Corticosteroid Resistance in Smokers—A Substudy Analysis of the CORTICO-COP Randomised Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
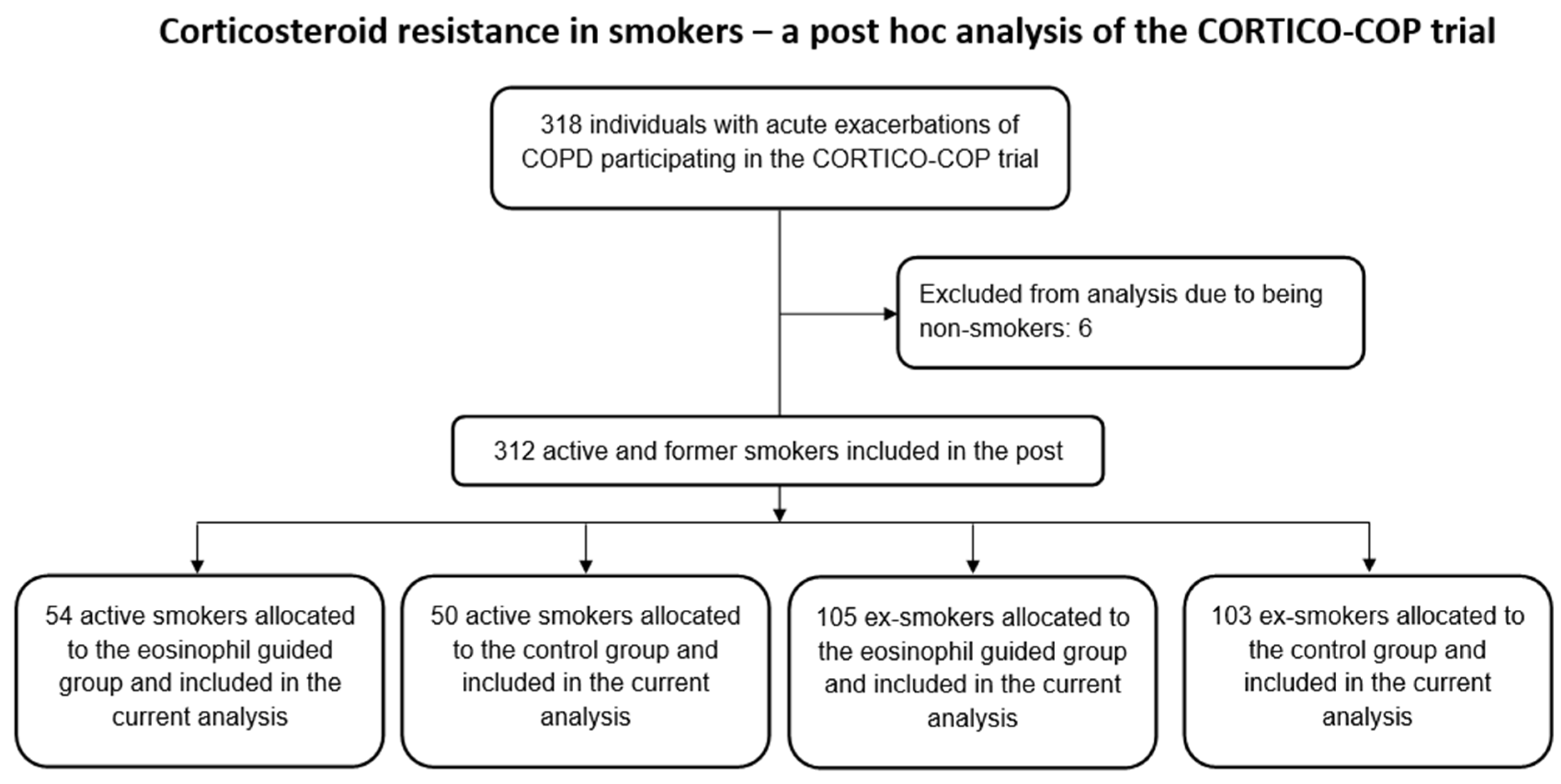
2.2. Study Population
2.3. Statistical Analysis
2.4. Sample Size
3. Results
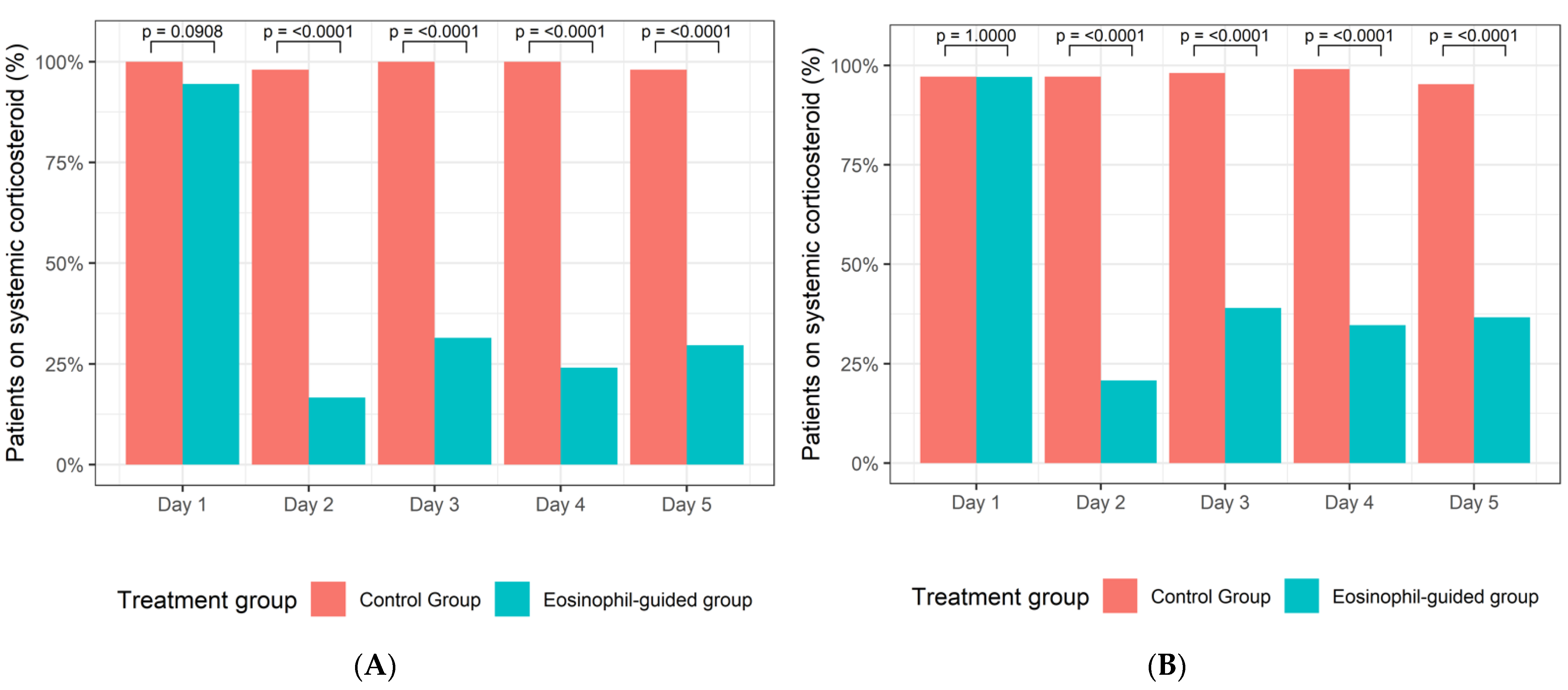
3.1. Primary Outcome
3.2. Secondary Outcomes
3.3. Sensitivity Analysis in Ex-Smokers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, D.; Agusti, A.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Criner, G.J.; Frith, P.; Halpin, D.M.G.; Han, M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease: The GOLD science committee report 2019. Eur. Respir. J. 2019, 53, 1900164. [Google Scholar] [CrossRef]
- Pavord, I.D.; Jones, P.W.; Burgel, P.R.; Rabe, K.F. Exacerbations of COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 21–30. [Google Scholar]
- Global Initiative for Chronic Obstructive Lung Disease. 2021 GOLD Report. 2020. Available online: https://goldcopd.org/wp-content/uploads/2019/12/GOLD-2020-FINAL-ver1.2-03Dec19_WMV.pdf (accessed on 9 April 2021).
