Physical Activity in Patients with Prader-Willi Syndrome—A Systematic Review of Observational and Interventional Studies
Abstract
1. Introduction
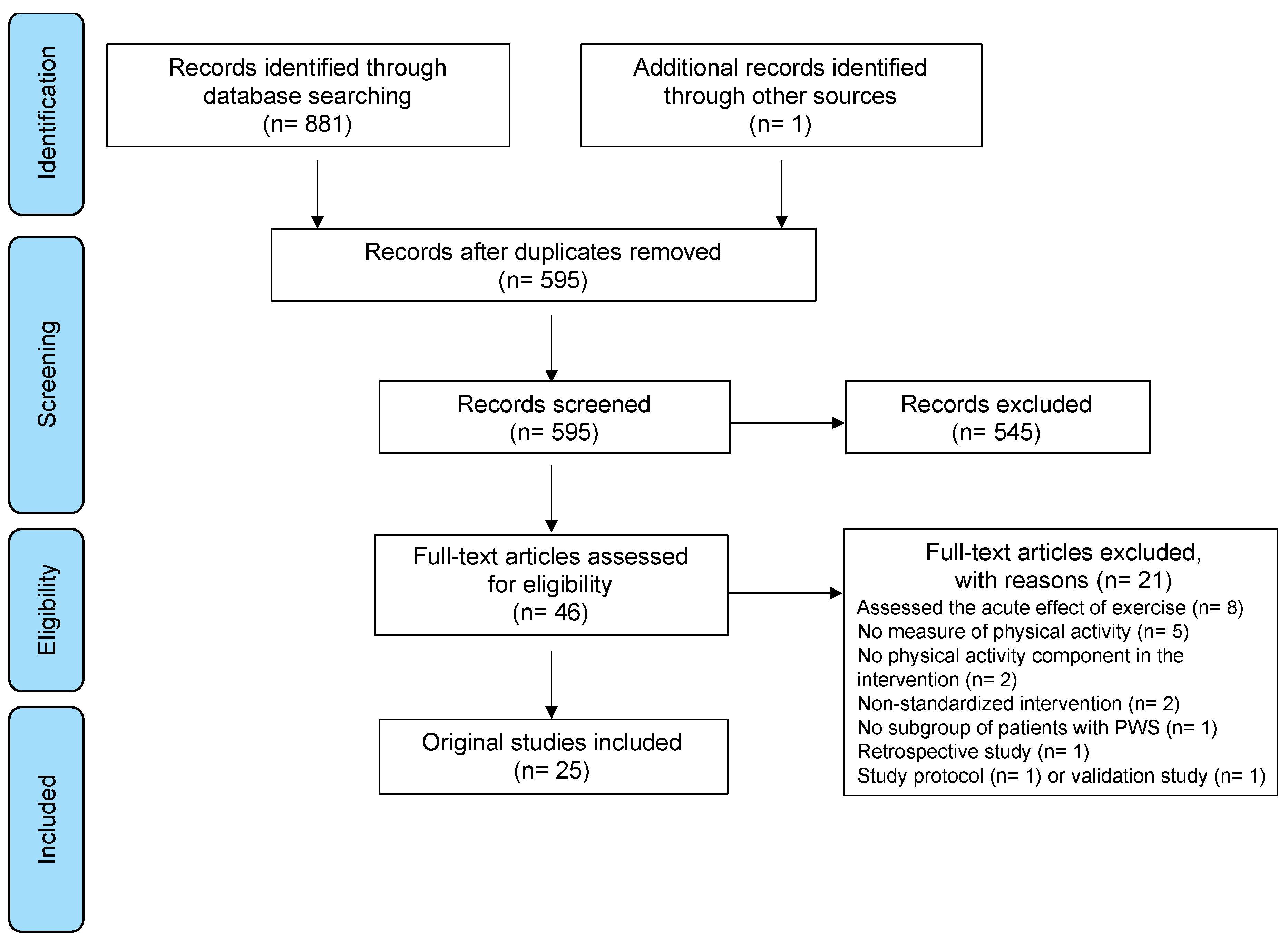
2. Methods
2.1. Search Strategy
2.2. Study Selection, Inclusion, and Exclusion Criteria
2.3. Data Extraction and Synthesis
2.4. Quality Assessment
3. Results
3.1. Study Characteristics and Quality
3.2. Description of Habitual PA and Sedentary Behavior in Patients with PWS
3.3. Relations between Habitual PA and Health Outcomes
3.4. Effectiveness of PA Interventions in Patients with PWS
3.4.1. Description of Interventions
3.4.2. Effect of Interventions on Health Outcomes Status
3.4.3. Implementation of Interventions
- Attendance, satisfaction, enjoyment, adverse events
- Adaptation of intervention
- Supervision
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Driscoll, D.J.; Miller, J.L.; Schwartz, S.; Cassidy, S.B. Prader-Willi Syndrome. In GeneReviews (®); Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Bar, C.; Diene, G.; Molinas, C.; Bieth, E.; Casper, C.; Tauber, M. Early Diagnosis and Care Is Achieved but Should Be Improved in Infants with Prader-Willi Syndrome. Orphanet. J. Rare. Dis. 2017, 12, 118. [Google Scholar] [CrossRef]
- Morales, J.S.; Valenzuela, P.L.; Pareja-Galeano, H.; Rincón-Castanedo, C.; Rubin, D.A.; Lucia, A. Physical Exercise and Prader-Willi Syndrome: A Systematic Review. Clin. Endocrinol. Oxf. 2019, 90, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, S.B.; Schwartz, S.; Miller, J.L.; Driscoll, D.J. Prader-Willi Syndrome. Genet. Med. 2012, 14, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, A.P.; Holland, A.J.; Hauffa, B.P.; Hokken-Koelega, A.C.; Tauber, M. Speakers contributors at the Second Expert Meeting of the Comprehensive Care of Patients with, P.W.S. Recommendations for the Diagnosis and Management of Prader-Willi Syndrome. J. Clin. Endocrinol. Metab. 2008, 93, 4183–4197. [Google Scholar] [CrossRef]
- Castner, D.M.; Tucker, J.M.; Wilson, K.S.; Rubin, D.A. Patterns of Habitual Physical Activity in Youth with and without Prader-Willi Syndrome. Res. Dev. Disabil. 2014, 35, 3081–3088. [Google Scholar] [CrossRef]
- McAlister, K.L.; Fisher, K.L.; Dumont-Driscoll, M.C.; Rubin, D.A. The Relationship between Metabolic Syndrome, Cytokines and Physical Activity in Obese Youth with and without Prader-Willi Syndrome. J. Pediatr. Endocrinol. Metab. 2018, 31, 837–845. [Google Scholar] [CrossRef]
- Bellicha, A.; Coupaye, M.; Hocquaux, L.; Speter, F.; Oppert, J.-M.; Poitou, C. Increasing Physical Activity in Adult Women with Prader-Willi Syndrome: A Transferability Study. J. Appl. Res. Intellect. Disabil. 2020, 33, 258–267. [Google Scholar] [CrossRef]
