Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida
Abstract
1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Fecal Sample Collection and 16S rRNA Gene Sequencing
2.3. Statistical Analysis
3. Results
3.1. Characteristics in the SB Patients Compared with Healthy Controls
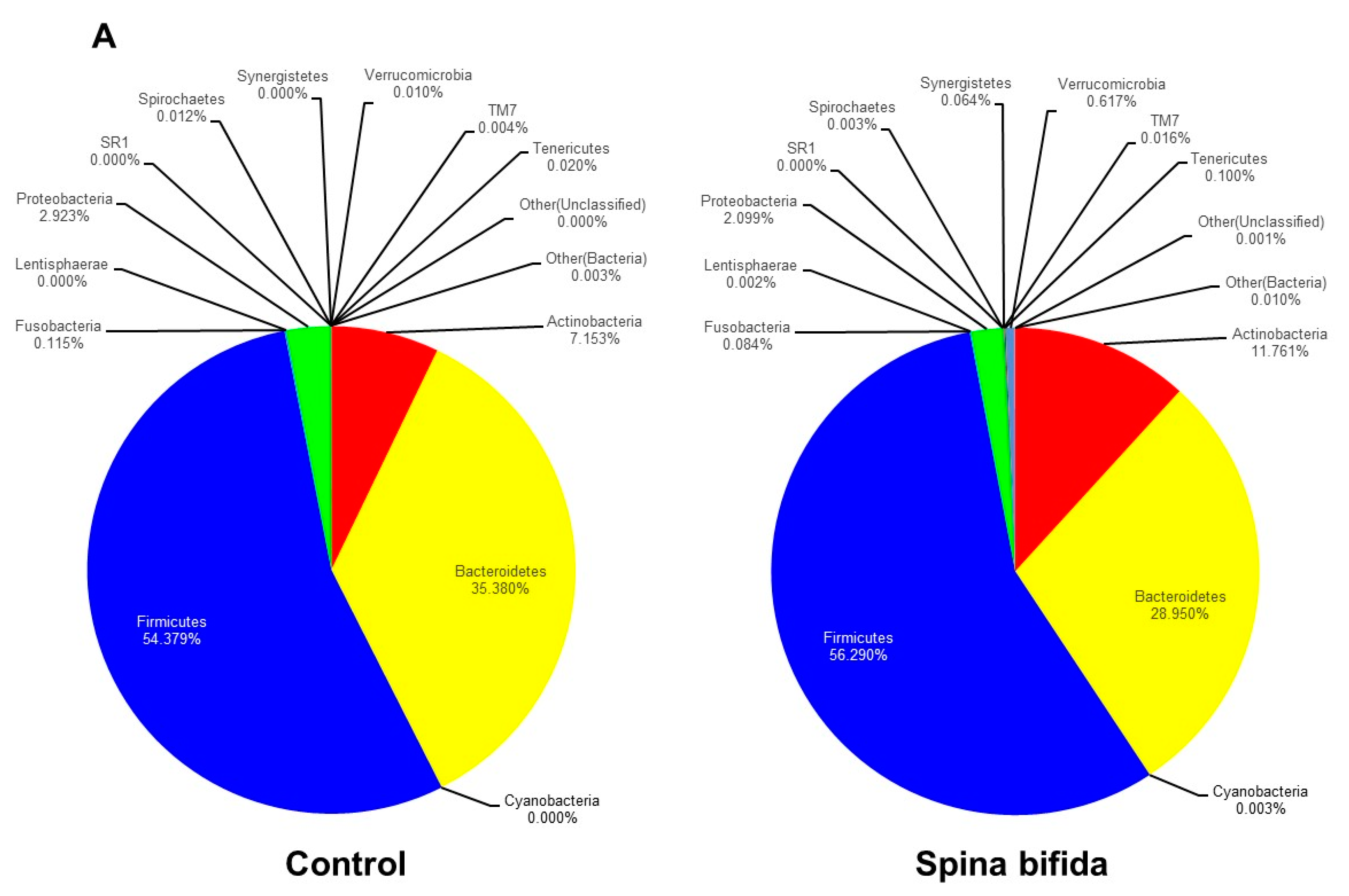
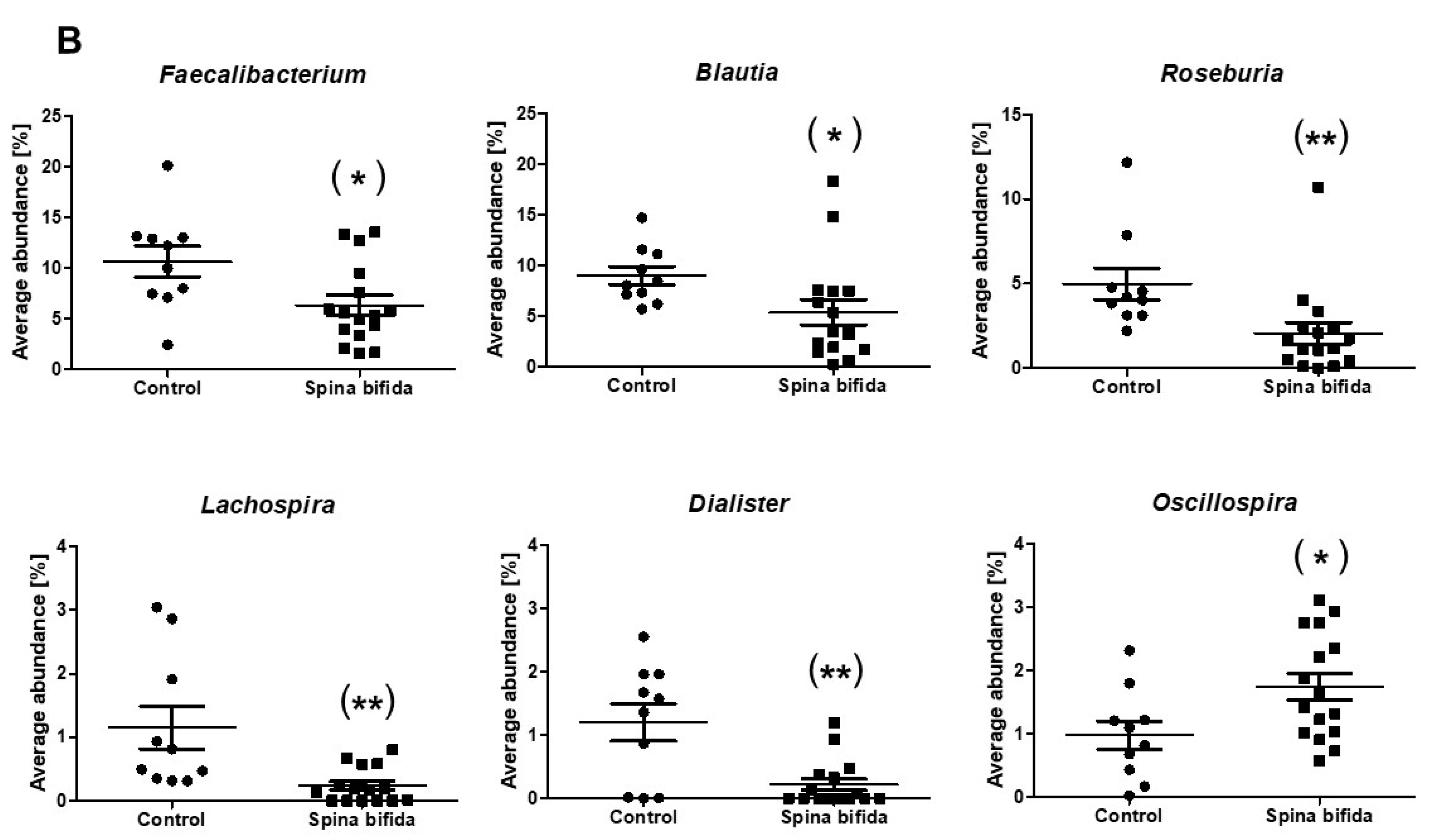
3.2. Comparison of Gut Microbiota in the SB Patients and Healthy Controls
3.3. Outcome Measures before and after TAI in the SB Patients
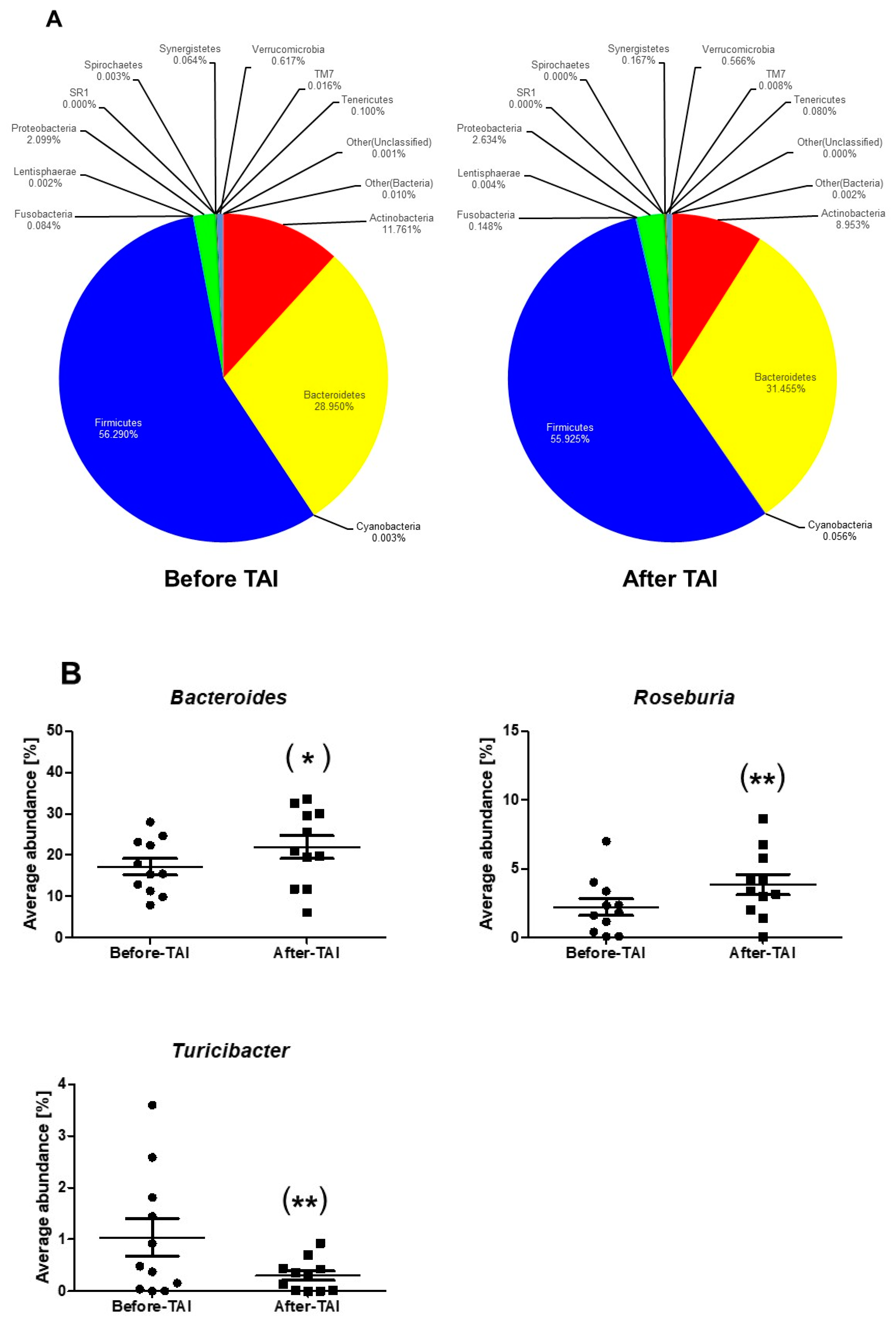
3.4. Changes in Gut Microbiota after TAI in the SB Patients
3.5. Correlation of Gut Microbiota and Bristol Scale
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| CIC | clean intermittent catheterization |
| IBS | irritable bowel syndrome |
| NBD | neurogenic bowel dysfunction |
| OUT | operational taxonomic unit |
| PCR | polymerase chain reaction |
| RDP | Ribosomal Database Project |
| SCFAs | short-chain fatty acids |
| SCI | spinal cord injury |
| TAI | transanal irrigation |
| UTI | urinary tract infection. |
References
- Christensen, P.; Bazzocchi, G.; Coggrave, M.; Abel, R.; Hultling, C.; Krogh, K.; Media, S.; Laurberg, S. A randomized, controlled trial of transanal irrigation versus conservative bowel management in spinal cord-injured patients. Gastroenterology 2006, 131, 738–747. [Google Scholar] [CrossRef] [PubMed]
- Ausili, E.; Focarelli, B.; Tabacco, F.; Murolo, D.; Sigismondi, M.; Gasbarrini, A.; Rendeli, C. Transanal irrigation in myelomeningocele children: An alternative, safe and valid approach for neurogenic constipation. Spinal Cord. 2010, 48, 560–565. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 2007, 449, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; deRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Merritt, J.L. Residual urine volume: Correlate of urinary tract infection in patients with spinal cord injury. Arch. Phys. Med. Rehabil. 1981, 62, 558–561. [Google Scholar]
