Fundamentals, Diagnostic Capabilities, and Perspective of Narrow Band Imaging for Early Gastric Cancer
Abstract
:1. Introduction
2. Basic Observation Methods and Points of ME-NBI for Gastric Cancer
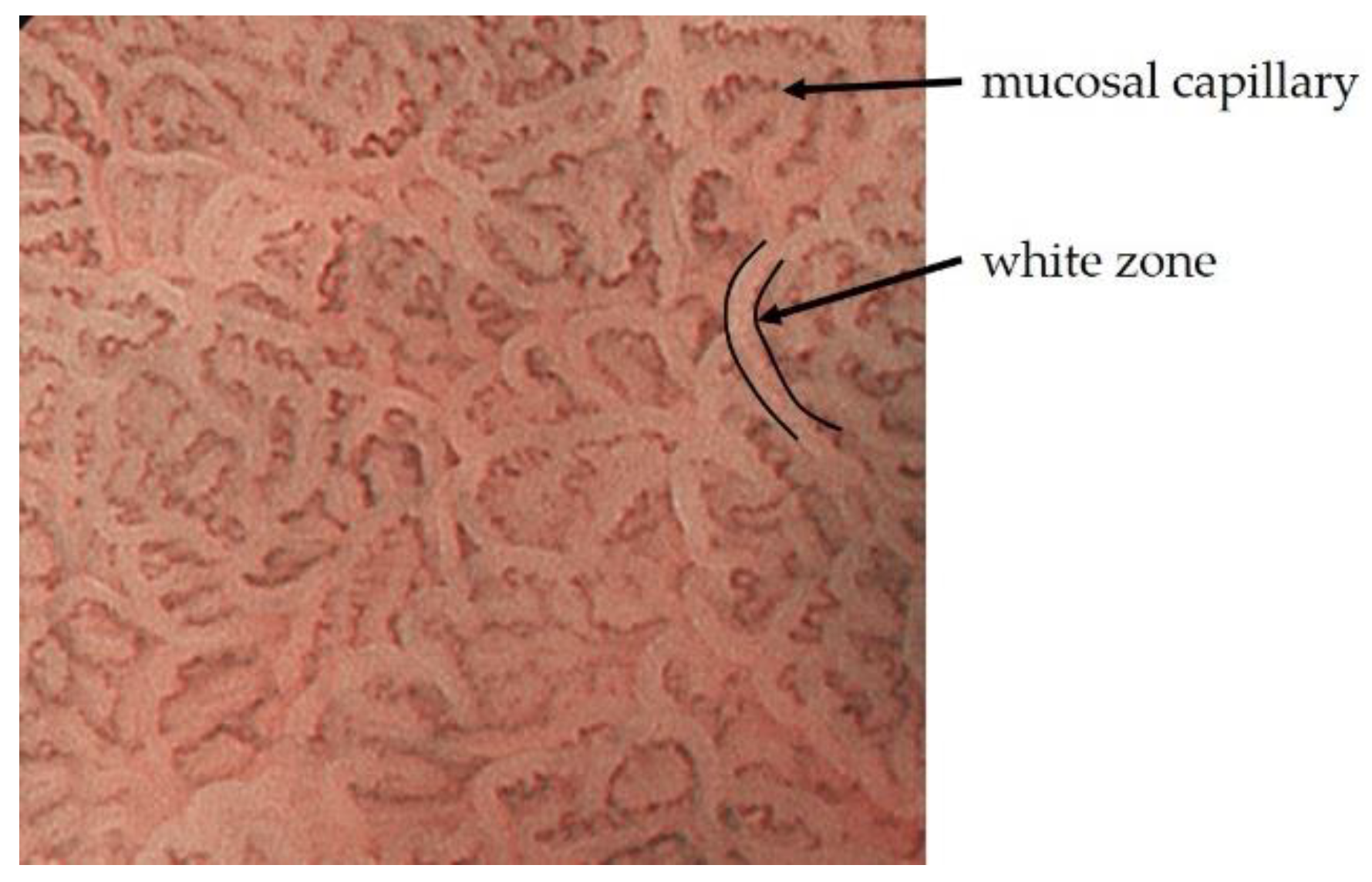
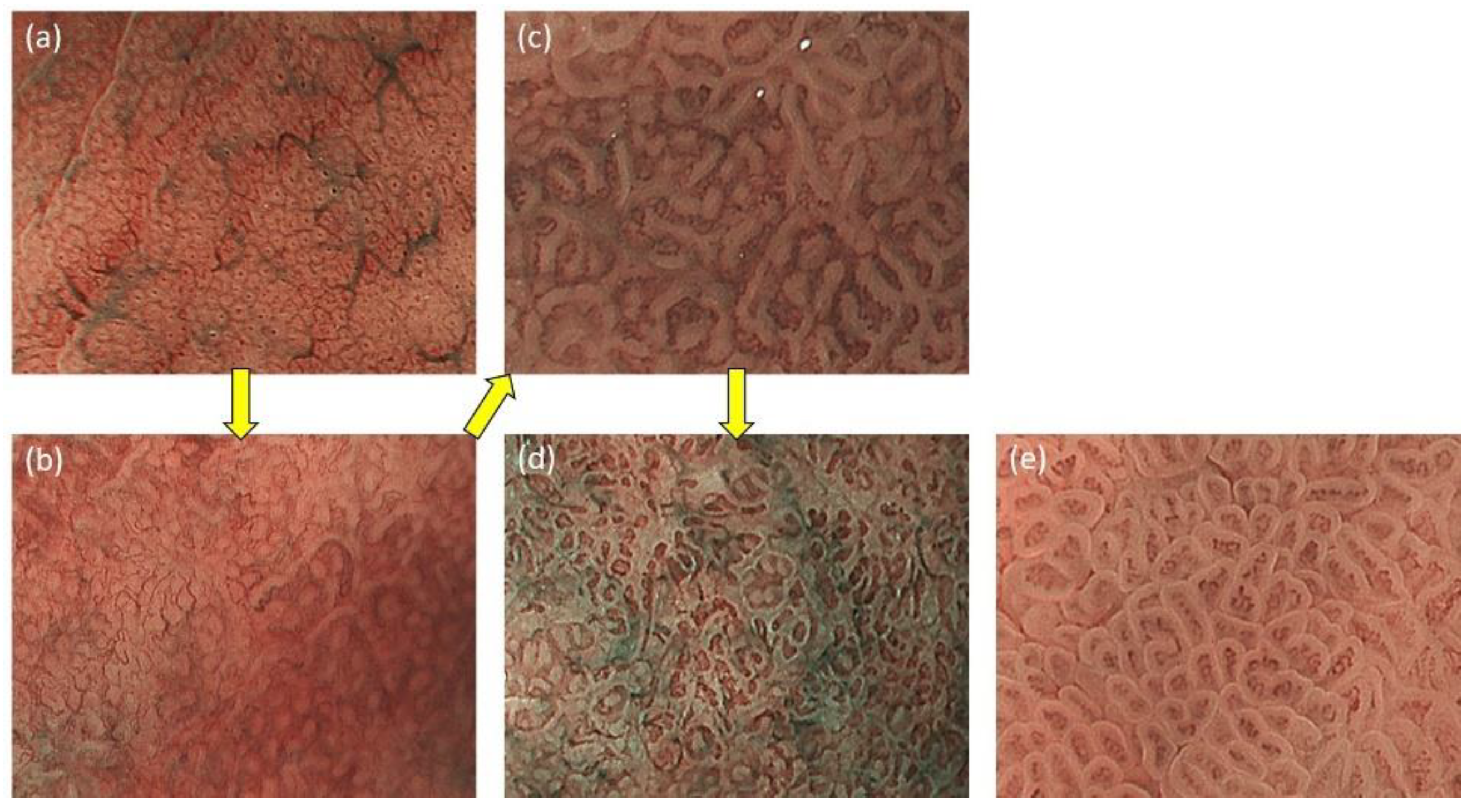
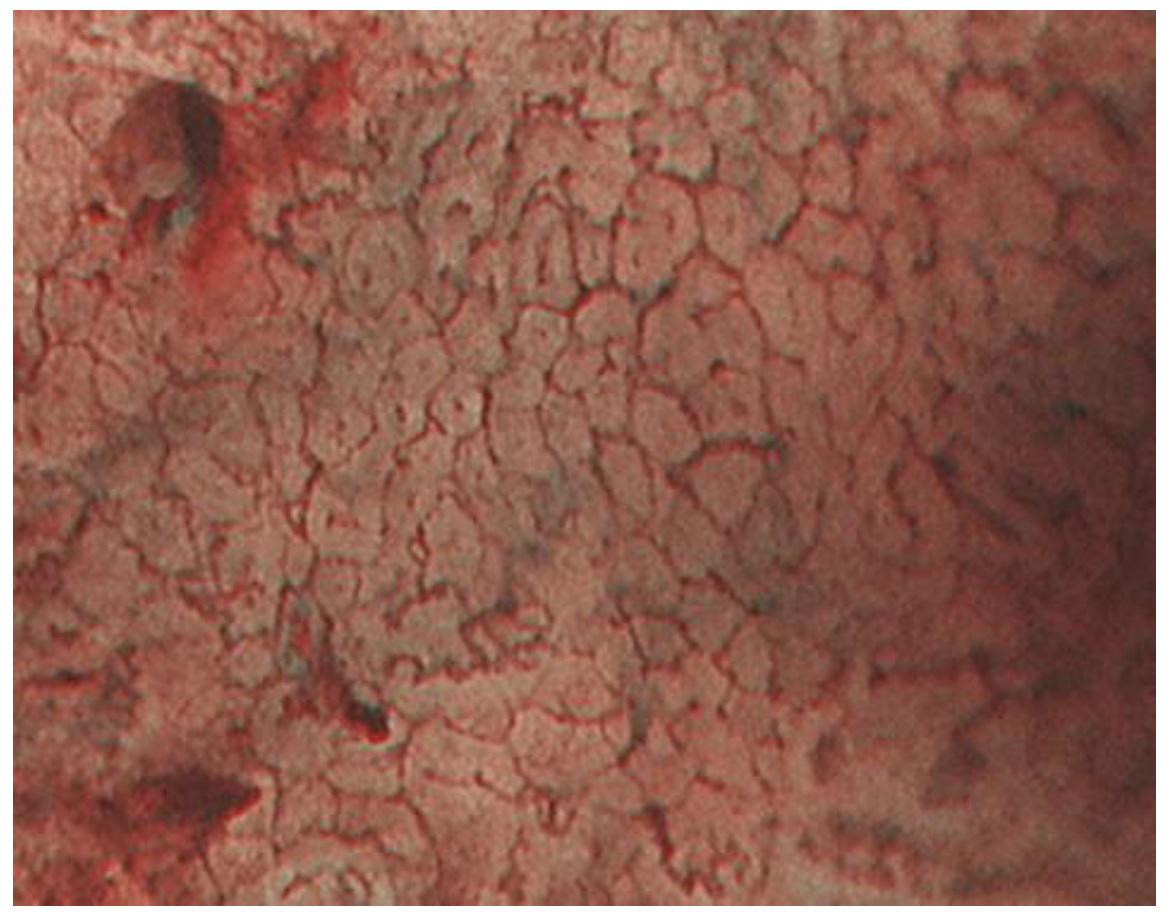
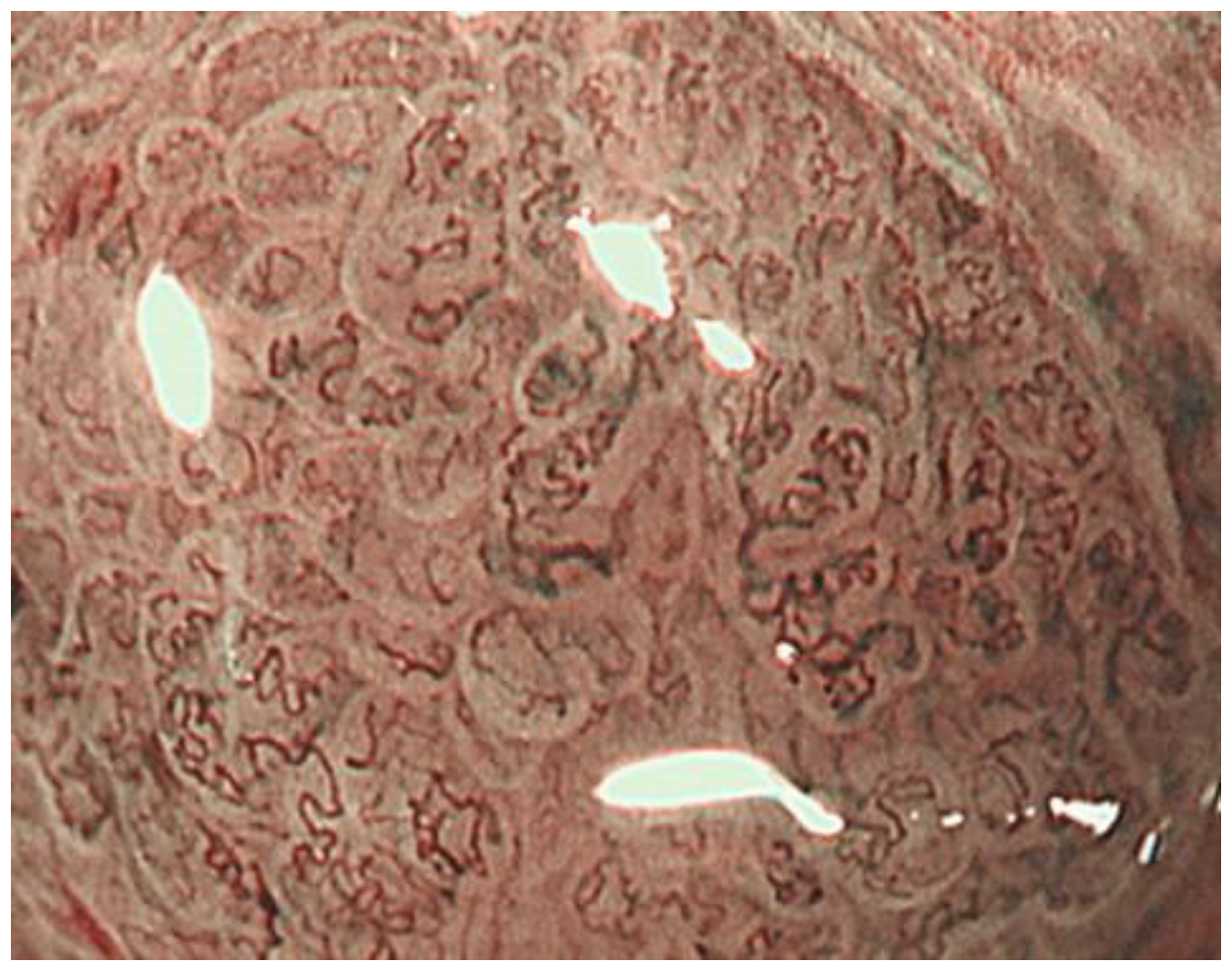
2.1. ME-NBI for Gastric Non-Cancerous Mucosa
2.2. Diagnostic Algorithm for Early Gastric Cancer Using ME-NBI
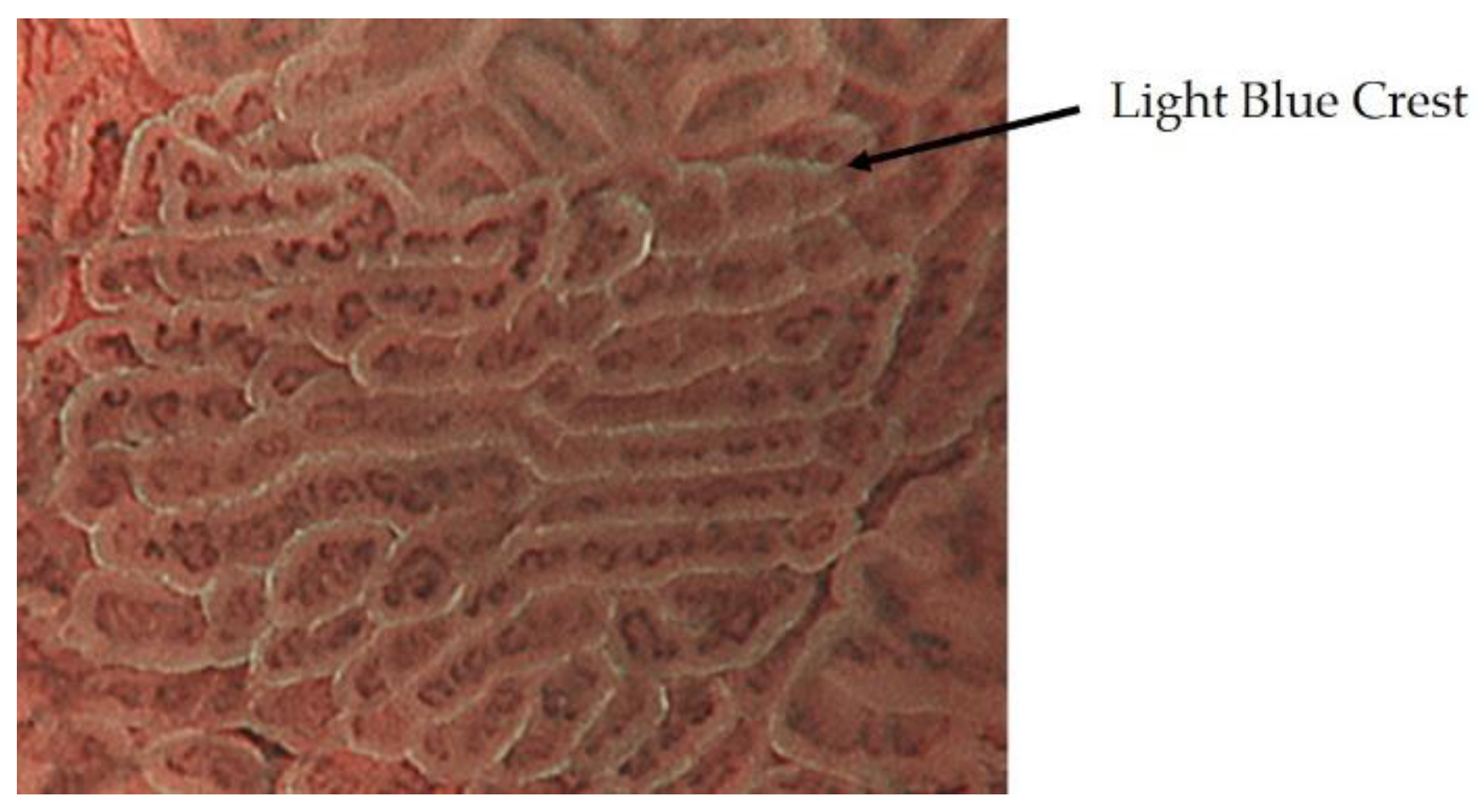
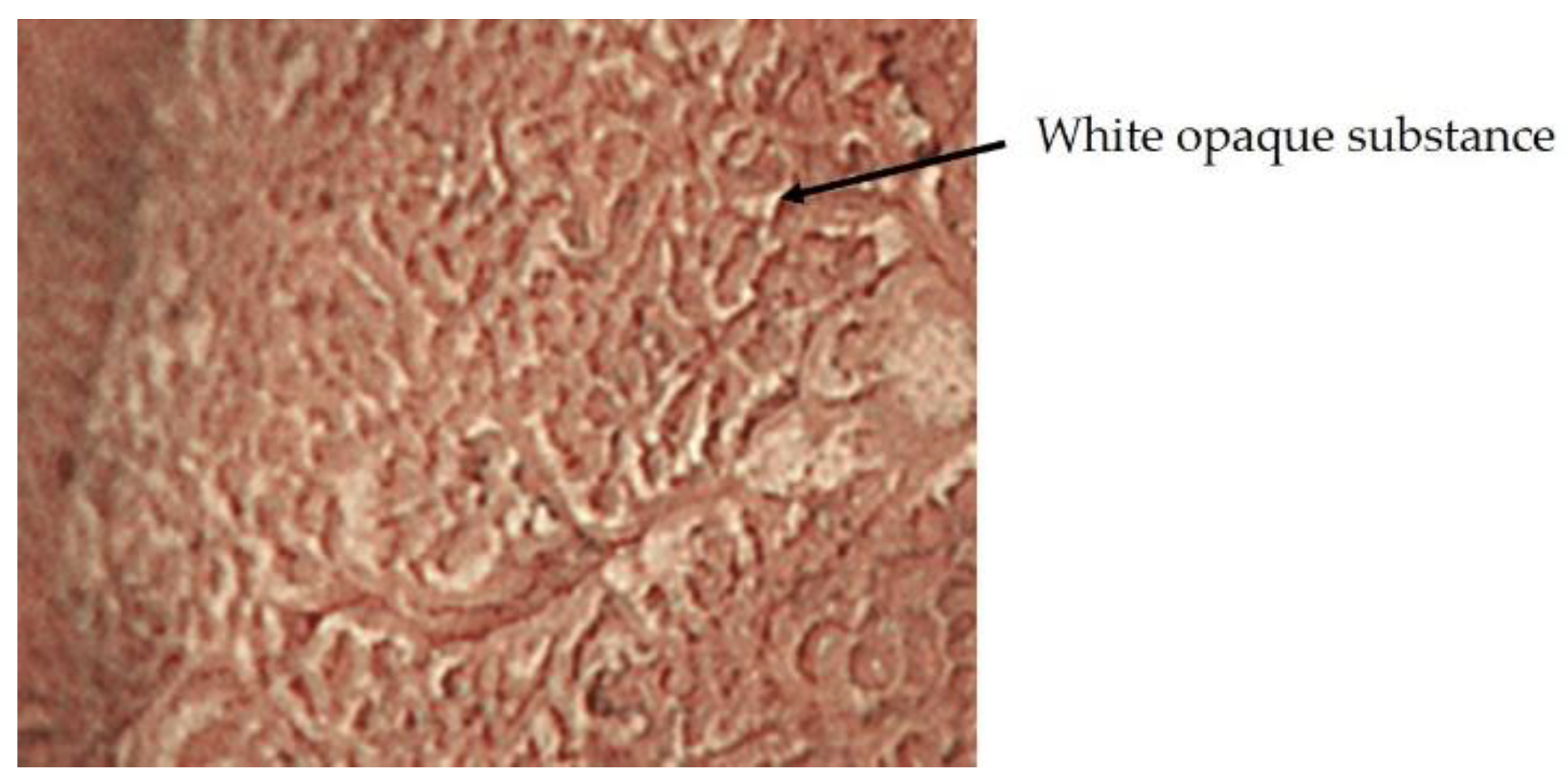
2.3. Inference of Histopathological Type of Gastric Cancer by ME-NBI
2.4. Efficacy of ME-NBI for Early Gastric Cancer
2.4.1. ME-NBI in Screening Tests for Gastric Cancer
2.4.2. ME-NBI in Histological Diagnosis of Gastric Cancer
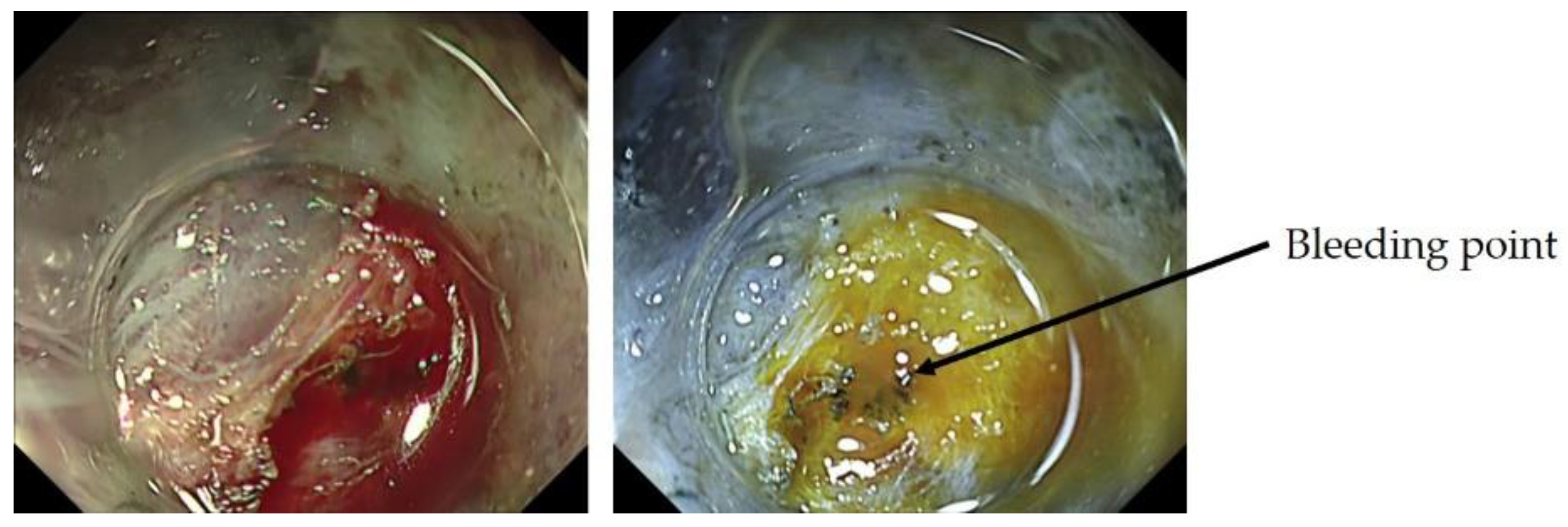
2.4.3. ME-NBI in Determining the Horizontal Extent of Gastric Cancer
2.4.4. ME-NBI in Estimating the Depth of Invasion of Early Gastric Cancer
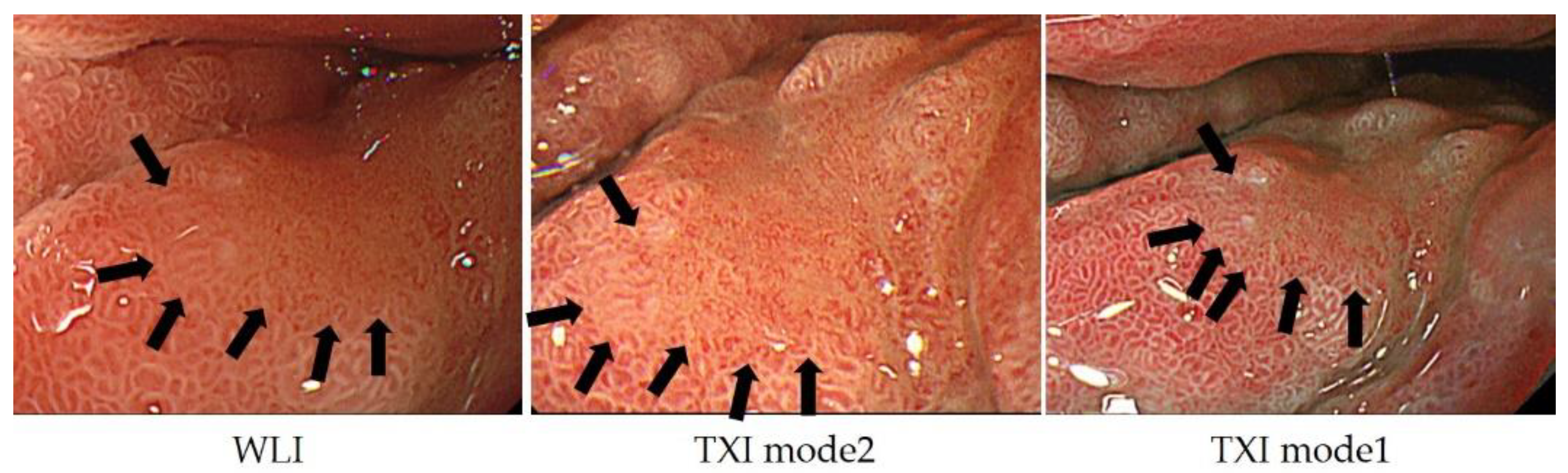
3. Comparison of ME-NBI with Other Modalities
4. Future Prospects
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Isomoto, H.; Shikuwa, S.; Yamaguchi, N.; Fukuda, E.; Ikeda, K.; Nishiyama, H.; Ohnita, K.; Mizuta, Y.; Shiozawa, J.; Kohno, S. Endoscopic submucosal dissection for early gastric cancer: A large-scale feasibility study. Gut 2009, 58, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Emura, F.; Saito, Y.; Ikematsu, H. Narrow-band imaging optical chromocolonoscopy: Advantages and limitations. World J. Gastroenterol. 2008, 14, 4867–4872. [Google Scholar] [CrossRef] [PubMed]
- Arima, M.; Tada, M.; Arima, H. Evaluation of microvascular patterns of superficial esophageal cancers by magnifying endoscopy. Esophagus 2005, 2, 191–197. [Google Scholar] [CrossRef]
