Prevalence of the Absence of Cirrhosis in Subjects with NAFLD-Associated Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction
2.3. Study Quality Assessment
2.4. Data Analysis
3. Results
3.1. Study Characteristics
3.2. Qualitative Analysis
3.3. Quantitative Analysis
3.4. Study Quality Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic as-sessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [Green Version]
- Parrish, N.F.; Feurer, I.D.; Matsuoka, L.K.; Rega, S.A.; Perri, R.; Alexopoulos, S.P. The Changing Face of Liver Transplantation in the United States: The Effect of HCV Antiviral Eras on Transplantation Trends and Outcomes. Transplant. Direct 2019, 5, e427. [Google Scholar] [CrossRef]
- Belli, L.S.; Perricone, G.; Adam, R.; Cortesi, P.A.; Strazzabosco, M.; Facchetti, R.; Karam, V.; Salizzoni, M.; Andujar, R.L.; Fondevila, C.; et al. Impact of DAAs on liver transplantation: Major effects on the evolution of indications and results. An ELITA study based on the ELTR registry. J. Hepatol. 2018, 69, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; LaVine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2017, 67, 328–357. [Google Scholar] [CrossRef]
- Frenette, C.T.; Isaacson, A.J.; Bargellini, I.; Saab, S.; Singal, A.G. A Practical Guideline for Hepatocellular Carcinoma Screening in Patients at Risk. Mayo Clin. Proc. Innov. Qual. Outcomes 2019, 3, 302–310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kokudo, N.; Takemura, N.; Hasegawa, K.; Takayama, T.; Kubo, S.; Shimada, M.; Nagano, H.; Hatano, E.; Izumi, N.; Kaneko, S.; et al. Clinical practice guidelines for hepatocellular carcinoma: The Japan Society of Hepatology 2017 (4th JSH-HCC guidelines) 2019 update. Hepatol. Res. 2019, 49, 1109–1113. [Google Scholar] [CrossRef]
- Singal, A.G.; Lampertico, P.; Nahon, P. Epidemiology and surveillance for hepatocellular carcinoma: New trends. J. Hepatol. 2020, 72, 250–261. [Google Scholar] [CrossRef] [Green Version]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Heart, Lung and Blood Institute. Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 1 April 2021).
