Identifying the Critical Threshold for Long-Term Pediatric Neurological Hospitalizations of the Offspring in Preterm Delivery
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
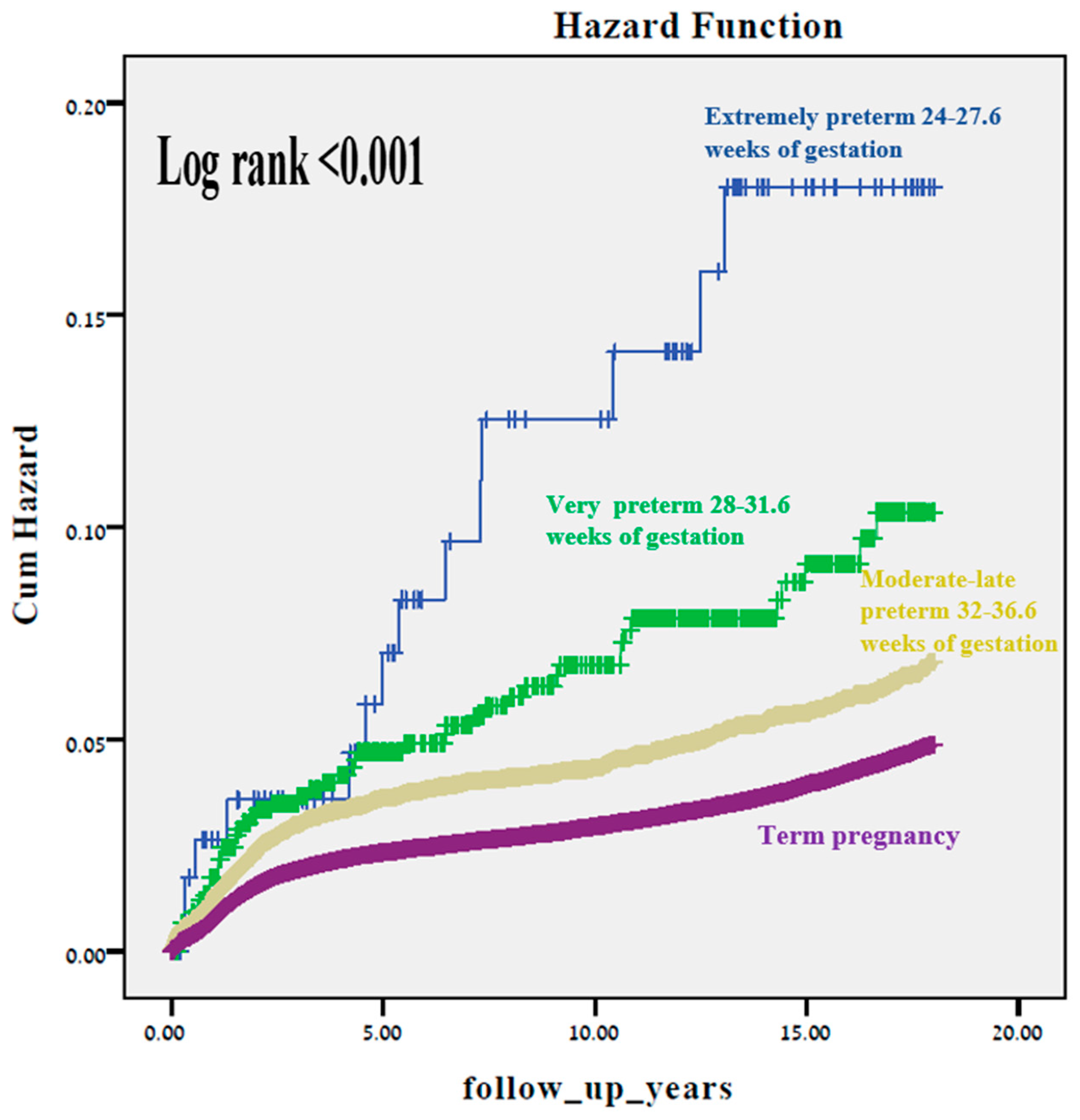
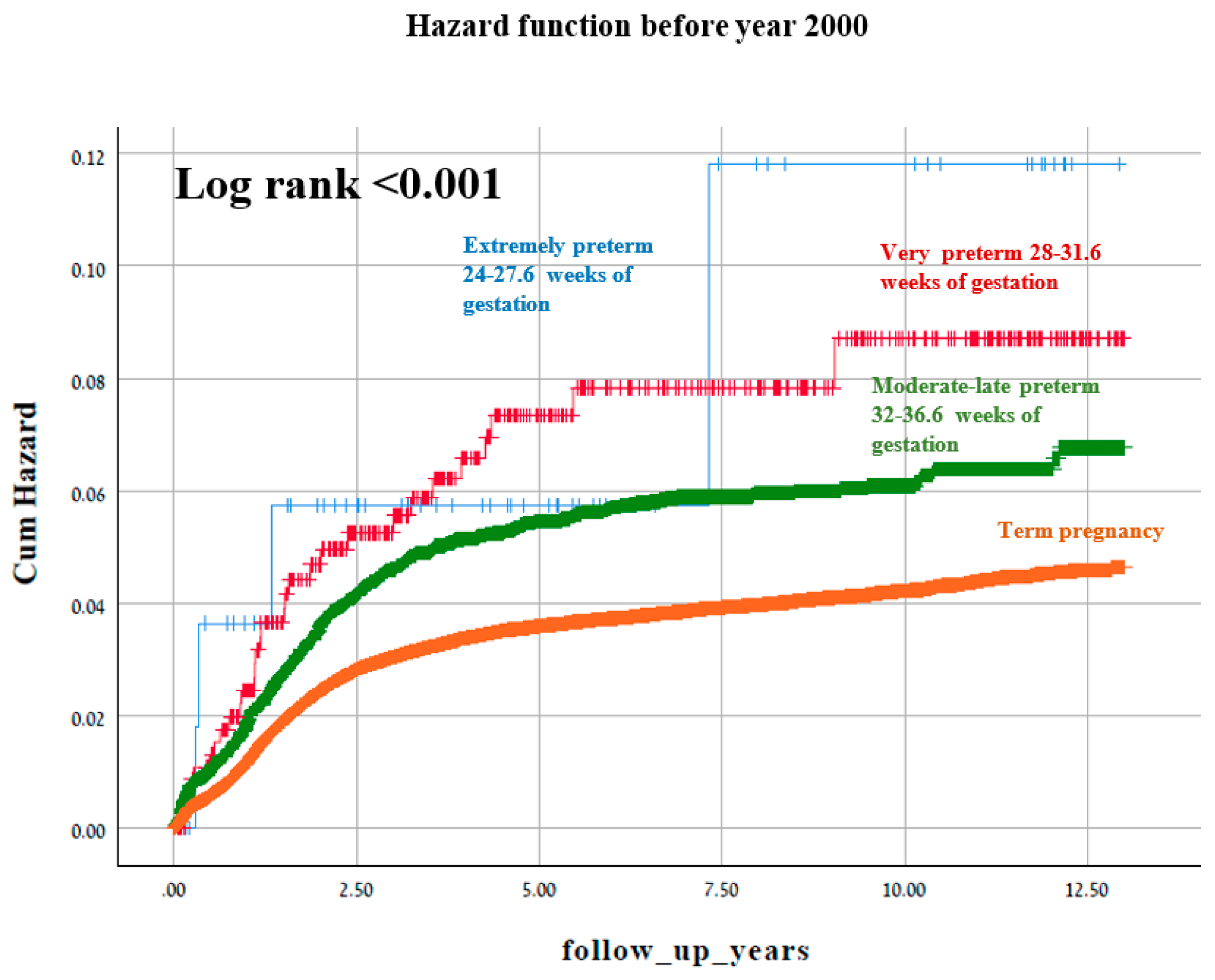
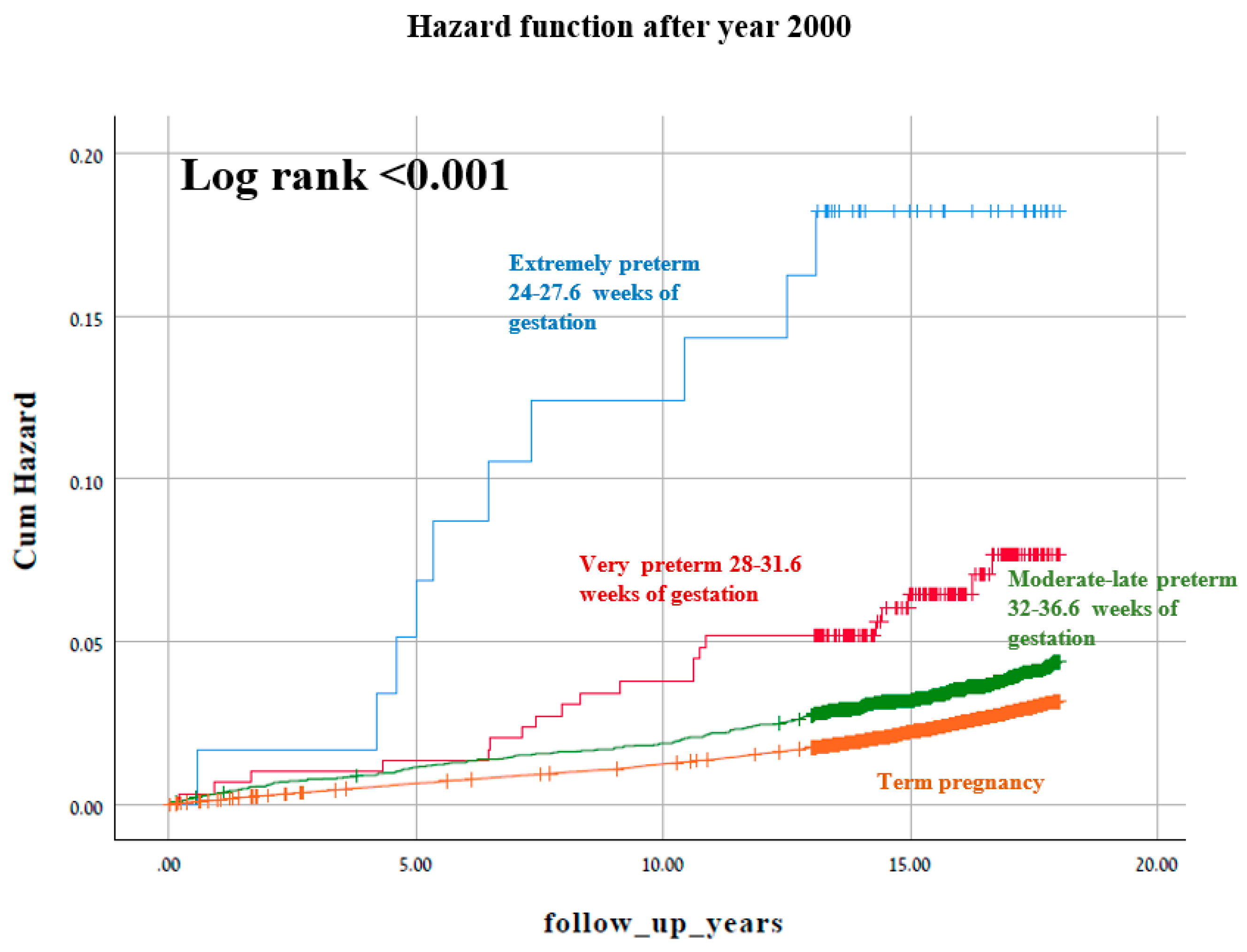
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. Born Too Soon: The Global Action Report on Preterm Birth. Available online: http://www.who.int/pmnch/media/news/2012/preterm_birth_report/en/ (accessed on 23 May 2021).