- Inoue, H.; Kaga, M.; Ikeda, H.; Sato, C.; Sato, H.; Minami, H.; Santi, E.G.; Hayee, B.; Eleftheriadis, N. Magnification endoscopy in esophageal squamous cell carcinoma: A review of the intrapapillary capillary loop classification. Ann. Gastroenterol. 2015, 28, 41–48. [Google Scholar] [PubMed]
- Oyama, T.; Inoue, H.; Arima, M.; Momma, K.; Omori, T.; Ishihara, R.; Hirasawa, D.; Takeuchi, M.; Tomori, A.; Goda, K. Prediction of the invasion depth of superficial squamous cell carcinoma based on microvessel morphology: Magnifying endoscopic classification of the Japan Esophageal Society. Esophagus 2017, 14, 105–112. [Google Scholar] [CrossRef] [Green Version]
- Morita, F.H.A.; Bernardo, W.M.; Ide, E.; Rocha, R.S.P.; Aquino, J.C.M.; Minata, M.K.; Yamazaki, K.; Marques, S.B.; Sakai, P.; Moura, E.G.H. Narrow band imaging versus lugol chromoendoscopy to diagnose squamous cell carcinoma of the esophagus: A systematic review and meta-analysis. BMC Cancer 2017, 17, 54–68. [Google Scholar] [CrossRef] [Green Version]
- Gruner, M.; Denis, A.; Masliah, C.; Amil, M.; Metivier-Cesbron, E.; Luet, D.; Kaasis, M.; Coron, E.; Le Rhun, M.; Lecleire, S.; et al. Narrow-band imaging versus Lugol chromoendoscopy for esophageal squamous cell cancer screening in normal endoscopic practice: Randomized controlled trial. Endoscopy 2020. [Google Scholar] [CrossRef]
- Yagi, K.; Nozawa, Y.; Endou, S.; Nakamura, A. Diannosis of Early Gastric Cancer by Magnifying Endoscopy with NBI from Viewpoint of Histological Imaging: Mucosal Patterning in terms of White Zone Visibility and Its Relationship to Histology. Diagn. Ther. Endosc. 2012, 2012, 954809. [Google Scholar] [CrossRef]
- Yao, K.; Anagnostopoulos, G.K.; Ragunath, K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy 2009, 41, 462–467. [Google Scholar] [CrossRef] [Green Version]