- Nordstrom, M.; Hansen, B.H.; Paus, B.; Kolset, S.O. Accelerometer-Determined Physical Activity and Walking Capacity in Persons with Down Syndrome, Williams Syndrome and Prader-Willi Syndrome. Res. Dev. Disabil. 2013, 34, 4395–4403. [Google Scholar] [CrossRef] [PubMed]
- Woods, S.G.; Knehans, A.; Arnold, S.; Dionne, C.; Hoffman, L.; Turner, P.; Baldwin, J. The Associations between Diet and Physical Activity with Body Composition and Walking a Timed Distance in Adults with Prader-Willi Syndrome. Food Nutr. Res. 2018, 62, 1343–1354. [Google Scholar] [CrossRef]
- Duran, A.T.; Wilson, K.S.; Castner, D.M.; Tucker, J.M.; Rubin, D.A. Association between Physical Activity and Bone in Children with Prader-Willi Syndrome. J. Pediatr. Endocrinol. Metab. JPEM 2016, 29, 819–826. [Google Scholar] [CrossRef]
- Strath, S.J.; Kaminsky, L.A.; Ainsworth, B.E.; Ekelund, U.; Freedson, P.S.; Gary, R.A.; Richardson, C.R.; Smith, D.T.; Swartz, A.M. American Heart Association Physical Activity Committee of the Council on, L.; et al. Guide to the Assessment of Physical Activity: Clinical and Research Applications: A Scientific Statement from the American Heart Association. Circulation 2013, 128, 2259–2279. [Google Scholar] [CrossRef]
- Shields, N.; Westle, A.; Bennell, K.L.; Taylor, N.F. Physiotherapists Perceived Developing Positive Rapport Facilitates Participation in Exercise among People with Prader-Willi Syndrome: A Qualitative Study. Disabil. Rehabil. 2020, 42, 3475–3489. [Google Scholar] [CrossRef]
- Shields, N.; Bennell, K.L.; Radcliffe, J.; Taylor, N.F. Is Strength Training Feasible for Young People with Prader-Willi Syndrome? A Phase I Randomised Controlled Trial. Physiotherapy 2020, 106, 136–144. [Google Scholar] [CrossRef]
- Rubin, D.A.; Wilson, K.S.; Orsso, C.E.; Gertz, E.R.; Haqq, A.M.; Castner, D.M.; Dumont-Driscoll, M. A 24-Week Physical Activity Intervention Increases Bone Mineral Content without Changes in Bone Markers in Youth with PWS. Genes 2020, 11, 984. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.A.; Wilson, K.S.; Honea, K.E.; Castner, D.M.; McGarrah, J.G.; Rose, D.J.; Dumont-Driscoll, M. An Evaluation of the Implementation of a Parent-Led, Games-Based Physical Activity Intervention: The Active Play at Home Quasi-Randomized Trial. Health Educ. Res. 2019, 34, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Diene, G.; Mimoun, E.; Feigerlova, E.; Caula, S.; Molinas, C.; Grandjean, H.; Tauber, M. French Reference Centre for PWS Endocrine Disorders in Children with Prader-Willi Syndrome--Data from 142 Children of the French Database. Horm. Res. Paediatr. 2010, 74, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. 2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014, 129, S102–S138. [Google Scholar] [CrossRef]
