Improving Adherence to Myofunctional Therapy in the Treatment of Sleep-Disordered Breathing
Abstract
:1. Introduction
2. Materials and Methods
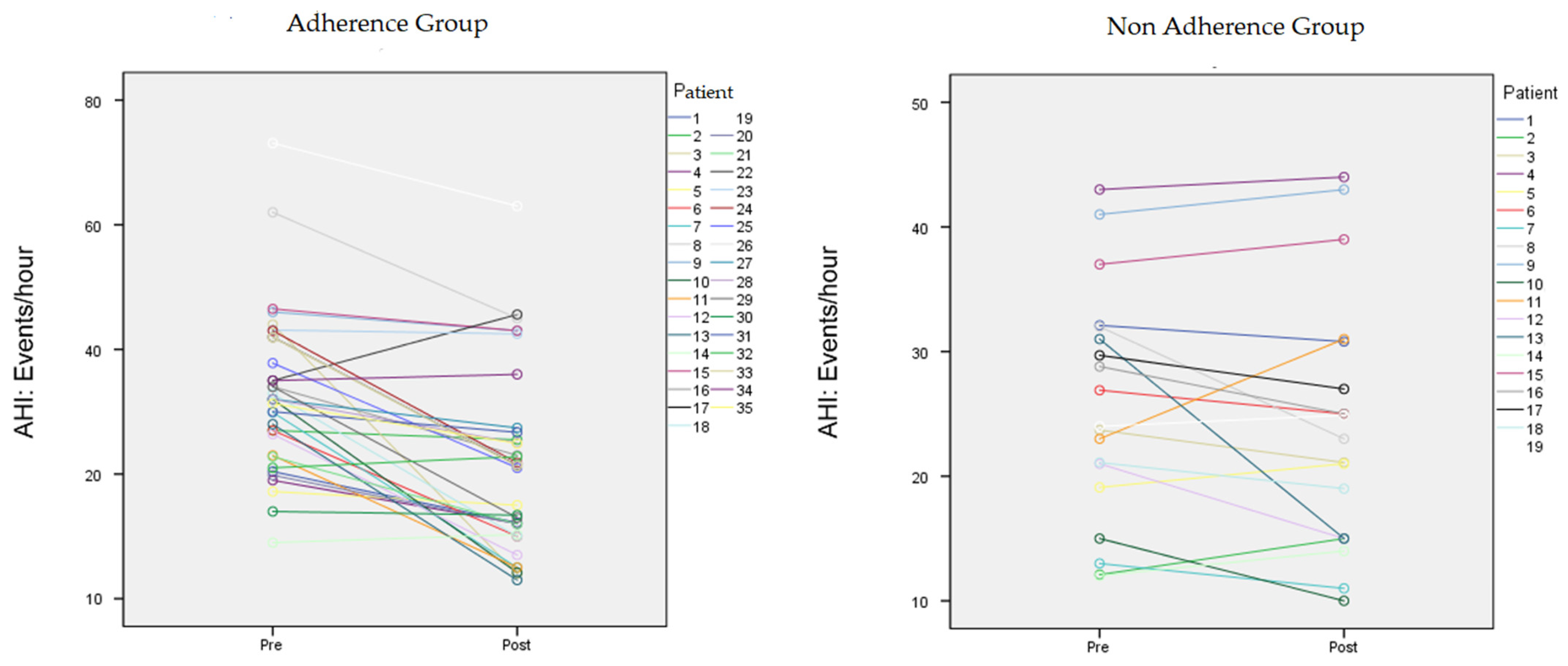
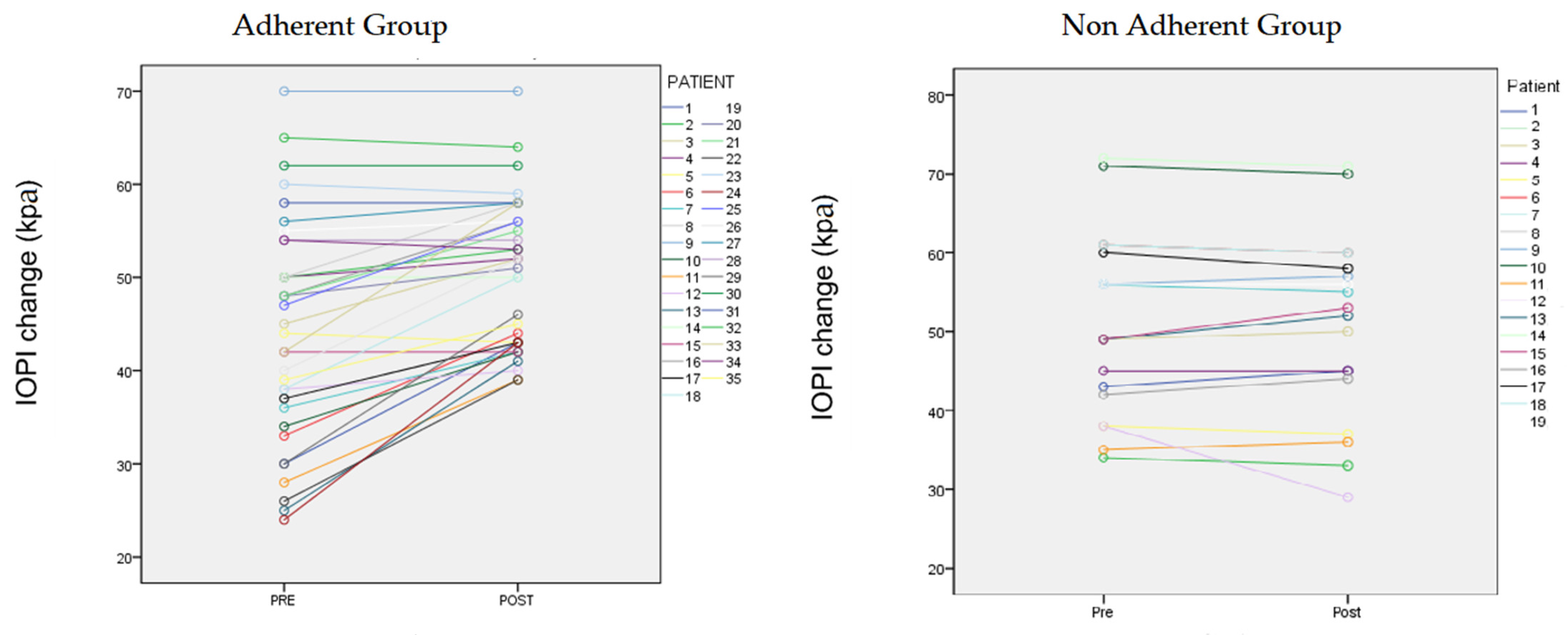
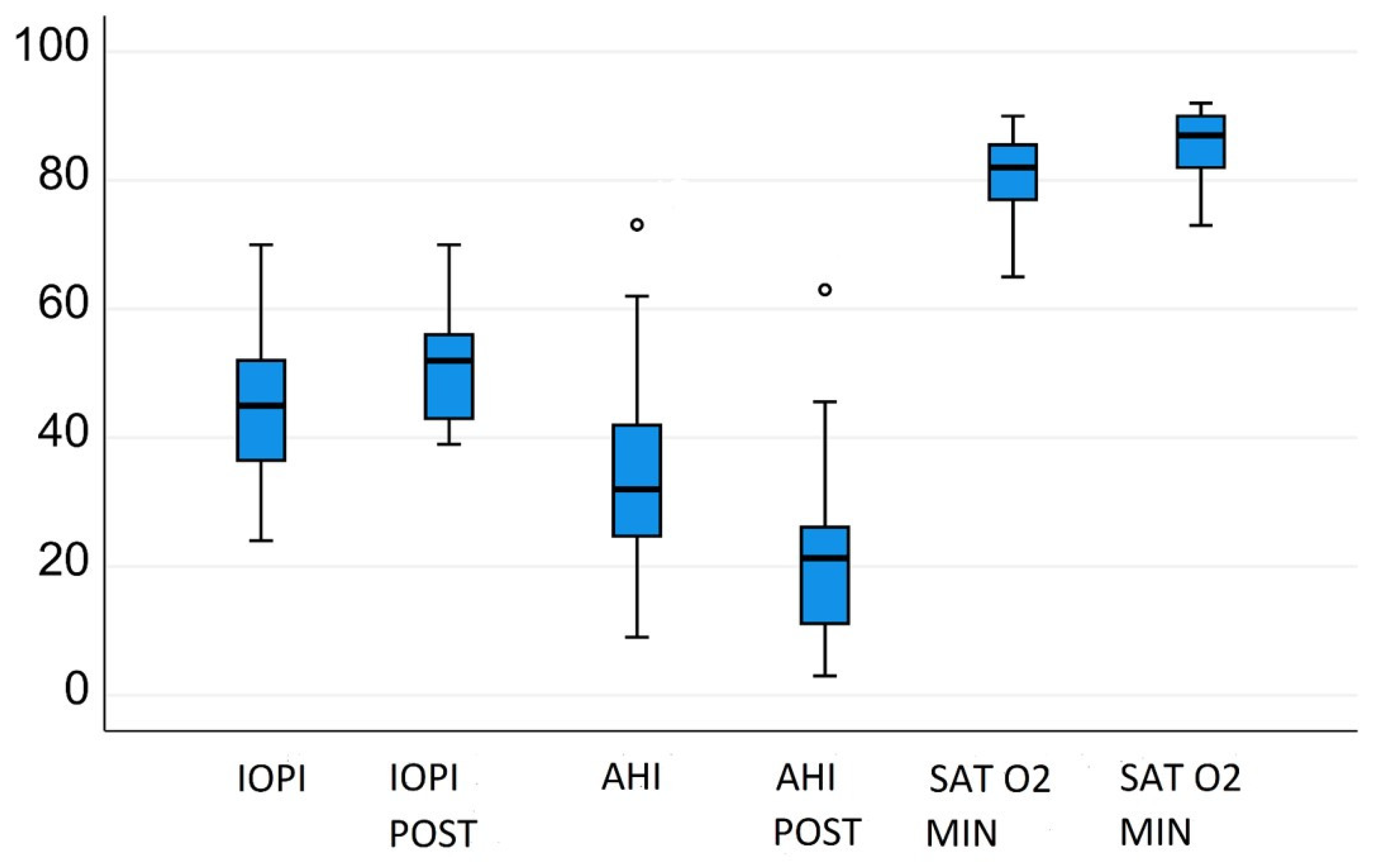
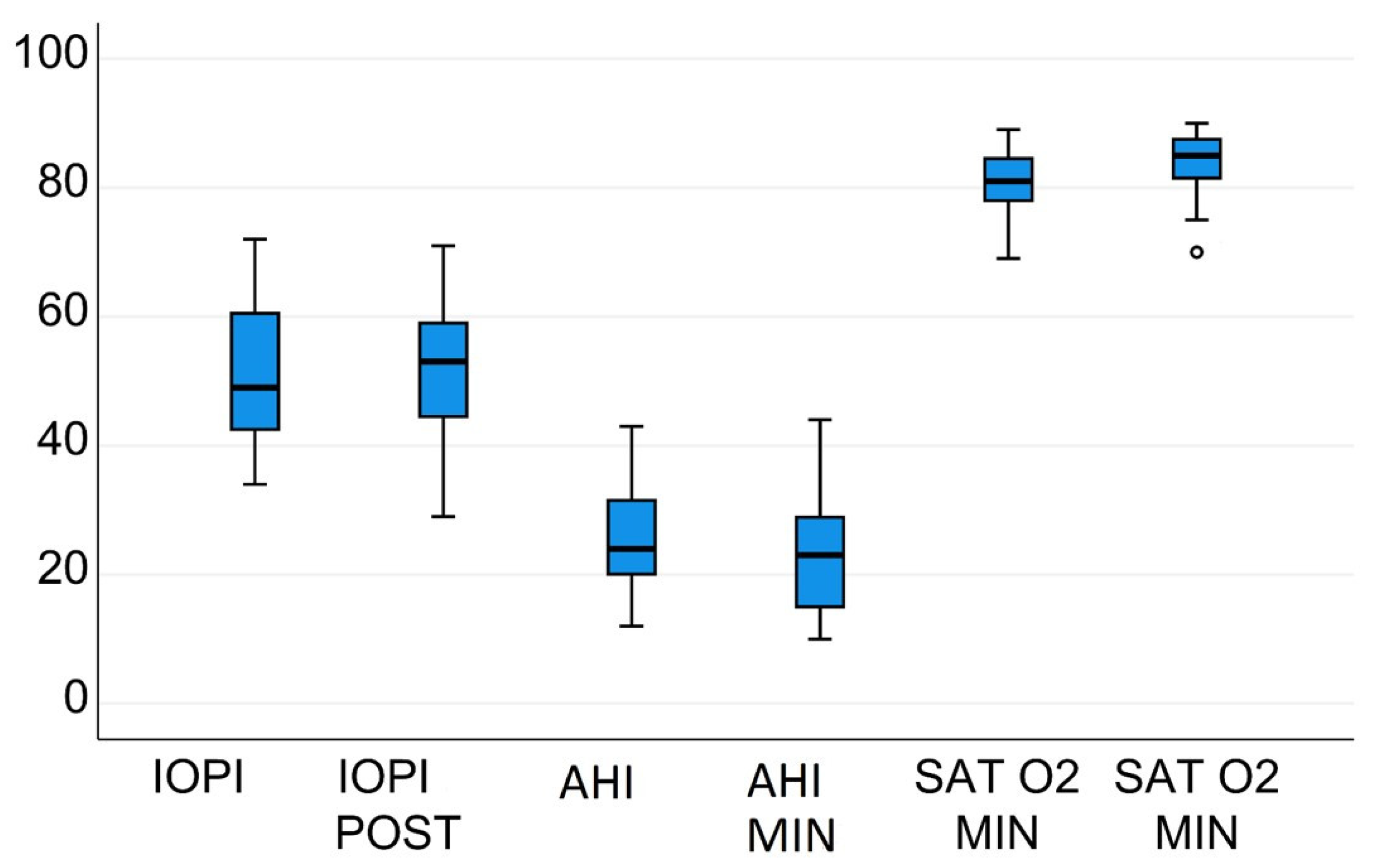
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mediano, O.; González Mangado, N.; Montserrat, J.M.; Alonso-Álvarez, M.L.; Almendros, I.; Alonso-Fernández, A.; Barbé, F.; Borsini, E.; Caballero-Eraso, C.; Cano-Pumarega, I.; et al. International consensus document on obstructive sleep apnea. Arch. Bronconeum. 2021. [Google Scholar] [CrossRef]
- Osman, A.M.; Carter, S.G.; Carberry, J.C.; Eckert, D.J. Obstructive sleep apnea: Current perspectives. Nat. Sci. Sleep 2018, 10, 21–34. [Google Scholar] [CrossRef] [Green Version]
- Eckert, D.J. Phenotypic approaches to obstructive sleep apnoea—New pathways for targeted therapy. Sleep Med. Rev. 2018, 37, 45–59. [Google Scholar] [CrossRef]
- Guilleminault, C.; Huang, Y.S.; Quo, S. Apraxia in children and adults with obstructive sleep apnea syndrome. Sleep 2019, 42, zsz168. [Google Scholar] [CrossRef]
- Aman, J.E.; Elangovan, N.; Yeh, I.L.; Konczak, J. The effectiveness of proprioceptive training for improving motor function: A systematic review. Front. Hum. Neurosci. 2015, 8, 1075. [Google Scholar] [CrossRef] [Green Version]