- Weld, K.J.; Dmochowski, R.R. Effect of bladder management on urological complications in spinal cord injured patients. J. Urol. 2000, 163, 768–772. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef]
- Nishijima, S.; Suda, W.; Oshima, K.; Kim, S.W.; Hirose, Y.; Morita, H.; Hattori, M. The gut microbiome of healthy Japanese and its microbial and functional uniqueness. DNA Res. 2016, 23, 125–133. [Google Scholar] [CrossRef]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef]
- Ohkusa, T.; Koido, S.; Nishikawa, Y.; Sato, N. Gut Microbiota and Chronic Constipation: A Review and Update. Front. Med. 2019, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.; Blaser, M.J. The human microbiome: At the interface of health and disease. Nat. Rev. Genet. 2012, 13, 260–270. [Google Scholar] [CrossRef]
- Macfarlane, S.; Macfarlane, G.T. Regulation of short-chain fatty acid production. Proc. Nutr. Soc. 2003, 62, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Pryde, S.E.; Duncan, S.H.; Hold, G.L.; Stewart, C.S.; Flint, H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol. Lett. 2002, 217, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Meehan, C.J.; Beiko, R.G. A phylogenomic view of ecological specialization in the Lachnospiraceae, a family of digestive tract-associated bacteria. Genome Biol. Evol. 2014, 6, 703–713. [Google Scholar] [CrossRef]
- Zhuang, M.; Shang, W.; Ma, Q.; Strappe, P.; Zhou, Z. Abundance of Probiotics and Butyrate-Production Microbiome Manages Constipation via Short-Chain Fatty Acids Production and Hormones Secretion. Mol. Nutr. Food Res. 2019, 63, e1801187. [Google Scholar] [CrossRef]
