors by Activating ERK1/2 and p38 Pathways. Cancer Lett. 2010, 288, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Kidd, M.; Drozdov, I.; Joseph, R.; Pfragner, R.; Culler, M.; Modlin, I. Differential Cytotoxicity of Novel Somatostatin and dopamine Chimeric compounds on Bronchopulmonary and Small intestinal Neuroendocrine tumor cell lines Cancer. Cancer 2008, 113, 690–700. [Google Scholar] [CrossRef]
- Halem, H.A.; Hochgeschwender, U.; Rih, J.K.; Nelson, R.; Johnson, G.A.; Thiagalingam, A.; Culler, M.D. TBR-760, a Dopamine-Somatostatin Compound, Arrests Growth of Aggressive Nonfunctioning Pituitary Adenomas in Mice. Endocrinology 2020, 161, bqaa101. [Google Scholar] [CrossRef]
- Froehlich, J.; Ramis, J.; Lesage, C.; Obach, R. Safety, Pharmacokinetics (PK) and Pharmacodynamics (PD) after Subcutaneous (s. c.) Administration (adm) of BIM23A760, a Chimeric Compound Combining Dopaminergic Agonist and Somatostatin Analogue, in Healthy Male Volunteers. In Proceedings of the 91st Annual Meeting of Endocrine Society, Washington, DC, USA, 10–13 June 2009; pp. 3–685. [Google Scholar]
- Lesage, C.; Seymour, C.; Urbanavivius, G.; Colao, A.M. A Phase II Exploratory Study of BIM23A760 in Acromegalic Patients: Preliminary Results of Safety and Efficacy after a Single-Dose Administration. In Proceedings of the 91st Annual Meeting of the Endocrine Society, Washington, DC, USA, 10–13 June 2009; pp. 3–673. [Google Scholar]
- Saveanu, A.; Datta, R.; Zhang, S.; Shen, Y.; Dong, J.Z.; Graillon, T.; Desfilles, C.; Landsman, T.; Halem, H.; Enjalbert, A.; et al. Novel somatostatin-dopamine chimeric compound demonstrates superior efficacy in suppressing growth hormone secretion from human acromegalic tumors partially responsive to current somatostatin and dopamine therapies. In Proceedings of the Endocrine Society’s 98th Annual Meeting and Expo, Boston, MA, USA, 1–4 April 2016. [Google Scholar]
- Vázquez-Borrego, M.C.; L-López, F.; Gálvez-Moreno, M.A.; Fuentes-Fayos, A.C.; Venegas-Moreno, E.; Herrera-Martínez, A.D.; Blanco-Acevedo, C.; Solivera, J.; Landsman, T.; Gahete, M.D.; et al. A New Generation Somatostatin-Dopamine Analogue Exerts Potent Antitumoral Actions on Pituitary Neuroendocrine Tumor Cells. Neuroendocrinology 2020, 110, 70–82. [Google Scholar] [CrossRef]
- Herrera-Martínez, A.D.; van den Dungen, R.; Dogan-Oruc, F.; van Koetsveld, P.M.; Culler, M.D.; de Herder, W.W.; Luque, R.M.; Feelders, R.A.; Hofland, L.J. Effects of Novel Somatostatin-Dopamine Chimeric Drugs in 2D and 3D Cell Culture Models of Neuroendocrine Tumors. Endocr. Relat. Cancer 2019, 26, 585–599. [Google Scholar] [CrossRef]
- Dicitore, A.; Cantone, M.C.; Gaudenzi, G.; Saronni, D.; Carra, S.; Borghi, M.O.; Albertelli, M.; Ferone, D.; Hofland, L.J.; Persani, L.; et al. Efficacy of a novel second-generation somatostatin-dopamine chimera (TBR-065) in human medullary thyroid cancer: A preclinical study. Neuroendocrinology 2020. online ahead of print. [Google Scholar] [CrossRef]
- Cuny, T.; Graillon, T.; Defilles, C.; Datta, R.; Zhang, S.; Figarella-Branger, D.; Dufour, H.; Mougel, G.; Brue, T.; Landsman, T.; et al. Characterization of the ability of a, second-generation SST-DA chimeric molecule, TBR-065, to suppress GH secretion from human GH-secreting adenoma cells. Pituitary 2021. advance online publication. [Google Scholar] [CrossRef]
- Halem, H.; Zhang, S.; Datta, R.; Bastille, A.; Beech, J.; Marques, M.; Patel, S.; Shen, Y.; Dong, J.; Culler, M.D. A novel somatostatin-dopamine chimeric compound induces dose-related suppression of GHRH-stimulated growth hormone secretion and increases insulin sensitivity in normal rats. In Proceedings of the Endocrine Society’s 98th Annual Meeting and Expo, Boston, MA, USA, 1–4 April 2016. [Google Scholar]
- De Boon, W.; van Esdonk, M.J.; Stuurman, F.E.; Biermasz, N.R.; Pons, L.; Paty, I.; Burggraaf, J. A Novel Somatostatin-Dopamine Chimera (BIM23B065) Reduced GH Secretion in a First-in-Human Clinical Trial. J. Clin. Endocrinol. Metab. 2019, 104, 883–891. [Google Scholar] [CrossRef]
- Van Esdonk, M.J.; Stevens, J.; Stuurman, F.E.; de Boon, W.; Dehez, M.; van der Graaf, P.H.; Burggraaf, J. The Pharmacodynamic Effects of a Dopamine–Somatostatin Chimera Agonist on the Cardiovascular System Reduced GH Secretion in a First in-Human Clinical Trial. J. Cardiovasc. Pharmacol. 2019, 74, 128–136. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantone, M.C.; Dicitore, A.; Vitale, G. Somatostatin-Dopamine Chimeric Molecules in Neuroendocrine Neoplasms. J. Clin. Med. 2021, 10, 501. https://doi.org/10.3390/jcm10030501
Cantone MC, Dicitore A, Vitale G. Somatostatin-Dopamine Chimeric Molecules in Neuroendocrine Neoplasms. Journal of Clinical Medicine. 2021; 10(3):501. https://doi.org/10.3390/jcm10030501
Chicago/Turabian StyleCantone, Maria Celeste, Alessandra Dicitore, and Giovanni Vitale. 2021. "Somatostatin-Dopamine Chimeric Molecules in Neuroendocrine Neoplasms" Journal of Clinical Medicine 10, no. 3: 501. https://doi.org/10.3390/jcm10030501
APA StyleCantone, M. C., Dicitore, A., & Vitale, G. (2021). Somatostatin-Dopamine Chimeric Molecules in Neuroendocrine Neoplasms. Journal of Clinical Medicine, 10(3), 501. https://doi.org/10.3390/jcm10030501