- Guimarães, K.C.; Drager, L.F.; Genta, P.R.; Marcondes, B.F.; Lorenzi-Filho, G. Effects of oropharyngeal exercises on patients with moderate obstructive sleep apnea syndrome. Am. J. Respir. Crit. Care Med. 2009, 179, 962–966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaferia, G.; Badke, L.; Santos-Silva, R.; Bommarito, S.; Tufik, S.; Bittencourt, L. Effect of speech therapy as adjunct treatment to continuous positive airway pressure on the quality of life of patients with obstructive sleep apnea. Sleep Med. 2013, 14, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Ieto, V.; Kayamori, F.; Montes, M.I.; Hirata, R.P.; Gregório, M.G.; Alencar, A.M.; Drager, L.F.; Genta, P.R.; Lorenzi-Filho, G. Effects of oropharyngeal exercises on snoring: A randomized trial. Chest 2015, 148, 683–691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camacho, M.; Certal, V.; Abdullatif, J.; Zaghi, S.; Ruoff, C.M.; Capasso, R.; Kushida, C.A. Myofunctional therapy to treat obstructive sleep apnea: A systematic review and meta-analysis. Sleep 2015, 38, 669–675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diaféria, G.; Santos-Silva, R.; Truksinas, E.; Haddad, F.L.M.; Santos, R.; Bommarito, S.; Gregório, L.C.; Tufik, S.; Bittencourt, L. Myofunctional therapy improves adherence to continuous positive airway pressure treatment. Sleep Breath. 2017, 21, 387–395. [Google Scholar] [CrossRef] [PubMed]
- de Felício, C.M.; da Silva Dias, F.V.; Trawitzki, L. Obstructive sleep apnea: Focus on myofunctional therapy. Nat. Sci. Sleep 2018, 10, 271–286. [Google Scholar] [CrossRef] [Green Version]
- Wishney, M.; Darendeliler, M.A.; Dalci, O. Myofunctional therapy and prefabricated functional appliances: An overview of the history and evidence. Aust. Dent. J. 2019, 64, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Okamoto, T.; Akagi, Y.; Matsui, K.; Sekiguchi, H.; Satoya, N.; Inoue, Y.; Tatsuta, A.; Hagiwara, N. Efficacy of oral myofunctional therapy in middle-aged to elderly patients with obstructive sleep apnoea treated with continuous positive airway pressure. J. Oral Rehabil. 