- Liu, L.; Oza, S.; Hogan, D.; Perin, J.; Rudan, I.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, Regional, and National Causes of Child Mortality in 2000–13, with Projections to Inform Post-2015 Priorities: An Updated Systematic Analysis. Lancet 2015, 385, 430–440. [Google Scholar] [CrossRef]
- Newman, D.E.; Paamoni-Keren, O.; Press, F.; Wiznitzer, A.; Mazor, M.; Sheiner, E. Neonatal outcome in preterm deliveries between 23 and 27 weeks’ gestation with and without preterm premature rupture of membranes. Arch. Gynecol. Obstet. 2009, 280, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Jarjour, I.T. Neurodevelopmental outcome after extreme prematurity: A review of the literature. Pediatr. Neurol. 2015, 52, 143–152. [Google Scholar] [CrossRef]
- Lin, C.Y.; Hsu, C.H.; Chang, J.H. Neurodevelopmental outcomes at 2 and 5 years of age in very-low-birth-weight preterm infants born between 2002 and 2009: A prospective cohort study in Taiwan. Pediatr. Neonatol. 2020, 61, 36–44. [Google Scholar] [CrossRef]
- Jameson, R.A.; Bernstein, H.B. Magnesium Sulfate and Novel Therapies to Promote Neuroprotection. Clin. Perinatol. 2019, 46, 187–201. [Google Scholar] [CrossRef]
- Hollanders, J.J.; Schaëfer, N.; Van Der Pal, S.M.; Oosterlaan, J.; Rotteveel, J.; Finken, M.J.J. Long-Term Neurodevelopmental and Functional Outcomes of Infants Born Very Preterm and/or with a Very Low Birth Weight. Neonatology 2019, 115, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Pariente, G.; Wainstock, T.; Walfisch, A.; Landau, D.; Sheiner, E. Placental abruption and long-term neurological hospitalisations in the offspring. Paediatr. Perinat. Epidemiol. 2019, 33, 215–222. [Google Scholar] [CrossRef]
- Bachnas, M.A.; Akbar, M.I.A.; Dachlan, E.G.; Dekker, G. The role of magnesium sulfate (MgSO4) in fetal neuroprotection. J. Matern. Neonatal. Med. 2019, 34, 966–978. [Google Scholar] [CrossRef]
- Hadar, O.; Sheiner, E.; Wainstock, T. The Association Between Delivery of Small-for-Gestational-Age Neonate and Their Risk for Long-Term Neurological Morbidity. J. Clin. Med. 2020, 9, 3199. [Google Scholar] [CrossRef]
- Bashiri, A.; Burstein, E.; Mazor, M. Cerebral palsy and fetal inflammatory response syndrome: A review. J. Perinat. Med. 2006, 34, 5–12. [Google Scholar] [CrossRef]
- Gutvirtz, G.; Wainstock, T.; Masad, R.; Landau, D.; Sheiner, E. Does nuchal cord at birth increase the risk for cerebral palsy? Early Hum. Dev. 2019, 133, 1–4. [Google Scholar] [CrossRef]