- Muto, M.; Yao, K.; Kaise, M.; Kato, M.; Uedo, N.; Yagi, K.; Tajiri, H. Magnifying endoscopy simple diagnostic algorithm for early gastric cancer (MESDA-G). Dig. Endosc. 2016, 28, 379–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uedo, N.; Ishihara, R.; Iishi, H.; Yamamoto, S.; Yamamoto, S.; Yamada, T.; Imanaka, K.; Takeuchi, Y.; Higashino, K.; Ishiguro, S.; et al. A new method of diagnosing gastric intestinal metaplasia: Narrow-band imaging with magnifying endoscopy. Endoscopy 2006, 38, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Togo, K.; Ueo, T.; Yao, K.; Wada, K.; Honda, H.; Inoue, S.; Fukuda, M.; Yanai, Y.; Yonemasu, H.; Murakami, K. White opaque substance visualized by magnifying narrow-band imaging is associated with intragastric acid conditions. Endosc. Int. Open 2018, 7, E830–E837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
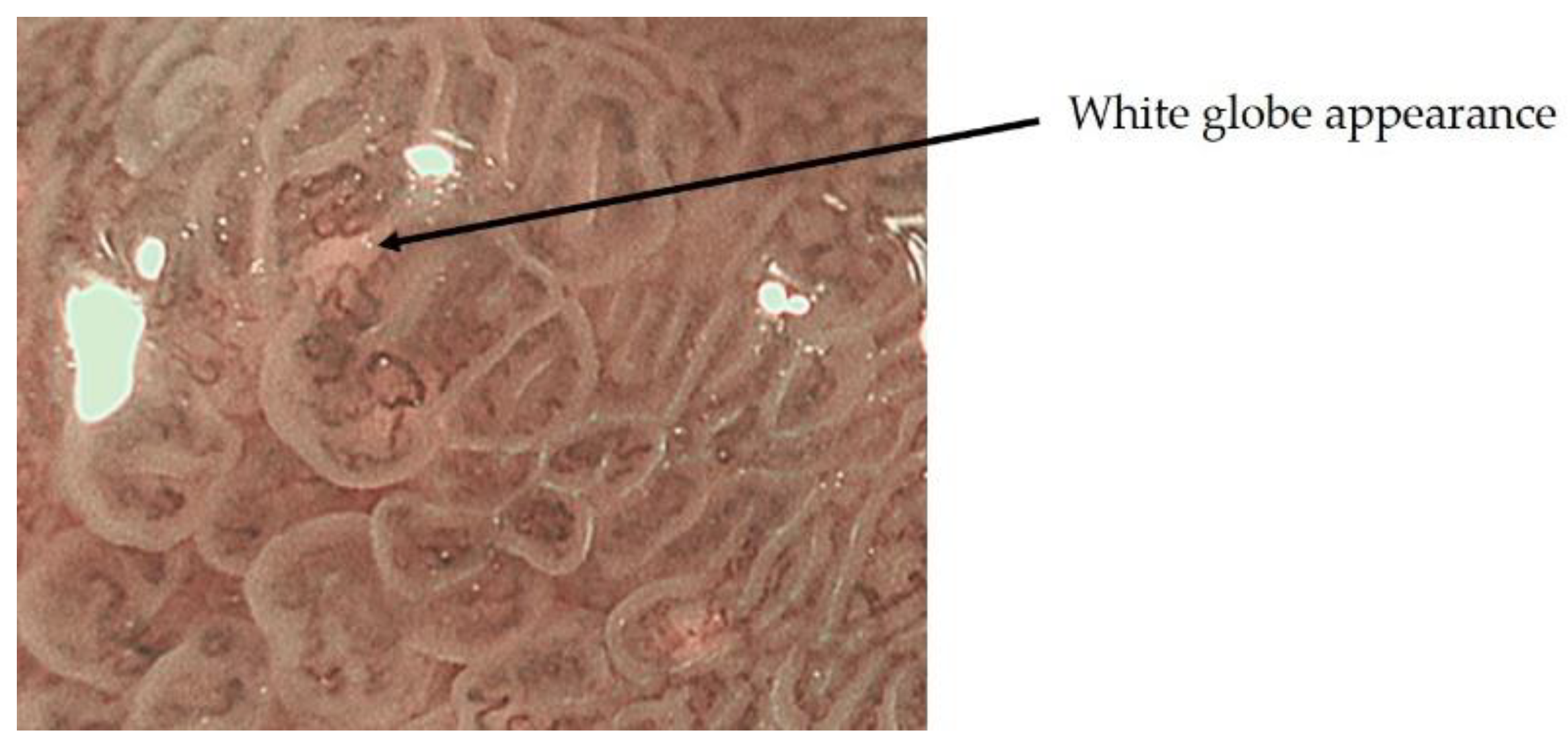
- Yao, K.; Iwashita, A.; Nambu, M.; Tanabe, H.; Nagahama, T.; Maki, S.; Ishikawa, H.; Matsui, T.; Enjoji, M. Nature of white opaque substance in gastric epithelial neoplasia as visualized by magnifying endoscopy with narrow-band imaging. Dig. Endosc. 2012, 24, 419–425. [Google Scholar] [CrossRef]
- Yao, K.; Iwashita, A.; Tanabe, H.; Nishimata, N.; Nagahama, T.; Maki, S.; Takaki, Y.; Hirai, F.; Hisabe, T.; Nishimura, T.; et al. White opaque substance within superficial elevated gastric neoplasia as visualized by magnification endoscopy with narrow-band imaging: A new optical sign for differentiating between adenoma and carcinoma. Gastrointest. Endosc. 2008, 68, 574–580. [Google Scholar] [CrossRef]
- Ueo, T.; Yonemasu, H.; Yao, K.; Ishida, T.; Togo, K.; Yanai, Y.; Fukuda, M.; Motomura, M.; Narita, R.; Murakami, K. Histologic differentiation and mucin phenotype in white opaque substance-positive gastric neoplasias. Endosc. Int. Open 2015, 03, E597–E604. [Google Scholar] [CrossRef] [Green Version]
