Benefits of Steroid Therapy in COVID-19 Patients with Different PaO2/FiO2 Ratio at Admission
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
2.3. Variables
2.4. Bias
2.5. Statistical Methods
3. Results
3.1. Study Population
3.2. Use of Steroids
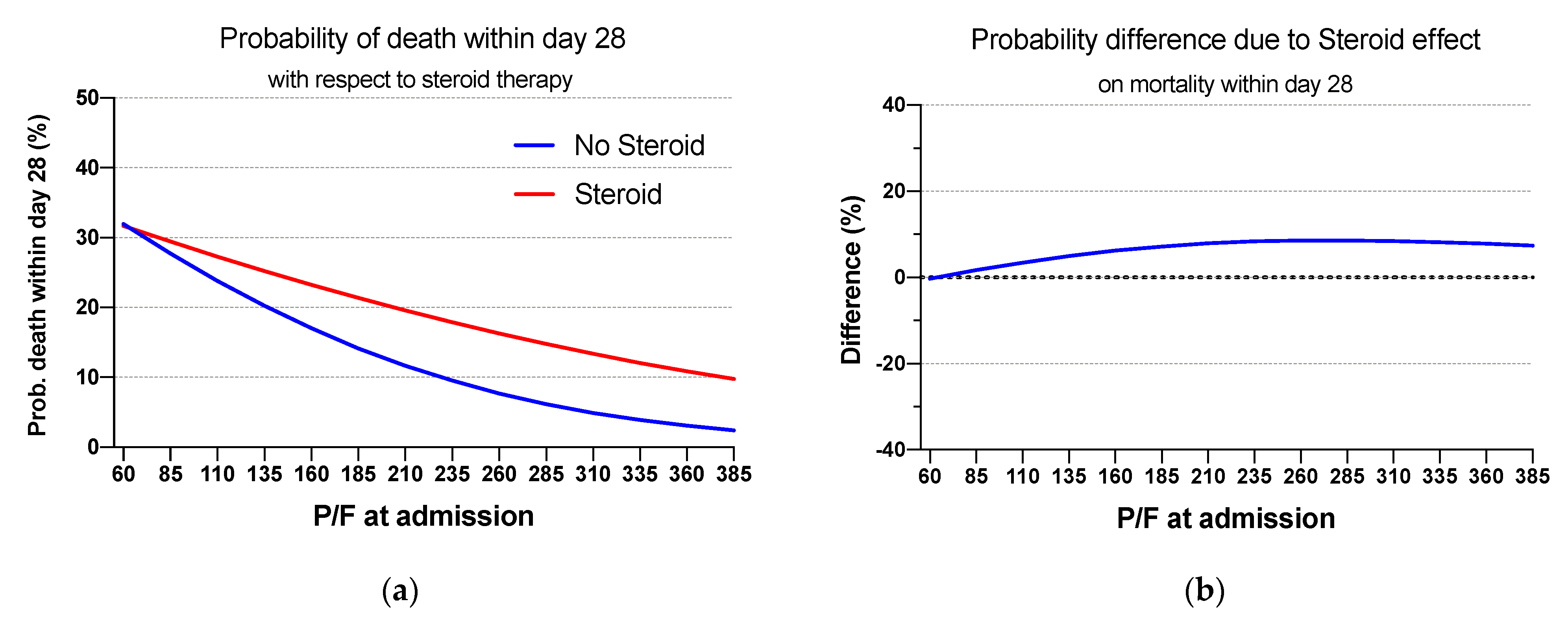
3.3. Mortality at Day 28
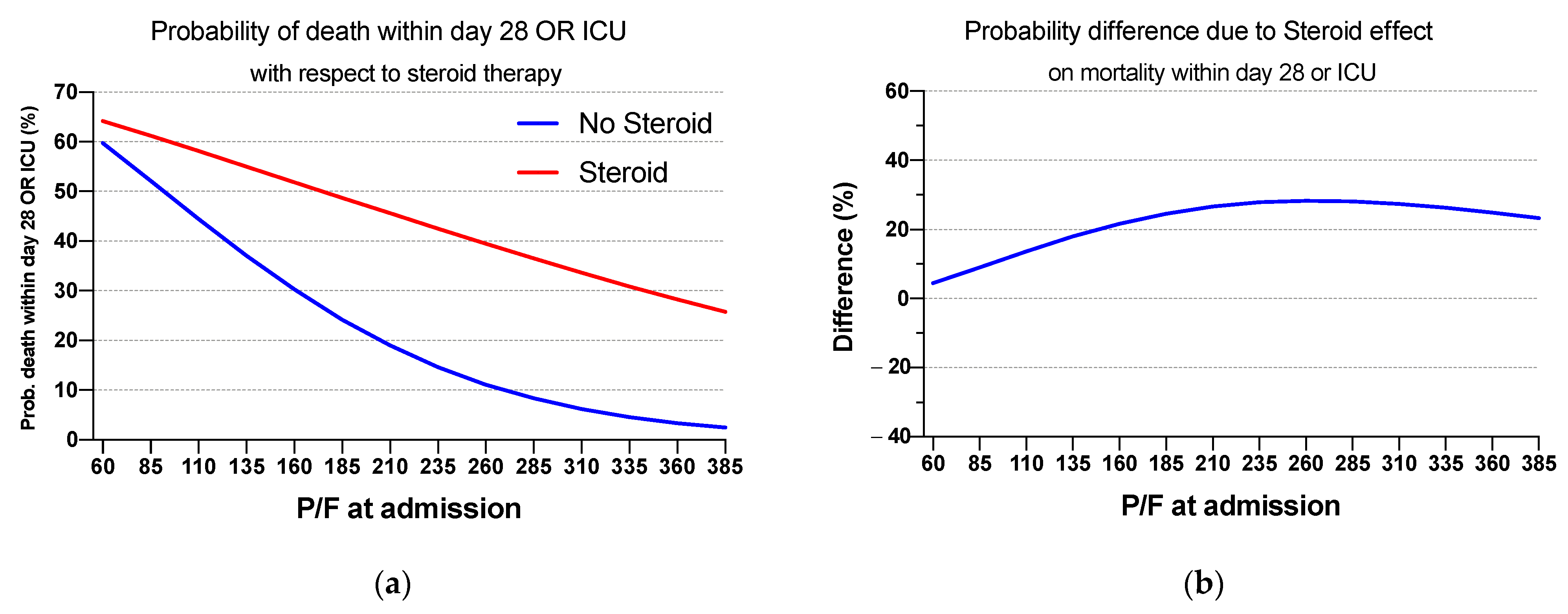
3.4. Admission to Intensive Care Unit or Mortality at day 28
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| P/F | Steroids | Prob. of Death within 28 Days | ||
|---|---|---|---|---|
| Average (%) (95% CI) | Difference (%) (95% CI) | p-Value | ||
| 60 | No | 31.9 (2.7–61.2) | −0.3 (−32.2–31.6) | 0.986 |
| Yes | 31.7 (18.2–45.2) | |||
| 85 | No | 27.7 (2.9–52.5) | 1.7 (−25.5–28.9) | 0.903 |
| Yes | 29.4 (17.7–41.2) | |||
| 110 | No | 23.8 (3.3–44.4) | 3.5 (−19.2–26.1) | 0.765 |
| Yes | 27.3 (17.2–37.3) | |||
| 135 | No | 20.2 (3.6–36.9) | 5.0 (−13.6–23.5) | 0.600 |
| Yes | 25.2 (16.7–33.8) | |||
| 160 | No | 17. (3.9–30.2) | 6.2 (−8.7–21.1) | 0.413 |
| Yes | 23.2 (16–30.4) | |||
| 185 | No | 14.2 (4–24.3) | 7.2 (−4.6–18.9) | 0.231 |
| Yes | 21.3 (15.2–27.5) | |||
| 210 | No | 11.7 (4–19.3) | 7.9 (−1.4–17.1) | 0.095 |
| Yes | 19.5 (14.3–24.8) | |||
| 235 | No | 9.5 (3.8–15.3) | 8.3 (0.9–15.7) | 0.027 |
| Yes | 17.8 (13.1–22.6) | |||
| 260 | No | 7.7 (3.3–12.) | 8.6 (2.4–14.8) | 0.007 |
| Yes | 16.3 (11.8–20.7) | |||
| 285 | No | 6.2 (2.8–9.6) | 8.6 (3.0–14.2) | 0.003 |
| Yes | 14.8 (10.3–19.2) | |||
| 310 | No | 4.9 (2.1–7.7) | 8.5 (3.1–13.8) | 0.002 |
| Yes | 13.4 (8.8–17.9) | |||
| 335 | No | 3.9 (1.4–6.3) | 8.2 (2.8–13.6) | 0.003 |
| Yes | 12.1 (7.3–16.8) | |||
| 360 | No | 3.1 (0.8–5.3) | 7.8 (2.4–13.3) | 0.005 |
| Yes | 10.9 (5.9–15.8) | |||
| 385 | No | 2.4 (0.4–4.4) | 7.4 (1.8–12.9) | 0.009 |
| Yes | 9.8 (4.6–14.9) | |||
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- McGonagle, D.; Sharif, K.; O’Regan, A.; Bridgewood, C. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun. Rev. 2020, 19, 102537. [Google Scholar] [CrossRef]