- Spittle, A.J.; Morgan, C.; Olsen, J.E.; Novak, I.; Cheong, J.L.Y. Early Diagnosis and Treatment of Cerebral Palsy in Children with a History of Preterm Birth. Clin. Perinatol. 2018, 45, 409–420. [Google Scholar] [CrossRef]
- Hirvonen, M.; Ojala, R.; Korhonen, P.; Haataja, P.; Eriksson, K.; Gissler, M.; Luukkaala, T.; Tammela, O. Cerebral palsy among children born moderately and late preterm. Pediatrics 2014, 134, e1584–e1593. [Google Scholar] [CrossRef]
- Bax, M.; Goldstein, M.; Rosenbaun, P.; Leviton, A.; Paneth, N.; Dan, B.; Jacobsson, B.; Damiano, D. Proposed definition and classification of cerebral palsy, April 2005. Dev. Med. Child Neurol. 2005, 47, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Oskoui, M.; Coutinho, F.; Dykeman, J.; Jetté, N.; Pringsheim, T. An update on the prevalence of cerebral palsy: A systematic review and meta-analysis. Dev. Med. Child Neurol. 2013, 55, 509–519. [Google Scholar] [CrossRef]
- Himpens, E.; Oostra, A.; Franki, I.; Vansteelandt, S.; Vanhaesebrouck, P.; den Broeck, C.V. Predictability of cerebral palsy in a high-risk NICU population. Early Hum. Dev. 2010, 86, 413–417. [Google Scholar] [CrossRef]
- Hagberg, H.; Gressens, P.; Mallard, C. Inflammation during fetal and neonatal life: Implications for neurologic and neuropsychiatric disease in children and adults. Ann. Neurol. 2012, 71, 444–457. [Google Scholar] [CrossRef] [PubMed]
- Fahey, M.C.; Maclennan, A.H.; Kretzschmar, D.; Gecz, J.; Kruer, M.C. The genetic basis of cerebral palsy. Dev. Med. Child Neurol. 2017, 59, 462–469. [Google Scholar] [CrossRef]
- Harding, D.R.; Humphries, S.E.; Whitelaw, A.; Marlow, N.; Montgomery, H.E. Cognitive outcome and cyclo-oxygenase-2 gene (−765 G/C) variation in the preterm infant. Arch. Dis. Child Fetal. Neonatal Ed. 2007, 92, 108–112. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Casavant, S.G.; Cong, X.; Moore, J.; Starkweather, A. Associations between preterm infant stress, epigenetic alteration, telomere length and neurodevelopmental outcomes: A systematic review. Early Hum. Dev. 2019, 131, 63–74. [Google Scholar] [CrossRef]
- Carlo, W.A.; McDonald, S.A.; Tyson, J.E.; Stoll, B.J.; Ehrenkranz, R.A.; Shankaran, S.; Goldberg, R.N.; Das, A.; Schendel, D.; Thorsen, P.; et al. Cytokines and neurodevelopmental outcomes in extremely low birth weight infants. J. Pediatr. 2011, 159, 919–925. [Google Scholar] [CrossRef]