- Ahn, S.Y.; Kim, S.B.; Song, I.H. Clinical Patterns and Outcome of Hepatocellular Carcinoma in Patients with Nonalcoholic Fatty Liver Disease. Can. J. Gastroenterol. Hepatol. 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Amaddeo, G.; Brustia, R.; Allaire, M.; Lequoy, M.; Hollande, C.; Regnault, H.; Blaise, L.; Ganne-Carrié, N.; Séror, O.; Larrey, E.; et al. Impact of COVID-19 on the management of hepatocellular carcinoma in a high-prevalence area. JHEP Rep. 2021, 3, 100199. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, B.; Stål, P.; Wahlin, S.; Björkström, N.K.; Hagström, H. Characteristics and outcome of hepatocellular carcinoma in patients with NAFLD without cirrhosis. Liver Int. 2019, 39, 1098–1108. [Google Scholar] [CrossRef] [PubMed]
- Benhammou, J.N.; Aby, E.; Shirvanian, G.; Manansala, K.; Hussain, S.K.; Tong, M.J. Improved survival after treatments of patients with nonalcoholic fatty liver disease associated hepatocellular carcinoma. Sci. Rep. 2020, 10, 9902. [Google Scholar] [CrossRef]
- Best, J.; Bechmann, L.P.; Sowa, J.P.; Sydor, S.; Dechêne, A.; Pflanz, K.; Bedreli, S.; Schotten, C.; Geier, A.; Berg, T.; et al. GALAD Score Detects Early Hepatocellular Carcinoma in an International Cohort of Patients with Nonalcoholic Steatohepatitis. Clin. Gastroenterol. Hepatol. 2020, 18, 728–735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Billeter, A.T.; Müller, P.C.; Albrecht, T.; Roessler, S.; Löffler, M.; Lemekhova, A.; Mehrabi, A.; Müller-Stich, B.P.; Hoffmann, K. Impact of Type 2 Diabetes on Oncologic Outcomes of Hepatocellular Carcinomas in Non-Cirrhotic, Non-alcoholic Steatohepatitis: A Matched-Pair Analysis. J. Gastrointest. Surg. 2021, 25, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Canbay, A.; Kachru, N.; Haas, J.S.; Sowa, J.-P.; Meise, D.; Ozbay, A.B. Patterns and predictors of mortality and disease progression among patients with non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2020, 52, 1185–1194. [Google Scholar] [CrossRef]
- Cotrim, H.P.; Oliveira, C.P.; Coelho, H.S.M.; Alvares-Da-Silva, M.R.; Nabuco, L.; Parise, E.R.; Ivantes, C.; Martinelli, A.L.; Galizzi-Filho, J.; Carrilho, F.J. Nonalcoholic steatohepatitis and hepatocellular carcinoma: Brazilian survey. Clinics 2016, 71, 281–284. [Google Scholar] [CrossRef]
