Evidence of a Muscle–Brain Axis by Quantification of the Neurotrophic Myokine METRNL (Meteorin-Like Protein) in Human Cerebrospinal Fluid and Serum
Abstract
:1. Introduction
- -
- to quantify the protein concentrations of the neurotrophic adipo-myokine METRNL by ELISA in paired serum and CSF samples from a well- characterized cohort of patients (n = 260) with various diseases who underwent neurological evaluation, including lumbar puncture in a single and tertiary care centre;
- -
- to correlate serum/CSF METRNL levels with anthropometric parameters, routine laboratory parameters and neurological and internal medicine diseases;
- -
- to obtain insight into the basal and inducible regulation of BBB function with respect to the migration of METRNL by providing novel and specific CSF/serum ratios and classical Reibergrams;
- -
- to provide evidence whether or not METRNL fulfils the biochemical and physiological conditions to act as a mediator of the muscle–brain axis.
2. Materials and Methods
2.1. Study Population
2.2. Measurement of CSF and Serum Parameters
2.3. High-Sensitive Quantification of METRNL in Serum and CSF
2.4. Statistics
3. Results
3.1. Characteristics of the Study Population and Concentrations of METRNL in Serum and CSF
3.2. Correlation of Serum and CSF METRNL Concentrations with Numerical Standard Variables
3.3. CSF Concentrations and CSF/Serum Ratios of METRNL Significantly Increase with Biochemical Parameters Blood–Brain Barrier (BBB) Dysfunction
3.4. Detailed Evaluation of Classical Reibergrams for the Quantitative Description of BBB Dysfunction
3.5. Correlation of Serum and CSF METRNL Levels with Respect to the Metabolic Syndrome Complex
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hopfinger, A.; Berghoff, M.; Karrasch, T.; Schmid, A.; Schaffler, A. Systematic quantification of neurotrophic adipokines rbp4, pedf, and clusterin in human cerebrospinal fluid and serum. J. Clin. Endocrinol. Metab. 2021, 106, 2239–2250. [Google Scholar] [CrossRef]
- Ahima, R.S.; Saper, C.B.; Flier, J.S.; Elmquist, J.K. Leptin regulation of neuroendocrine systems. Front. Neuroendocrinol. 2000, 21, 263–307. [Google Scholar] [CrossRef]
- McMillen, I.C.; Adam, C.L.; Muhlhausler, B.S. Early origins of obesity: Programming the appetite regulatory system. J. Physiol. 2005, 565, 9–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schnabl, K.; Li, Y.; Klingenspor, M. The gut hormone secretin triggers a gut-brown fat-brain axis in the control of food intake. Exp. Physiol. 2020, 105, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- De Kloet, A.D.; Herman, J.P. Fat-brain connections: Adipocyte glucocorticoid control of stress and metabolism. Front. Neuroendocrinol. 