- Cetral Bureau of Statistics. Population—Statistical Abstract of Israel 2020—No.71. Available online: https://www.cbs.gov.il/en/publications/Pages/2020/Population-Statistical-Abstract-of-Israel-2020-No-71.aspx (accessed on 30 September 2020).
- Pascal, A.; Govaert, P.; Oostra, A.; Naulaers, G.; Ortibus, E.; Van den Broeck, C. Neurodevelopmental outcome in very preterm and very-low-birthweight infants born over the past decade: A meta-analytic review. Dev. Med. Child Neurol. 2018, 60, 342–355. [Google Scholar] [CrossRef]
- Herber-Jonat, S.; Streiftau, S.; Knauss, E.; Voigt, F.; Flemmer, A.W.; Hummler, H.D.; Schulze, A.; Bode, H. Long-term outcome at age 7–10 years after extreme prematurity-a prospective, two centre cohort study of children born before 25 completed weeks of gestation (1999–2003). J. Matern. Neonatal. Med. 2014, 27, 1620–1626. [Google Scholar] [CrossRef]
- Ream, M.A.; Lehwald, L. Neurologic Consequences of Preterm Birth. Curr. Neurol. Neurosci. Rep. 2017, 18, 1–10. [Google Scholar] [CrossRef]
- Davidesko, S.; Wainstock, T.; Sheiner, E.; Pariente, G. Long-Term Infectious Morbidity of Premature Infants: Is There a Critical Threshold? J. Clin. Med. 2020, 9, 3008. [Google Scholar] [CrossRef]
- Ohana, O.; Wainstock, T.; Sheiner, E.; Leibson, T.; Pariente, G. Long-Term digestive hospitalizations of premature infants (besides necrotizing enterocolitis): Is there a critical threshold? Arch. Gynecol. Obstet. 2021. [Google Scholar] [CrossRef] [PubMed]
- Fallang, B.; Hadders-Algra, M. Postural behavior in children born preterm. Neural Plast. 2005, 12, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Ancel, P.Y.; Livinec, F.; Larroque, B.; Marret, S.; Arnaud, C.; Pierrat, V.; Dehan, M.; Sylvie, N.; Escande, B.; Burguet, A.; et al. Cerebral palsy among very preterm children in relation to gestational age and neonatal ultrasound abnormalities: The EPIPAGE cohort study. Pediatrics 2006, 117, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Volpe, J.J. Overview: Normal and abnormal human brain development. Ment. Retard. Dev. Disabil. Res. Rev. 2000, 5, 1–5. [Google Scholar] [CrossRef]
- Lin, G.G.; Scott, J.G. Cytokines and Neurodevelopmental Outcomes in Extremely Low Birth Weight Infants. J. Pediatr. 2012, 100, 130–134. [Google Scholar]