- Wong, J.J.M.; Leong, J.Y.; Lee, J.H.; Albani, S.; Yeo, J.G. Insights into the immuno-pathogenesis of acute respiratory distress syndrome. Ann. Transl. Med. 2019, 7, 504. [Google Scholar] [CrossRef]
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017, 39, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Mokra, D.; Mikolka, P.; Kosutova, P.; Mokry, J. Corticosteroids in Acute Lung Injury: The Dilemma Continues. Int. J. Mol. Sci. 2019, 20, 4765. [Google Scholar] [CrossRef] [Green Version]
- Annane, D.; Pastores, S.M.; Arlt, W.; Balk, R.A.; Beishuizen, A.; Briegel, J.; Carcillo, J.; Christ-Crain, M.; Cooper, M.S.; Marik, P.E.; et al. Critical illness-related corticosteroid insufficiency (CIRCI): A narrative review from a Multispecialty Task Force of the Society of Critical Care Medicine (SCCM) and the European Society of Intensive Care Medicine (ESICM). Intensive Care Med. 2017, 43, 1781–1792. [Google Scholar] [CrossRef] [PubMed]
- Meduri, G.U.; Annane, D.; Chrousos, G.P.; Marik, P.E.; Sinclair, S.E. Activation and regulation of systemic inflammation in ARDS: Rationale for prolonged glucocorticoid therapy. Chest 2009, 136, 1631–1643. [Google Scholar] [CrossRef] [PubMed]
- Recovery Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Diaz, J.; Villar, J.; Murthy, S.; Slutsky, A.S.; Perner, A.; Perner, A.; Jüni, P.; Angus, D.C.; Annane, D.; et al. Corticosteroid therapy for critically ill patients with COVID-19: A structured summary of a study protocol for a prospective meta-analysis of randomized trials. Trials 2020, 21, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, S.; Dong, X.; Li, Z.; Xu, Q.; Feng, H.; Cai, J.; Huang, S.; Guo, J.; Zhang, L.; et al. Corticosteroid treatment in severe COVID-19 patients with acute respiratory distress syndrome. J. Clin. Investig. 2020, 130, 6417–6428. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liu, J.; Zhou, Y.; Zhao, X.; Zhao, Q.; Liu, J. The effect of corticosteroid treatment on patients with coronavirus infection: A systematic review and meta-analysis. J. Infect. 2020, 81, e13–e20. [Google Scholar] [CrossRef] [PubMed]