- Mancabelli, L.; Milani, C.; Lugli, G.A.; Turroni, F.; Mangifesta, M.; Viappiani, A.; Ticinesi, A.; Nouvenne, A.; Meschi, T.; van Sinderen, D.; et al. Unveiling the gut microbiota composition and functionality associated with constipation through metagenomic analyses. Sci. Rep. 2017, 7, 9879. [Google Scholar] [CrossRef]
- Chassard, C.; Dapoigny, M.; Scott, K.P.; Crouzet, L.; Del’homme, C.; Marquet, P.; Martin, J.C.; Pickering, G.; Ardid, D.; Eschalier, A.; et al. Functional dysbiosis within the gut microbiota of patients with constipated-irritable bowel syndrome. Aliment. Pharmacol. Ther. 2012, 35, 828–838. [Google Scholar] [CrossRef]
- Gobert, A.P.; Sagrestani, G.; Delmas, E.; Wilson, K.T.; Verriere, T.G.; Dapoigny, M.; Del’homme, C.; Bernalier-Donadille, A. The human intestinal microbiota of constipated-predominant irritable bowel syndrome patients exhibits anti-inflammatory properties. Sci. Rep. 2016, 6, 39399. [Google Scholar] [CrossRef]
- Gungor, B.; Adiguzel, E.; Gursel, I.; Yilmaz, B.; Gursel, M. Intestinal Microbiota in Patients with Spinal Cord Injury. PLoS ONE 2016, 11, e0145878. [Google Scholar] [CrossRef]
- Liu, C.; Li, J.; Zhang, Y.; Philip, A.; Shi, E.; Chi, X.; Meng, J. Influence of glucose fermentation on CO2 assimilation to acetate in homoacetogen Blautia coccoides GA-1. J. Ind. Microbiol. Biotechnol. 2015, 42, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Morotomi, M.; Nagai, F.; Sakon, H.; Tanaka, R. Dialister succinatiphilus sp. nov. and Barnesiella intestinihominis sp. nov., isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2008, 58, 2716–2720. [Google Scholar] [CrossRef] [PubMed]
- Canani, R.B.; Costanzo, M.D.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Huda-Faujan, N.; Abdulamir, A.S.; Fatimah, A.B.; Anas, O.M.; Shuhaimi, M.; Yazid, A.M.; Loong, Y.Y. The impact of the level of the intestinal short chain Fatty acids in inflammatory bowel disease patients versus healthy subjects. Open Biochem. J. 2010, 4, 53–58. [Google Scholar] [CrossRef]