- Woods, J.A.; Wheeler, J.S.; Finch, C.K.; Pinner, N.A. Corticosteroids in the treatment of acute exacerbations of chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2014, 9, 421–430. [Google Scholar] [CrossRef] [Green Version]
- Saeed, M.I.; Eklof, J.; Achir, I.; Sivapalan, P.; Meteran, H.; Lokke, A.; Biering-Sorensen, T.; Knop, F.K.; Jensen, J.S. Use of inhaled corticosteroids and the risk of developing type 2 diabetes in patients with chronic obstructive pulmonary disease. Diabetes Obes. Metab. 2020, 22, 1348–1356. [Google Scholar] [CrossRef]
- Waljee, A.K.; Rogers, M.A.; Lin, P.; Singal, A.G.; Stein, J.D.; Marks, R.M.; Ayanian, J.Z.; Nallamothu, B.K. Short term use of oral corticosteroids and related harms among adults in the United States: Population based cohort study. BMJ 2017, 357, j1415. [Google Scholar] [CrossRef] [Green Version]
- Broersen, L.H.; Pereira, A.M.; Jorgensen, J.O.; Dekkers, O.M. Adrenal Insufficiency in Corticosteroids Use: Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2015, 100, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Walsh, L.J.; Wong, C.A.; Oborne, J.; Cooper, S.; Lewis, S.A.; Pringle, M.; Hubbard, R.; Tattersfield, A.E. Adverse effects of oral corticosteroids in relation to dose in patients with lung disease. Thorax 2001, 56, 279–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baker, J.M.; Pace, H.A.; Ladesich, J.B.; Simon, S.D. Evaluation of the Impact of Corticosteroid Dose on the Incidence of Hyperglycemia in Hospitalized Patients with an Acute Exacerbation of Chronic Obstructive Pulmonary Disease. Hosp. Pharm. 2016, 51, 296–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Staa, T.P.; Leufkens, H.G.; Cooper, C. The epidemiology of corticosteroid-induced osteoporosis: A meta-analysis. Osteoporos. Int. 2002, 13, 777–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leuppi, J.D.; Schuetz, P.; Bingisser, R.; Bodmer, M.; Briel, M.; Drescher, T.; Duerring, U.; Henzen, C.; Leibbrandt, Y.; Maier, S.; et al. Short-term vs. conventional glucocorticoid therapy in acute exacerbations of chronic obstructive pulmonary disease: The REDUCE randomized clinical trial. JAMA 2013, 309, 2223–2231. [Google Scholar] [CrossRef] [PubMed]
- Sivapalan, P.; Lapperre, T.S.; Janner, J.; Laub, R.R.; Moberg, M.; Bech, C.S.; Eklof, J.; Holm, F.S.; Armbruster, K.; Sivapalan, P.; et al. Eosinophil-guided corticosteroid therapy in patients admitted to hospital with COPD exacerbation (CORTICO-COP): A multicentre, randomised, controlled, open-label, non-inferiority trial. Lancet Respir. Med. 2019, 7, 699–709. [Google Scholar] [CrossRef]
- Bafadhel, M.; McKenna, S.; Terry, S.; Mistry, V.; Pancholi, M.; Venge, P.; Lomas, D.A.; Barer, M.R.; Johnston, S.L.; Pavord, I.D.; et al. Blood eosinophils to direct corticosteroid treatment of exacerbations of chronic obstructive pulmonary disease: A randomized placebo-controlled trial. Am. J. Respir. Crit. Care. Med. 2012, 186, 48–55. [Google Scholar] [CrossRef] [Green Version]
