The Impact of Neurocognitive Functioning on the Course of Posttraumatic Stress Symptoms following Civilian Traumatic Brain Injury
Abstract
:1. Introduction
2. Materials and Methods
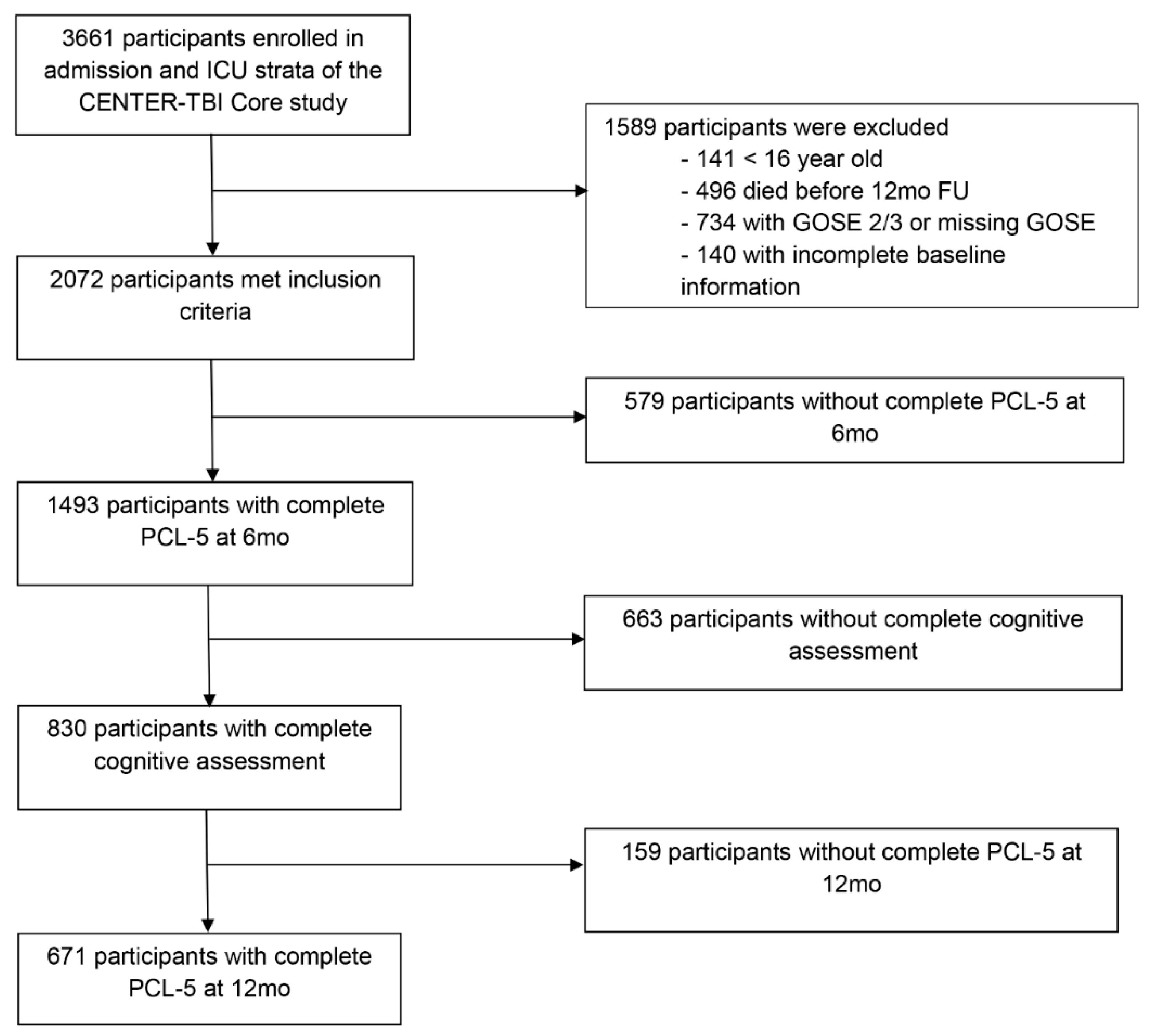
2.1. Study Participants and Procedure
2.2. Measures
2.3. Statistical Analysis
3. Results
3.1. Study Sample and Characteristics
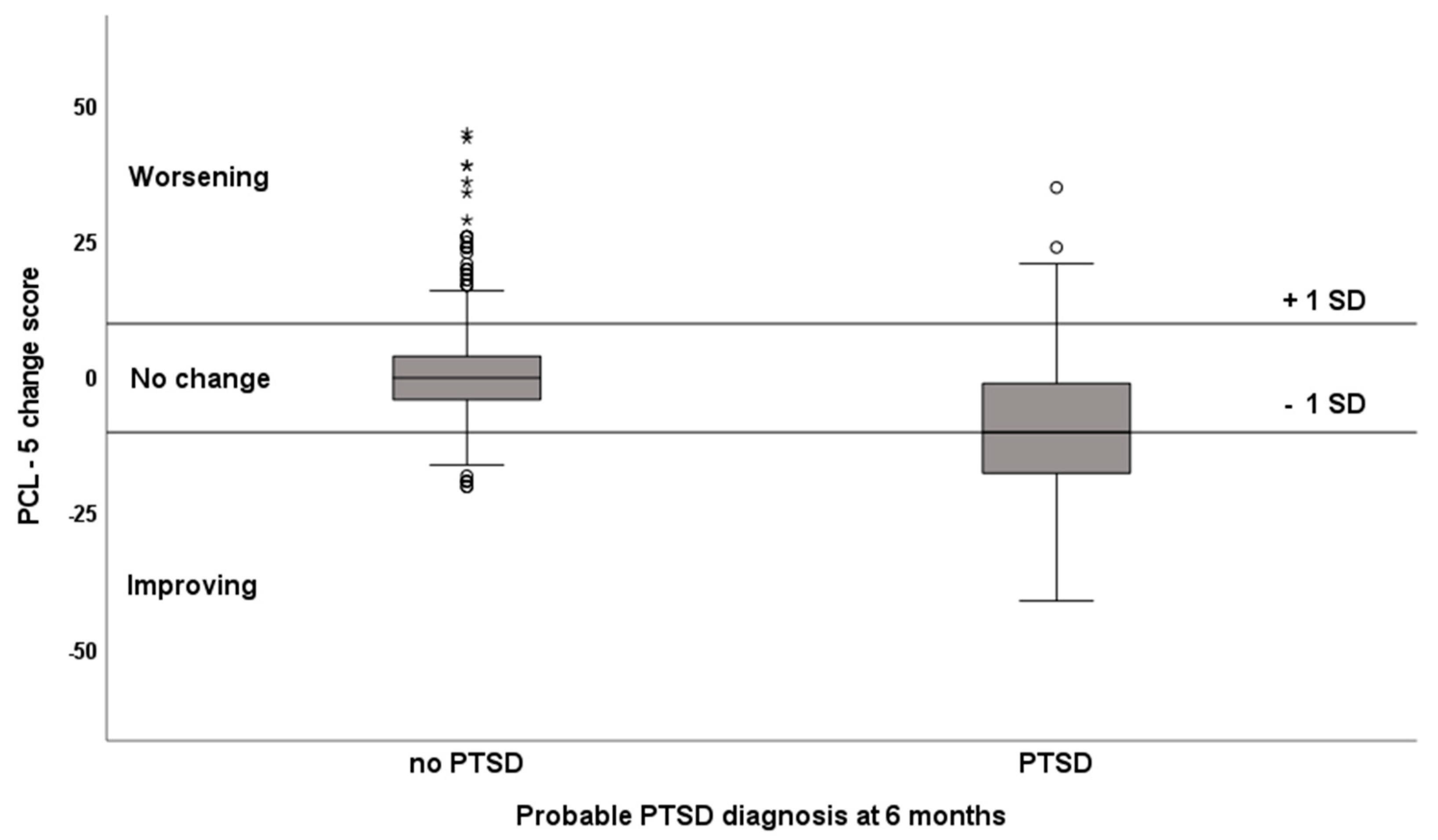
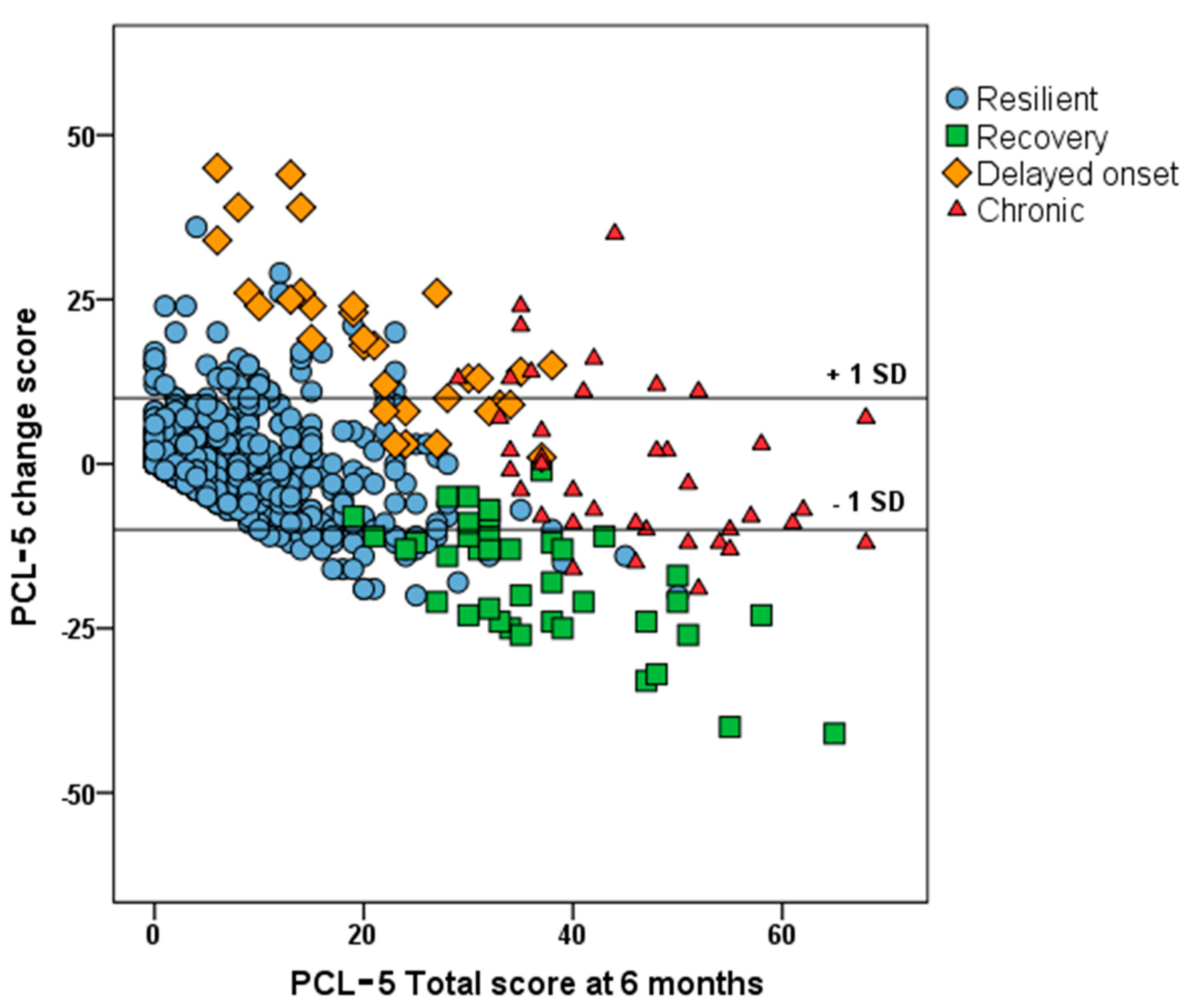
3.2. Course of PTSD Symptoms
3.3. Cognitive Functioning and the Course of PTSD Symptoms
3.4. Cognitive Functioning and the Course of PTSD Cluster Symptoms
4. Discussion
4.1. Course of PTSD Symptoms
4.2. Cognitive Functioning and the Course of PTSD Symptoms
4.3. Cognitive Functioning and the Course of PTSD Cluster Symptoms
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cristofori, I.; Levin, H.S. Traumatic brain injury and cognition. Handb. Clin. Neurol. 2015, 128, 579–611. [Google Scholar] [CrossRef]
- Rabinowitz, A.R.; Levin, H.S. Cognitive sequelae of traumatic brain injury. Psychiatr. Clin. N. Am. 2014, 37, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, L.; Stewart, W.; Dams-O’Connor, K.; Diaz-Arrastia, R.; Horton, L.; Menon, D.K.; Polinder, S. The chronic and evolving neurological consequences of traumatic brain injury. Lancet Neurol. 2017, 16, 813–825. [Google Scholar] [CrossRef] [Green Version]