- Vismara, L.; Cimolin, V.; Grugni, G.; Galli, M.; Parisio, C.; Sibilia, O.; Capodaglio, P. Effectiveness of a 6-Month Home-Based Training Program in Prader-Willi Patients. Res. Dev. Disabil. 2010, 31, 1373–1379. [Google Scholar] [CrossRef]
- Eiholzer, U.; Nordmann, Y.; l’Allemand, D.; Schlumpf, M.; Schmid, S.; Kromeyer-Hauschild, K. Improving Body Composition and Physical Activity in Prader-Willi Syndrome. J. Pediatr. 2003, 142, 73–78. [Google Scholar] [CrossRef]
- Rubin, D.A.; Wilson, K.S.; Dumont-Driscoll, M.; Rose, D.J. Effectiveness of a Parent-Led Physical Activity Intervention in Youth with Obesity. Med. Sci. Sports Exerc. 2019, 51, 805–813. [Google Scholar] [CrossRef]
- Schlumpf, M.; Eiholzer, U.; Gygax, M.; Schmid, S.; van der Sluis, I.; l’Allemand, D. A Daily Comprehensive Muscle Training Programme Increases Lean Mass and Spontaneous Activity in Children with Prader-Willi Syndrome after 6 Months. J. Pediatr. Endocrinol. Metab. 2006, 19, 65–74. [Google Scholar] [CrossRef]
- Grolla, E.; Andrighetto, G.; Parmigiani, P.; Hladnik, U.; Ferrari, G.; Bernardelle, R.; Lago, M.D.; Albarello, A.; Baschirotto, G.; Filippi, G.; et al. Specific Treatment of Prader-Willi Syndrome through Cyclical Rehabilitation Programmes. Disabil. Rehabil. 2011, 33, 1837–1847. [Google Scholar] [CrossRef]
- Hsu, W.-L.; Chiu, V.J.-Y.; Chang, W.-H.; Lin, M.-C.; Wei, J.-T.; Tzeng, I.-S. Hand Strength and Dexterity in Patients with Prader-Willi Syndrome: A Pilot Intervention Study. J. Int. Med. Res. 2018, 46, 4669–4677. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.A.; Wilson, K.S.; Castner, D.M.; Dumont-Driscoll, M.C. Changes in Health-Related Outcomes in Youth With Obesity in Response to a Home-Based Parent-Led Physical Activity Program. J. Adolesc. Health 2019. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.A.; Duran, A.T.; Haqq, A.M.; Gertz, E.R.; Dumont-Driscoll, M. Changes in Cardiometabolic Markers in Children with Prader-Willi Syndrome and Nonsyndromic Obesity Following Participation in a Home-Based Physical Activity Intervention. Pediatr. Obes. 2018, 13, 734–743. [Google Scholar] [CrossRef]
- Capodaglio, P.; Cimolin, V.; Vismara, L.; Grugni, G.; Parisio, C.; Sibilia, O.; Galli, M. Postural Adaptations to Long-Term Training in Prader-Willi Patients. J. Neuroeng. Rehabil. 2011, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Borland, R.L.; Hu, N.; Tonge, B.; Einfeld, S.; Gray, K.M. Participation in Sport and Physical Activity in Adults with Intellectual Disabilities. J. Intellect. Disabil. Res. 2020, 64, 908–922. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.G.; Theodoro, M.F.; Bittel, D.C.; Donnelly, J.E. Energy Expenditure and Physical Activity in Prader-Willi Syndrome: Comparison with Obese Subjects. Am. J. Med. Genet. A 2007, 143A, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Sellinger, M.H.; Hodapp, R.M.; Dykens, E.M. Leisure Activities of Individuals with Prader-Willi, Williams, and Down Syndromes. J. Dev. Phys. Disabil. 2006, 18, 59–71. [Google Scholar] [CrossRef]