- Debes, J.D.; Chan, A.J.; Balderramo, D.; Kikuchi, L.; Ballerga, E.G.; Prieto, J.E.; Tapias, M.; Idrovo, V.; Davalos, M.B.; Cairo, F.; et al. Hepatocellular carcinoma in South America: Evaluation of risk factors, demographics and therapy. Liver Int. 2018, 38, 136–143. [Google Scholar] [CrossRef]
- Dyson, J.; Jaques, B.; Chattopadyhay, D.; Lochan, R.; Graham, J.; Das, D.; Aslam, T.; Patanwala, I.; Gaggar, S.; Cole, M.; et al. Hepatocellular cancer: The impact of obesity, type 2 diabetes and a multidis-ciplinary team. J. Hepatol. 2014, 60, 110–117. [Google Scholar] [CrossRef]
- Gawrieh, S.; Dakhoul, L.; Miller, E.; Scanga, A.; Delemos, A.; Kettler, C.; Burney, H.; Liu, H.; Abu-Sbeih, H.; Chalasani, N.; et al. Characteristics, aetiologies and trends of hepatocellular carcinoma in patients without cirrhosis: A United States multicentre study. Aliment. Pharmacol. Ther. 2019, 50, 809–821. [Google Scholar] [CrossRef]
- Hester, C.A.; Rich, N.E.; Singal, A.G.; Yopp, A.C. Comparative Analysis of Nonalcoholic Steatohepatitis—Versus Viral Hepatitis—and Alcohol-Related Liver Disease–Related Hepatocellular Carcinoma. J. Natl. Compr. Cancer Netw. 2019, 17, 322–329. [Google Scholar] [CrossRef] [Green Version]
- Kanwal, F.; Kramer, J.R.; Mapakshi, S.; Natarajan, Y.; Chayanupatkul, M.; Richardson, P.A.; Li, L.; Desiderio, R.; Thrift, A.P.; Asch, S.M.; et al. Risk of Hepatocellular Cancer in Patients with Non-Alcoholic Fatty Liver Disease. Gastroenterology 2018, 155, 1828–1837.e2. [Google Scholar] [CrossRef] [Green Version]
- Kodama, K.; Kawaguchi, T.; Hyogo, H.; Nakajima, T.; Ono, M.; Seike, M.; Takahashi, H.; Nozaki, Y.; Kawanaka, M.; Tanaka, S.; et al. Clinical features of hepatocellular carcinoma in nonalcoholic fatty liver disease patients without advanced fibrosis. J. Gastroenterol. Hepatol. 2019, 34, 1626–1632. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.X.; Tan, H.J.; Liew, Y.X.; Syn, N.; Teo, J.Y.; Lee, S.Y.; Goh, B.K.P.; Goh, G.B.B.; Chan, C.Y. Liver Resection for Nonalcoholic Fatty Liver Disease-Associated Hepatocellular Carcinoma. J. Am. Coll. Surg. 2019, 229, 467–478.e1. [Google Scholar] [CrossRef]