- Scholten, A.C.; Haagsma, J.A.; Cnossen, M.C.; Olff, M.; van Beeck, E.F.; Polinder, S. Prevalence of and risk factors for anxiety and depressive disorders after traumatic brain injury: A systematic review. J. Neurotrauma 2016, 33, 1969–1994. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, D.; Cnossen, M.C.; Polinder, S.; Wilson, L.; Maas, A. Post-Traumatic Stress Disorder after Civilian Traumatic Brain Injury: A Systematic Review and Meta-Analysis of Prevalence Rates. J. Neurotrauma 2019, 36, 3220–3232. [Google Scholar] [CrossRef]
- Bryant, R. Post-traumatic stress disorder vs traumatic brain injury. Dialogues Clin. Neurosci. 2011, 13, 251–262. [Google Scholar] [CrossRef]
- Howlett, J.R.; Stein, M.B. Post-Traumatic Stress Disorder: Relationship to Traumatic Brain Injury and Approach to Treatment. In Translational Research in Traumatic Brain Injury; Laskowitz, D., Grant, G., Eds.; CRC Press/Taylor and Francis Group: Boca Raton, FL, USA, 2016. [Google Scholar]
- Merz, Z.C.; Roskos, P.T.; Gfeller, J.D.; Bucholz, R.D. Impact of psychiatric symptomatology on neuropsychological assessment performance in persons with TBI: A comparison of OEF/OIF veteran and civilian samples. Brain Inj. 2017, 31, 1422–1428. [Google Scholar] [CrossRef]
- Scott, J.C.; Matt, G.E.; Wrocklage, K.M.; Crnich, C.; Jordan, J.; Southwick, S.M.; Krystal, J.H.; Schweinsburg, B.C. A quantitative meta-analysis of neurocognitive functioning in posttraumatic stress disorder. Psychol. Bull. 2015, 141, 105–140. [Google Scholar] [CrossRef]