- Bartoletti, M.; Marconi, L.; Scudeller, L.; Pancaldi, L.; Tedeschi, S.; Giannella, M.; PREDICO Study Group. Efficacy of corticosteroid treatment for hospitalized patients with severe COVID-19: A multicentre study. Clin. Microbiol. Infect. 2021, 27, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Ann. Intern. Med. 2007, 147, 573–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alhazzani, W.; Møller, M.H.; Arabi, Y.M.; Loeb, M.; Gong, M.N.; Fan, E.; Oczkowski, S.; Levy, M.M.; Derde, L.; Dzierba, A.; et al. Surviving Sepsis Campaign: Guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med. 2020, 46, 854–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeronimo, C.M.P.; Farias, M.E.L.; Val, F.F.A.; Sampaio, V.S.; Alexandre, M.A.A.; Melo, G.C.; Safe, I.P.; Borba, M.G.S.; Abreu-Netto, R.L.; Maciel, A.B.S.; et al. Methylprednisolone as adjunctive therapy for patients hospitalized with COVID-19 (Metcovid): A randomised, double-blind, phase IIb, placebo-controlled trial. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Lee, N.; Allen Chan, K.C.; Hui, D.S.; Ng, E.K.O.; Wu, A.; Chiu, R.W.K. Effects of early corticosteroid treatment on plasma SARS-associated coronavirus RNA concentrations in adult patients. J. Clin. Virol. 2004, 31, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Mandourah, Y.; Al-Hameed, F.; Sindi, A.A.; Almekhlafi, G.A.; Hussein, M.A. Corticosteroid therapy for critically ill patients with middle east respiratory syndrome. Am. J. Respir. Crit. Care Med. 2018, 197, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.; Xu, S.; Lin, Y.; Tian, D.; Zhu, Z.; Dai, F.; Wu, F.; Song, Z.; Huang, W.; Chen, J.; et al. The persistence and clearance of viral RNA in 2019 novel coronavirus disease survivors. Chin. Med. J. 2020. [Google Scholar] [CrossRef] [PubMed]
| Patients Feature | Total N (%) | CS Yes N (%) | CS No N (%) | OR (95% CI) | p-Value | Decd N (%) | Surviv. N (%) | OR (95% CI) | p-Value | |
|---|---|---|---|---|---|---|---|---|---|---|
| Age | <50 Years | 110 (21.5) | 29 (26.4) | 81 (73.6) | <0.001 | 3 (2.7) | 107 (97.3) | <0.001 | ||