- Leung, C.; Yeoh, S.W.; Patrick, D.; Ket, S.; Marion, K.; Gow, P.; Angus, P.W. Characteristics of hepatocellular carcinoma in cirrhotic and non-cirrhotic non-alcoholic fatty liver disease. World J. Gastroenterol. 2015, 21, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-L.; Patman, G.L.; Leathart, J.B.S.; Piguet, A.-C.; Burt, A.D.; Dufour, J.-F.; Day, C.P.; Daly, A.K.; Reeves, H.L.; Anstee, Q.M. Carriage of the PNPLA3 rs738409 C >G polymorphism confers an increased risk of non-alcoholic fatty liver disease associated hepatocellular carcinoma. J. Hepatol. 2014, 61, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, B.; Shah, V.; Onyshchenko, M.; ElShamy, M.; Aucejo, F.; Lopez, R.; Hanouneh, I.A.; Alhaddad, R.; Alkhouri, N. Characterization of hepatocellular carcinoma (HCC) in non-alcoholic fatty liver disease (NAFLD) patients without cirrhosis. Hepatol. Int. 2015, 10, 632–639. [Google Scholar] [CrossRef]
- Piñero, F.; Pages, J.; Marciano, S.; Fernandez, N.; Silva, J.; Anders, M.; Zerega, A.; Ridruejo, E.; Ameigeiras, B.; D’Amico, C.; et al. Fatty liver disease, an emerging etiology of hepatocellular carcinoma in Argentina. World J. Hepatol. 2018, 10, 41–50. [Google Scholar] [CrossRef]
- Piscaglia, F.; Svegliati-Baroni, G.; Barchetti, A.; Pecorelli, A.; Marinelli, S.; Tiribelli, C.; Bellentani, S. HCC-NAFLD Italian Study Group Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: A multicenter prospective study. Hepatology 2016, 63, 827–838. [Google Scholar] [CrossRef]
- Sanyal, A.; Poklepovic, A.; Moyneur, E.; Barghout, V. Population-based risk factors and resource utilization for HCC: US perspective. Curr. Med. Res. Opin. 2010, 26, 2183–2191. [Google Scholar] [CrossRef]
- Tateishi, R.; Okanoue, T.; Fujiwara, N.; Okita, K.; Kiyosawa, K.; Omata, M.; Kumada, H.; Hayashi, N.; Koike, K. Clinical characteristics, treatment, and prognosis of non-B, non-C hepatocellular carcinoma: A large retrospective multicenter cohort study. J. Gastroenterol. 2015, 50, 350–360. [Google Scholar] [CrossRef] [Green Version]
- Ni Than, N.; Ghazanfar, A.; Hodson, J.; Tehami, N.; Coldham, C.; Mergental, H.; Manas, D.; Shah, T.; Newsome, P.N.; Reeves, H.; et al. Comparing clinical presentations, treatments and outcomes of hepatocellular carcinoma due to hepatitis C and non-alcoholic fatty liver disease. Qjm Int. J. Med. 2017, 110, 73–81. [Google Scholar] [CrossRef] [Green Version]
- Tobari, M.; Hashimoto, E.; Taniai, M.; Kodama, K.; Kogiso, T.; Tokushige, K.; Yamamoto, M.; Takayoshi, N.; Satoshi, K.; Tatsuo, A. The characteristics and risk factors of hepatocellular carcinoma in nonalcoholic fatty liver disease without cirrhosis. J. Gastroenterol. Hepatol. 2020, 35, 862–869. [Google Scholar] [CrossRef]