- Andrews, W.W.; Cliver, S.P.; Biasini, F.; Peralta-Carcelen, A.M.; Rector, R.; Alriksson-Schmidt, A.I.; Faye-Petersen, O.; Carlo, W.; Goldenberg, R.; Hauth, J.C. Early preterm birth: Association between in utero exposure to acute inflammation and severe neurodevelopmental disability at 6 years of age. Am. J. Obstet. Gynecol. 2008, 198, 466.e1. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, C.N.; Tsimis, M.; Burd, I. Infections and brain development. Obstet. Gynecol. Surv. 2015, 70, 644–655. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Extreme PTB (n = 118) (%) | Very PTB (n = 776) (%) | Moderate-Late PTB (n = 13,308) (%) | Term Birth (n = 206,361) (%) | p-Value |
|---|---|---|---|---|---|
| Maternal Age, years (mean ± SD) | 28.47 ± 6.34 | 28.36 ± 6.37 | 28.31 ± 6.24 | 28.24 ± 5.76 | 0.482 |
| Gravidity | |||||
| 1 | 29.7 | 24.5 | 23.3 | 20.2 | <0.001 |
| 2–4 | 46.6 | 44.8 | 44.5 | 48.7 | |
| 5+ | 23.7 | 30.7 | 32.1 | 31.2 | |
| Parity | |||||
| 1 | 38.1 | 31.4 | 29.0 | 24.2 | |
| 2–4 | 48.3 | 47.7 | 47.7 | 52.0 | <0.001 |
| 5+ | 13.6 | 20.9 | 23.3 | 23.8 | |
| Smokers | 4.2 | 1 | 1.5 | 1 | <0.001 |
| Fertility treatments | <0.001 | ||||
| In vitro fertilization | 3.4 | 2.6 | 2.3 | 1.1 | |
| Ovulation induction | 1.7 | 1.8 | 1.1 | 0.8 | |
| Hypertensive disorders * | 8.5 | 19.1 | 12.9 | 4.7 | <0.001 |
| Maternal diabetes ** | 0.0 | 6.2 | 8.1 | 5.2 | <0.001 |
| Induction of labor | 14.3 | 4 | 27.3 | 31.8 | <0.001 |
| Type of birth | <0.001 | ||||
| Vaginal delivery | 48.3 | 46.5 | 67.3 | 83.9 | |
| Assisted vaginal delivery | 0 | 0.6 | 1.7 | 3.3 | |
| Cesarean delivery | 51.7 | 52.8 | 31.0 | 12.7 | |
| Small for gestational age (SGA) | 3.4 | 1.8 | 3.6 | 4.4 | <0.001 |
| Apgar score 1 min < 7 | 49.2 | 28.1 | 10.2 | 4.1 | <0.001 |
| Apgar score 5 min < 7 | 14.4 | 6.2 | 2.5 | 1.4 | <0.001 |
| Neurological Morbidity of the Offspring | Extreme PTB (n = 118) (%) | Very PTB (n = 776) (%) | Moderate-Late PTB (n = 13,308) (%) | Term Birth n= (206,361) (%) | p-Value |
|---|---|---|---|---|---|
| Movement disorders | 4.2 | 3.5 | 2.7 | 1.8 | <0.001 |
| Cerebral palsy | 4.2 | 0.9 | 0.2 | 0.1 | <0.001 |
| Psychiatric disorders | 2.5 | 0.9 | 0.7 | 0.5 | <0.001 |
| Developmental disorders | 0.8 | 0.3 | 0.2 | 0.1 | <0.001 |
| Degenerative disorders | 0 | 0.6 | 0.1 | 0.1 | <0.001 |
| Total Neurologic Related Hospitalizations | Cerebral Palsy | |||||
|---|---|---|---|---|---|---|
| Gestational Age | aHR * | 95% CI | p-Value | aHR ** | 95% CI | p-Value |
| Term delivery (reference) >37 gestational weeks | 1 | - | - | 1 | - | - |
| Moderate to late preterm | 1.3 | 1.2–1.5 | <0.001 | 2.5 | 1.6–3.9 | <0.001 |
| Very preterm | 1.9 | 1.4–2.5 | <0.001 | 13.4 | 6.2–28.7 | <0.001 |
