Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Subjects
2.3. Procedure
2.4. Physiological Measures of Autonomic Function
2.5. Statistics
3. Results
3.1. Subjects
3.2. Exercise-Related Outcomes
3.3. Autonomic Function
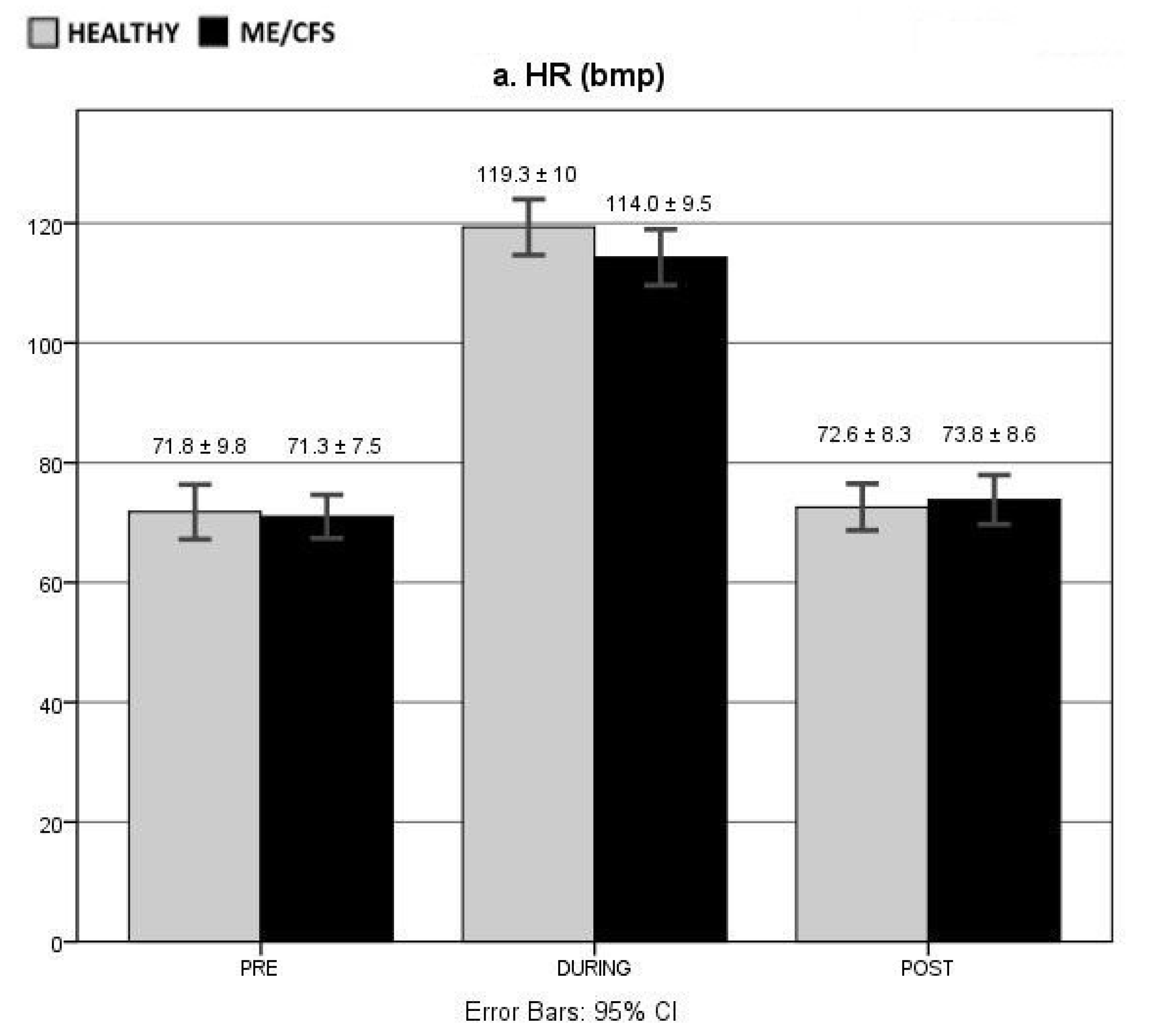
3.4. HR
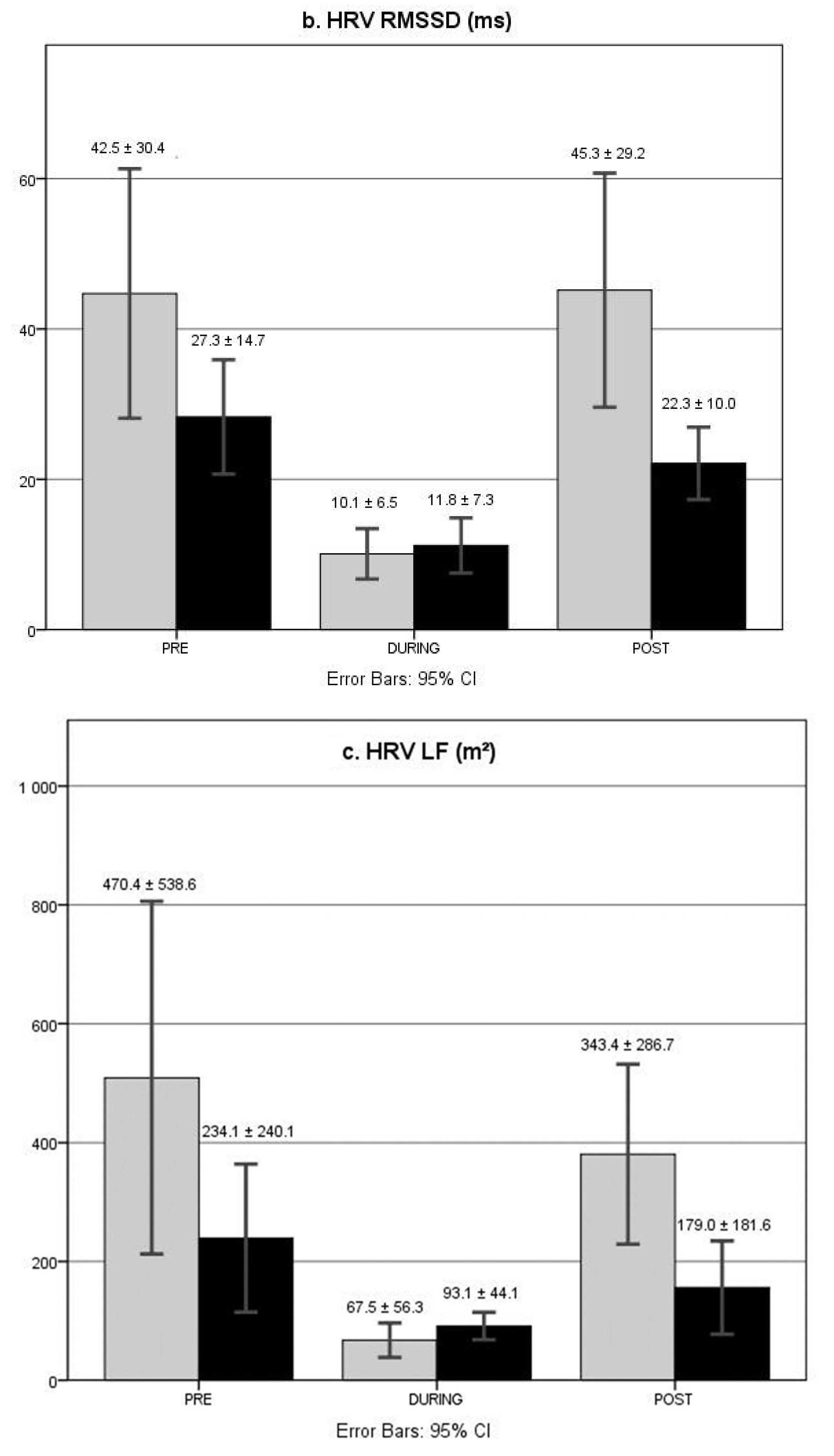
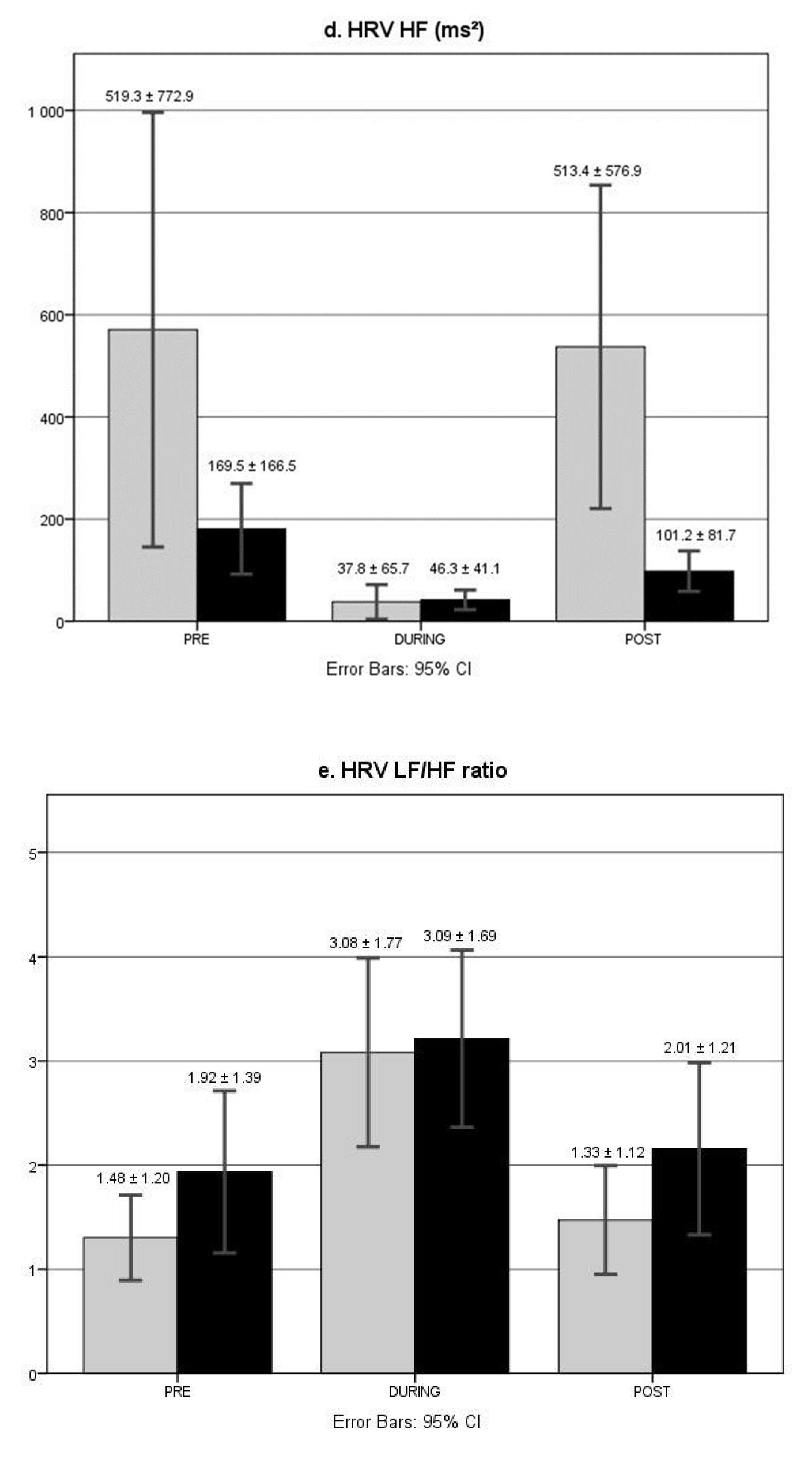
3.5. HRV
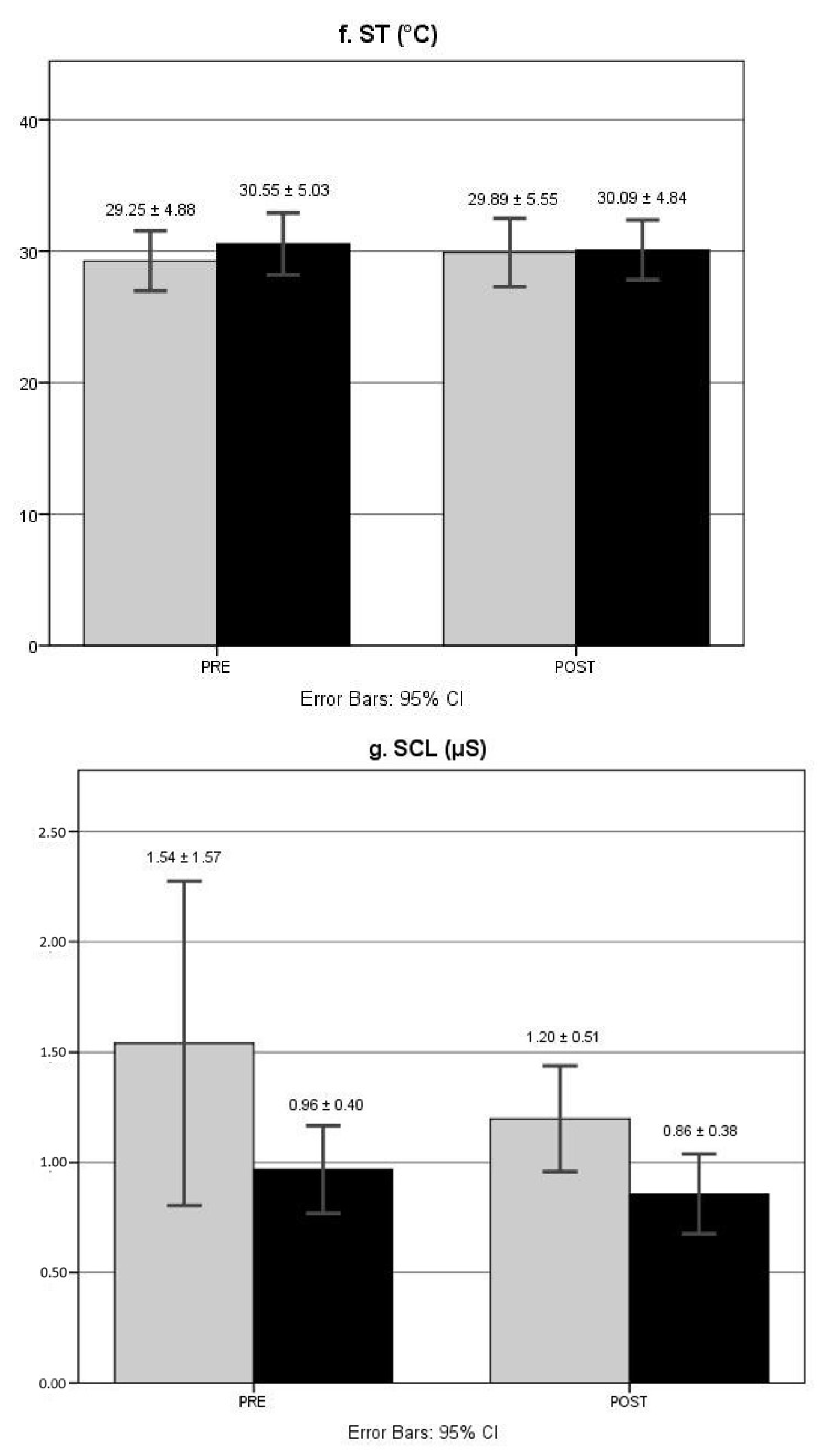
3.6. Electrodermal Responses
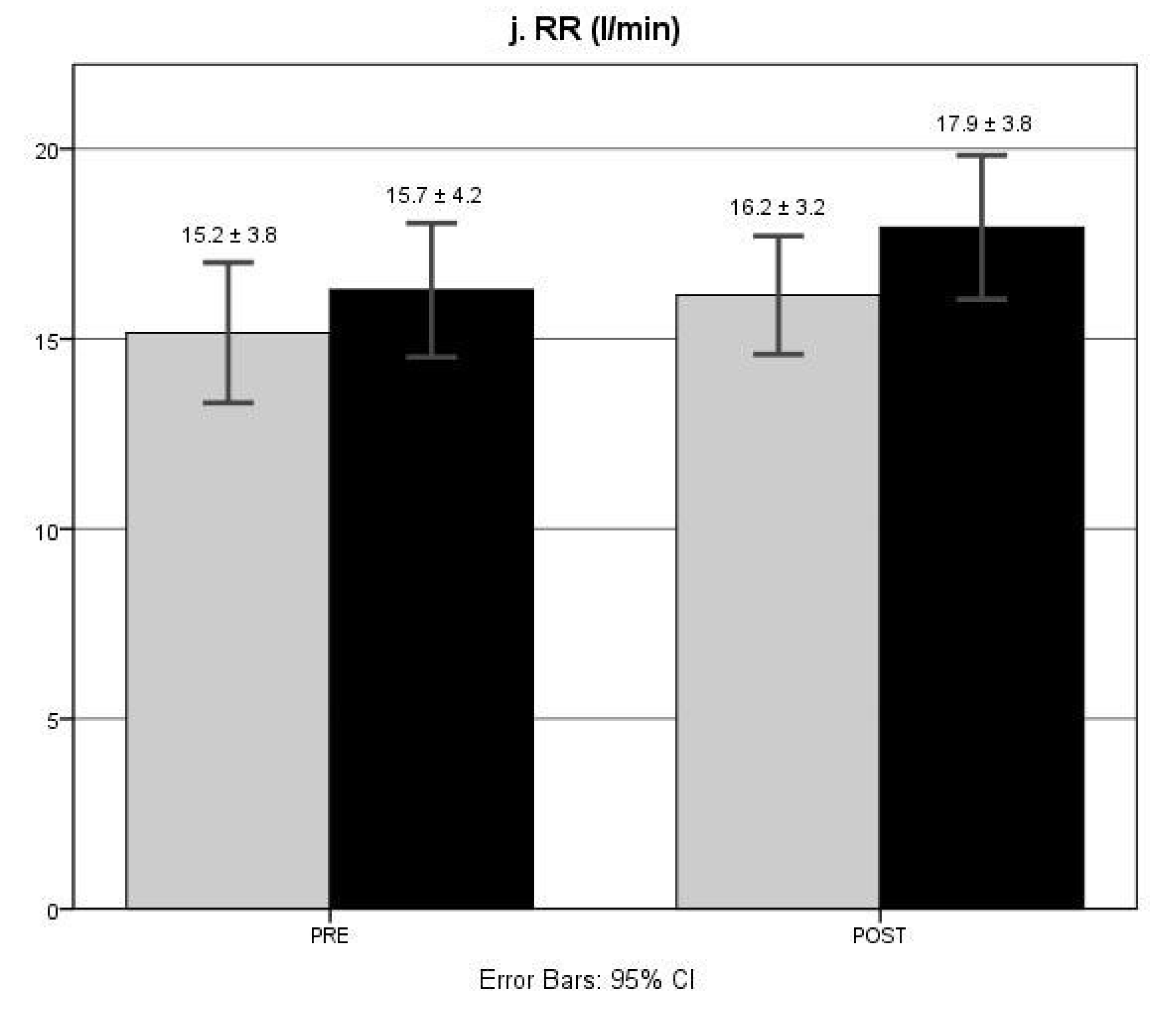
3.7. RR
3.8. BP
4. Discussion
4.1. Autonomic Function at Rest in ME/CFS
4.2. Autonomic Function during an Acute Aerobic Exercise Bout in ME/CFS
4.3. Autonomic Function during Recovery from Exercise in ME/CFS
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANS | autonomic nervous system |
| ECG | electrocardiogram |
| CFS | chronic fatigue syndrome |
| HR | heart rate |
| HRV | heart rate variability |
| HF | high frequency |
| LF | low frequency |
| ME | myalgic encephalomyelitis |
| RMSSD | root mean square of successive differences between NN intervals |
| RPE | ratings of perceived exertion |
| RR | respiration rate |
| SC | skin conductance |
| SCL | skin conductance level |
| SCR | skin conductance responses |
| ST | skin temperature |
| W | workload |
References
- Carruthers, B.M.; van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Int. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef]
- Freeman, R.; Komaroff, A.L. Does the Chronic Fatigue Syndrome Involve the Autonomic Nervous System? Am. J. Med. 1997, 102, 357–364. [Google Scholar] [CrossRef]
- Newton, J.; Okonkwo, O.; Sutcliffe, K.; Seth, A.; Shin, J.; Jones, D. Symptoms of autonomic dysfunction in chronic fatigue syndrome. QJM Int. J. Med. 2007, 100, 519–526. [Google Scholar] [CrossRef]
