Estimation of the Actual Incidence of Coronavirus Disease (COVID-19) in Emergent Hotspots: The Example of Hokkaido, Japan during February–March 2020
Abstract
1. Introduction
2. Methods
2.1. Epidemiological Data
2.2. Modeling Commuter Movement between Subprefectures of Hokkaido
2.3. Estimating Actual Incidence of COVID-19 Cases in Hokkaido Prefecture and Sapporo City
- (i)
- Estimating incidence in Hokkaido by using imported cases among travelers outbound from Hokkaido
- (ii)
- Estimating incidence in Sapporo using cases imported within Hokkaido
2.4. Reporting Delay between Illness Onset and Confirmation
2.5. Simulation Platform
3. Results
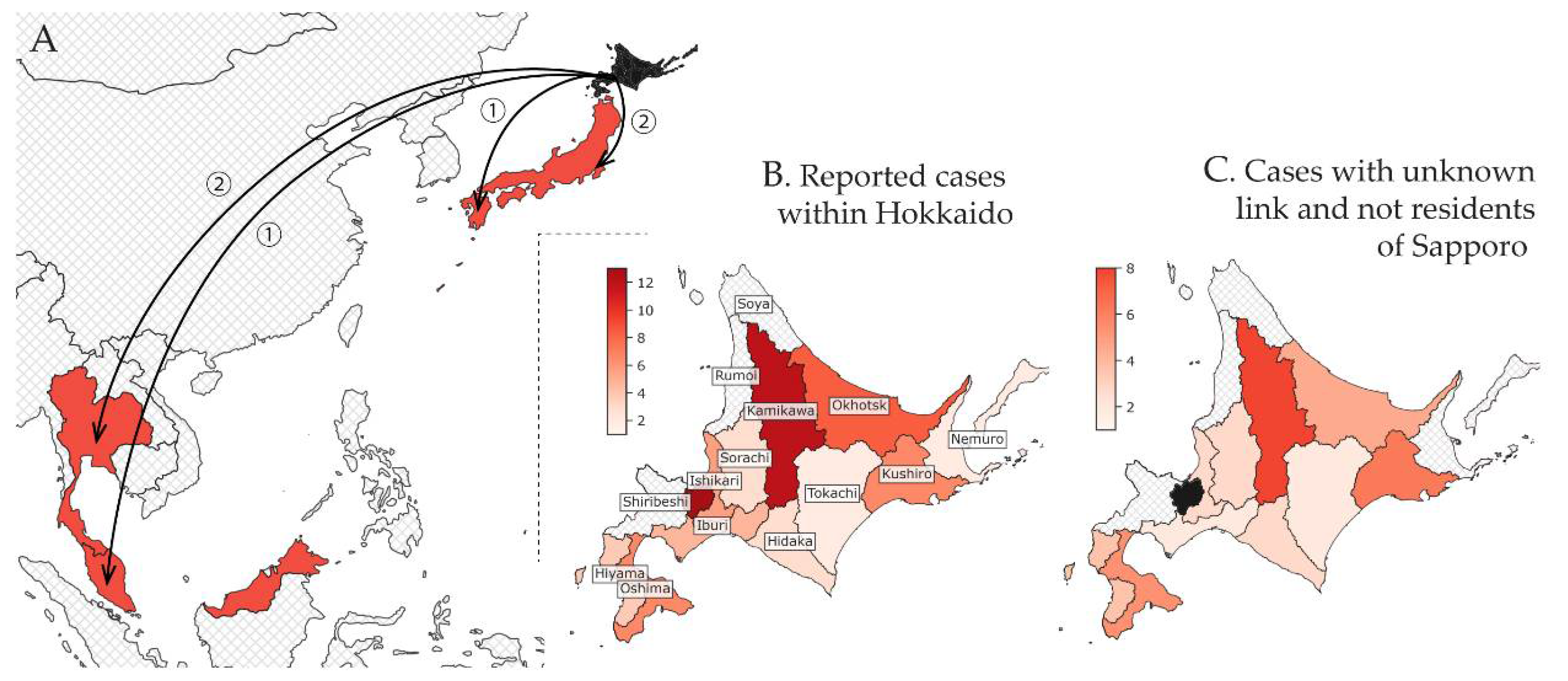
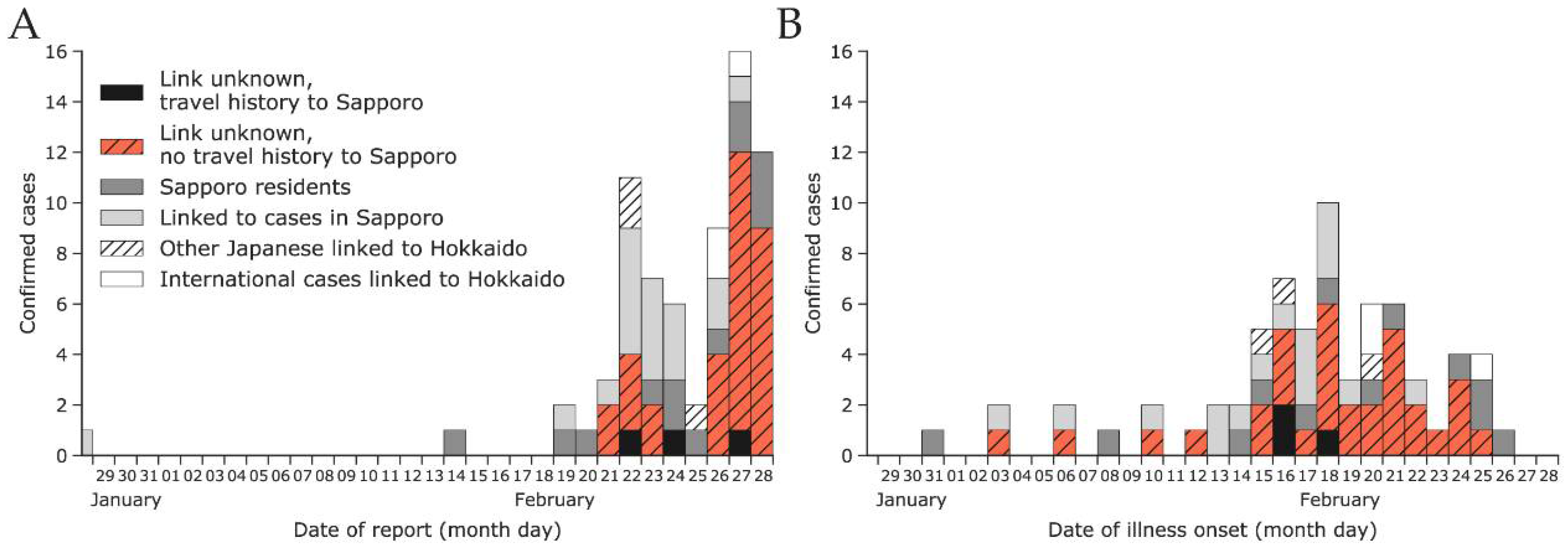
3.1. Epidemiological Situation
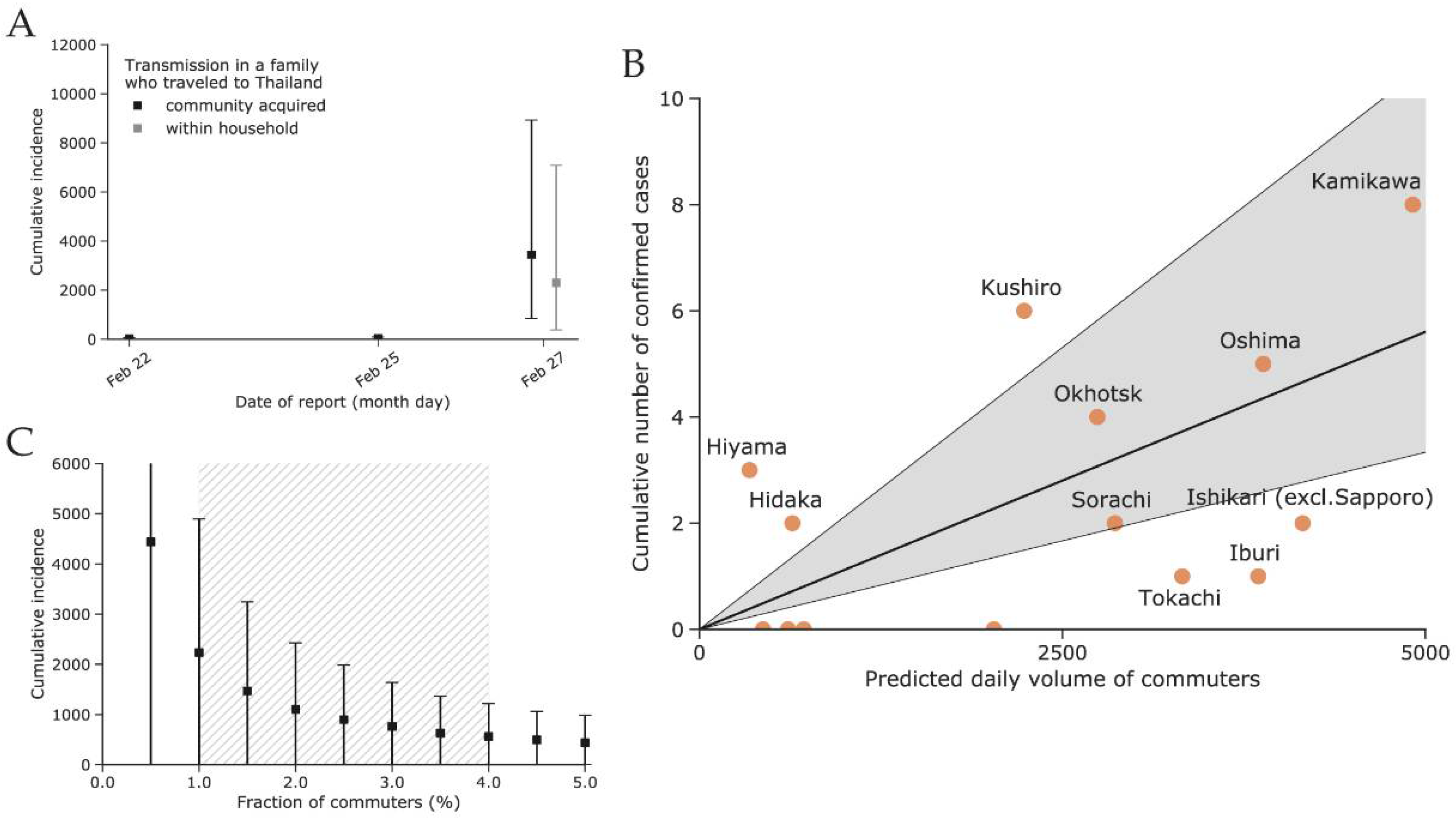
3.2. Estimated Incidence in Hokkaido Using Confirmed Cases Diagnosed Outside Hokkaido (Method (i))
3.3. Estimated Incidence in Sapporo Using Confirmed Cases Diagnosed within Hokkaido (Method (ii))