- van den Berg-Emons, R.; Festen, D.; Hokken-Koelega, A.; Bussmann, J.; Stam, H. Everyday Physical Activity and Adiposity in Prader-Willi Syndrome. J. Pediatr. Endocrinol. Metab. 2008, 21, 1041–1048. [Google Scholar] [CrossRef]
- van Mil, E.; Westerterp, K.R.; Kester, A.D.M.; Curfs, L.M.G.; Gerver, W.J.M.; Schrander-Stumpel, C.; Saris, W.H.M. Activity Related Energy Expenditure in Children and Adolescents with Prader-Willi Syndrome. Int. J. Obes. 2000, 24, 429–434. [Google Scholar] [CrossRef] [PubMed]
- van Mil, E.G.; Westerterp, K.R.; Gerver, W.J.; Van Marken Lichtenbelt, W.D.; Kester, A.D.; Saris, W.H. Body Composition in Prader-Willi Syndrome Compared with Nonsyndromal Obesity: Relationship to Physical Activity and Growth Hormone Function. J. Pediatr. 2001, 139, 708–714. [Google Scholar] [CrossRef] [PubMed][Green Version]
- WHO. WHO Guidelines on Physical Activity and Sedentary Behavior; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Diaz, K.M.; Duran, A.T.; Colabianchi, N.; Judd, S.E.; Howard, V.J.; Hooker, S.P. Potential Effects on Mortality of Replacing Sedentary Time With Short Sedentary Bouts or Physical Activity: A National Cohort Study. Am. J. Epidemiol. 2019, 188, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Ekelund, U.; Tarp, J.; Fagerland, M.W.; Johannessen, J.S.; Hansen, B.H.; Jefferis, B.J.; Whincup, P.H.; Diaz, K.M.; Hooker, S.; Howard, V.J.; et al. Joint Associations of Accelero-Meter Measured Physical Activity and Sedentary Time with All-Cause Mortality: A Harmonised Meta-Analysis in More than 44 000 Middle-Aged and Older Individuals. Br. J. Sports Med. 2020, 54, 1499–1506. [Google Scholar] [CrossRef]
- Bellicha, A.; Van Baak, M.A.; Battista, F.; Beaulieu, K.; Blundell, J.E.; Busetto, L.; Carraça, E.V.; Dicker, D.; Encantado, J.; Ermolao, A.; et al. Effect of Exercise Training on Weight Loss, Body Composition Changes and Weight Maintenance in Adults with Overweight or Obesity: An Overview of 12 Systematic Reviews Ans 149 Studies. Obes. Rev. 2021, e13256. [Google Scholar] [CrossRef]
- Shaw, K.; Gennat, H.; O’Rourke, P. Del Mar C Exercise for Overweight or Obesity. Cochrane Database Syst. Rev. 2006, CD003817. [Google Scholar] [CrossRef]
- Alves, C.; Franco, R.R. Prader-Willi Syndrome: Endocrine Manifestations and Management. Arch. Endocrinol. Metab. 2020, 64, 223–234. [Google Scholar] [CrossRef]