- Meeus, M.; Goubert, D.; De Backer, F.; Struyf, F.; Hermans, L.; Coppieters, I.; De Wandele, I.; Da Silva, H.; Calders, P. Heart rate variability in patients with fibromyalgia and patients with chronic fatigue syndrome: A systematic review. Semin. Arthrit. Rheum. 2013, 43, 279–287. [Google Scholar] [CrossRef]
- Van Cauwenbergh, D.; Nijs, J.; Kos, D.; Van Weijnen, L.; Struyf, F.; Meeus, M. Malfunctioning of the autonomic nervous system in patients with chronic fatigue syndrome: A systematic literature review. Eur. J. Clin. Investig. 2014, 44, 516–526. [Google Scholar] [CrossRef]
- Nijs, J.; Nees, A.; Paul, L.; De Kooning, M.; Ickmans, K.; Meeus, M.; Van Oosterwijck, J. Altered immune response to exercise in patients with chronic fatigue syndrome/myalgic encephalomyelitis: A systematic literature review. Exerc. Immunol. Rev. 2014, 20, 94–116. [Google Scholar] [PubMed]
- De Becker, P.; Roeykens, J.; Reynders, M.; McGregor, N.; De Meirleir, K. Exercise Capacity in Chronic Fatigue Syndrome. Arch. Int. Med. 2000, 160, 3270–3277. [Google Scholar] [CrossRef] [PubMed]
- Gibson, H.; Carroll Clague, J.E.; Edwards, R.H. Exercise performance and fatiguability in patients with chronic fatigue syndrome. J. Neurol. Neurosurg. Psychiatry 1993, 56, 993–998. [Google Scholar] [CrossRef]
- Montague, T.J.; Marrie, T.J.; Klassen, G.A.; Bewick, D.J.; Horacek, B.M. Cardiac Function at Rest and with Exercise in the Chronic Fatigue Syndrome. Chest 1989, 95, 779–784. [Google Scholar] [CrossRef] [PubMed]
- Sisto, S.A.; LaManca, J.; Cordero, D.L.; Bergen, M.T.; Ellis, S.P.; Drastal, S.; Bodaad, W.L.; Tappac, W.N.; Natelsonac, B.H. Metabolic and cardiovascular effects of a progressive exercise test in patients with chronic fatigue syndrome. Am. J. Med. 1996, 100, 634–640. [Google Scholar] [CrossRef]