- Tokushige, K.; Hashimoto, E.; Kodama, K. Hepatocarcinogenesis in non-alcoholic fatty liver disease in Japan. J. Gastroenterol. Hepatol. 2013, 28, 88–92. [Google Scholar] [CrossRef] [Green Version]
- Van Meer, S.; van Erpecum, K.J.; Sprengers, D.; Coenraad, M.J.; Klümpen, H.J.; Jansen, P.L.M.; Ijzermans, J.N.M.; Verheij, J.; van Nieuwkerk, C.M.J.; Siersema, P.D.; et al. Hepatocellular carcinoma in cirrhotic versus noncirrhotic livers: Results from a large cohort in the Netherlands. Eur. J. Gastroenterol. Hepatol. 2016, 28, 352–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, C.; Njei, B.; Nguyen, M.H.; Nguyen, A.; Lim, J.K. Survival after treatment with curative intent for hepatocellular carcinoma among patients with vs without non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2017, 46, 1061–1069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, T.; Hu, L.Y.; Li, Z.L.; Liu, K.; Wu, H.; Xing, H.; Lau, W.Y.; Pawlik, T.M.; Zeng, Y.-Y.; Zhou, Y.-H.; et al. Liver Resection for Hepatocellular Carcinoma in Non-alcoholic Fatty Liver Disease: A Multicenter Propensity Matching Analysis with HBV-HCC. J. Gastrointest. Surg. 2020, 24, 320–329. [Google Scholar] [CrossRef]
- Yasui, K.; Hashimoto, E.; Komorizono, Y.; Koike, K.; Arii, S.; Imai, Y.; Shima, T.; Kanbara, Y.; Saibara, T.; Mori, T.; et al. Characteristics of Patients with Nonalcoholic Steatohepatitis Who Develop Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2011, 9, 428–433. [Google Scholar] [CrossRef]
- Yoon, C.H.; Jin, Y.J.; Lee, J.W. Nonalcoholic fatty liver disease-associated hepatocellular carcinoma in a hepatitis B virus-endemic area. Eur. J. Gastroenterol. Hepatol. 2018, 30, 1090–1096. [Google Scholar] [CrossRef]
- Angulo, P. Nonalcoholic fatty liver disease. N. Engl. J. Med. 2002, 346, 1221–1231. [Google Scholar] [CrossRef] [Green Version]
- Stine, J.G.; Wentworth, B.J.; Zimmet, A.; Rinella, M.E.; Loomba, R.; Caldwell, S.H.; Argo, C.K. Systematic review with meta-analysis: Risk of hepatocellular carcinoma in non-alcoholic steatohepatitis without cirrhosis compared to other liver diseases. Aliment. Pharmacol. Ther. 2018, 48, 696–703. [Google Scholar] [CrossRef]