- Coupaye, M.; Lorenzini, F.; Lloret-Linares, C.; Molinas, C.; Pinto, G.; Diene, G.; Mimoun, E.; Demeer, G.; Labrousse, F.; Jauregi, J.; et al. Growth Hormone Therapy for Children and Adolescents with Prader-Willi Syndrome Is Associated with Improved Body Composition and Metabolic Status in Adulthood. J. Clin. Endocrinol. Metab. 2013, 98, E328–E335. [Google Scholar] [CrossRef]
| Observational Studies | ||||
| Reference (Country) | Study Design and Quality | Characteristics of Patients with PWS | Characteristics of the Control Group | Outcomes of Interest (Methods) |
| Borland 2020 [28] (Australia) | Comparative cross-sectional study | Adults with PWS (N = 30, 11 F + 19 M) Data are mean (SD) (min–max) Age, year: 30.9 (7.8) (18–46) GHRT: not reported | Adults with Down Syndrome (N = 64, 39 F + 25 M) Age, year: 27.9 (4.5) (20–36) General population (N = 316, 133 F + 183) Age, year: 28.8 (4.5) (19–39) |
|
| Butler 2007 [29] (USA) | Comparative cross-sectional study | Children/adults with PWS (N = 48, 27 F + 21 M) Data are mean (SD) (min–max) Age, year: 23 (9) (10–45) BMI, kg/m2: 34 (9) Body fat, %: 51 (8) Deletion, n (%): 27 (56%) Uniparental disomy: 21 (44%) Type 2 diabetes, n (%): 8 (17%) Not currently on GHRT (N = 48) | Children/adults with NSO (N = 24, 15 F + 9 M) Age, year: 27 (13) (11–49) BMI, kg/m2: 41 (8) Body fat, %: 50 (7) Type 2 diabetes, n (%): 3 (13%) |
|
| Castner 2014 [6] (USA) | Comparative cross-sectional study | Children with PWS (N = 24, 12 F + 12 M) Data are mean (SD) (min–max) Age, year: 11.2 (2.3) (8–16) BMI, kg/m2: 29.4 (12.7) Body fat, %: 45.8 (11.0) Deletion, n (%): 10 (42%) Uniparental disomy: 3 (13%) Imprinting defect: 3 (13%) Unknown subtype, n (%): 8 (33%) Type 2 diabetes: 1 (4.2%) Currently on GHRT (N = 15), had previously been on GHRT (N = 6) | Children with NSO (N = 40, 19 F + 21 M) Age, year: 9.8 (1.1) (8–11) BMI, kg/m2: 27.3 (4.0) Body fat, %: 44.1 (5.7) |
|
| Duran 2016 [11] (USA) | Cross-sectional study | Children with PWS (N = 23, 12 F + 11 M) Data are mean (SD) (min–max) Age, year: 11.0 (2.0) (8–14) BMI, kg/m2: 29.4 (13.0) Body fat, %: 45.9 (11.2) Deletion, n (%): 10 (43%) Uniparental disomy, n (%): 3 (13%) Unknown subtype, n (%): 10 (43%) Currently on GHRT (N = 15), had previously been on GHRT (N = 5), had never been on GHRT (N = 3) | -- |
|
| McAlister 2018 [7] (USA) | Comparative cross-sectional study | Children with PWS (N = 21, 12 F + 9 M) Data are mean (SD) (min–max) Age, year: 10.7 (2.6) (8–15) BMI, kg/m2: 28.2 (10.0) Body fat, %: 46.0 (8.9) Deletion, n (%): 9 (43%) Uniparental disomy, n (%): 5 (24%) Unknown subtype, n (%): 7 (33%) Currently on GHRT (N = 16) | Children with NSO (N = 34, 17 F + 17 M) Age, year: 9.6 (1.0) (8–15) BMI, kg/m2: 29.0 (5.1) Body fat, %: 45.4 (6.4) |
|