- Rajilić-Stojanović, M.; Biagi, E.; Heilig, H.G.; Kajander, K.; Kekkonen, R.A.; Tims, S.; de Vos, W.M. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1792–1801. [Google Scholar] [CrossRef]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Chen, Y.R.; Zheng, H.M.; Zhang, G.X.; Chen, F.L.; Chen, L.D.; Yang, Z.C. High Oscillospira abundance indicates constipation and low BMI in the Guangdong Gut Microbiome Project. Sci. Rep. 2020, 10, 9364. [Google Scholar] [CrossRef]
- Allen, J.M.; Berg Miller, M.E.; Pence, B.D.; Whitlock, K.; Nehra, V.; Gaskins, H.R.; White, B.A.; Fryer, J.D.; Woods, J.A. Voluntary and forced exercise differentially alters the gut microbiome in C57BL/6J mice. J. Appl. Physiol. 2015, 118, 1059–1066. [Google Scholar] [CrossRef]
- Erdogan, A.; Rao, S.S.; Thiruvaiyaru, D.; Lee, Y.Y.; Coss Adame, E.; Valestin, J.; O’Banion, M. Randomised clinical trial: Mixed soluble/insoluble fibre vs. psyllium for chronic constipation. Aliment. Pharmacol. Ther. 2016, 44, 35–44. [Google Scholar] [CrossRef]
- Jalanka, J.; Major, G.; Murray, K.; Singh, G.; Nowak, A.; Kurtz, C.; Silos-Santiago, I.; Johnston, J.M.; de Vos, W.M.; Spiller, R. The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls. Int. J. Mol. Sci. 2019, 20, 433. [Google Scholar] [CrossRef]
| Healthy Control | Spina Bifida | p Value | |
|---|---|---|---|
| Participants (Number) | 10 | 16 | |
| Male/Female (Number) | 5/5 | 5/11 | 0.339 |
| Age (Mean ± SD ) | 12.6 ± 2.5 | 10.8 ± 3.3 | 0.223 |
| BMI (Mean ± SD ) | 17.5 ± 1.6 | 18.9 ± 3.8 | 0.429 |
| CIC (Number) | 0 | 16 | |
| Bristol scale (Mean ± SD ) | 3.8 ± 0.4 | 1.8 ± 1.0 | 0.001 |
| NBD score (Mean ± SD ) | 0.5 ± 1.0 | 14.1 ± 4.7 | 0.001 |
| Very mild (Number) | 10 | 0 | |
| Mild (Number) | 0 | 3 | |
| Moderate (Number) | 0 | 4 | |