- Soetekouw, P.M.; Lenders, J.W.; Bleijenberg, G.; Thien, T.; van der Meer, J.W. Autonomic function in patients with chronic fa-tigue syndrome. Clin. Auton. Res. 1999, 9, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Vanness, J.M.; Snell, C.R.; Strayer, D.R.; Dempsey, L., IV; Stevens, S.R. Subclassifying chronic fatigue syndrome through exer-cise testing. Med. Sci. Sports Exerc. 2003, 35, 908–913. [Google Scholar] [CrossRef] [PubMed]
- Fink, G. Stress Science: Neuroendocrinology; Academic Press, Elsevier Ltd.: San Diego, CA, USA, 2009; p. 829. [Google Scholar]
- Cole, C.R.; Blackstone, E.H.; Pashkow, F.J.; Snader, C.E.; Lauer, M.S. Heartrate recovery immediately after exercise as a preditor of mortality. N. Engl. J. Med. 1999, 341, 1351–1357. [Google Scholar] [CrossRef] [PubMed]
- Jouven, X.; Empana, J.P.; Schwartz, P.J.; Desnos, M.; Courbon, D.; Ducimetière, P. Heart-Rate Profile during Exercise as a Predictor of Sudden Death. N. Engl. J. Med. 2005, 352, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Lauer, M.S. Autonomic function and prognosis. Clevel. Clin. J. Med. 2009, 76, S18–S22. [Google Scholar] [CrossRef]
- Nishime, E.O.; Cole, C.R.; Blackstone, E.H.; Pashkow, F.J.; Lauer, M. Heart Rate Recovery and Treadmill Exercise Score as Predictors of Mortality in Patients Referred for Exercise ECG. JAMA 2000, 284, 1392–1398. [Google Scholar] [CrossRef]