- Mohanty, S.R.; Troy, T.N.; Huo, D.; O’Brien, B.L.; Jensen, D.M.; Hart, J. Influence of ethnicity on histological differences in non-alcoholic fatty liver disease. J. Hepatol. 2009, 50, 797–804. [Google Scholar] [CrossRef]
- Chan, T.T.; Chan, W.K.; Wong, G.L.H.; Chan, A.W.H.; Mustapha, N.R.N.; Chan, S.L.; Chong, C.C.-N.; Mahadeva, S.; Shu, S.S.-T.; Lai, P.B.-S.; et al. Positive Hepatitis B Core Antibody Is Associated with Cirrhosis and Hepato-cellular Carcinoma in Nonalcoholic Fatty Liver Disease. Am. J. Gastroenterol. 2020, 115, 867–875. [Google Scholar] [CrossRef]
- Jiang, W.; Huang, S.; Teng, H.; Wang, P.; Wu, M.; Zhou, X.; Ran, H. Diagnostic accuracy of point shear wave elastography and transient elastography for staging hepatic fibrosis in patients with non-alcoholic fatty liver disease: A meta-analysis. BMJ Open 2018, 8, e021787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loomba, R.; Lim, J.K.; Patton, H.; El-Serag, H.B. AGA Clinical Practice Update on Screening and Surveillance for Hepatocellular Carcinoma in Patients With Nonalcoholic Fatty Liver Disease: Expert Review. Gastroenterology 2020, 158, 1822–1830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chou, R.; Cuevas, C.; Fu, R.; Devine, B.; Wasson, N.; Ginsburg, A.; Zakher, B.; Pappas, M.; Graham, E.; Sullivan, S.D. Imaging Techniques for the Diagnosis of Hepatocellular Carcinoma: A Systematic Review and Meta-analysis. Ann. Intern. Med. 2015, 162, 697–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolf, E.; Rich, N.E.; Marrero, J.A.; Parikh, N.D.; Singal, A.G. Use of Hepatocellular Carcinoma Surveillance in Patients with Cirrhosis: A Systematic Review and Meta-Analysis. Hepatology 2021, 73, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Orci, L.A.; Sanduzzi-Zamparelli, M.; Caballol, B.; Sapena, V.; Colucci, N.; Torres, F.; Bruix, J.; Reig, M.; Toso, C. Incidence of Hepatocellular Carcinoma in Patients with Non-alcoholic Fatty Liver Disease: A Systematic Review, Meta-analysis, and Meta-regression. Clin. Gastroenterol. Hepatol. 2021, S1542-3565(21)00505-X. [Google Scholar] [CrossRef]