2021, 48, 176–182. [Google Scholar] [CrossRef]
- Atilgan, E.; Kunter, E.; Algun, Z.C. Are oropharyngeal exercises effective in obstructive sleep apnea syndrome? J. Back Musculoskelet. Rehabil. 2020, 33, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Mario Diaz, S.; Ana Salazar, C.; Felipe Bravo, G.; Ocampo-Garcés, A. Tratamiento del síndrome de apneas e hipopneas obstructivas del sueño con terapia miofuncional orofaríngea: Experiencia en hospital público de Chile. Rev. Otorrinolaringol. Cir. Cabeza Cuello 2019, 79, 395–403. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Oh, E.G.; Choi, M.; Choi, S.J.; Joo, E.Y.; Lee, H.; Kim, H.Y. Development and evaluation of myofunctional therapy support program (MTSP) based on self-efficacy theory for patients with obstructive sleep apnea. Sleep Breath. 2020, 24, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.; Emperumal, C.P.; Grbach, V.X.; Padilla, M.; Enciso, R. Effects of respiratory muscle therapy on obstructive sleep apnea: A systematic review and meta-analysis. J. Clin. Sleep Med. 2020, 16, 785–801. [Google Scholar] [CrossRef]
- Rueda, J.R.; Mugueta-Aguinaga, I.; Vilaró, J.; Rueda-Etxebarria, M. Myofunctional therapy (oropharyngeal exercises) for obstructive sleep apnoea. Cochrane Database Syst. Rev. 2020, 11, CD013449. [Google Scholar] [CrossRef]
- McKeown, P.; O’Connor-Reina, C.; Plaza, G. Breathing re-education and phenotypes of sleep apnea: A review. J. Clin. Med. 2021, 10, 471. [Google Scholar] [CrossRef] [PubMed]