| 50–70 Years | 198 (38.7) | 73 (36.9) | 125 (63.1) | 1.63 (0.98–2.72) | 9 (4.6) | 189 (95.5) | 1.70 (0.45–6.41) | |||
| >70 Years | 203 (39.7) | 98 (48.3) | 105 (51.7) | 2.61 (1.57–4.32) | 39 (19.2) | 164 (80.8) | 8.48 (2.56–28.14) | |||
| Sex | Female | 181 (35.4) | 65 (35.9) | 116 (64.1) | ref | 0.267 | 19 (10.5) | 162 (89.5) | ref | 0.774 |
| Male | 330 (64.6) | 135 (40.9) | 195 (59.1) | 1.24 (0.85–1.8) | 32 (9.7) | 298 (90.3) | 0.92 (0.5–1.67) | |||
| use of Antivirals | No | 185 (36.2) | 40 (21.6) | 145 (78.4) | ref | <0.001 | 15 (8.1) | 170 (91.9) | ref | 0.281 |
| Yes | 326 (63.8) | 160 (49.1) | 166 (50.9) | 3.49 (2.31–5.27) | 36 (11.0) | 290 (89.0) | 1.41 (0.75–2.65) | |||
| use of Aminoquinoline | No | 218 (42.7) | 80 (36.7) | 138 (63.3) | ref | 0.329 | 22 (10.1) | 196 (89.9) | ref | 0.942 |
| Yes | 293 (57.3) | 120 (41.0) | 173 (59.0) | 1.20 (0.83–1.72) | 29 (9.9) | 264 (90.1) | 0.98 (0.55–1.76) | |||
| use of Azithromycin | No | 327 (64.0) | 115 (35.2) | 212 (64.8) | ref | 0.015 | 29 (8.9) | 298 (91.1) | ref | 0.269 |
| Yes | 184 (36.0) | 85 (46.2) | 99 (53.8) | 1.58 (1.1–2.29) | 22 (12.0) | 162 (88.0) | 1.40 (0.78–2.51) | |||
| use of Monoclonal antib. | No | 408 (79.8) | 126 (30.9) | 282 (69.1) | ref | <0.001 | 40 (9.8) | 368 (90.2) | ref | 0.793 |
| Yes | 103 (20.2) | 74 (71.8) | 29(28.2) | 5.71 (3.54–9.21) | 11 (10.7) | 92 (89.3) | 1.10 (0.54–2.23) | |||
| Diabetes | No | 430 (84.1) | 162 (37.7) | 268 (62.3) | ref | 0.121 | 34 (7.9) | 396 (92.1) | ref | 0.001 |
| Yes | 81 (15.9) | 38 (46.9) | 43 (53.1) | 1.46 (0.91–2.36) | 17 (21.0) | 64 (79.0) | 3.09 (1.63–5.86) | |||
| Hypertension | No | 295 (57.7) | 96 (32.5) | 199 (67.5) | ref | <0.001 | 16 (5.4) | 279 (94.6) | ref | <0.001 |
| Yes | 216 (42.3) | 104 (48.2) | 112 (51.9) | 1.93 (1.34–2.76) | 35 (16.2) | 181 (83.8) | 3.37 (1.81–6.27) | |||
| Renal failure | No | 482 (94.3) | 185 (38.4) | 297 (61.6) | ref | 0.158 | 45 (9.3) | 437 (90.7) | ref | 0.076 |
| Yes | 29 (5.7) | 15 (51.7) | 14 (48.3) | 1.72 (0.81–3.65) | 6 (20.7) | 23 (79.3) | 2.53 (0.98–6.55) | |||