- Van Oosterwijck, J.; Uros, M.; De Wandele, I.; Meeus, M.; Paul, L.; Lambrecht, L.; Moorkens, G. Nijs Reduced Parasympathetic Reactivation During Recovery from Exercise in Myalgic Encephalomyelitis (ME)/chronic Fatigue Syndrome (CFS). Physiothera-py 2015, 101, e1091–e1092. [Google Scholar] [CrossRef][Green Version]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.C.; Dobbins, J.G.; Komaroff, A. The Chronic Fatigue Syndrome, a comprehensive approach to its definition and study. Ann. Int. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, M.S.; Morabia, A.; Sloutskis, D. Definition and prevalence of sedentarism in an urban population. Am. J. Public Health 1999, 89, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Bleijenberg, G. Chronic fatigue and chronic fatigue syndrome in the general population. Health Qual. Life Outcomes 2003, 1, 52. [Google Scholar] [CrossRef] [PubMed]
- Sheel, A.W.; Richards, J.C.; Foster, G.; Guenette, J.A. Sex Differences in Respiratory Exercise Physiology. Sports Med. 2004, 34, 567–579. [Google Scholar] [CrossRef]
- Wallman, K.; Goodman, C.; Morton, A.; Grove, R.; Dawson, B. Test-Retest Reliability of the Aerobic Power Index Test in Patients with Chronic Fatigue Syndrome. J. Chronic Fatigue Syndr. 2003, 11, 19–32. [Google Scholar] [CrossRef]
- Wallman, K.; Goodman, C.; Morton, A.; Grove, R.; Dawson, B. Test-retest reliability of the aerobic power index component of the tri-level fitness profile in a sedentary population. J. Sci. Med. Sport 2003, 6, 443–454. [Google Scholar] [CrossRef]
- Wallman, K.E.; Morton, A.R.; Goodman, C.; Grove, R. Physiological Responses during a Submaximal Cycle Test in Chronic Fatigue Syndrome. Med. Sci. Sports Exerc. 2004, 36, 1682–1688. [Google Scholar] [CrossRef]