- Sivapalan, P.; Bikov, A.; Jensen, J.U. Using Blood Eosinophil Count as a Biomarker to Guide Corticosteroid Treatment for Chronic Obstructive Pulmonary Disease. Diagnostics 2021, 11, 236. [Google Scholar] [CrossRef]
- Barnes, P.J. Oxidative stress-based therapeutics in COPD. Redox. Biol. 2020, 33, 101544. [Google Scholar] [CrossRef] [PubMed]
- Roche, N.; Postma, D.S.; Colice, G.; Burden, A.; Guilbert, T.W.; Israel, E.; Martin, R.J.; van Aalderen, W.M.; Grigg, J.; Hillyer, E.V.; et al. Differential effects of inhaled corticosteroids in smokers/ex-smokers and nonsmokers with asthma. Am. J. Respir. Crit. Care Med. 2015, 191, 960–964. [Google Scholar] [CrossRef]
- Chalmers, G.W.; Macleod, K.J.; Little, S.A.; Thomson, L.J.; McSharry, C.P.; Thomson, N.C. Influence of cigarette smoking on inhaled corticosteroid treatment in mild asthma. Thorax 2002, 57, 226–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamimi, A.; Serdarevic, D.; Hanania, N.A. The effects of cigarette smoke on airway inflammation in asthma and COPD: Therapeutic implications. Respir. Med. 2012, 106, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Sohal, S.S.; Reid, D.; Soltani, A.; Weston, S.; Muller, H.K.; Wood-Baker, R.; Walters, E.H. Changes in airway histone deacetylase2 in smokers and COPD with inhaled corticosteroids: A randomized controlled trial. PLoS ONE 2013, 8, e64833. [Google Scholar] [CrossRef] [Green Version]
- Van Overveld, F.J.; Demkow, U.; Gorecka, D.; De Backer, W.A.; Zielinski, J. Differences in responses upon corticosteroid therapy between smoking and non-smoking patients with COPD. J. Physiol. Pharmacol. 2006, 57 (Suppl. 4), 273–282. [Google Scholar] [PubMed]
- Sonnex, K.; Alleemudder, H.; Knaggs, R. Impact of smoking status on the efficacy of inhaled corticosteroids in chronic obstructive pulmonary disease: A systematic review. BMJ Open 2020, 10, e037509. [Google Scholar] [CrossRef] [Green Version]
- Whittaker, H.R.; Jarvis, D.; Sheikh, M.R.; Kiddle, S.J.; Quint, J.K. Inhaled corticosteroids and FEV1 decline in chronic obstructive pulmonary disease: A systematic review. Respir. Res. 2019, 20, 277. [Google Scholar] [CrossRef]
- Pauwels, R.A.; Lofdahl, C.G.; Laitinen, L.A.; Schouten, J.P.; Postma, D.S.; Pride, N.B.; Ohlsson, S.V. Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European Respiratory Society Study on Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 1999, 340, 1948–1953. [Google Scholar] [CrossRef]
- Shaker, S.B.; Dirksen, A.; Ulrik, C.S.; Hestad, M.; Stavngaard, T.; Laursen, L.C.; Maltbaek, N.; Clementsen, P.; Skjaerbaek, N.; Nielsen, L.; et al. The effect of inhaled corticosteroids on the development of emphysema in smokers assessed by annual computed tomography. COPD 2009, 6, 104–111. [Google Scholar] [CrossRef]