| Nordstrom 2013 [9] (Norway) | Comparative cross-sectional study | Adults with PWS (N = 22, 13 F + 9 M) Data are mean (SD) Age, year: 28.1 (7.5) BMI, kg/m2: 30.7 (6.2) Deletion, n (%): 15 (68%) Uniparental disomy, n (%): 5 (23%) Unknown subtype, n (%): 1 (5%) Non-genetically confirmed: 1 (5%) GHRT: not reported | Adults with Down Syndrome (N = 40, 25 F + 15 M) Age, year: 26.8 (7.5) BMI, kg/m2: 31.8 (6.5) Adults with Williams syndrome (N = 25, 16 F + 9 M) Age, y: 31.5 (6.2) BMI, kg/m2: 26.6 (6.5) |
|
| Sellinger 2006 [30] (USA) | Comparative cross-sectional study | Children/adults with PWS (N = 29, 11 F + 18 M) Data are mean (SD) Age, year: 16.8 (7.0) GHRT: not reported | Children/adults with Down Syndrome (N = 104, 38 F + 66 M) Age, year: 17.0 (9.9) Children/adults with Williams Syndrome (N= 90, 48 F + 42 M) Age, y: 14.2 (9.6) |
|
| van den Berg-Emons 2008 [31] (the Netherlands) | Comparative cross-sectional study | Children with PWS (N = 12, 7 F + 5 M) Data are mean (SD) Age, year: 11.4 (2.4) (7–16) Body fat, %: 46.4 (6.7) (30.1–52.5) All children were enrolled in a trial assessing the effect of GHRT | Children without obesity (N = 12, 7 F + 5 M) Age, year: 11.1 (2.1) [8,9,10,11,12,13,14,15,16] |
|
| Van Mil 2000 [32](the Netherlands) | Comparative cross-sectional study | Children with PWS (N = 17, 10 F + 7 M) Data are mean (SD) (min–max) Age, year: 11.9 (3.4) (7–19) BMI, kg/m2: 23.5 (6.0) (15.2–38.1) Body fat, %: 43.7 (7.9) (29.4–59.5) Had never been on GHRT (N = 17) | Children with NSO (N = 17, 10 F + 7 M) Age, year: 11.3 (2.6) (6–15) BMI, kg/m2: 26.0 (6.5) (13.5–39.4) Body fat, %: 39.1 (8.8) (16.3–46.7) |
|
| Van Mil 2001 [33] (the Netherlands) | Same design as [32] | Same participants as [32] Had never been on GHRT (N = 17) | Same intervention as [32] |
|
| Woods 2018 [10] (USA) | Cross-sectional study | Adults with PWS (N = 19, 8 F + 11 M) Data are mean (SEM) (min–max) Age, year: 34.5 (4.3) (18–62) BMI, kg/m2: 26.7 (1.3) (19.5–35.0) Body fat, %: 26.8 (1.7) (16.6–41.9) GHRT: not reported | -- |
|
| Intervention Studies | ||||
| Reference (Country) | Study Design and Quality | Characteristics of Patients with PWS Participating to the PA Intervention | Description of Intervention | Outcomes (Method) |
| Bellicha 2020 [8] (France) | Single-group intervention (control group for baseline measures) Study quality: fair | Adults with PWS (N = 10 F) Data are median (P25–P75) (min–max) Age, year: 28.8 (24.2; 33.0) (19–48) BMI, kg/m2: 37.2 (34.3; 45.8) (31.8–52.8) Body fat, %: 51.9 (49.2; 54.7) (41.1–62.4) Deletion: 9 (90%), Uniparental disomy: 1 (10%) Type 2 diabetes: 2 (20%) Had previously been on GHRT (N = 4), had never been on GHRT (N = 6) |
|
|
| Eiholzer 2003 [20] (Switzerland) | NRCT Study quality: poor | Children with PWS (N = 17, 8 F + 9 M) Data are mean (min–max) Age, year: 10.5 (4–18) Currently or previously on GHRT for at least 3 years (N = 17) |
|
|