2018, 48, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Berghoff, M.; Hochberg, A.; Schmid, A.; Schlegel, J.; Karrasch, T.; Kaps, M.; Schaffler, A. Quantification and regulation of the adipokines resistin and progranulin in human cerebrospinal fluid. Eur. J. Clin. Invest. 2016, 46, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Neumeier, M.; Weigert, J.; Buettner, R.; Wanninger, J.; Schaffler, A.; Muller, A.M.; Killian, S.; Sauerbruch, S.; Schlachetzki, F.; Steinbrecher, A.; et al. Detection of adiponectin in cerebrospinal fluid in humans. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E965–E969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmid, A.; Berghoff, M.; Hochberg, A.; Schaffler, A.; Karrasch, T. Ctrp-3 is permeable to the blood-brain barrier and is not regulated by glucose or lipids in vivo. Eur. J. Clin. Invest. 2017, 47, 203–212. [Google Scholar] [CrossRef]
- Giudice, J.; Taylor, J.M. Muscle as a paracrine and endocrine organ. Curr. Opin. Pharmacol. 2017, 34, 49–55. [Google Scholar] [CrossRef]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, bone, and fat crosstalk: The biological role of myokines, osteokines, and adipokines. Curr. Osteoporos. Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef]
- Pedersen, B.K. Muscle as a secretory organ. Compr. Physiol. 2013, 3, 1337–1362. [Google Scholar] [PubMed]
- Yoon, J.H.; Kim, J.; Song, P.; Lee, T.G.; Suh, P.G.; Ryu, S.H. Secretomics for skeletal muscle cells: A discovery of novel regulators? Adv. Biol. Regul. 2012, 52, 340–350. [Google Scholar] [CrossRef]
- Weigert, C.; Hartwig, S.; Lehr, S. Methods for proteomics-based analysis of the human muscle secretome using an in vitro exercise model. Methods Mol. Biol. 2021, 2261, 433–442. [Google Scholar] [PubMed]
- Tok, O.; Kisioglu, S.V.; Ersoz, H.O.; Kahveci, B.; Goktas, Z. Effects of increased physical activity and/or weight loss diet on serum myokine and adipokine levels in overweight adults with impaired glucose metabolism. J. Diabetes Complicat. 2021, 35, 107892. [Google Scholar] [CrossRef] [PubMed]
- Townsend, L.K.; Knuth, C.M.; Wright, D.C. Cycling our way to fit fat. Physiol. Rep. 2017, 5, e13247. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.R.; Long, J.Z.; White, J.P.; Svensson, K.J.; Lou, J.; Lokurkar, I.; Jedrychowski, M.P.; Ruas, J.L.; Wrann, C.D.; Lo, J.C.; et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 2014, 157, 1279–1291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.O.; Byun, W.S.; Kang, M.J.; Han, J.A.; Moon, J.; Shin, M.J.; Lee, H.J.; Chung, J.H.; Lee, J.S.; Son, C.G.; et al. The myokine meteorin-like (metrnl) improves glucose tolerance in both skeletal muscle cells and mice by targeting ampkalpha2. FEBS J. 2020, 287, 2087–2104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eaton, M.; Granata, C.; Barry, J.; Safdar, A.; Bishop, D.; Little, J.P. Impact of a single bout of high-intensity interval exercise and short-term interval training on interleukin-6, fndc5, and metrnl mrna expression in human skeletal muscle. J. Sport Health Sci. 2018, 7, 191–196. [Google Scholar] [CrossRef]
- Ost, M.; Coleman, V.; Kasch, J.; Klaus, S. Regulation of myokine expression: Role of exercise and cellular stress. Free Radic. Biol. Med. 2016, 98, 78–89. [Google Scholar] [CrossRef]
- Jung, T.W.; Lee, S.H.; Kim, H.C.; Bang, J.S.; Abd El-Aty, A.M.; Hacimuftuoglu, A.; Shin, Y.K.; Jeong, J.H. Metrnl attenuates lipid-induced inflammation and insulin resistance via ampk or ppardelta-dependent pathways in skeletal muscle of mice. Exp. Mol. Med. 2018, 50, 1–11. [Google Scholar]
- Ushach, I.; Arrevillaga-Boni, G.; Heller, G.N.; Pone, E.; Hernandez-Ruiz, M.; Catalan-Dibene, J.; Hevezi, P.; Zlotnik, A. Meteorin-like/meteorin-beta is a novel immunoregulatory cytokine associated with inflammation. J. Immunol. 2018, 201, 3669–3676. [Google Scholar] [CrossRef] [Green Version]
- Wen, D.; Xiao, Y.; Vecchi, M.M.; Gong, B.J.; Dolnikova, J.; Pepinsky, R.B. Determination of the disulfide structure of murine meteorin, a neurotrophic factor, by lc-ms and electron transfer dissociation-high-energy collisional dissociation analysis of proteolytic fragments. Anal. Chem. 2017, 89, 4021–4030. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.Y. Aerobic exercise increases meteorin-like protein in muscle and adipose tissue of chronic high-fat diet-induced obese mice. Biomed. Res. Int. 2018, 2018, 6283932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bae, J.Y.; Woo, J.; Kang, S.; Shin, K.O. Effects of detraining and retraining on muscle energy-sensing network and meteorin-like levels in obese mice. Lipids Health Dis. 2018, 17, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huh, J.Y. The role of exercise-induced myokines in regulating metabolism. Arch. Pharm. Res. 2018, 41, 14–29. [Google Scholar] [CrossRef] [PubMed]
| Study Population (n = 260) | |
|---|---|
| Age (years) [range] | 50.5 ± 17.5 [18–90] |
| Males n (%) | 107 (41.2) |
| Females n (%) | 153 (58.8) |
| Meteorin-like | |
| METRNL in serum (pg/mL) [range] | 801.2 ± 378.3 [203.1–2685.1] |
| METRNL in cerebrospinal fluid (pg/mL) [range] | 1007.2 ± 624.2 [230.0–4275.3] |
| METRNL CSF/serum ratio [range] | 1.4 ± 0.8 [0.2–6.7] |