- Koka, V.; De Vito, A.; Roisman, G.; Petitjean, M.; Filograna Pignatelli, G.R.; Padovani, D.; Randerath, W. Orofacial myofunctional therapy in obstructive sleep apnea syndrome: A pathophysiological perspective. Medicina 2021, 57, 323. [Google Scholar] [CrossRef]
- Carrasco-Llatas, M.; O’Connor-Reina, C.; Calvo-Henríquez, C. The role of myofunctional therapy in treating sleep-disordered breathing: A state-of-the-art review. Int. J. Environ. Res. Public Health 2021, 18, 7291. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.T.; Sternbach, J.M.; Guilleminault, C. Continuous positive airway pressure therapy in obstructive sleep apnea: Benefits and alternatives. Expert Rev. Respir. Med. 2017, 11, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Weaver, T.E.; Grunstein, R.R. Adherence to continuous positive airway pressure therapy: The challenge to effective treatment. Proc. Am. Thorac. Soc. 2008, 5, 173–178. [Google Scholar] [CrossRef] [Green Version]
- Rowland, S.; Aiyappan, V.; Hennessy, C.; Catcheside, P.; Chai-Coezter, C.L.; McEvoy, R.D.; Antic, N.A. Comparing the efficacy, mask leak, patient adherence, and patient preference of three different CPAP interfaces to treat moderate-severe obstructive sleep apnea. J. Clin. Sleep Med. 2018, 14, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Rosa, T.; Bellardi, K.; Viana, A., Jr.; Ma, Y.; Capasso, R. Digital health and sleep-disordered breathing: A systematic review and meta-analysis. J. Clin. Sleep Med. 2018, 14, 1605–1620. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Giron, M.; Garmendia, O.; Lugo, V.; Ruiz, C.; Salord, N.; Alsina, X.; Farré, R.; Montserrat, J.M.; Torres, M. Mobile health application to support CPAP therapy in obstructive sleep apnoea: Design, feasibility and perspectives. ERJ Open Res. 2020, 6, 00220–02019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Connor Reina, C.; Plaza, G.; Ignacio-Garcia, J.M.; Jardin, P.B.; Garcia-Iriarte, M.T.; Casado-Morente, J.C.; Gonzalez, E.D.; Rodriguez-Reina, A. New mHealth application software based on myofunctional therapy applied to sleep-disordered breathing in non-compliant subjects. Sleep Sci. Pract. 2020, 4, 3. [Google Scholar] [CrossRef]
- O’Connor-Reina, C.; Ignacio Garcia, J.M.; Rodriguez Ruiz, E.; Morillo Dominguez, M.; Ignacio Barrios, V.; Baptista Jardin, P.; Casado Morente, J.C.; Garcia Iriarte, M.T.; Plaza, G. Myofunctional therapy app for severe apnea–hypopnea sleep obstructive syndrome: Pilot randomized controlled trial. JMIR mHealth uHealth 2020, 8, e23123. [Google Scholar] [CrossRef]
- Kim, H.; Cho, N.B.; Kim, J.; Kim, K.M.; Kang, M.; Choi, Y.; Kim, M.; You, H.; Nam, S.I.; Shin, S. Implementation of a home-based mHealth app intervention program with human mediation for swallowing tongue pressure strengthening exercises in older adults: Longitudinal observational study. JMIR mHealth uHealth 2020, 8, e22080. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Salapatas, A.M.; Bonzelaar, L.B. Updated Friedman staging system for obstructive sleep apnea. Adv. Otorhinolaryngol. 2017, 80, 41–48. [Google Scholar] [CrossRef] [PubMed]
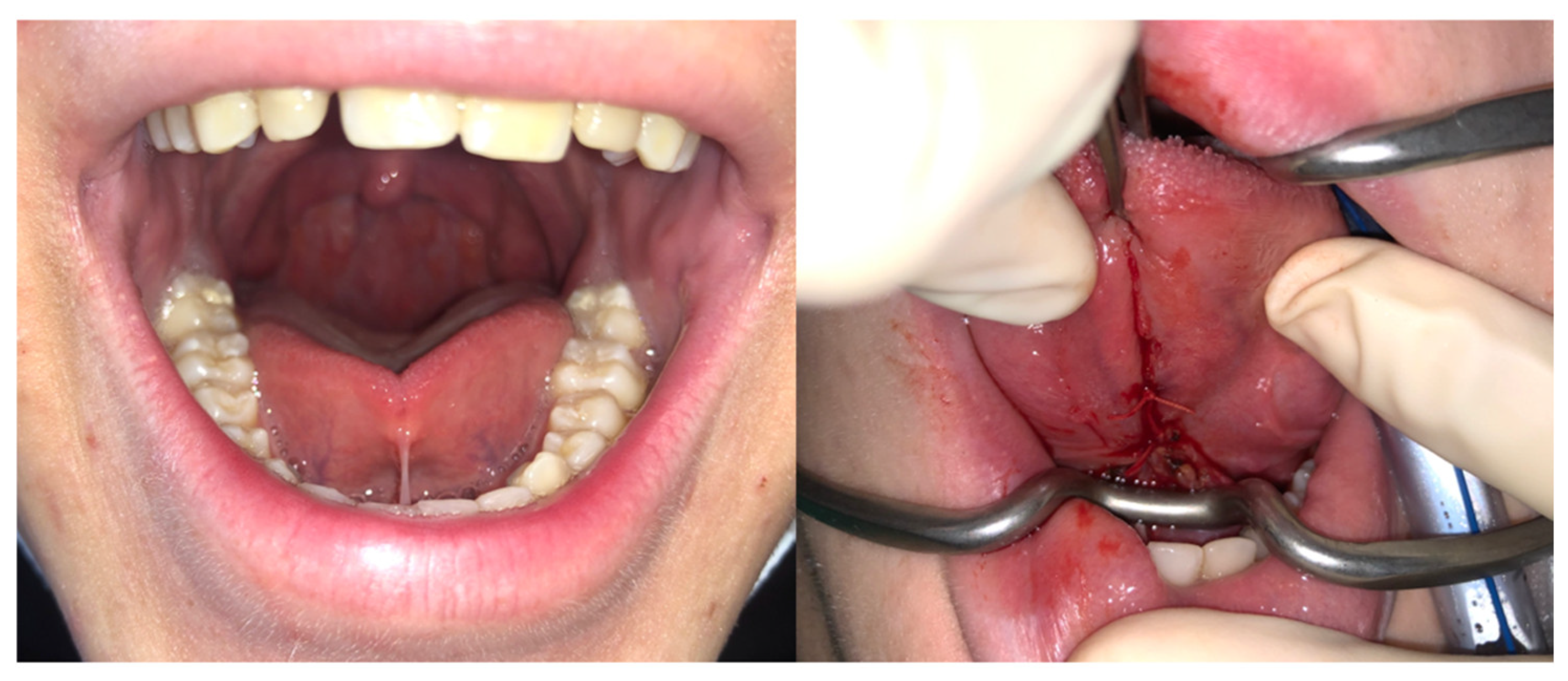