| Severe (Number) | 0 | 9 |
| Phylum Level | |||
|---|---|---|---|
| Family Level | Control | Spina Bifida | p Value |
| Genus Level | (Mean) | (Mean) | |
| Firmicutes/Bacteroidetes ratio | 1.58 | 2.38 | 0.246 |
| Firmicutes | 54.38 | 56.29 | 0.654 |
| Ruminococcaceae | |||
| Ruminococcus | 2.79 | 6.30 | 0.108 |
| Faecalibacterium | 10.64 | 6.33 | 0.033 |
| Oscillospira | 0.98 | 1.74 | 0.029 |
| Lachospiraceae | |||
| Blautia | 9.00 | 5.38 | 0.017 |
| Ruminococcus | 8.44 | 8.53 | 0.693 |
| Roseburia | 4.99 | 2.06 | 0.001 |
| Dorea | 1.71 | 1.73 | 0.937 |
| Lachnospira | 1.15 | 0.24 | 0.002 |
| Coprococcus | 1.07 | 0.55 | 0.654 |
| Lactobacillus | 0.03 | 1.18 | 0.346 |
| Streptococcaceae | |||
| Streptococcus | 1.98 | 1.82 | 0.304 |
| Clostridiaceae | |||
| SMB53 | 1.15 | 2.50 | 0.120 |
| Veillonellaceae | |||
| Veillonella | 1.22 | 0.97 | 0.051 |
| Dialister | 1.20 | 0.22 | 0.003 |
| Bacillaceae | |||
| Bacillus | 0.04 | 1.89 | 0.593 |
| Erysipelotrichaceae | |||
| Eubacterium | 0.20 | 0.61 | 0.055 |
| Turicibacteraceae | |||
| Turicibacter | 0.24 | 0.80 | 0.144 |
| Bacteroidetes | 35.38 | 28.95 | 0.147 |
| Bacteroidaceae | |||
| Bacteroides | 27.66 | 20.11 | 0.087 |
| Porphyromonadaceae | |||
| Parabacteroides | 2.34 | 3.69 | 0.257 |
| Prevotellaceae | |||
| Prevotella | 3.05 | 0.06 | 0.477 |
| Actinobacteria | 7.15 | 11.76 | 0.133 |
| Bifidobacteriaceae | |||
| Bifidobacterium | 5.89 | 8.67 | 0.280 |
| Coriobacteriaceae | |||
| Collinsella | 0.96 | 2.01 | 0.684 |
| Proteobacteria | 2.92 | 2.09 | 0.414 |
| Alcaligenaceae | |||
| Sutterella | 1.81 | 1.07 | 0.087 |
| Enterobacteriaceae | |||
| Trabulsiella | 0.36 | 0.41 | 0.385 |
| Before TAI | After TAI | p Value | |
|---|---|---|---|
| Bristol scale ( Mean ± SD ) | 1.9 ± 1.2 | 3.6 ± 1.2 | 0.001 |
| Total NBD score ( Mean ± SD ) | 15.6 ± 4.1 | 11.1 ± 4.6 | 0.009 |