- Doyama, H.; Yoshida, N.; Tsuyama, S. The “white globe appearance”(WGA): A novel marker for a correct diagnosis of early gastric cancer by magnifying endoscopy with narrowband imaging(MNBI). Gastroenterol. Endosc. 2015, 57, 2577. [Google Scholar] [CrossRef] [Green Version]
- Yagi, K.; Nakamura, A.; Sekine, A.; Umezu, H. Magnifying endoscopy with narrow band imaging for early differentiated gastric adenocarcinoma. Dig. Endosc. 2008, 20, 115–122. [Google Scholar] [CrossRef]
- Nakayoshi, T.; Tajiri, H.; Matsuda, K.; Kaise, M.; Ikegami, M.; Sasaki, H. Magnifying endoscopy combined with narrow band imaging system for early gastric cancer: Correlation of vascular pattern with histopathology (including video). Endoscopy 2004, 36, 1080–1084. [Google Scholar] [CrossRef]
- Kanemitsu, T.; Yao, K.; Nagahama, T.; Fujiwara, S.; Takaki, Y.; Ono, Y.; Matsushima, Y.; Matsui, T.; Tanabe, H.; Ota, A.; et al. The vessels within epithelial circle (VEC) pattern as visualized by magnifying endoscopy with narrow-band imaging (ME-NBI) is a useful marker for the diagnosis of papillary adenocarcinoma: A case-controlled study. Gastric Cancer Off. J. Int. Gastric Cancer Assoc. Jpn. Gastr. Cancer Assoc. 2014, 17, 469–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaibara, N.; Kimura, O.; Nishidoi, H.; Makino, M.; Kawasumi, H.; Koga, S. High incidence of liver metastasis in gastric cancer with medullary growth pattern. J. Surg. Oncol. 1985, 3, 195–198. [Google Scholar] [CrossRef]
- Mita, T.; Shimoda, T. Risk factors for lymph node metastasis of submucosal invasive differentiated type gastric carcinoma: Clinical significance of histological heterogeneity. J. Gastroenterol. 2001, 10, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Uefuji, K.; Ichikura, T.; Tamakuma, S. Clinical and prognostic characteristics of papillary clear cell carcinoma of stomach. Jpn. J. Surg. 1996, 26, 15–163. [Google Scholar]