| Anthropometric parameters | |
| Mean BMI (kg/m²) ° | 26.5 ± 5.0 [17.4–47.7] |
| BMI < 25.0 kg/m² n (%) | 126 (48.5) |
| BMI ≥ 25.0 kg/m² n (%) | 128 (49.2) |
| Neurological diseases/Clinical subgroups | |
| Infectious CNS disease n (%) | 13 (5.0) |
| Multiple sclerosis n (%) | 40 (15.4) |
| Vascular disease n (%) | 22 (8.5) |
| Epilepsie n (%) | 26 (10.0) |
| Headache/facial pain n (%) | 23 (8.8) |
| Neuropathy/cranial nerve palsy n (%) | 46 (17.7) |
| Others * n (%) | 90 (34.6) |
| * consisting of dementia (8), psychiatric disorders (12), normal pressure hydrocephalus (8), and patients undergoing spinal puncture for exclusion of other diseases (53) | |
| Serum METRNL | |
| Males | 745.3 ± 350.9 * |
| Females | 840.2 ± 392.8 * (p = 0.031) |
| BMI < 25 | 774.7 ± 343.8 |
| BMI ≥ 25 | 827.3 ± 411.3 |
| Diabetes mellitus | 945.0 ± 565.3 |
| Non-Diabetes | 784.3 ± 343.6 |
| Hypertension | 835.9 ± 433.7 |
| Normotension | 778.5 ± 330.5 |
| Smoker | 783.2 ± 320.9 |
| Non-Smoker | 807.1 ± 389.9 |
| CSF METRNL | |
| Males | 1071.2 ± 603.4 |
| Females | 962.4 ± 636.5 |
| BMI < 25 | 970.9 ± 637.4 |
| BMI ≥ 25 | 1041.6 ± 606.8 |
| Diabetes mellitus | 1128.1 ± 569.1 |
| Non-Diabetes | 993.9 ± 630.7 |
| Hypertension | 1117.5 ± 673.1 * |
| Normotension | 929.6 ± 575.2 * (p = 0.023) |
| Smoker | 915.5 ± 447.0 |
| Non-Smoker | 1029.7 ± 655.5 |
| Serum METRNL [pg/mL] | CSF METRNL [pg/mL] | |
|---|---|---|
| Correlation with | ||
| Age [years] | n. s. | rho = +0.237 p < 0.001 |
| BMI [kg/m2) | n. s. | n. s. |
| Serum parameters | ||
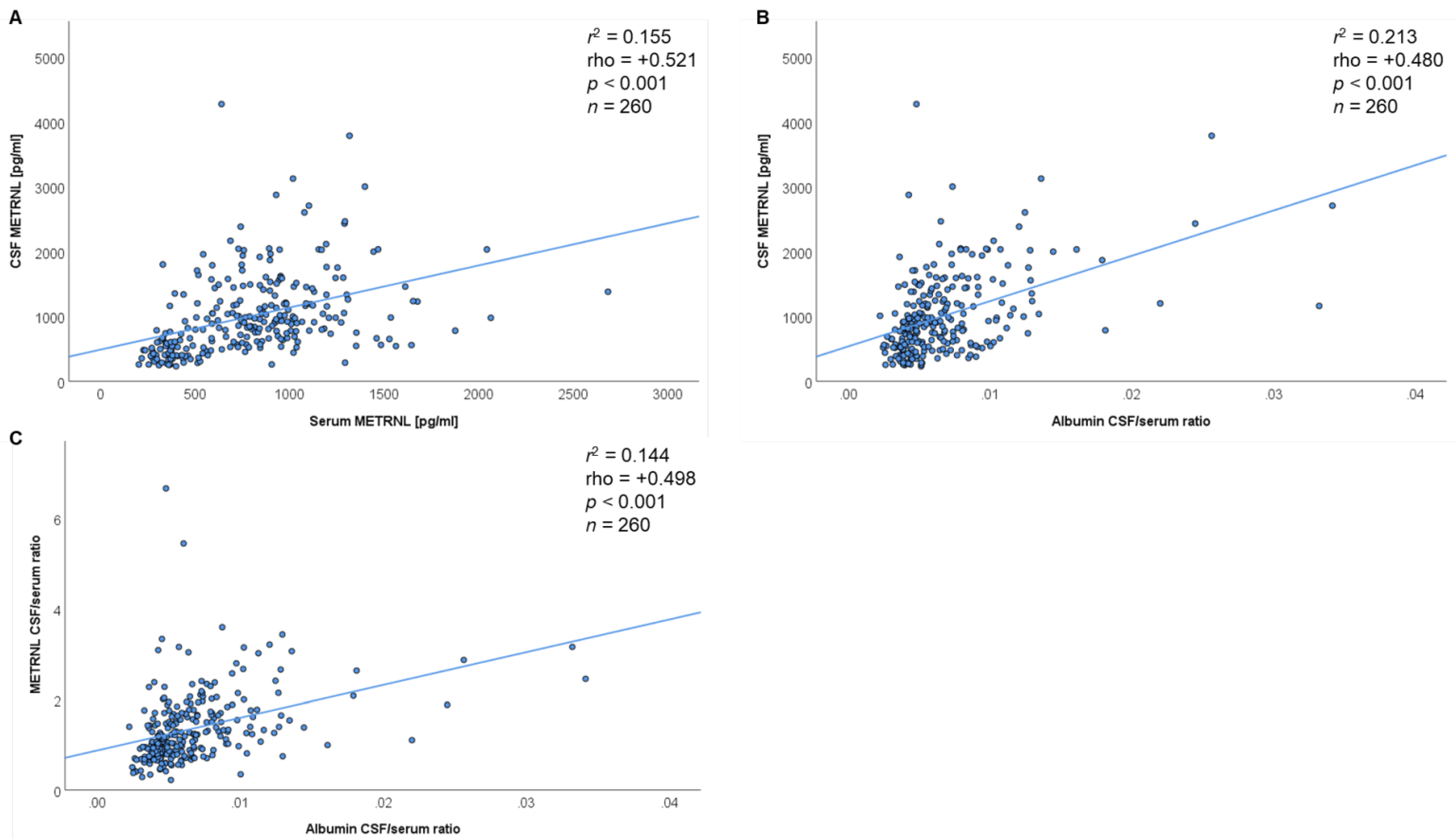
| METRNL [pg/mL] | - | rho = +0.521 p < 0.001 |
| Leukocyte [giga/L] | n. s. | n. s. |
| Hemoglobin [g/L] | rho = −0.231 p < 0.001 | n. s. |
| CRP [mg/dL] | rho = +0.159 p = 0.011 | n. s. |
| ALT [U/L] | n. s. | rho = +0.169 p = 0.009 |
| AST [U/L] | n. s. | rho = +0.255 p < 0.001 |
| Creatinine [mg/dL] | n. s. | rho = -0.151 p = 0.016 |
| Urea [mg/dL] | n. s. | rho = +0.142 p = 0.025 |
| Albumin [g/L] | rho = −0.172 p = 0.005 | n. s. |
| CSF/serum albumin ratio | n. s. | rho = +0.480 p < 0.001 |
| Lipoprotein-Metabolism (triglycerides, LDL cholesterol, HDL cholesterol) | n. s. | n. s. |
| Carbohydrate-Metabolism (Glucose or HbA1c) | n. s. | n. s. |
| CSF parameters | ||
| Total protein [g/L] | n. s. | rho = +0.420 p < 0.001 |
| Albumin [g/L] | n. s. | rho = +0.463 p < 0.001 |
| IgM [g/L] | n. s. | rho = +0.349 p = 0.003 |
| IgG [g/L] | n. s. | rho = +0.357 p < 0.001 |
| IgA [g/L] | rho = +0.348 p = 0.003 | rho = +0.430 p < 0.001 |
| Lactate [mmol/L] | n. s. | rho = +0.210 p = 0.001 |
| Serum METRNL [pg/mL] | CSF METRNL [pg/mL] | C/S METRNL | |