| Cancer | No | 458 (89.6) | 178 (38.9) | 280 (61.1) | ref | 0.710 | 41 (9.0) | 417 (91.0) | ref | 0.037 |
| Yes | 53 (10.4) | 22 (41.5) | 31 (58.5) | 1.12 (0.63–1.99) | 10 (18.9) | 43 (81.1) | 2.37 (1.11–5.05) | |||
| CVD | No | 358 (70.1) | 123 (34.4) | 76 (53.1) | ref | 0.027 | 8 (2.2) | 350 (97.8) | ref | <0.001 |
| Yes | 143 (28.0) | 67 (46.9) | 282 (69.1) | 1.56 (1.05–2.3) | 33 (23.1) | 110 (76.9) | 5.83 (3.16–10.77) | |||
| COPD | No | 460 (90.0) | 172 (37.4) | 288 (62.6) | ref | 0.016 | 40 (8.7) | 420 (91.3) | ref | 0.009 |
| Yes | 51 (10.0) | 28 (54.9) | 23 (45.1) | 2.04 (1.14–3.65) | 11 (21.6) | 40 (78.4) | 2.89 (1.38–6.06) | |||
| Obesity | No | 447 (87.5) | 168 (37.6) | 279 (62.4) | ref | 0.060 | 37 (8.3) | 410 (91.7) | ref | 0.002 |
| Yes | 64 (12.5) | 32 (50.0) | 32 (50.0) | 1.66 (0.98–2.81) | 14 (21.9) | 50 (78.1) | 3.10 (1.57–6.13) | |||
| Overall | - | 511 (100.0) | 200 (39.1) | 311 (60.9) | - | - | 51 (10.0) | 460 (90.0) | - | - |
| P/F at admission (Median–IQR) * | 343 (271–406) | 283 (199–344) | 376 (319–438) | 0.95 (0.94–0.96) | <0.001 | 221 (144–329) | 352 (289–410) | 0.95 (0.94–0.98) | <0.001 | |
| P/F Ratio at Admission (mmHg) | Total N (%) | CS No N (%) | CS Yes N (%) |
|---|---|---|---|
| <100 | 9 (1.8) | 1 (11.1) | 8 (88.9) |
| 100–150 | 24 (4.7) | 2 (8.3) | 22 (91.7) |
| 150–200 | 28 (5.5) | 8 (28.6) | 20 (71.4) |
| 200–300 | 103 (20.2) | 40 (38.8) | 63 (61.2) |
| >300 | 347 (67.9) | 260 (74.9) | 87 (25.1) |
| Total N (%) | 511 (100) | 311 (60.9) | 200 (39.1) |
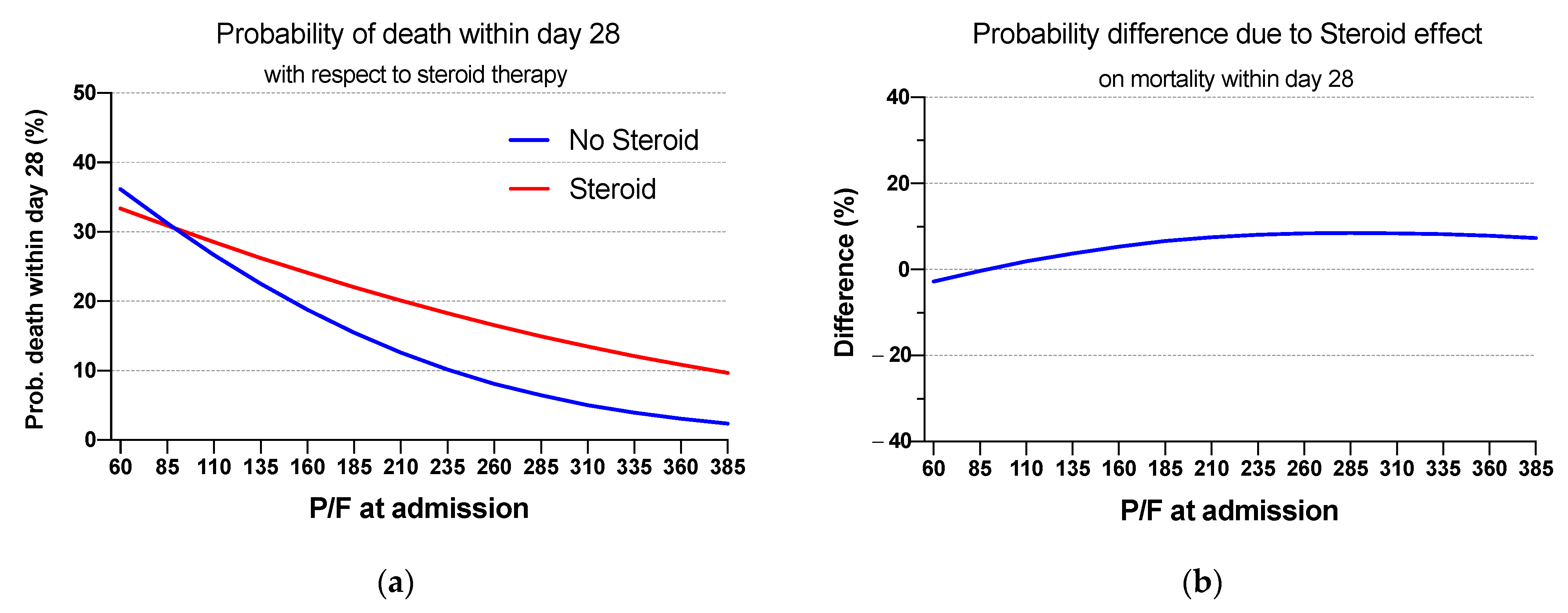
| P/F | Steroids | Prob. of Death within 28 Days | ||
|---|---|---|---|---|
| Average (%) (95% CI) | Difference (%) (95% CI) | p-Value | ||
| 60 | No | 36.2 (3.9–68.4) | −2.8 (−37.8; 32.2) | 0.875 |
| Yes | 33.4 (18.6–48.1) | |||
| 85 | No | 31.2 (3.9–58.5) | −0.3 (−30.1; 29.4) | 0.982 |
| Yes | 30.9 (18.1–43.7) | |||
| 110 | No | 26.7 (4.1–49.2) | 1.9 (−22.9; 26.6) | 0.884 |
| Yes | 28.5 (17.6–39.5) | |||
| 135 | No | 22.5 (4.3–40.7) | 3.7 (−16.5; 23.9) | 0.716 |