- Ezoe, Y.; Muto, M.; Uedo, N.; Doyama, H.; Yao, K.; Oda, I.; Kaneko, K.; Kawahara, Y.; Yokoi, C.; Sugiura, Y.; et al. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology 2011, 141, 2017–2025.e3. [Google Scholar] [CrossRef] [Green Version]
- Yao, K.; Doyama, H.; Gotoda, T.; Ishikawa, H.; Nagahama, T.; Yokoi, C.; Oda, I.; Machida, H.; Uchita, K.; Tabuchi, M. Diagnostic performance and limitations of magnifying narrow-band imaging in screening endoscopy of early gastric cancer: A prospective multicenter feasibility study. Gastr. Cancer 2014, 17, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carrasco, M.; Esposito, G.; Libânio, D.; Pimentel-Nunes, P.; Dinis-Ribeiro, M. Image-enhanced endoscopy for gastric preneoplastic conditions and neoplastic lesions: A systematic review and meta-analysis. Endoscopy 2020, 52, 1048–1065. [Google Scholar] [CrossRef]
- Nonaka, K.; Arai, S.; Ban, S.; Kitada, H.; Namoto, M.; Nagata, K.; Ochiai, Y.; Togawa, O.; Nakao, M.; Nishimura, M.; et al. Prospective study of the evaluation of the usefulness of tumor typing by narrow band imaging for the differential diagnosis of gastric adenoma and well-differentiated adenocarcinoma. Dig. Endosc. Off. J. Japan Gastroenterol. Endosc. Soc 2011, 23, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, K. Usefulness of the DL in ME with NBI for determining the expanded area of early-stage differentiated gastric carcinoma. World J. Gastrointest. Endosc. 2012, 4, 362. [Google Scholar] [CrossRef]
- Horii, Y.; Dohi, O.; Naito, Y.; Takayama, S.; Ogita, K.; Terasaki, K.; Nakano, T.; Majima, A.; Yoshida, N.; Kamada, K.; et al. Efficacy of Magnifying Narrow Band Imaging for Delineating Horizontal Margins of Early Gastric Cancer. Digestion 2019, 100, 93–99. [Google Scholar] [CrossRef]