- Amir, L.H.; James, J.P.; Donath, S.M. Reliability of the Hazelbaker assessment tool for lingual frenulum function. Int. Breastfeed. J. 2006, 1, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Connor-Reina, C.; Plaza, G.; Garcia-Iriarte, M.T.; Ignacio-Garcia, J.M.; Baptista, P.; Casado-Morente, J.C.; De Vicente, E. Tongue peak pressure: A tool to aid in the identification of obstruction sites in patients with obstructive sleep apnea/hypopnea syndrome. Sleep Breath. 2020, 24, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.L. Proprioception, Squire, Encyclopedia of Neuroscience; Larry, R., Ed.; Academic Press: Cambridge, MA, USA, 2009; pp. 1143–1149. ISBN 9780080450469. [Google Scholar] [CrossRef]
- Rodríguez-Alcalá, L.; Martínez, J.M.; Baptista, P.; Ríos Fernández, R.; Francisco, J.G.; Parejo Santaella, J.; Plaza, G. Sensorimotor tongue evaluation and rehabilitation in patients with sleep-disordered breathing: A novel approach. J. Oral Rehabil. 2021, 48, 1363–1372. [Google Scholar] [CrossRef]
- Borrmann, P.F.; O’Connor-Reina, C.; Ignacio, J.M.; Rodriguez Ruiz, E.; Rodriguez Alcala, L.; Dzembrovsky, F.; Baptista, P.; Garcia Iriarte, M.T.; Casado Alba, C.; Plaza, G. Muscular assessment in patients with severe obstructive sleep apnea syndrome: Protocol for a case–control study. JMIR Res. Protoc. 2021, 10, e30500. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Alcalá, L.; Martín-Lagos Martínez, J.; O’Connor-Reina, C.; Plaza, G. Assessment of muscular tone of the tongue using a digital measure spoon in a healthy population: A pilot study. PLoS ONE 2021, 16, e0245901. [Google Scholar] [CrossRef] [PubMed]