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huang, C.; Wang, Y.; Li, X.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; Cheng, Z.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Hui, D.S.I.; Azhar, E.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Ippolito, G.; McHugh, T.D.; Memish, Z.A.; Drosten, C.; et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef]
- Nishiura, H.; Jung, S.; Linton, N.M.; Ryo, K.; Yichi, Y.; Katsuma, H.; Tetsuro, K.; Baoyin, Y.; Andrei, R.A. The extent of transmission of novel coronavirus in Wuhan, China, 2020. J. Clin. Med. 2020, 9, 330. [Google Scholar] [CrossRef]
- Chinazzi, M.; Davis, J.T.; Ajelli, M. The effect of travel restrictions on the spread of the 2019 novel coronavirus (COVID-19) outbreak. Science 2020, 395–400. [Google Scholar] [CrossRef] [PubMed]
- WHO Coronavirus disease 2019 (COVID-19) Situation Report 53. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200313-sitrep-53-covid-19.pdf (accessed on 13 May 2020).
- Omori, R.; Mizumoto, K.; Chowell, G. Changes in testing rates could mask the novel coronavirus disease (COVID-19) growth rate. Int. J. Infect. Dis. 2020, 94, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Oshitani, H. The Expert Members of The National COVID-19 Cluster Taskforce at The Ministry of Health, Labour and Welfare, Japan. Cluster-Based Approach to Coronavirus Disease 2019 (COVID-19) Response in Japan, from February to April 2020. Jpn. J. Infect. Dis. 2020, 73, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Rothe, C.; Schunk, M.; Sothmann, P. Transmission of 2019-nCoV Infection from an Asymptomatic Contact in Germany. N. Engl. J. Med. 2020, 382, 970–971. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Hu, N.; Lou, J. Characteristics of COVID-19 infection in Beijing. J. Infect. 2020, 80, 401–406. [Google Scholar] [CrossRef]
- WHO Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). WHO Report 16–24 February 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf (accessed on 5 March 2020).