- Thompson, W.H.; Nielson, C.P.; Carvalho, P.; Charan, N.B.; Crowley, J.J. Controlled trial of oral prednisone in outpatients with acute COPD exacerbation. Am. J. Respir. Crit. Care Med. 1996, 154, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Zhu, L. Update on molecular mechanisms of corticosteroid resistance in chronic obstructive pulmonary disease. Pulm. Pharmacol. Ther. 2016, 37, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Milara, J.; Diaz-Platas, L.; Contreras, S.; Ribera, P.; Roger, I.; Ballester, B.; Montero, P.; Cogolludo, A.; Morcillo, E.; Cortijo, J. MUC1 deficiency mediates corticosteroid resistance in chronic obstructive pulmonary disease. Respir. Res. 2018, 19, 226. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Corticosteroid resistance in patients with asthma and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2013, 131, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Bafadhel, M.; Davies, L.; Calverley, P.M.; Aaron, S.D.; Brightling, C.E.; Pavord, I.D. Blood eosinophil guided prednisolone therapy for exacerbations of COPD: A further analysis. Eur. Respir. J. 2014, 44, 789–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, D.; Kolsum, U.; Brightling, C.E.; Locantore, N.; Agusti, A.; Tal-Singer, R.; ECLIPSE investigators. Eosinophilic inflammation in COPD: Prevalence and clinical characteristics. Eur. Respir. J. 2014, 44, 1697–1700. [Google Scholar] [CrossRef] [Green Version]
- Csoma, B.; Bikov, A.; Toth, F.; Losonczy, G.; Muller, V.; Lazar, Z. Blood eosinophils on hospital admission for COPD exacerbation do not predict the recurrence of moderate and severe relapses. ERJ Open Res. 2021, 7, 00543-2020. [Google Scholar] [CrossRef]
- Kostikas, K.; Papathanasiou, E.; Papaioannou, A.I.; Bartziokas, K.; Papanikolaou, I.C.; Antonakis, E.; Makou, I.; Hillas, G.; Karampitsakos, T.; Papaioannou, O.; et al. Blood eosinophils as predictor of outcomes in patients hospitalized for COPD exacerbations: A prospective observational study. Biomarkers 2021, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.; Angus, R.M.; Calverley, P.M. Oral corticosteroids in patients admitted to hospital with exacerbations of chronic obstructive pulmonary disease: A prospective randomised controlled trial. Lancet 1999, 354, 456–460. [Google Scholar] [CrossRef]

| All Current Smokers | Current Smokers Control Group | Current Smokers Eosinophil-Guided Group | All Ex-Smokers | Ex-Smokers Control Group | Ex-Smokers Eosinophil-Guided Group | |
|---|---|---|---|---|---|---|
| Number, n (%) | 104 (100) | 50 (100) | 54 (100) | 208 (100) | 105 (100) | 103 (100) |
| Age, median (IQR), years | 72 (66–78) | 73 (66–78) | 70 (66–77) | 76 (71–83) | 76 (71–83) | 77 (72–83) |
| Male sex, n (%) | 47 (45) | 20 (40) | 27 (50) | 94 (45) | 48 (46) | 46 (45) |