- Verfaellie, M.; Lafleche, G.; Spiro, A.; Bousquet, K. Neuropsychological outcomes in OEF/OIF veterans with self-report of blast exposure: Associations with mental health, but not MTBI. Neuropsychology 2014, 28, 337–346. [Google Scholar] [CrossRef] [Green Version]
- Pineau, H.; Marchand, A.; Guay, S. Objective neuropsychological deficits in post-traumatic stress disorder and mild traumatic brain injury: What remains beyond symptom similarity? Behav. Sci. 2014, 4, 471–486. [Google Scholar] [CrossRef] [Green Version]
- Shandera-Ochsner, A.L.; Berry, D.T.; Harp, J.P.; Edmundson, M.; Graue, L.O.; Roach, A.; High, W.M. Neuropsychological effects of self-reported deployment-related mild TBI and current PTSD in OIF/OEF veterans. Clin. Neuropsychol. 2013, 27, 881–907. [Google Scholar] [CrossRef]
- Van Praag, D.; Van Den Eede, F.; Wouters, K.; Wilson, L.; Maas, A.I.R. Neurocognitive correlates of posttraumatic stress disorder following traumatic brain injury. J. Psychosom. Res. 2021, 145, 110448. [Google Scholar] [CrossRef]
- Marx, B.P.; Doron-Lamarca, S.; Proctor, S.P.; Vasterling, J.J. The influence of pre-deployment neurocognitive functioning on post-deployment PTSD symptom outcomes among Iraq-deployed Army soldiers. J. Int. Neuropsychol. Soc. 2009, 15, 840–852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parslow, R.A.; Jorm, A.F. Pretrauma and posttrauma neurocognitive functioning and PTSD symptoms in a community sample of young adults. Am. J. Psychiatry 2007, 164, 509–515. [Google Scholar] [CrossRef]