| Yes | 26.2 (17.0–35.5) | |||
| 160 | No | 18.8 (4.5–33.1) | 5.3 (−10.8; 21.4) | 0.518 |
| Yes | 24.1 (16.3–31.9) | |||
| 185 | No | 15.5 (4.5–26.40) | 6.6 (−6.1; 19.2) | 0.309 |
| Yes | 22.0 (15.4–28.6) | |||
| 210 | No | 12.6 (4.4–20.8) | 7.5 (−2.4; 17.4) | 0.138 |
| Yes | 20.1 (14.4–25.70) | |||
| 235 | No | 10.2 (4.1–16.2) | 8.1 (0.3; 15.9) | 0.043 |
| Yes | 18.3 (13.2–23.3) | |||
| 260 | No | 8.1 (3.6–12.6) | 8.4 (1.9; 14.9) | 0.011 |
| Yes | 16.5 (11.9–21.2) | |||
| 285 | No | 6.4 (2.9–9.9) | 8.5 (2.7; 14.3) | 0.004 |
| Yes | 14.9 (10.3–19.5) | |||
| 310 | No | 5.0 (2.2–7.9) | 8.4 (2.9; 13.9) | 0.003 |
| Yes | 13.5 (8.8–18.2) | |||
| 335 | No | 3.9 (1.4–6.4) | 8.2 (2.7; 13.6) | 0.004 |
| Yes | 12.1 (7.2–16.9) | |||
| 360 | No | 3.0 (0.8–5.3) | 7.8 (2.3; 13.3) | 0.006 |
| Yes | 10.8 (5.8–15.9) | |||
| 385 | No | 2.3 (0.3–4.4) | 7.3 (1.8; 12.9) | 0.010 |
| Yes | 9.7 (4.5–14.9) | |||
| Patients Feature | Total N (%) | Death or ICU N (%) | Not Death or ICU N(%) | OR (95% CI) | p-Value | LR Test p-Value | |
|---|---|---|---|---|---|---|---|
| Age | <50 Years | 110 (21.5) | 12 (10.9) | 98 (89.1) | ref | 0.002 | 0.424 |
| 50–70 Years | 198 (38.7) | 31 (15.7) | 167 (84.3) | 1.52 (0.744–3.09) | |||
| >70 Years | 203 (39.7) | 52 (25.6) | 151 (74.4) | 2.81 (2.56–28.14) | |||
| Sex | Female | 181 (35.4) | 33 (18.2) | 148 (81.8) | ref | 0.877 | 0.676 |
| Male | 330 (64.6) | 62 (18.8) | 268 (81.2) | 1.04 (0.65–1.66) | |||
| Use of Antivirals | No | 185 (36.2) | 24 (13.0) | 161 (87.0) | ref | 0.012 | 0.440 |
| Yes | 326 (63.8) | 71 (21.8) | 255 (78.2) | 1.87 (1.13–3.09) | |||
| Use of Aminoquinoline | No | 218 (42.7) | 43 (19.7) | 175 (80.3) | ref | 0.571 | 0.355 |
| Yes | 293 (57.3) | 52 (17.7) | 241 (82.3) | .88 (0.56–1.36) | |||
| Use of Azithromycin | No | 327 (64.0) | 52 (15.9) | 275 (84.1) | ref | 0.040 | 0.579 |
| Yes | 184 (36.0) | 43 (23.4) | 141 (76.6) | 1.61 (1.03–2.53) | |||
| Use of monoclonal antib. | No | 408 (79.8) | 62 (15.2) | 346 (84.8) | ref | <0.001 | 0.788 |
| Yes | 103 (20.2) | 33 (32.0) | 70 (68.0) | 2.63 (1.6–4.31) | |||