|---|---|---|---|
| Disease groups | |||
| Infectious disease (n = 13) | 935.2 ± 434.6 | 1674.8 ± 1072.0 | 1.8 ± 0.8 |
| Multiple Sclerosis (n = 40) | 759.1 ± 367.3 | 909.2 ± 537.1 | 1.3 ± 0.7 |
| Vascular disease (n = 22) | 640.2 ± 391.6 | 879.5 ± 599.1 | 1.4 ± 0.6 |
| Epilepsy (n = 26) | 758.5 ± 292.8 | 927.6 ± 485.2 | 1.4 ± 1.0 |
| Headache/facial pain (n = 23) | 732.5 ± 312.6 | 864.0 ± 450.8 | 1.2 ± 0.4 |
| Neuropathy/ cranial nerve palsy (n = 46) | 800.3 ± 347.2 | 1068.7 ± 560.4 | 1.4 ± 0.6 |
| Others (n = 90) | 870.2 ± 412.6 | 1013.7 ± 640.3 | 1.3 ± 0.9 |
| BBB dysfunction | |||
| Grade 0 (n = 206) | 800.4 ± 375.5 | 912.1 ± 545.9 | 1.2 ± 0.7 |
| Grade 1 (n = 21) | 765.4 ± 321.8 | 1151.5 ± 588.3 | 1.6 ± 0.7 |
| Grade 2 (n = 31) | 827.9 ± 431.8 | 1446.6 ± 749.3 | 1.9 ± 0.8 |
| Grade 3 (n = 2) | 842.3 ± 672.1 | 2473.9 ± 1857.2 | 3.0 ± 0.2 |
| p < 0.001 | p < 0.001 | ||
| CSF cell count/µL | |||
| 0–5 cells (n = 226) | 784.4 ± 376.8 | 959.5 ± 561.7 | 1.4 ± 0.8 |
| >5 cells (n = 34) | 912.9 ± 374.7 | 1324.6 ± 887.4 | 1.4 ± 0.7 |
| p = 0.028 | p = 0.035 | ||
| Oligoclonal bands | |||
| negative (n = 207) | 791.8 ± 375.4 | 1010.6 ± 609.1 | 1.4 ± 0.8 |
| positive (n = 41) | 806.9 ± 385.2 | 1070.9 ± 715.4 | 1.4 ± 0.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berghoff, M.; Höpfinger, A.; Rajendran, R.; Karrasch, T.; Schmid, A.; Schäffler, A. Evidence of a Muscle–Brain Axis by Quantification of the Neurotrophic Myokine METRNL (Meteorin-Like Protein) in Human Cerebrospinal Fluid and Serum. J. Clin. Med. 2021, 10, 3271. https://doi.org/10.3390/jcm10153271
Berghoff M, Höpfinger A, Rajendran R, Karrasch T, Schmid A, Schäffler A. Evidence of a Muscle–Brain Axis by Quantification of the Neurotrophic Myokine METRNL (Meteorin-Like Protein) in Human Cerebrospinal Fluid and Serum. Journal of Clinical Medicine. 2021; 10(15):3271. https://doi.org/10.3390/jcm10153271
Chicago/Turabian StyleBerghoff, Martin, Alexandra Höpfinger, Ranjithkumar Rajendran, Thomas Karrasch, Andreas Schmid, and Andreas Schäffler. 2021. "Evidence of a Muscle–Brain Axis by Quantification of the Neurotrophic Myokine METRNL (Meteorin-Like Protein) in Human Cerebrospinal Fluid and Serum" Journal of Clinical Medicine 10, no. 15: 3271. https://doi.org/10.3390/jcm10153271
APA StyleBerghoff, M., Höpfinger, A., Rajendran, R., Karrasch, T., Schmid, A., & Schäffler, A. (2021). Evidence of a Muscle–Brain Axis by Quantification of the Neurotrophic Myokine METRNL (Meteorin-Like Protein) in Human Cerebrospinal Fluid and Serum. Journal of Clinical Medicine, 10(15), 3271. https://doi.org/10.3390/jcm10153271