| Extremely preterm | 3.9 | 2.3–6.6 | <0.001 | 62.4 | 25.6–152.4 | <0.001 |
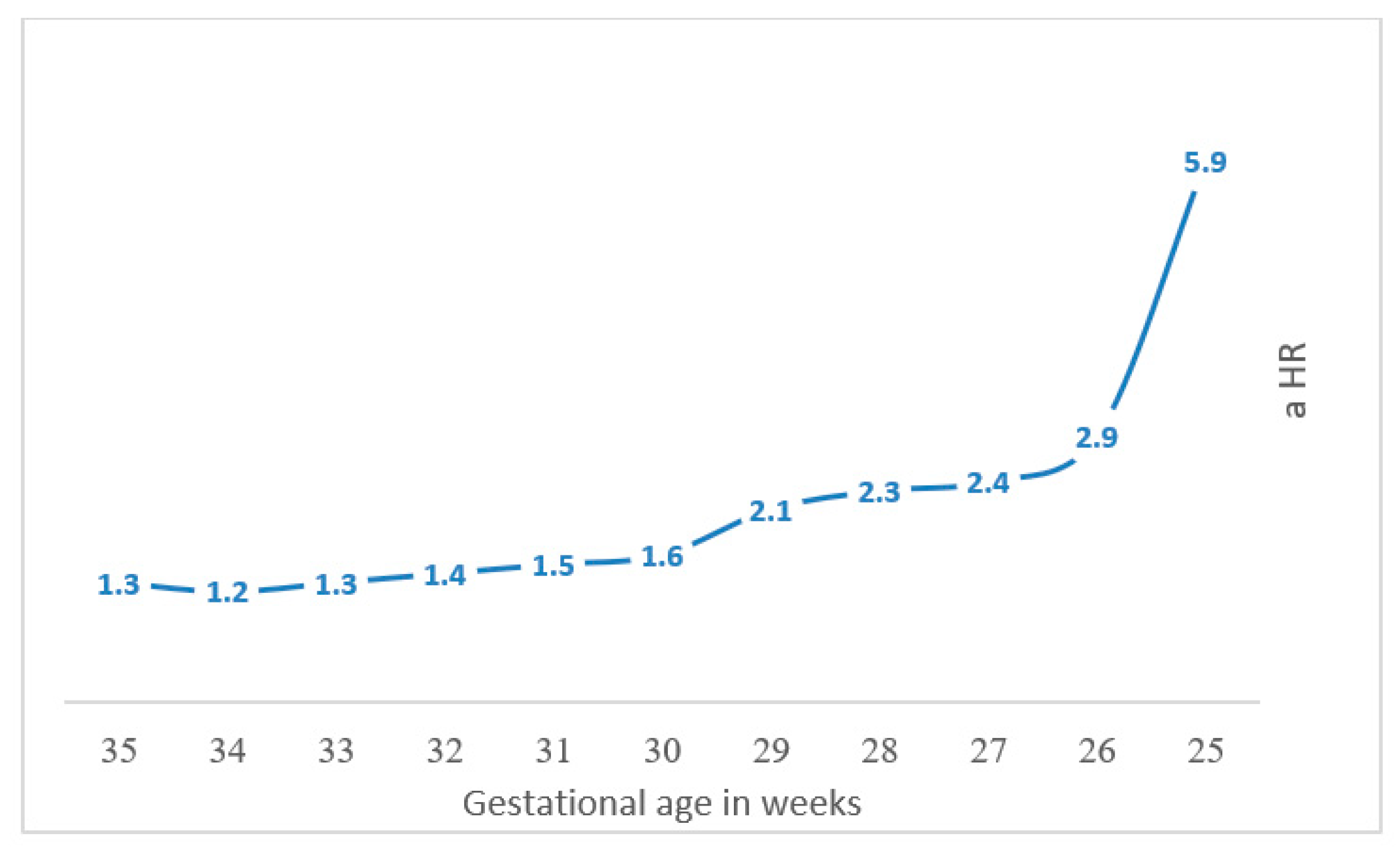
| Total Neurologic Related Hospitalizations | ||||
|---|---|---|---|---|
| Gestational Age | aHR * | 95% CI | p-Value | |
| Model 1 | PTB 25 gestational week versus other later weeks of PTB | 5.9 | 2.2–15.9 | <0.001 |
| Model 2 | PTB 26 gestational week versus other later weeks of PTB | 2.9 | 1.4–6.2 | <0.004 |
| Model 3 | PTB 27 gestational week versus other later weeks of PTB | 2.4 | 1.4–4.2 | <0.001 |
| Model 4 | PTB 28 gestational week versus other later weeks of PTB | 2.3 | 1.5–3.5 | <0.001 |
| Model 5 | PTB 29 gestational week versus other later weeks of PTB | 2.1 | 1.4–3.0 | <0.001 |
| Model 6 | PTB 30 gestational week versus other later weeks of PTB | 1.6 | 1.2–2.2 | <0.002 |
| Model 7 | PTB 31 gestational week versus other later weeks of PTB | 1.5 | 1.1–1.9 | <0.002 |
| Model 8 | PTB 32 gestational week versus other later weeks of PTB | 1.4 | 1.1–1.7 | <0.003 |
| Model 9 | PTB 33 gestational week versus other later weeks of PTB | 1.3 | 1.1–1.6 | <0.003 |
| Model 10 | PTB 34 gestational week versus other later weeks of PTB | 1.2 | 1.0–1.4 | <0.03 |
| Model 11 | PTB 35 gestational week versus other later weeks of PTB | 1.3 | 1.1–1.5 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zer, S.; Wainstock, T.; Sheiner, E.; Miodownik, S.; Pariente, G. Identifying the Critical Threshold for Long-Term Pediatric Neurological Hospitalizations of the Offspring in Preterm Delivery. J. Clin. Med. 2021, 10, 2919. https://doi.org/10.3390/jcm10132919
Zer S, Wainstock T, Sheiner E, Miodownik S, Pariente G. Identifying the Critical Threshold for Long-Term Pediatric Neurological Hospitalizations of the Offspring in Preterm Delivery. Journal of Clinical Medicine. 2021; 10(13):2919. https://doi.org/10.3390/jcm10132919
Chicago/Turabian StyleZer, Shiran, Tamar Wainstock, Eyal Sheiner, Shayna Miodownik, and Gali Pariente. 2021. "Identifying the Critical Threshold for Long-Term Pediatric Neurological Hospitalizations of the Offspring in Preterm Delivery" Journal of Clinical Medicine 10, no. 13: 2919. https://doi.org/10.3390/jcm10132919
APA StyleZer, S., Wainstock, T., Sheiner, E., Miodownik, S., & Pariente, G. (2021). Identifying the Critical Threshold for Long-Term Pediatric Neurological Hospitalizations of the Offspring in Preterm Delivery. Journal of Clinical Medicine, 10(13), 2919. https://doi.org/10.3390/jcm10132919