- Vasterling, J.J.; Aslan, M.; Lee, L.O.; Proctor, S.P.; Ko, J.; Jacob, S.; Concato, J. Longitudinal associations among Posttraumatic stress disorder symptoms, traumatic brain injury and neurocognitive functioning in Army Soldiers deployed to the Iraq War. J. Int. Neuropsychol. Soc. 2018, 24, 311–323. [Google Scholar] [CrossRef]
- Brewin, C.R.; Gregory, J.D.; Lipton, M.; Burgess, N. Intrusive images in psychological disorders: Characteristics, neural mechanisms, and treatment implications. Psychol. Rev. 2010, 117, 210–232. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Edmed, S.L.; Anderson, V.; Kenardy, J. Neurocognitive predictors of posttraumatic stress disorder symptoms in children 6 months after traumatic brain injury: A prospective study. Neuropsychology 2017, 31, 84–92. [Google Scholar] [CrossRef]
- Alway, Y.; Gould, K.R.; McKay, A.; Johnston, L.; Ponsford, J. The Evolution of Post-Traumatic Stress Disorder following Moderate-to-Severe Traumatic Brain Injury. J. Neurotrauma 2016, 33, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Visser, E.; Gosens, T.; Den Oudsten, B.L.; De Vries, J. The course, prediction, and treatment of acute and posttraumatic stress in trauma patients: A systematic review. J. Trauma Acute Care Surg. 2017, 82, 1158–1183. [Google Scholar] [CrossRef]