| Frequency of bowel movements ( Mean ± SD ) | 1.7 ± 2.4 | 1.0 ± 0.0 | 0.279 |
| Time used for defecation ( Mean ± SD ) | 0.9 ± 2.1 | 1.1 ± 1.5 | 0.828 |
| Headache or perspiration during defecation ( Mean ± SD ) | 0.6 ± 0.9 | 0.1 ± 0.5 | 0.082 |
| Use of tablets against constipation ( Mean ± SD ) | 1.1 ± 1.0 | 0.4 ± 0.9 | 0.019 |
| Use of drops against constipation ( Mean ± SD ) | 0.4 ± 0.9 | 0.0 ± 0.0 | 0.082 |
| Digital stimulation or evacuation ( Mean ± SD ) | 4.3 ± 2.8 | 3.4 ± 3.1 | 0.336 |
| Frequency of fecal incontinence ( Mean ± SD ) | 5.0 ± 3.7 | 3.7 ± 3.4 | 0.108 |
| Use of tablets against fecal incontinence ( Mean ± SD ) | 0.3 ± 1.1 | 0.0 ± 0.0 | 0.336 |
| Flatus incontinence ( Mean ± SD ) | 2.0 ± 0.0 | 2.0 ± 0.0 | 1.000 |
| Perianal skin problems ( Mean ± SD ) | 0.9 ± 1.4 | 0.2 ± 0.8 | 0.083 |
| Urinary tract infection (Number, %) | 9 (82%) | 6 (55%) | 0.082 |
| Causative bacteria (Number) Escherichia coli | 9 | 6 |
| Phylum Level | |||
|---|---|---|---|
| Family Level | Before TAI | After TAI | p Value |
| Genus Level | (Mean) | (Mean) | |
| Firmicutes/Bacteroidetes ratio | 2.53 | 2.36 | 0.638 |
| Firmicutes | 58.57 | 57.28 | 0.638 |
| Ruminococcaceae | |||
| Ruminococcus | 7.48 | 7.48 | 1.000 |
| Faecalibacterium | 7.30 | 9.27 | 0.320 |
| Oscillospira | 1.72 | 1.83 | 0.700 |
| Lachospiraceae | |||
| Blautia | 6.11 | 7.45 | 0.240 |
| Ruminococcus | 8.70 | 8.05 | 0.966 |
| Roseburia | 2.22 | 3.86 | 0.007 |
| Dorea | 2.28 | 2.12 | 0.638 |
| Coprococcus | 0.75 | 0.54 | 1.000 |
| Streptococcaceae | |||
| Streptococcus | 1.49 | 2.00 | 0.700 |
| Clostridiaceae | |||
| SMB53 | 2.98 | 1.00 | 0.067 |
| 02d06 | 0.90 | 0.53 | 0.695 |
| Veillonellaceae | |||
| Phascolarctobacterium | 0.50 | 0.47 | 0.938 |
| Erysipelotrichaceae | |||
| Eubacterium | 0.74 | 0.83 | 0.898 |
| Turicibacteraceae | |||