- Lee, T.; Wu, J.; Yu, S.; Lin, J.; Wu, M.-S.; Wu, C. The occurrence of hepatocellular carcinoma in different risk stratifications of clinically noncirrhotic nonalcoholic fatty liver disease. Int. J. Cancer 2017, 141, 1307–1314. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.S.; Byoun, Y.-S.; Jeong, S.-H.; Woo, B.H.; Jang, E.S.; Kim, J.-W.; Kim, H.Y. Role of the PNPLA3 I148M Polymorphism in Nonalcoholic Fatty Liver Disease and Fibrosis in Korea. Dig. Dis. Sci. 2014, 59, 2967–2974. [Google Scholar] [CrossRef]
- Sorino, P.; Caruso, M.G.; Misciagna, G.; Bonfiglio, C.; Campanella, A.; Mirizzi, A.; Franco, I.; Bianco, A.; Buongiorno, C.; Liuzzi, R.; et al. Selecting the best machine learning algorithm to support the diagnosis of Non-Alcoholic Fatty Liver Disease: A meta learner study. PLoS ONE 2020, 15, e0240867. [Google Scholar] [CrossRef]
- Reddy, S.K.; Steel, J.L.; Chen, H.W.; DeMateo, D.J.; Cardinal, J.; Behari, J.; Humar, A.; Marsh, J.W.; Geller, D.A.; Tsung, A.; et al. Outcomes of curative treatment for hepatocellular cancer in nonalcoholic steatohepatitis versus hepatitis C and alcoholic liver disease. Hepatology 2012, 55, 1809–1819. [Google Scholar] [CrossRef] [PubMed]

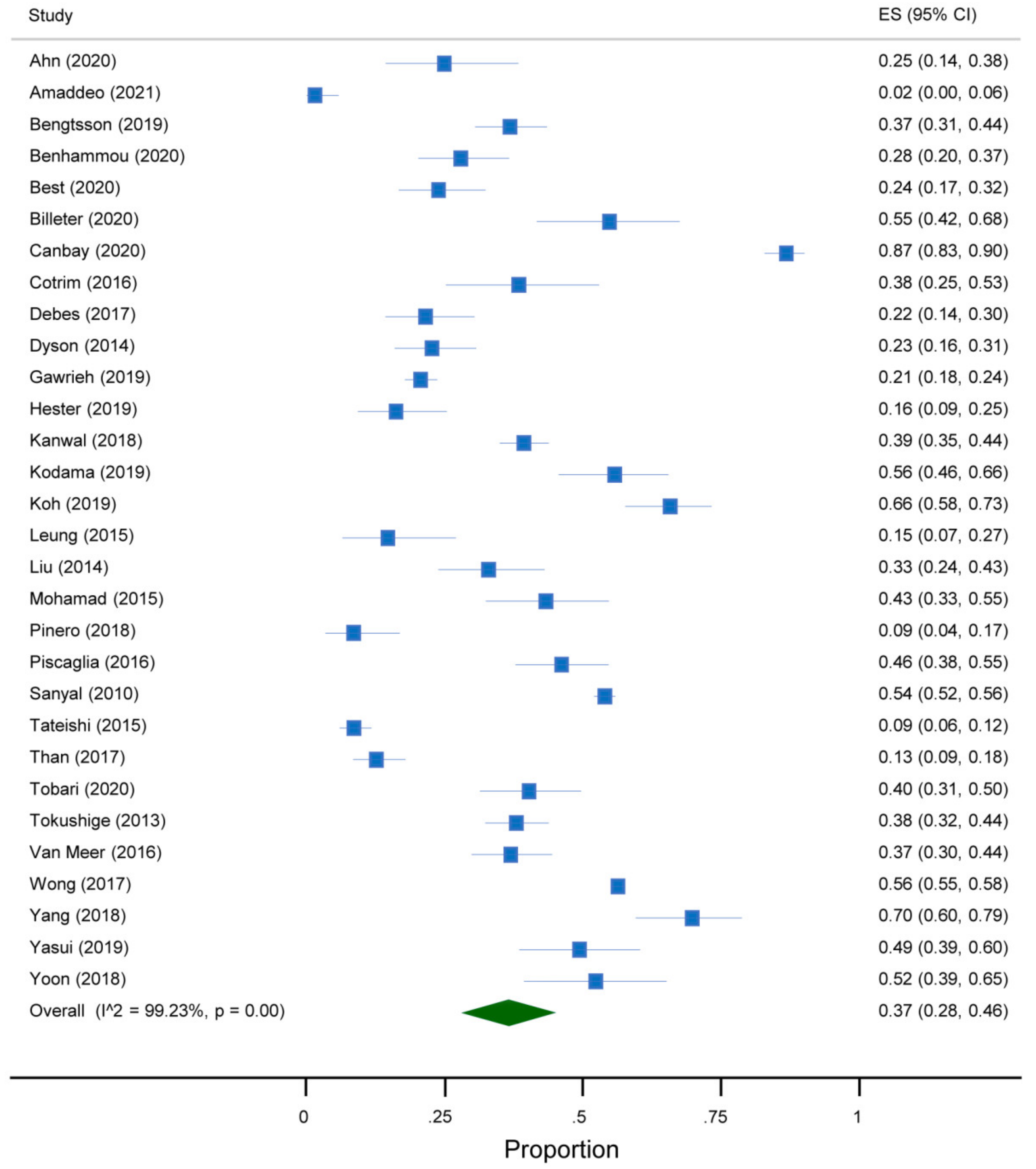
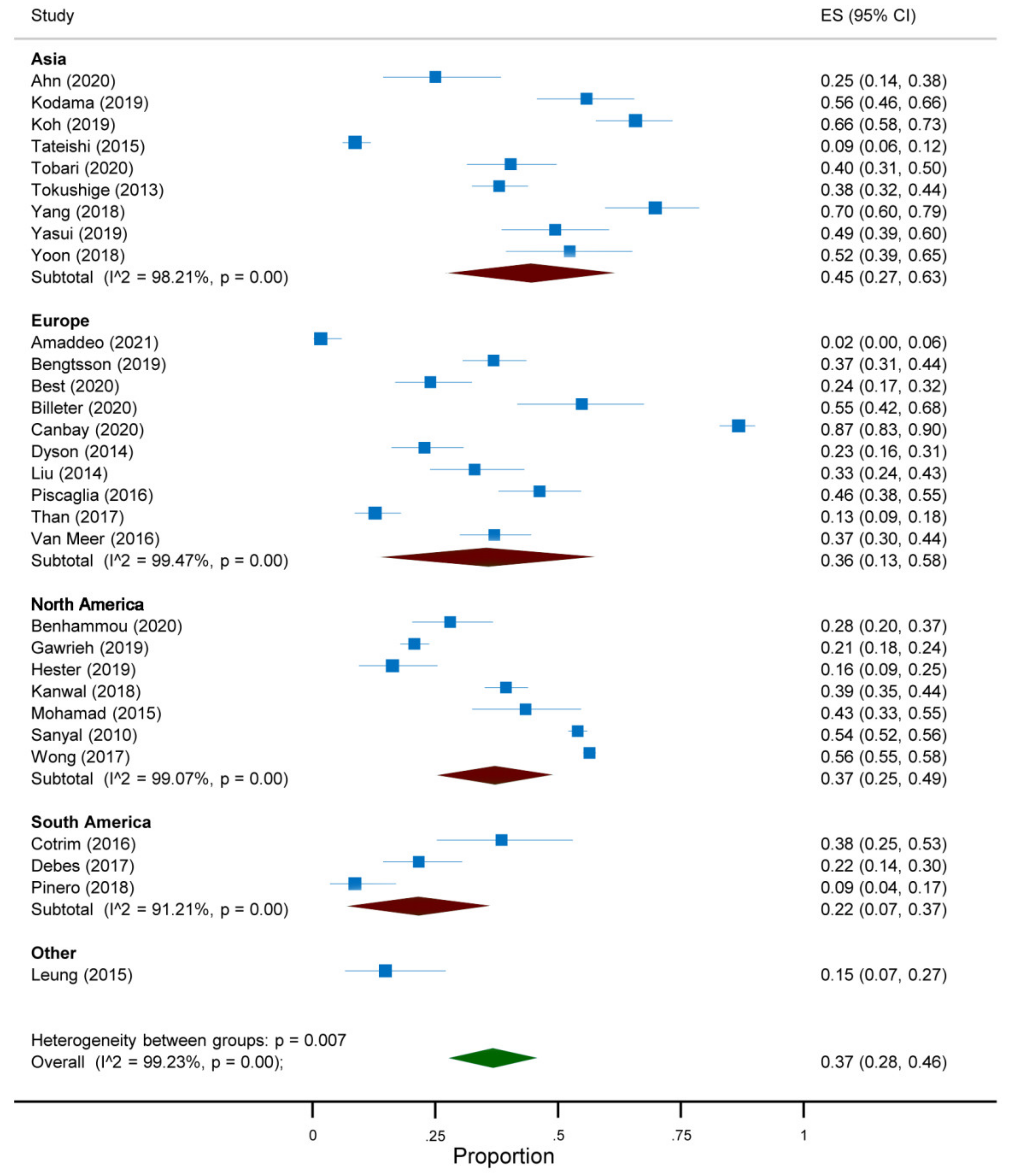
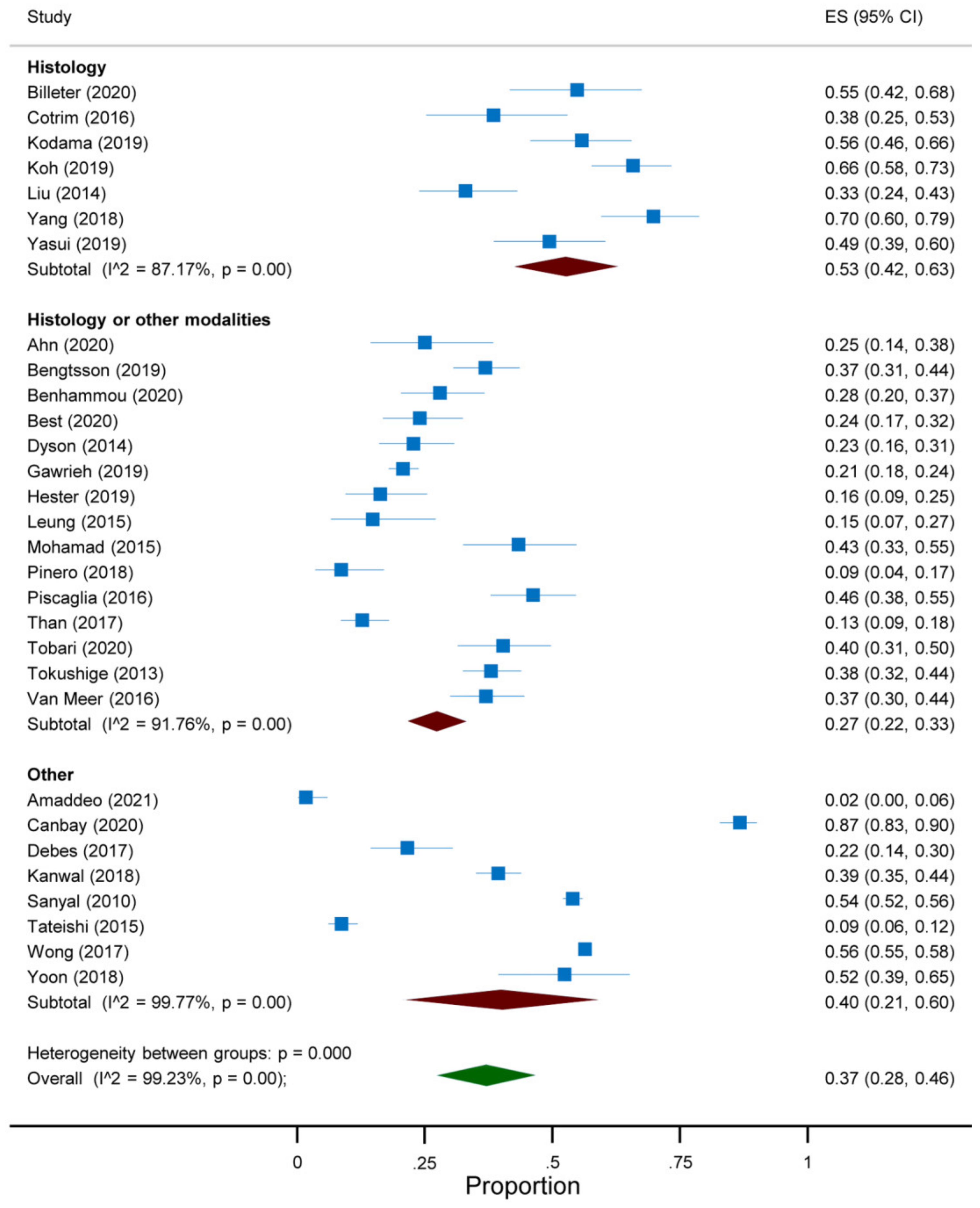

| First Author, Year | Country | Study Design | Years | Target Condition a | Participants (n) |
|---|---|---|---|---|---|
| Ahn, 2020 [10] | Republic of Korea | RCS | 2000–2016 | NAFLD | 56 |
| Amaddeo, 2021 [11] | France | RCS | 2019–2020 | NASH | 119 |
| Bengtsson, 2019 [12] | Sweden | RCS | 2004–2017 | NAFLD | 225 |
| Benhammou, 2020 [13] | USA | RCS | 2000–2016 | NAFLD | 125 |
| Best, 2020 [14] b | Germany | RCS | 2001–2016 | NASH | 125 |
| Billeter, 2020 [15] | Germany | PCS | 2001–2017 | NASH | 62 |
| Canbay, 2020 [16] | Germany | RCS | 2011–2016 | NAFLD | 363 |
| Cotrim, 2016 [17] | Brazil | RCS | - | NAFLD | 52 |
| Debes, 2017 [18] | Argentina, Brazil, Colombia, Ecuador, Peru, Uruguay | RCS | 2005–2015 | NAFLD | 111 |
| Dyson, 2014 [19] | United Kingdom | PCS | 2000–2010 | NAFLD | 136 |
| Gawrieh, 2019 [20] | USA | RCS | 2000–2014 | NAFLD | 767 |
| Hester, 2019 [21] | USA | RCS | 2008–2016 | NASH | 92 |
| Kanwal, 2018 [22] | USA | RCS | 2003–2011 | NAFLD | 490 |
| Kodama, 2019 [23] | Japan | RCS | 2000–2016 | NAFLD | 104 |
| Koh, 2019 [24] | Singapore | PCS | 2000–2015 | NAFLD | 152 |