- Maas, A.I.; Menon, D.K.; Steyerberg, E.W.; Citerio, G.; Lecky, F.; Manley, G.T.; Hill, S.; Legrand, V.; Sorgner, A.; CENTER-TBI Participants and Investigators. Collaborative European NeuroTrauma Effectiveness Research in Traumatic Brain Injury (CENTER-TBI): A prospective longitudinal observational study. Neurosurgery 2015, 76, 67–80. [Google Scholar] [CrossRef] [Green Version]
- Steyerberg, E.W.; Wiegers, E.; Sewalt, C.; Buki, A.; Citerio, G.; De Keyser, V.; Ercole, A.; Kunzmann, K.; Lanyon, L.; Lecky, F.; et al. Case-mix, care pathways, and outcomes in patients with traumatic brain injury in CENTER-TBI: A European prospective, multicenter, longitudinal, cohort study. Lancet Neurol. 2019, 18, 923–934. [Google Scholar] [CrossRef]
- Wilson, J.T.; Pettigrew, L.E.; Teasdale, G.M. Structured interviews for the Glasgow Outcome Scale and the extended Glasgow Outcome Scale: Guidelines for their use. J. Neurotrauma 1998, 15, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Bagiella, E.; Novack, T.A.; Ansel, B.; Diaz-Arrastia, R.; Dikmen, S.; Hart, T.; Temkin, N. Measuring outcome in traumatic brain injury treatment trials: Recommendations from the traumatic brain injury clinical trials network. J. Head Trauma Rehabil. 2010, 25, 375–382. [Google Scholar] [CrossRef] [Green Version]
- Weathers, F.L.; Litz, B.T.; Keane, T.M.; Palmieri, P.A.; Marx, B.P.; Schnurr, P.P. The PTSD Checklist for DSM-5 (PCL-5). 2013. Available online: https://www.ptsd.va.gov (accessed on 1 July 2021).