- Van Oosterwijck, J.; Nijs, J.; Meeus, M.; Lefever, I.; Huybrechts, L.; Lambrecht, L.; Paul, L. Pain inhibition and postexertional malaise in myalgic encephalomyelitis/chronic fatigue syndrome: An experimental study. J. Int. Med. 2010, 268, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Koenig, A.; Omlin, X.; Zimmerli, L.; Sapa, M.; Krewer, C.; Bolliger, M.; Müller, F.; Riener, R. Psychological state estimation from physiological recordings during robot-assisted gait rehabilitation. J. Rehabilit. Res. Dev. 2011, 48, 367–385. [Google Scholar] [CrossRef] [PubMed]
- Guideline: Standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [CrossRef]
- DeGiorgio, C.M.; Miller, P.; Meymandi, S.; Chin, A.; Epps, J.; Gordon, S.; Gornbein, J.; Harper, R.M. RMSSD, a measure of vagusmediated heart rate variability, is associated with risk factors for SUDEP: The SUDEP-7 Inventory. Epilepsy Behav. 2010, 19, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Novak, D.; Mihelj, M.; Munih, M. Psychophysiological responses to different levels of cognitive and physical workload in haptic interaction. Robotica 2010, 29, 367–374. [Google Scholar] [CrossRef]
- Hoaglin, D.C.; Iglewicz, B. Fine tuning some resistant rules for outlier labelling. J. Am. Statist. Assoc. 1987, 82, 1147–1149. [Google Scholar] [CrossRef]
- Francica, J.V.; Bigongiari, A.; Mochizuki, L.; Scapini, K.B.; Moraes, O.A.; Mostarda, C.; Caperuto, É.C.; Irigoyen, M.C.; De Angelis, K.; Rodrigues, B. Cardiac autonomic dysfunction in chronic stroke women is attenuated after submaximal exercise test, as evaluated by linear and nonlinear analysis. BMC Cardiovasc. Disord. 2015, 15, 105. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.S.; Bentho, O.; Park, M.Y.; Sharabi, Y. Low-frequency power of heart rate variability is not a measure of cardiac sympathetic tone but may be a measure of modulation of cardiac autonomic outflows by baroreflexes. Exp. Physiol. 2011, 96, 1255–1261. [Google Scholar] [CrossRef] [PubMed]