| Diabetes | No | 430 (84.1) | 73 (17.0) | 357 (83.0) | ref | 0.038 | 0.326 |
| Yes | 81 (15.9) | 22 (27.2) | 59 (72.8) | 1.82 (1.05–3.16) | |||
| Hypertension | No | 295 (57.7) | 41 (13.9) | 254 (86.1) | ref | 0.002 | 0.215 |
| Yes | 216 (42.3) | 54 (25.0) | 162 (75.0) | 2.07 (1.32–3.24) | |||
| Renal failure | No | 482 (94.3) | 85 (17.6) | 397 (82.4) | ref | 0.036 | 0.202 |
| Yes | 29 (5.7) | 10 (34.5) | 19 (65.5) | 2.46 (1.1–5.47) | |||
| Cancer | No | 458 (89.6) | 82 (17.9) | 376 (82.1) | ref | 0.037 | 0.367 |
| Yes | 53 (10.4) | 13 (24.5) | 40 (75.5) | 1.49 (0.76–2.91) | |||
| CVD | No | 358 (70.1) | 41 (11.5) | 317 (88.5) | ref | <0.001 | <0.001 |
| Yes | 143 (28.0) | 44 (30.8) | 99 (69.2) | 2.76 (1.74–4.39) | |||
| COPD | No | 460 (90.0) | 80 (17.4) | 380 (82.6) | ref | 0.047 | 0.606 |
| Yes | 51 (10.0) | 15 (29.4) | 36 (70.6) | 1.98 (1.03–3.79) | |||
| Obesity | No | 447 (87.5) | 73 (16.3) | 374 (83.7) | ref | 0.001 | 0.013 |
| Yes | 64 (12.5) | 22 (34.4) | 42 (65.6) | 2.68 (1.51–4.76) | |||
| Overall | - | 511 (100.0) | 95 (18.6) | 416 (81.4) | - | - | |
| P/F at admission (Median–IQR) * | 343 (271–406) | 244 (148–329) | 356 (296–419) | 0.95 (0.94–0.96) | <0.001 | - | |
| P/F | Steroids | Prob. of Death within 28 Days or ICU | ||
|---|---|---|---|---|
| Average (%) (95% CI) | Difference (%) (95% CI) | p-Value | ||
| 60 | No | 59.7 (24.0–95.3) | 4.5 (−34.2; 43.1) | 0.821 |
| Yes | 64.1 (48.6–79.7) | |||
| 85 | No | 52.1 (18.8–85.4) | 9.1 (−26.9; 45.1) | 0.622 |
| Yes | 61.2 (46.7–75.6) | |||
| 110 | No | 44.5 (15.0–73.9) | 13.7 (−18.4; 45.7) | 0.403 |
| Yes | 58.1 (44.9–71.3) | |||
| 135 | No | 37.1 (12.4–61.8) | 17.9 (−9.3; 45.1) | 0.197 |
| Yes | 55.0 (43.0–66.9) | |||
| 160 | No | 30.3 (10.6–50.0) | 21.6 (−0.6; 43.8) | 0.056 |
| Yes | 51.9 (41.2–62.5) | |||
| 185 | No | 24.2 (9.2–39.2) | 24.5 (7.0; 42.0) | 0.006 |
| Yes | 48.7 (39.3–58.1) | |||
| 210 | No | 19.0 (8.0–29.9) | 26.6 (13.0; 40.2) | <0.001 |
| Yes | 45.6 (37.3–53.8) | |||
| 235 | No | 14.6 (6.8–22.5) | 27.9 (17.1; 38.6) | <0.001 |
| Yes | 42.5 (35.0–49.9) | |||