| BMI, median (IQR), kg/m2 | 23.2 (20.0–27.7) | 21.9 (19.3–27.5) | 23.7 (20.6–27.5) | 24.2 (20,8–27.4) | 24.4 (20.8–27.9) | 24.2 (20.8–26.3) |
| Pack-years history of current or prior smoker (median, IQR), years | 50 (40–60) | 50 (40–60) | 50 (40–60) | 40 (30–50) | 45 (30–50) | 40 (28–50) |
| Blood eosinophil count, median (IQR), ×109 cells/L | 0.09 (0.01–0.20) | 0.07 (0.01–0.15) | 0.10 (0.01–0.29) | 0.07 (0.01–0.27) | 0.06 (0.01–0.23) | 0.10 (0.01–0.30) |
| FEV1, median (IQR), % | 32 (23–40) | 30 (22–40) | 32 (25–39) | 30 (23–39) | 30 (24–40) | 30 (23–38) |
| CAT scores | 23 (18–27) | 21 (18–27) | 23 (18–28) | 21 (16–25) | 21 (14–25) | 21 (17–25) |
| MRC, median (IQR) | 4 (3–5) | 4 (3–5) | 4 (3–5) | 4 (3–5) | 4 (3–4) | 4 (3–5) |
| MRC 1, n (%) | 3 (3) | 0 (0) | 3 (6) | 5 (2) | 3 (3) | 2 (2) |
| MRC 2, n (%) | 9 (9) | 4 (8) | 5 (10) | 14 (7) | 5 (5) | 9 (9) |
| MRC 3, n (%) | 26 (25) | 16 (32) | 10 (19) | 64 (31) | 34 (33) | 30 (30) |
| MRC 4, n (%) | 30 (29) | 11 (22) | 19 (37) | 69 (34) | 39 (38) | 30 (30) |
| MRC 5, n (%) | 34 (33) | 19 (38) | 15 (29) | 52 (25) | 23 (22) | 29 (29) |
| Treatment with LABA alone, n (%) | 19 (18) | 7 (14) | 12 (22) | 65 (31) | 26 (25) | 39 (38) |
| Treatment with LAMA alone, n (%) | 9 (9) | 4 (8) | 5 (9) | 20 (10) | 15 (14) | 5 (5) |
| Treatment with ICS alone, n (%) | 5 (5) | 2 (4) | 3 (6) | 4 (2) | 1 (1) | 3 (3) |
| Treatment with ICS/LABA, n (%) | 58 (56) | 31 (62) | 27 (50) | 106 (51) | 60 (57) | 46 (45) |
| None of the above, n (%) | 13 (12) | 6 (12) | 7 (13) | 13 (6) | 3 (3) | 10 (10) |
| 0 severe exacerbations 12 months prior to baseline, n (%) | 72 (69) | 35 (70) | 37 (69) | 136 (65) | 68 (65) | 68 (66) |
| 1 severe exacerbation 12 months prior to baseline, n (%) | 17 (16) | 8 (16) | 9 (17) | 41 (20) | 20 (19) | 21 (20) |
| ≥2 severe exacerbations 12 months prior to baseline, n (%) | 15 (14) | 7 (14) | 8 (15) | 31 (15) | 17 (16) | 14 (14) |
| Ischaemic heart disease, n (%) | 15 (14) | 8 (16) | 7 (13) | 21 (10) | 6 (6) | 15 (15) |
| Hypertension, n (%) | 39 (38) | 18 (36) | 21 (39) | 82 (39) | 41 (39) | 41 (40) |
| Hypercholesterolemia, n (%) | 15 (14) | 10 (20) | 5 (9) | 22 (11) | 9 (9) | 13 (13) |
| Chronic renal failure, n (%) | 2 (2) | 2 (4) | 0 (0) | 19 (9) | 7 (7) | 12 (12) |
| Heart failure, n (%) | 6 (6) | 4 (8) | 2 (4) | 24 (12) | 9 (9) | 15 (15) |
| Osteoporosis, n (%) | 18 (17) | 9 (18) | 9 (17) | 39 (19) | 15 (14) | 24 (23) |
| All n = 104 | Control Group n = 54 | Eosinophil-Guided Group n = 50 | p-Value | |
|---|---|---|---|---|
| DAOH14, mean (95% CI) | 9.2 (4.1) | 9.8 (8.7–10.8) | 8.7 (7.5–9.9) | 0.34 |
| Exacerbation or death within 6 months, n (%) | 61 (59) | 30 (60) | 31 (57) | |
| HR (95% CI) | Reference | 0.99 (0.60–1.64) | 0.98 | |
| ICU or death within 6 months, n (%) | 38 (37) | 18 (36) | 20 (37) | |
| OR (95% CI) | Reference | 1.05 (0.47–2.33) | 0.91 | |
| FEV1 pct | ||||