- Wortmann, J.H.; Jordan, A.H.; Weathers, F.W.; Resick, P.A.; Dondanville, K.A.; Hall-Clark, B.; Foa, E.B.; Young-McCaughan, S.; Yarvis, J.S.; Hembree, E.A.; et al. Psychometric analysis of the PTSD Checklist-5 (PCL-5) among treatment-seeking military service members. Psychol. Assess. 2016, 28, 1392–1403. [Google Scholar] [CrossRef]
- Hoge, C.W.; Riviere, L.A.; Wilk, J.E.; Herrell, R.K.; Weathers, F.W. The prevalence of post-traumatic stress disorder (PTSD) in US combat soldiers: A head-to-head comparison of DSM-5 versus DSM-IV-TR symptom criteria with the PTSD checklist. Lancet Psychiatry 2014, 1, 269–277. [Google Scholar] [CrossRef]
- Reitan, R.M. Trail Making Test: Manual for Administration and Scoring; Reitan Neuropsychology Laboratory: Tucson, AZ, USA, 1992. [Google Scholar]
- Rey, A. L’examen psychologique dans les cas d’encéphalopathie traumatique. Arch. Psychol. 1941, 28, 21. [Google Scholar]
- Schmidt, M. Rey Auditory Verbal Learning Test: A Handbook; Western Psychological Services: Los Angeles, CA, USA, 1996. [Google Scholar]
- Cambridge Cognition. CANTAB Research Suite 6: Test Administration Guide; Cambridge Cognition Ltd.: Cambridge, UK, 2014. [Google Scholar]
- IBM Corp. IBM SPSS Statistics for Windows, Version 25.0; IBM Corp: Armonk, NY, USA, 2017. [Google Scholar]
- Teasdale, G.; Jennett, B. Assessment of coma and impaired consciousness. A practical scale. Lancet (London England) 1974, 2, 81–84. [Google Scholar] [CrossRef]
- Alway, Y.; McKay, A.; Gould, K.R.; Johnston, L.; Ponsford, J. Factors associated with posttraumatic stress disorder following moderate to severe traumatic brain injury: A prospective study. Depress. Anxiety 2016, 33, 19–26. [Google Scholar] [CrossRef]
- Nijdam, M.J.; Gersons, B.P.R.; Olff, M. The role of major depression in neurocognitive functioning in patients with posttraumatic stress disorder. Eur. J. Psychotraumatol. 2013, 4, 19979. [Google Scholar] [CrossRef]
- Ben-Zion, Z.; Fine, N.B.; Keynan, N.J.; Admon, R.; Green, N.; Halevi, M.; Fonzo, G.A.; Achituv, M.; Merin, O.; Sharon, H.; et al. Cognitive Flexibility Predicts PTSD Symptoms: Observational and Interventional Studies. Front. Psychiatry 2018, 9, 477. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Moghaddam, B. Impact of anxiety on prefrontal cortex encoding of cognitive flexibility. Neuroscience 2017, 345, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Shin, L.M.; Whalen, P.J.; Pitman, R.K.; Bush, G.; Macklin, M.L.; Lasko, N.B.; Orr, S.P.; McInerney, S.C.; Rauch, S.L. An fMRI study of anterior cingulate function in posttraumatic stress disorder. Biol. Psychiatry 2001, 50, 932–942. [Google Scholar] [CrossRef]
- Wisdom, N.M.; Pastorek, N.J.; Miller, B.I.; Booth, J.E.; Romesser, J.M.; Linck, J.F.; Sim, A.H. PTSD and cognitive functioning: Importance of including performance validity testing. Clin. Neuropsychol. 2014, 28, 128–145. [Google Scholar] [CrossRef]