- Horiuchi, Y.; Fujisaki, J.; Yamamoto, N.; Shimizu, T.; Omae, M.; Ishiyama, A.; Yoshio, T.; Hirasawa, T.; Yamamoto, Y.; Tsuchida, T.; et al. Accuracy of diagnostic demarcation of undifferentiated-type early gastric cancer for magnifying endoscopy with narrow-band imaging: Surgical cases. Surg. Endosc. 2017, 31, 1906–1913. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hashimoto, S.; Nishikura, K.; Mizuno, K.; Takeuchi, M.; Sato, Y.; Ajioka, Y.; Aoyagi, Y. Magnifying narrow-band imaging of surface maturation in early differentiated-type gastric cancers after Helicobacter pylori eradication. J. Gastroenterol. 2013, 12, 1332–1342. [Google Scholar] [CrossRef]
- Saka, A.; Yagi, K.; Nimura, S. Endoscopic and histological features of gastric cancers after successful Helicobacter pylori eradication therapy. Gastr. Cancer 2016, 19, 524–530. [Google Scholar] [CrossRef]
- Akazawa, Y.; Ueyama, H.; Yao, T.; Komori, H.; Takeda, T.; Matsumoto, K.; Matsumoto, K.; Asaoka, D.; Hojo, M.; Watanabe, S.; et al. Usefulness of Demarcation of Differentiated-Type Early Gastric Cancers after Helicobacter pylori Eradication by Magnifying Endoscopy with Narrow-Band Imaging. Digestion 2018, 98, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, N.; Tahara, T.; Kawamura, T.; Okubo, M.; Tahara, S.; Nagasaka, M.; Nakagawa, Y.; Shibata, T.; Ohmiya, N. A Comparative Study of White Light Endoscopy, Chromoendoscopy and Magnifying Endoscopy with Narrow Band Imaging in the Diagnosis of Early Gastric Cancer after Helicobacter pylori Eradication. J. Gastrointestin. Liver Dis. 2017, 26, 357–362. [Google Scholar] [CrossRef] [Green Version]
- Kikuchi, D.; Iizuka, T.; Hoteya, S.; Yamada, A.; Furuhata, T.; Yamashita, S.; Domon, K.; Nakamura, M.; Matsui, A.; Mitani, T.; et al. Usefulness of magnifying endoscopy with narrow-band imaging for determining tumor invasion depth in early gastric cancer. Gastroenterol. Res. Pract. 2013. [Google Scholar] [CrossRef]