- De Felício, C.M.; Folha, G.A.; Ferreira, C.L.; Medeiros, A.P. Expanded protocol of orofacial myofunctional evaluation with scores: Validity and reliability. Int. J. Pediatr. Otorhinolaryngol. 2010, 74, 1230–1239. [Google Scholar] [CrossRef]
- Esteller, E.; Carrasco, M.; Díaz-Herrera, M.Á.; Vila, J.; Sampol, G.; Juvanteny, J.; Sieira, R.; Farré, A.; Vilaseca, I. Clinical Practice Guideline recommendations on examination of the upper airway for adults with suspected obstructive sleep apnoea–hypopnoea syndrome. Acta Otorrinolaringol. Esp. 2019, 70, 364–372. [Google Scholar] [CrossRef] [PubMed]
| Non-Adherent (n = 19) | Adherent (n = 35) | p Value | |
|---|---|---|---|
| Anthropometric data | |||
| Age (years) | 50.26 (36.2–64.224) | 45.9 (28.17–63.77) | NS |
| Female (%) | 7 (36.8%) | 6 (17.14%) | NS |
| BMI (kg/m2) | 25.1 ± 2.8 | 25.8 (22.7–29.1) | NS |
| Friedman stage | 5 (I). 4 (II). 3 (III). 3 (IV) | 4 (I). 11 (II). 14 (III). 6 (IV) | NS |
| Polysomnography data | |||
| AHI/h | 25.5 ± 9.2 | 32.97 ± 1.8/h | NS |
| Sat O2 Min | 80.68 ± 5.6 | 80.91% ± 6.1% | NS |
| Tone measures (kPa) | |||
| IOPI max tongue | 51.3 ± 11.4 | 44.4 ± 11.08 | 0.04 |
| 27 | |||
| Tongue-tie examination | |||
| Hazlebaker score | 10.79 ± 2.4 | 13.03 ± 1.5 | 0.04 |
| Non-Adherent (n = 19) | Airway Gym Group (n = 35) | |||||
|---|---|---|---|---|---|---|
| Baseline | After 3 Months | p Value | Baseline | After 3 Months | p Value | |
| Anthropometric data | ||||||
| BMI (kg/m2) | 25.1 ± 2.8 | 24.6 ± 2.5 | n.s | 25.8 (22.7–29.1) | 25.1.2(22.4–28) | n.s |
| Polysomnographic data | ||||||
| AHI/h | 25.5 ± 9.2 | 23.8 ± 10.1 | n.s | 32.97 ± 1.8/h | 21.9 ± 14.5/h | 0.01 |