- Boone, K.B.; Lu, P.; Wen, J. Comparison of various RAVLT scores in the detection of noncredible memory performance. Arch. Clin. Neuropsychol. 2015, 20, 301–319. [Google Scholar] [CrossRef] [Green Version]
- Iverson, G.L.; Lange, R.T.; Green, P.; Franzen, M.D. Detecting exaggeration and malingering with the Trail Making Test. Clin. Neuropsychol. 2002, 16, 398–406. [Google Scholar] [CrossRef]
- Sherman, E.M.S.; Brooks, B.L.; Iverson, G.L.; Slick, D.J.; Strauss, E. Reliability and Validity in Neuropsychology. In The Little Black Book of Neuropsychology; Schoenberg, M., Scott, J., Eds.; Springer: Boston, MA, USA, 2011. [Google Scholar] [CrossRef]
- Jak, A.J.; Jurick, S.; Crocker, L.D.; Sanderson-Cimino, M.; Aupperle, R.; Rodgers, C.S.; Thomas, K.R.; Boyd, B.; Norman, S.B.; Lang, A.J.; et al. SMART-CPT for veterans with comorbid post-traumatic stress disorder and history of traumatic brain injury: A randomised controlled trial. J. Neurol. Neurosurg. Psychiatry 2019, 90, 333–341. [Google Scholar] [CrossRef]
| Demographic Characteristics | Median (IQR) 1 or n (%) |
|---|---|
| Age (years) >65 years | 49 (31–61) 102/671 (15.2) |
| Male Highest educational level: College/University Married or living with partner | 463/671 (69.0) 188/614 (30.6) 369/648 (56.9) |
| Injury-related characteristics | |
| Glasgow Coma Scale: Mild TBI (13–15) Moderate TBI (9–12) Severe TBI (3–8) | 474/671 (70.6) 67/671 (10.0) 130/671 (19.4) |
| Cause of injury: Road traffic incident Incidental fall Violence/assault/act of mass violence Suicide attempt | 321/656 (48.9) 244/656 (37.2) 24/656 (3.7) 12/656 (1.8) |
| Care pathway: Admitted to hospital Intensive Care Unit | 32/671 (47.7) 351/671 (52.3) |
| Psychiatric history ² | |
| Psychiatric disorders Type of psychiatric disorder: Depression Anxiety Sleep disorder Substance abuse | 68/671 (10.1) 37/68 (54.4) 18/68 (26.5) 11/68 (16.2) 8/68 (11.8) |
| Psychiatric characteristics at follow-up | |
| PCL-5 3 total score 6 months PTSD probable diagnosis 6 months PCL-5 total score 12 months PTSD probable diagnosis 12 months PHQ-9 4 total score 6 months GAD-7 5 total score 6 months Psychotropic medication at 6 months Type of medication: Antidepressants Anxiolytics Antipsychotic agents | 8 (3–18) 79/671 (11.8) 8 (2–18) 71/671 (10.6) 3 (1–7) 2 (0–5) 143/633 (22.6) 38/143 (26.6) 21/143 (14.7) 11/143 (7.7) |
| Covariate | B | SE (B) | 95% CI 1 | p 4 | VIF 5 | |
|---|---|---|---|---|---|---|
| LL 2 | UL 3 | |||||
| (Intercept) | −2.45 | 1.94 | −6.25 | 1.35 | 0.21 | |
| Age | −0.032 | 0.022 | −0.075 | 0.012 | 0.15 | 1.22 |
| Psychiatric history | 2.14 | 1.18 | −0.18 | 4.46 | 0.071 | 1.01 |
| GCS 6 | −0.27 | 0.095 | −0.46 | −0.082 | 0.005 | 1.23 |
| PTSD diagnosis 6mo | −10.09 | 1.11 | −12.28 | −7.91 | <0.001 | 1.02 |
| CANTAB 7 RVP 8 | −1.04 | 0.40 | −1.82 | −0.26 | 0.009 | 1.16 |
| CANTAB AST 9 | 2.67 | 1.08 | 0.55 | 4.79 | 0.014 | 1.19 |
| GCS × CANTAB AST | −0.21 | 0.086 | −0.38 | −0.037 | 0.017 | |