- Kaneko, K.; Oono, Y.; Yano, T.; Ikematsu, H.; Odagaki, T.; Yoda, Y.; Yagishita, A.; Sato, A.; Nomura, S. Effect of novel bright image enhanced endoscopy using blue laser imaging (BLI). Endosc. Int. Open 2014, 4, E212–E219. [Google Scholar] [CrossRef] [Green Version]
- Le, H.; Wang, L.; Zhang, L.; Chen, P.; Xu, B.; Peng, D.; Yang, M.; Tan, Y.; Cai, C.; Li, H.; et al. Magnifying endoscopy in detecting early gastric cancer. Medicine 2021, 100, e23934. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, H.; Fan, C.; Chen, S.; Liu, A. Comparison of the diagnostic efficacy of blue laser imaging with narrow band imaging for gastric cancer and precancerous lesions: A meta-analysis. Rev. Esp. Enferm. Dig. 2020, 12, 649–658. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, Y.; Shen, Z.; Zhang, X.; Sang, J.; Ding, Y.; Yang, X.; Li, J.; Chen, M.; Jin, C.; et al. Convolutional neural network for the diagnosis of early gastric cancer based on magnifying narrow band imaging. Gastr. Cancer 2020, 23, 126–132. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.; Gong, L.; Dong, D.; Zhu, L.; Wang, M.; He, J.; Shu, L.; Cai, Y.; Cai, S.; Su, W.; et al. Identifying early gastric cancer under magnifying narrow-band images with deep learning: A multicenter study. Gastrointest. Endosc. 2021, 6, 1333–1341. [Google Scholar] [CrossRef]
- Shinozaki, S.; Osawa, H.; Hayashi, Y.; Lefor, A.K.; Yamamoto, H. Linked color imaging for the detection of early gastrointestinal neoplasms. Therap. Adv. Gastroenterol. 2019, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sato, T. TXI: Texture and Color Enhancement Imaging for Endoscopic Image Enhancement. J. Health Care. Eng. 2021, 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yorita, N.; Oka, S.; Tanaka, S.; Kotachi, T.; Nagasaki, N.; Hata, K.; Kuroki, K.; Masuda, K.; Kurihara, M.; Kiso, M.; et al. Clinical Usefulness of Dual Red Imaging in Gastric Endoscopic Submucosal Dissection: A Pilot Study. Clin. Endosc. 2020, 1, 54–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