| Grolla 2011 [23] (Italy) | Single-group intervention Study quality: poor | Adolescents/adults with PWS (N = 49, 21 F + 28 M) Data are mean (SEM) (min–max) Age, year: 23.7 (1.0) (13–42) BMI, kg/m2: 38.7 (1.4) (21.7–58.7) Deletion, n (%): 33 (67%)/ Uniparental disomy, n (%): 6 (12%)/ Unknown subtype, n (%): 10 (20%) GHRT: not reported |
|
|
| Hsu 2018 [24] (Taiwan) | Single-group intervention Study quality: fair | Adults with PWS (N = 6, 2 F + 4 M) Data are mean (SD) (min–max) Age, year: 26.1 (5.0) (20–32) BMI, kg/m2: mean not reported (20.7–38.4) Previously been on GHRT (N = 6) |
|
|
| Rubin, 2019 [21] (USA) | NRCT Study quality: good | Children with PWS (N = 34, 12 F + 22 M) Data are mean (SD) Age, year: 10.8 (2.5) Body fat, %: 45.9 (10.1) GHRT: not reported |
|
|
| Rubin 2019 [25] (USA) | Same design as [21] | Same participants as [21] Currently on GHRT (N = 33), had previously been on GHRT (N = 9), had never been on GHRT (N = 2) |
|
|
| Rubin 2018 [26] (USA) | Subsample of participants included in [21] Children with PWS (N = 18, 8 F + 10 M) Data are mean (SE) (min–max) Age, year: 10.5 (0.7) (8–16) Body fat, %: 44.6 (2.0) (26.2–55.2) Deletion, n (%): 7 (39%) Uniparental disomy, n (%): 5 (28%) Unknown subtype, n (%): 6 (33%) Currently on GHRT (N = 15), had previously been on GHRT (N = 3) |
|
| |
| Rubin 2020 [15] (USA) | Same design as [21] | Same participants as [21] Currently on GHRT (N = 33), had previously been on GHRT (N = 9), had never been on GHRT (N = 2) |
|
|
| Rubin 2019 [16] (USA) | Same design as [21] | Same participants as [21] GHRT: not reported |
|
|
| Schlumpf 2006 [22] (Switzerland) | NRCT Study quality: poor | Children with PWS (N = 7, 2 F + 5 M) Data are mean (SD) (min–max) Age, year: 8.9 (2.1) Currently or previously on GHRT (N = 7) |
|
|
| Shields 2020 [14] (Australia) | RCT Study quality: good | Children and adults with PWS (N = 16, 8 F + 8 M) Data are mean (SD) (min–max) Age, year: 25.0 (10.0) (13–39) BMI, kg/m2: 35.4 (9.4) (20.6–48.7) Deletion, n (%): 11 (69%) Uniparental disomy, n (%): 4 (25%) Unknown subtype, n (%): 1 (6%) Type 2 diabetes: 5 (31%) Currently on GHRT (N = 2) |
|
|
| Shields 2020 [13] (Australia) | Same design as [14] | Same participants as [14] Currently on GHRT (N = 2) |
|
|
| Vismara 2010 [19] (Italy) | NRCT (control group of healthy subjects for baseline measures) Study quality: fair | Adults with PWS (N = 11, 6 F + 5 M) Data are mean (SD) Age, year: 33.8 (4.3) BMI, kg/m2: 43.3 (5.9) Deletion: 10 (91%) Uniparental disomy: 1 (9%) GHRT: not reported |
|
|
| Capodaglio 2011 [27] (Italy) | Same design as [19] | Same participants as [19] GHRT: not reported |
|
|
| Reference—Population | PA Volume | LPA | MVPA | Sedentary Time | Meet PA Guidelines |
|---|---|---|---|---|---|
| Compared to patients with normal weight | |||||