| Covariates | Cluster Intrusion (5 Items) | Cluster Avoidance (2 Items) | Cluster Cognition/Mood (7 Items) | Cluster Arousal (6 Items) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B (SE) | p 1 | VIF 2 | B (SE) | p 1 | VIF 2 | B (SE) | p 1 | VIF 2 | B (SE) | p 1 | VIF 2 | |
| Intercept | −0.98 (0.58) | 0.092 | −0.79 (0.26) | 0.003 | 2.32 (0.84) | 0.006 | −0.84 (0.63) | 0.18 | ||||
| Age | −0.014 (0.010) | 0.16 | 1.13 | |||||||||
| Psychiatric history | 0.84 (0.38) | 0.027 | 1.01 | 0.25 (0.57) | 0.66 | 1.00 | 0.53 (0.41) | 0.20 | 1.01 | |||
| GCS 3 | −0.051 (0.029) | 0.080 | 1.02 | −0.029 (0.016) | 0.060 | 1.03 | −0.12 (0.046) | 0.007 | 1.11 | −0.092 (0.031) | 0.003 | 1.03 |
| PTSD 6mo | −2.64 (0.36) | <0.001 | 1.01 | −1.26 (0.19) | <0.001 | 1.01 | −2.67 (0.38) | <0.001 | 1.02 | |||
| CANTAB 4 RVP 5 | −0.23 (0.12) | 0.053 | 1.03 | −0.40 (0.19) | 0.033 | 1.08 | −0.31 (0.15) | 0.031 | 1.29 | |||
| CANTAB AST 6 | 0.70 (0.18) | <0.001 | 1.05 | 1.05 (0.38) | 0.005 | 1.11 | ||||||
| GCS × CANTAB AST | −0.050 (0.015) | 0.001 | −0.083 (0.030) | 0.006 | ||||||||
| CANTAB RTI 7 | 0.46 (0.17) | 0.006 | 1.14 | |||||||||
| GCS × CANTAB RTI | −0.034 (0.014) | 0.013 | ||||||||||
| RAVLT-Delayed recall 8 | 0.31 (0.14) | 0.023 | 1.21 | |||||||||
| F (df, between and within) | F(4, 666) = 16.10 | <0.001 | F(6, 664) = 12.68 | <0.001 | F(4, 666) = 19.05 | <0.001 | F(7, 663) = 11.07 | <0.001 | ||||
| Adjusted R² | 0.083 | 0.095 | 0.020 | 0.095 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Praag, D.L.G.; Van Den Eede, F.; Wouters, K.; Wilson, L.; Maas, A.I.R.; The CENTER-TBI Investigators and Participants. The Impact of Neurocognitive Functioning on the Course of Posttraumatic Stress Symptoms following Civilian Traumatic Brain Injury. J. Clin. Med. 2021, 10, 5109. https://doi.org/10.3390/jcm10215109
Van Praag DLG, Van Den Eede F, Wouters K, Wilson L, Maas AIR, The CENTER-TBI Investigators and Participants. The Impact of Neurocognitive Functioning on the Course of Posttraumatic Stress Symptoms following Civilian Traumatic Brain Injury. Journal of Clinical Medicine. 2021; 10(21):5109. https://doi.org/10.3390/jcm10215109
Chicago/Turabian StyleVan Praag, Dominique L. G., Filip Van Den Eede, Kristien Wouters, Lindsay Wilson, Andrew I. R. Maas, and The CENTER-TBI Investigators and Participants. 2021. "The Impact of Neurocognitive Functioning on the Course of Posttraumatic Stress Symptoms following Civilian Traumatic Brain Injury" Journal of Clinical Medicine 10, no. 21: 5109. https://doi.org/10.3390/jcm10215109
APA StyleVan Praag, D. L. G., Van Den Eede, F., Wouters, K., Wilson, L., Maas, A. I. R., & The CENTER-TBI Investigators and Participants. (2021). The Impact of Neurocognitive Functioning on the Course of Posttraumatic Stress Symptoms following Civilian Traumatic Brain Injury. Journal of Clinical Medicine, 10(21), 5109. https://doi.org/10.3390/jcm10215109