- Pazderka-Robinson, H.; Morrison, J.W.; Flor-Henry, P. Electrodermal dissociation of chronic fatigue and depression: Evidence for distinct physiological mechanisms. Int. J. Psychophysiol. 2004, 53, 171–182. [Google Scholar] [CrossRef]
- Nijs, J.; Van Oosterwijck, J.; Meeus, M.; Lambrecht, L.; Metzger, K.; Fremont, M.; Paul, L. Unravelling the nature of postexertional malaise in myalgic encephalomyelitis/chronic fatigue syndrome: The role of elastase, complement C4a and interleukin-1β. J. Int. Med. 2010, 267, 418–435. [Google Scholar] [CrossRef] [PubMed]
- Rowell, L.B. Human Circulation: Regulation During Physical Stress; Oxford University Press: New York, NY, USA, 1986. [Google Scholar]
- Billman, G.E.; Dujardin, J.P. Dynamic changes in cardiac vagal tone as measured by time-series analysis. Am. J. Physiol. Content 1990, 258, 896–902. [Google Scholar] [CrossRef] [PubMed]
- Breuer, H.W.; Skyschally, A.; Schulz, R.; Martin, C.; Wehr, M.; Heusch, G. Heart rate variability and circulating catecholamine concentrations during steady state exercise in healthy volunteers. Br. Heart J. 1993, 70, 144–149. [Google Scholar] [CrossRef]
- Tekin, G.; Tekin, A. Heart rate recovery and methodological issues. Anadolu Kardiyol. Dergisi/The Anatol. J. Cardiol. 2015, 15, 87–88. [Google Scholar] [CrossRef]
- Sugawara, J.; Murakami, H.; Maeda, S.; Kuno, S.; Matsuda, M. Change in post-exercise vagal reactivation with exercise train-ing and detraining in young men. Eur. J. Appl. Physiol. 2001, 85, 259–263. [Google Scholar] [CrossRef]