| Sat O 2Min | 80.68 ± 5.6 | 81.3 ± 5.7 | n.s | 80.91% ± 6.1% | 85.09% ± 5.3% | 0.01 |
| Tone measures (kPa) | ||||||
| IOPI max tongue | 51.3 ± 11.4 | 51.1 ± 11.7 | n.s | 44.4 ± 11.08 | 50.1 ± 10.2 kPa | 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Connor-Reina, C.; Ignacio Garcia, J.M.; Rodriguez Alcala, L.; Rodríguez Ruiz, E.; Garcia Iriarte, M.T.; Casado Morente, J.C.; Baptista, P.; Plaza, G. Improving Adherence to Myofunctional Therapy in the Treatment of Sleep-Disordered Breathing. J. Clin. Med. 2021, 10, 5772. https://doi.org/10.3390/jcm10245772
O’Connor-Reina C, Ignacio Garcia JM, Rodriguez Alcala L, Rodríguez Ruiz E, Garcia Iriarte MT, Casado Morente JC, Baptista P, Plaza G. Improving Adherence to Myofunctional Therapy in the Treatment of Sleep-Disordered Breathing. Journal of Clinical Medicine. 2021; 10(24):5772. https://doi.org/10.3390/jcm10245772
Chicago/Turabian StyleO’Connor-Reina, Carlos, Jose María Ignacio Garcia, Laura Rodriguez Alcala, Elisa Rodríguez Ruiz, María Teresa Garcia Iriarte, Juan Carlos Casado Morente, Peter Baptista, and Guillermo Plaza. 2021. "Improving Adherence to Myofunctional Therapy in the Treatment of Sleep-Disordered Breathing" Journal of Clinical Medicine 10, no. 24: 5772. https://doi.org/10.3390/jcm10245772
APA StyleO’Connor-Reina, C., Ignacio Garcia, J. M., Rodriguez Alcala, L., Rodríguez Ruiz, E., Garcia Iriarte, M. T., Casado Morente, J. C., Baptista, P., & Plaza, G. (2021). Improving Adherence to Myofunctional Therapy in the Treatment of Sleep-Disordered Breathing. Journal of Clinical Medicine, 10(24), 5772. https://doi.org/10.3390/jcm10245772