| Turicibacter | 1.04 | 0.30 | 0.003 |
| Bacteroidetes | 28.92 | 31.65 | 0.638 |
| Bacteroidaceae | |||
| Bacteroides | 17.35 | 21.92 | 0.048 |
| Porphyromonadaceae | |||
| Parabacteroides | 3.03 | 3.34 | 0.831 |
| Prevotellaceae | |||
| Prevotella | 1.96 | 2.33 | 0.557 |
| Odoribacteraceae | |||
| Odoribacter | 0.51 | 0.49 | 0.922 |
| Actinobacteria | 9.74 | 7.67 | 0.320 |
| Bifidobacteriaceae | |||
| Bifidobacterium | 6.79 | 5.93 | 0.413 |
| Coriobacteriaceae | |||
| Collinsella | 1.65 | 1.43 | 0.250 |
| Proteobacteria | 1.84 | 2.52 | 0.175 |
| Alcaligenaceae | |||
| Sutterella | 0.71 | 1.35 | 0.059 |
| Bacteria | Bristol Scale | p Value |
|---|---|---|
| (Genus level) | (Correlation coefficient) | |
| Bacteroides | 0.262 | 0.147 |
| Faecalibacterium | 0.239 | 0.187 |
| Blautia | 0.264 | 0.144 |
| Roseburia | 0.486 | 0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furuta, A.; Suzuki, Y.; Takahashi, R.; Jakobsen, B.P.; Kimura, T.; Egawa, S.; Yoshimura, N. Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida. J. Clin. Med. 2021, 10, 224. https://doi.org/10.3390/jcm10020224
Furuta A, Suzuki Y, Takahashi R, Jakobsen BP, Kimura T, Egawa S, Yoshimura N. Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida. Journal of Clinical Medicine. 2021; 10(2):224. https://doi.org/10.3390/jcm10020224
Chicago/Turabian StyleFuruta, Akira, Yasuyuki Suzuki, Ryosuke Takahashi, Birte Petersen Jakobsen, Takahiro Kimura, Shin Egawa, and Naoki Yoshimura. 2021. "Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida" Journal of Clinical Medicine 10, no. 2: 224. https://doi.org/10.3390/jcm10020224
APA StyleFuruta, A., Suzuki, Y., Takahashi, R., Jakobsen, B. P., Kimura, T., Egawa, S., & Yoshimura, N. (2021). Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida. Journal of Clinical Medicine, 10(2), 224. https://doi.org/10.3390/jcm10020224