| Baseline, mean (SD) | 33.3 (13,1) | 31.7 (12.3) | 34.7 (13.7) | |
| Day 90, mean (SD) | 42.2(15.7) | 40.0 (15.6) | 44.3 (15.7) | |
| Difference, mean (SD) | 9.1 (11.3) | 8.0 (9.3) | 10.1 (13.0) | 0.46 |
| CAT score | ||||
| Baseline, mean (SD) | 22.3 (7.6) | 22.2 (7.3) | 22.5 (7.9) | |
| Day 90, mean (SD) | 18.0 (7.8) | 19.2 (7.0) | 16.8 (8.4) | |
| Difference, mean (SD) | −4.0 (8.0) | −3.4 (9.4) | −4.5 (6.4) | 0.56 |
| All n = 208 | Control Group n = 105 | Eosinophil-Guided Group n = 103 | p-Value | |
|---|---|---|---|---|
| DAOH14, mean (95% CI) | 9.1 (8.6–9.7) | 9.3 (8.5–10.0) | 9.0 (8.2–9.8) | 0.61 |
| Exacerbation or death within 6 months, n (%) | 104 (50) | 51 (49) | 53 (51) | |
| HR (95% CI) | Reference | 1.17 (0.80–1.72) | 0.43 | |
| ICU or death within 6 months, n (%) | 85 (41) | 41 (39) | 44 (43) | |
| OR (95% CI) | Reference | 1.16 (0.62–1.64) | 0.59 | |
| FEV1 pct. | ||||
| Baseline, mean (SD) | 32.5 (13.2) | 32.3 (12.9) | 32.7 (13.5) | |
| Day 90, mean (SD) | 41.5 (17.5) | 41.2 (15.9) | 41.8 (19.3) | |
| Difference, mean (SD) | 6.0 (12.1) | 6.7 (12.4) | 5.1 (11.9) | 0.48 |
| CAT score | ||||
| Baseline, mean (SD) | 20.6 (7.2) | 20.3 (7.6) | 20.9 (6.7) | |
| Day 90, mean (SD) | 17.5 (7.1) | 17.1 (8.0) | 18.0 (5.6) | |
| Difference, mean (SD) | −3.2 (6.7) | −3.4 (7.0) | −3.0 (6.4) | 0.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sivapalan, P.; Bikov, A.; Suppli Ulrik, C.; Lapperre, T.S.; Mathioudakis, A.G.; Højberg Lassen, M.C.; Grundtvig Skaarup, K.; Biering-Sørensen, T.; Vestbo, J.; Jensen, J.-U.S. Corticosteroid Resistance in Smokers—A Substudy Analysis of the CORTICO-COP Randomised Controlled Trial. J. Clin. Med. 2021, 10, 2734. https://doi.org/10.3390/jcm10122734
Sivapalan P, Bikov A, Suppli Ulrik C, Lapperre TS, Mathioudakis AG, Højberg Lassen MC, Grundtvig Skaarup K, Biering-Sørensen T, Vestbo J, Jensen J-US. Corticosteroid Resistance in Smokers—A Substudy Analysis of the CORTICO-COP Randomised Controlled Trial. Journal of Clinical Medicine. 2021; 10(12):2734. https://doi.org/10.3390/jcm10122734
Chicago/Turabian StyleSivapalan, Pradeesh, Andras Bikov, Charlotte Suppli Ulrik, Therese Sophie Lapperre, Alexander G. Mathioudakis, Mats Christian Højberg Lassen, Kristoffer Grundtvig Skaarup, Tor Biering-Sørensen, Jørgen Vestbo, and Jens-Ulrik S. Jensen. 2021. "Corticosteroid Resistance in Smokers—A Substudy Analysis of the CORTICO-COP Randomised Controlled Trial" Journal of Clinical Medicine 10, no. 12: 2734. https://doi.org/10.3390/jcm10122734
APA StyleSivapalan, P., Bikov, A., Suppli Ulrik, C., Lapperre, T. S., Mathioudakis, A. G., Højberg Lassen, M. C., Grundtvig Skaarup, K., Biering-Sørensen, T., Vestbo, J., & Jensen, J.-U. S. (2021). Corticosteroid Resistance in Smokers—A Substudy Analysis of the CORTICO-COP Randomised Controlled Trial. Journal of Clinical Medicine, 10(12), 2734. https://doi.org/10.3390/jcm10122734