| Leung, 2015 [25] | Australia | RCS | 2000–2012 | NAFLD | 54 |
| Liu, 2014 [26] | Switzerland, United Kingdom | - | - | NAFLD | 100 |
| Mohamad, 2015 [27] | USA | RCS | 2003–2012 | NAFLD | 83 |
| Pinero, 2018 [28] | Argentina | PCS | 2009–2016 | NAFLD | 81 |
| Piscaglia, 2016 [29] | Italy | PCS | 2010–2012 | NAFLD | 145 |
| Sanyal, 2010 [30] | USA | RCS | 2002–2008 | NAFLD | 2578 |
| Tateishi, 2015 [31] | Japan | RCS | 1991–2010 | NAFLD | 403 |
| Than, 2017 [32] | United Kingdom | RCS | 2000–2014 | NAFLD | 212 |
| Tobari, 2020 [33] | Japan | PCS | 1991–2018 | NAFLD | 119 |
| Tokushige, 2013 [34] | Japan | RCS | 2006–2009 | NAFLD | 292 |
| van Meer, 2016 [35] | the Netherlands | - | 2005–2012 | NAFLD | 181 |
| Wong, 2017 [36] | USA | RCS | 1991–2011 | NAFLD | 5898 |
| Yang, 2018 [37] | China | RCS | 2003–2014 | NAFLD | 96 |
| Yasui, 2019 [38] | Japan | RCS | 1993–2010 | NASH | 87 |
| Yoon, 2018 [39] | Republic of Korea | RCS | 2005–2015 | NAFLD | 63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castellana, M.; Donghia, R.; Lampignano, L.; Castellana, F.; Zupo, R.; Sardone, R.; Pergola, G.D.; Giannelli, G. Prevalence of the Absence of Cirrhosis in Subjects with NAFLD-Associated Hepatocellular Carcinoma. J. Clin. Med. 2021, 10, 4638. https://doi.org/10.3390/jcm10204638
Castellana M, Donghia R, Lampignano L, Castellana F, Zupo R, Sardone R, Pergola GD, Giannelli G. Prevalence of the Absence of Cirrhosis in Subjects with NAFLD-Associated Hepatocellular Carcinoma. Journal of Clinical Medicine. 2021; 10(20):4638. https://doi.org/10.3390/jcm10204638
Chicago/Turabian StyleCastellana, Marco, Rossella Donghia, Luisa Lampignano, Fabio Castellana, Roberta Zupo, Rodolfo Sardone, Giovanni De Pergola, and Gianluigi Giannelli. 2021. "Prevalence of the Absence of Cirrhosis in Subjects with NAFLD-Associated Hepatocellular Carcinoma" Journal of Clinical Medicine 10, no. 20: 4638. https://doi.org/10.3390/jcm10204638
APA StyleCastellana, M., Donghia, R., Lampignano, L., Castellana, F., Zupo, R., Sardone, R., Pergola, G. D., & Giannelli, G. (2021). Prevalence of the Absence of Cirrhosis in Subjects with NAFLD-Associated Hepatocellular Carcinoma. Journal of Clinical Medicine, 10(20), 4638. https://doi.org/10.3390/jcm10204638