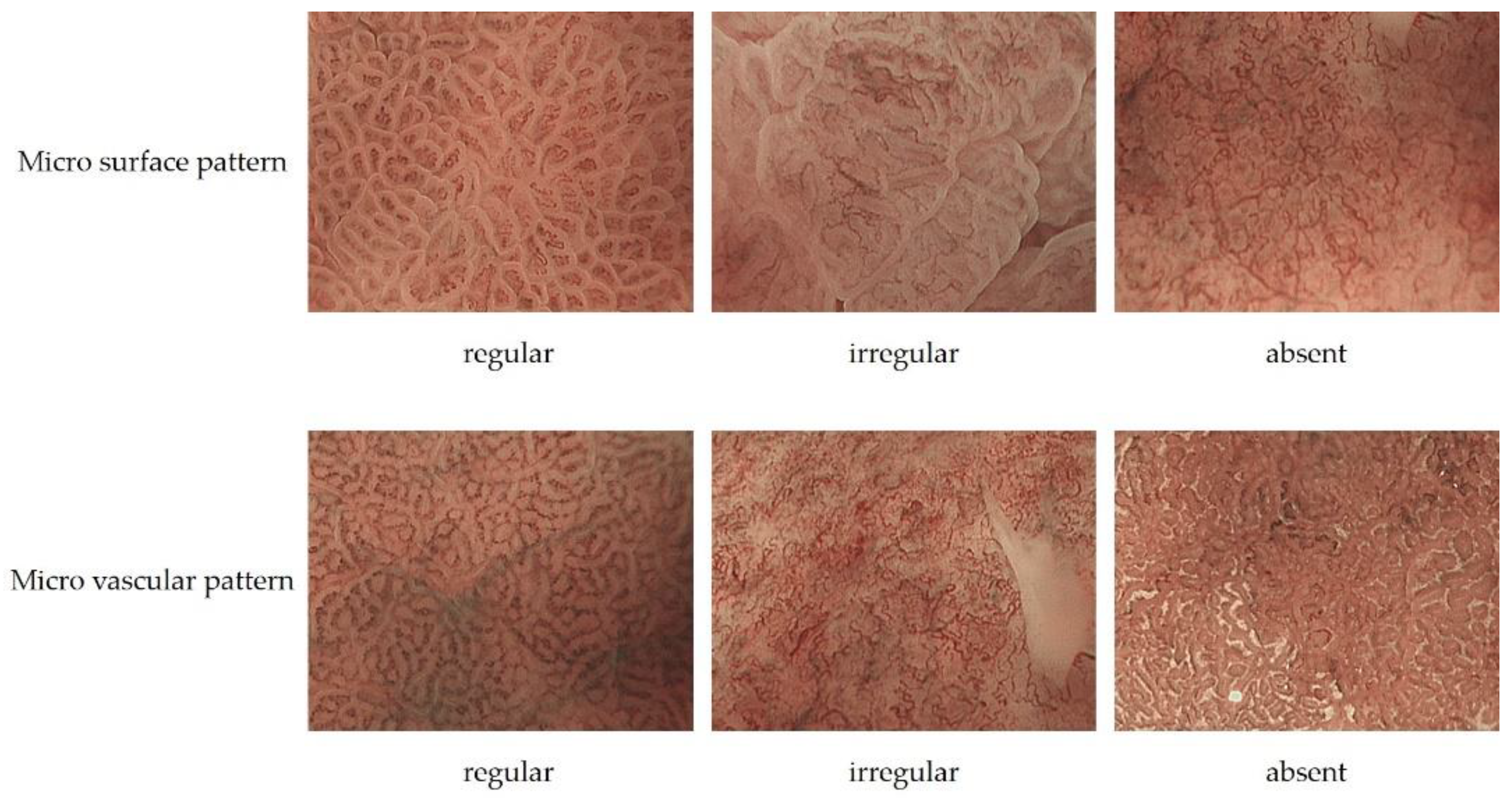
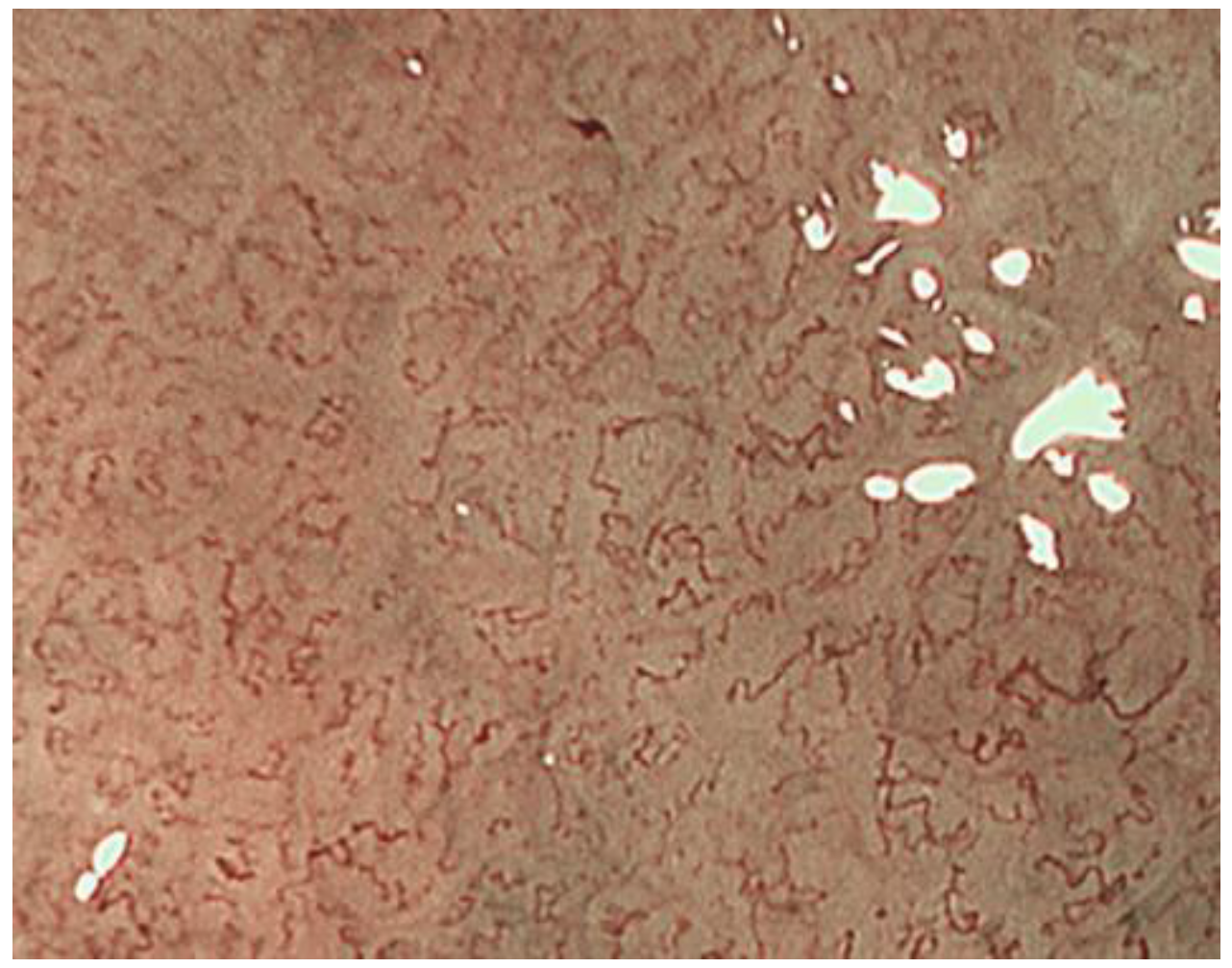
| Non Cancer | Cancer | |
|---|---|---|
| Micro surface pattern | morphology: homogeneous (uniform linear, curved, oval, circular) distribution: symmetric arrangement: regular | morphology: heterogeneous (irregular linear, curved, oval, circular) distribution: asymmetric arrangement: irregular |
| Micro vascular pattern | morphology: uniform distribution: symmetric arrangement: regular | morphology: heterogeneous (dilatation, heterogeneity in shape, abrupt caliber alteration, tortuousness) distribution: asymmetric arrangement: irregular |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurumi, H.; Nonaka, K.; Ikebuchi, Y.; Yoshida, A.; Kawaguchi, K.; Yashima, K.; Isomoto, H. Fundamentals, Diagnostic Capabilities, and Perspective of Narrow Band Imaging for Early Gastric Cancer. J. Clin. Med. 2021, 10, 2918. https://doi.org/10.3390/jcm10132918
Kurumi H, Nonaka K, Ikebuchi Y, Yoshida A, Kawaguchi K, Yashima K, Isomoto H. Fundamentals, Diagnostic Capabilities, and Perspective of Narrow Band Imaging for Early Gastric Cancer. Journal of Clinical Medicine. 2021; 10(13):2918. https://doi.org/10.3390/jcm10132918
Chicago/Turabian StyleKurumi, Hiroki, Kouichi Nonaka, Yuichiro Ikebuchi, Akira Yoshida, Koichiro Kawaguchi, Kazuo Yashima, and Hajime Isomoto. 2021. "Fundamentals, Diagnostic Capabilities, and Perspective of Narrow Band Imaging for Early Gastric Cancer" Journal of Clinical Medicine 10, no. 13: 2918. https://doi.org/10.3390/jcm10132918
APA StyleKurumi, H., Nonaka, K., Ikebuchi, Y., Yoshida, A., Kawaguchi, K., Yashima, K., & Isomoto, H. (2021). Fundamentals, Diagnostic Capabilities, and Perspective of Narrow Band Imaging for Early Gastric Cancer. Journal of Clinical Medicine, 10(13), 2918. https://doi.org/10.3390/jcm10132918