- Hautala, A.J.; Kiviniemi, A.; Tulppo, M. Individual responses to aerobic exercise: The role of the autonomic nervous system. Neurosci. Biobehav. Rev. 2009, 33, 107–115. [Google Scholar] [CrossRef]
- Tulppo, M.P.; Hautala, A.J.; Makikallio, T.H.; Laukkanen, R.T.; Nissila, S.; Hughson, R.L.; Huikuri, H.V. Effects of aerobic training on heart rate dynamics in sedentary subjects. J. Appl. Physiol. 2003, 95, 364–372. [Google Scholar] [CrossRef]

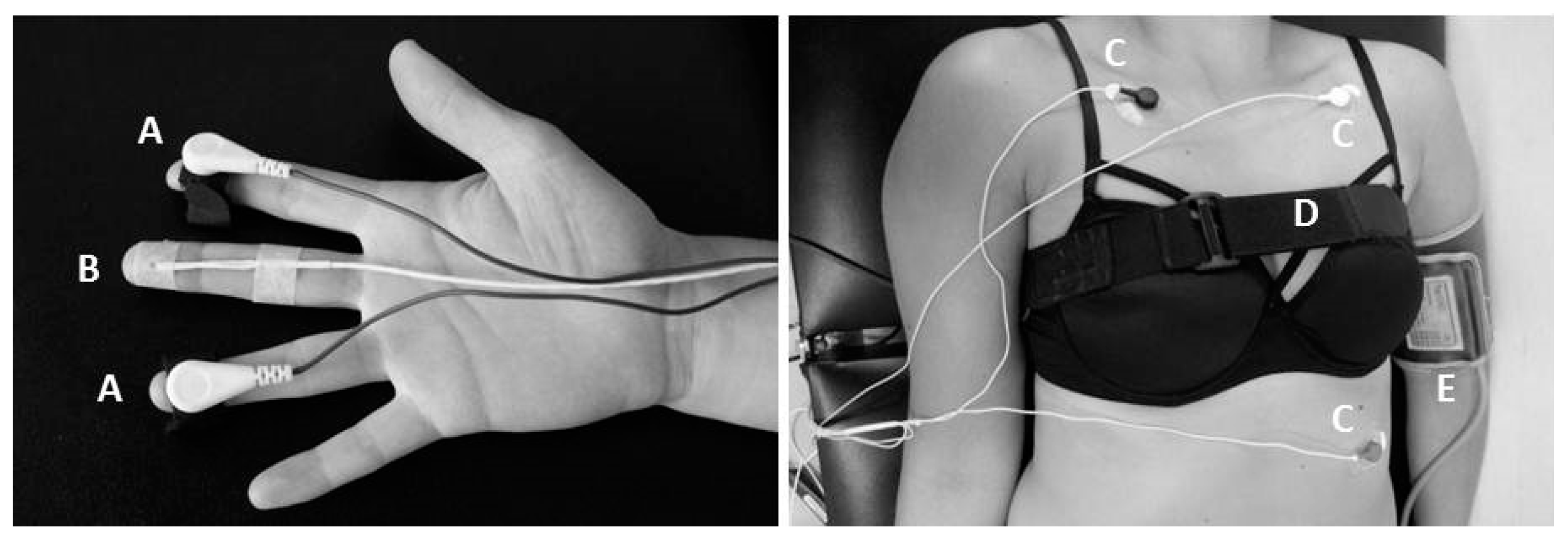

| ME/CFS Group (n = 20) | Healthy Group (n = 20) | Between-Group Comparison (p-Value) | |
|---|---|---|---|
| Age, years | 0.155 | ||
| Mean (SD) | 41.6 (9.8) | 34.6 (15.2) | |
| Length, cm | 0.935 | ||
| Mean (SD) | 168 (5) | 168 (8) | |
| Weight, kg | 0.168 | ||
| Mean (SD) | 68.1 (14.9) | 73.9 (15.6) | |
| Handedness | 1.000 | ||
| Right (n) | 17 | 16 | |
| Left (n) | 3 | 4 | |
| Employment status | 0.208 | ||
| Student (n) | 1 | 6 | |
| Retired (n) | 0 | 1 | |
| Full-time (n) | 4 | 2 | |
| Part-time (n) | 6 | 4 | |
| Non-employed (n) | 9 | 7 | |
| Years of education | 0.177 | ||
| Mean (SD) | 14.4 (2.8) | 15.6 (2.7) | |
| Highest degree of education | 0.054 | ||
| Primary school (n) | |||
| Secondary education (n) | 9 | 12 | |
| Higher education—university or college (n) | 2 | 1 | |
| Higher education—adult education social | 8 | 7 | |
| advancement course (n) | 1 | 0 | |
| Marital status | 0.609 | ||
| Single (n) | 9 | 12 | |
| Living together (n) | 2 | 1 | |
| Married (n) | 8 | 7 | |
| Widow (n) | 1 | 0 | |
| Children | |||
| Yes | 7 | 12 | 0.205 |
| No | 13 | 8 | |
| Mean number (SD) | 1 (1.0) | 1 (1.3) | 0.640 |
| Time from diagnosis, months | |||
| Mean (SD) | 70.3 (56.8) | NA | NA |