| 260 | No | 11.1 (5.5–16.8) | 28.3 (19.4; 37.2) | <0.001 |
| Yes | 39.4 (32.5–46.4) | |||
| 285 | No | 8.4 (94.2–12.5) | 28.1 (20.1; 36.1) | <0.001 |
| Yes | 36.5 (29.6–43.3) | |||
| 310 | No | 6.2 (2.9–9.5) | 27.4 (19.6; 35.2) | <0.001 |
| Yes | 33.6 (26.5–40.7) | |||
| 335 | No | 4.6 (1.9–7.3) | 26.3 (18.2; 34.3) | <0.001 |
| Yes | 30.9 (23.3–38.4) | |||
| 360 | No | 3.4 (1.0–5.7) | 24.9 (16.4; 33.4) | <0.001 |
| Yes | 28.3 (20.1–36.4) | |||
| 385 | No | 2.5 (0.5–4.4) | 23.3 (14.4; 32.2) | <0.001 |
| Yes | 25.8 (17.1–34.5) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vita, S.; Centanni, D.; Lanini, S.; Piselli, P.; Rosati, S.; Giancola, M.L.; Mondi, A.; Pinnetti, C.; Topino, S.; Chinello, P.; et al. Benefits of Steroid Therapy in COVID-19 Patients with Different PaO2/FiO2 Ratio at Admission. J. Clin. Med. 2021, 10, 3236. https://doi.org/10.3390/jcm10153236
Vita S, Centanni D, Lanini S, Piselli P, Rosati S, Giancola ML, Mondi A, Pinnetti C, Topino S, Chinello P, et al. Benefits of Steroid Therapy in COVID-19 Patients with Different PaO2/FiO2 Ratio at Admission. Journal of Clinical Medicine. 2021; 10(15):3236. https://doi.org/10.3390/jcm10153236
Chicago/Turabian StyleVita, Serena, Daniele Centanni, Simone Lanini, Pierluca Piselli, Silvia Rosati, Maria Letizia Giancola, Annalisa Mondi, Carmela Pinnetti, Simone Topino, Pierangelo Chinello, and et al. 2021. "Benefits of Steroid Therapy in COVID-19 Patients with Different PaO2/FiO2 Ratio at Admission" Journal of Clinical Medicine 10, no. 15: 3236. https://doi.org/10.3390/jcm10153236
APA StyleVita, S., Centanni, D., Lanini, S., Piselli, P., Rosati, S., Giancola, M. L., Mondi, A., Pinnetti, C., Topino, S., Chinello, P., Mosti, S., Gualano, G., Faraglia, F., Iacomi, F., Marchioni, L., Maritti, M., Girardi, E., Ippolito, G., Nicastri, E., & on behalf of the ReCOVeRI Study Group. (2021). Benefits of Steroid Therapy in COVID-19 Patients with Different PaO2/FiO2 Ratio at Admission. Journal of Clinical Medicine, 10(15), 3236. https://doi.org/10.3390/jcm10153236