| Eiholzer 2003 [20]—Children | (−) | ||||
| van den Berg-Emons 2008 [31]—Children | (−) | ||||
| Compared to patients with non-syndromic obesity | |||||
| Bellicha 2020 [8]—Adults | (−) | (−) | ns* | (+) | ns |
| Butler 2007 [29]—Children/adults | (−) | ||||
| Castner 2014 [6]—Children | (−) | ns* | ns | ||
| McAlister 2018 [7]—Children | (−) | ns | |||
| Rubin, 2019 [26]—Children | (−) | ns* | |||
| Van Mil 2000 [32]—Children | (−) | ||||
| Compared to patients with another neurodevelopmental disorder (i.e., Down Syndrome, Williams Syndrome) | |||||
| Nordstrom 2013 [9]—Adults | ns* | (−) | ns | (+) | |
| Reference—Population | Body Weight Fat Mass | LBM | Bone Parameters | Habitual PA | Habitual Sedentary Time | Physical Function | Cardio-Metabolic Markers | QOL |
|---|---|---|---|---|---|---|---|---|
| PA programs | ||||||||
| Bellicha 2020 [8]—Adults | ns | ns | (+) | ns | (+) | ns | ||
| Eiholzer 2003 [20]—Children | (+) | (+) | (+) | |||||
| Hsu 2018 [24]—Adults | (+) | |||||||
| Rubin, 2019 [21]—Children | ns | ns | (+) | |||||
| Rubin 2018 [26]—Children | ns | (+) | ns | ns | ||||
| Rubin 2019 [25]—Children | ns | ns | ns | ns | ||||
| Rubin 2020 [15]—Children | (+) | |||||||
| Schlumpf 2006 [22]—Children | ns | (+) | (+) | |||||
| Shields 2020 [14]—Children/adults | ns | ns | ns | |||||
| Combined PA program and dietary intervention | ||||||||
| Grolla 2011 [23]—Children/adults | (−) | (−) | ||||||
| Vismara 2010 [19]—Adults | (+) | |||||||
| Capodaglio 2011 [27]—Adults | ns | ns | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellicha, A.; Coupaye, M.; Mosbah, H.; Tauber, M.; Oppert, J.-M.; Poitou, C. Physical Activity in Patients with Prader-Willi Syndrome—A Systematic Review of Observational and Interventional Studies. J. Clin. Med. 2021, 10, 2528. https://doi.org/10.3390/jcm10112528
Bellicha A, Coupaye M, Mosbah H, Tauber M, Oppert J-M, Poitou C. Physical Activity in Patients with Prader-Willi Syndrome—A Systematic Review of Observational and Interventional Studies. Journal of Clinical Medicine. 2021; 10(11):2528. https://doi.org/10.3390/jcm10112528
Chicago/Turabian StyleBellicha, Alice, Muriel Coupaye, Héléna Mosbah, Maithé Tauber, Jean-Michel Oppert, and Christine Poitou. 2021. "Physical Activity in Patients with Prader-Willi Syndrome—A Systematic Review of Observational and Interventional Studies" Journal of Clinical Medicine 10, no. 11: 2528. https://doi.org/10.3390/jcm10112528
APA StyleBellicha, A., Coupaye, M., Mosbah, H., Tauber, M., Oppert, J.-M., & Poitou, C. (2021). Physical Activity in Patients with Prader-Willi Syndrome—A Systematic Review of Observational and Interventional Studies. Journal of Clinical Medicine, 10(11), 2528. https://doi.org/10.3390/jcm10112528