| ME/CFS Group (n = 20) | Healthy Group (n = 20) | Between-Group Comparison (p-Value) | |
|---|---|---|---|
| HR, bpm Theoretical target HR peak * Actual achieved HR peak Mean HR | 134 (7) 140 (9) 114 (10) | 140 (12) 142 (10) 119 (10) | 0.149 0.453 0.092 |
| Cycling time, min | 3.86 (1.00) | 4.15 (1.15) | 0.401 |
| Peak Workload | 109 (25) | 118 (25) | 0.327 |
| VO2 peak, mL/min/kg | 16.98 (4.25) | 19.96 (6.80) | 0.112 |
| VE peak, L/min | 31.81 (9.67) | 31.61 (11.30) | 0.758 |
| RER peak | 0.76 (0.89) | 72 (0.08) | 0.101 |
| RPE | 16 (0.3) | 12 (2) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Oosterwijck, J.; Marusic, U.; De Wandele, I.; Meeus, M.; Paul, L.; Lambrecht, L.; Moorkens, G.; Danneels, L.; Nijs, J. Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J. Clin. Med. 2021, 10, 4527. https://doi.org/10.3390/jcm10194527
Van Oosterwijck J, Marusic U, De Wandele I, Meeus M, Paul L, Lambrecht L, Moorkens G, Danneels L, Nijs J. Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Journal of Clinical Medicine. 2021; 10(19):4527. https://doi.org/10.3390/jcm10194527
Chicago/Turabian StyleVan Oosterwijck, Jessica, Uros Marusic, Inge De Wandele, Mira Meeus, Lorna Paul, Luc Lambrecht, Greta Moorkens, Lieven Danneels, and Jo Nijs. 2021. "Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome" Journal of Clinical Medicine 10, no. 19: 4527. https://doi.org/10.3390/jcm10194527
APA StyleVan Oosterwijck, J., Marusic, U., De Wandele, I., Meeus, M., Paul, L., Lambrecht, L., Moorkens, G., Danneels, L., & Nijs, J. (2021). Reduced Parasympathetic Reactivation during Recovery from Exercise in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Journal of Clinical Medicine, 10(19), 4527. https://doi.org/10.3390/jcm10194527