- He, X.; Lau, E.H.Y.; Wu, P.; Deng, X.; Wang, J.; Hao, X.; Lau, Y.C.; Wong, Y.J.; Guan, Y.; Tan, X. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 2020, 26, 672–675. [Google Scholar] [CrossRef]
- Cheng, H.-Y.; Jian, S.-W.; Liu, D.-P.; Ng, T.-C.; Huang, W.-T.; Lin, H.-H.; for the Taiwan COVID-19 Outbreak Investigation Team. Contact tracing assessment of COVID-19 transmission dynamics in Taiwan and risk at different exposure periods before and after symptom onset. JAMA Intern. Med. 2020, 180, 1156–1163. [Google Scholar] [CrossRef]
- Nishiura, H.; Kobayashi, T.; Yang, Y. The rate of underascertainment of novel coronavirus (2019-nCoV) infection: Estimation using Japanese passengers data on evacuation flights. J. Clin. Med. 2020, 9, 419. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H. Backcalculating the incidence of infection with COVID-19 on the Diamond Princess. J. Clin. Med. 2020, 9, 657. [Google Scholar] [CrossRef] [PubMed]
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance 2020, 25, 10. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.W.X.; Tan, Y.K.; Chia, P.Y.; Lee, H.T.; Oon, T.N.; Wong, Y.S.M.; Marimuthu, K. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020, 323, 1610–1612. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Zhu, B. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020, 26, 502–505. [Google Scholar] [CrossRef]
- Grantz, K.; Metcalf, C.; Lessler, J. Dispersion vs. Control. 2020. Available online: https://hopkinsidd.github.io/nCoV-Sandbox/DispersionExploration.html (accessed on 5 March 2020).
- Endo, A.; Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group; Abbott, S. Estimating the overdispersion in COVID-19 transmission using outbreak sizes outside China. Wellcome Open. Res. 2020, 5, 672. [Google Scholar] [CrossRef]
- Linton, N.M.; Kobayashi, T.; Yang, Y. Incubation period and other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data. J. Clin. Med. 2020, 9, 538. [Google Scholar] [CrossRef]
- Bi, Q.; Wu, Y.; Mei, S.; Ye, C.; Zou, X.; Zhang, Z.; Liu, X.; Wei, L.; Truelove, S.A.; Zhang, T.; et al. Epidemiology and transmission of COVID-19 in 391 cases and 1286 of their close contacts in Shenzhen, China: A retrospective cohort study. Lancet Infect. Dis. 2020, 20, 911–919. [Google Scholar] [CrossRef]
- Lee, J.-S.; Kim, S.Y.; Kim, T.S.; Hong, K.H.; Ryoo, N.-H.; Lee, J. Evidence of Severe Acute Respiratory Syndrome Coronavirus 2 reinfection after recovery from mild Coronavirus Disease 2019. Clin. Infect. Dis. 2020, ciaa1421. [Google Scholar] [CrossRef]
- Guan, W.; Ni, Z.; Hu, Y. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Hauser, A.; Counotte, M.J.; Margossian, C.C.; Konstantinoudis, G.; Low, N.; Althaus, C.; Riou, J. Estimation of SARS-CoV-2 mortality during the early stages of an epidemic: A modeling study in Hubei, China, and six regions in Europe. PLoS Med. 2020, 17, e1003189. [Google Scholar] [CrossRef]
- Li, R.; Pei, S.; Chen, B.; Song, Y.; Zhang, T.; Yang, W.; Shaman, J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2). Science 2020, 368, 489–493. [Google Scholar] [CrossRef]
- Anderson, R.M.; Heesterbeek, H.; Klinkenberg, D.; Hollingsworth, T. How will country-based mitigation measures influence the course of the COVID-19 epidemic? Lancet 2020, 395, 931–934. [Google Scholar] [CrossRef]
- Tuite, A.; Ng, V.; Rees, E.; Fisman, D. Estimation of COVID-19 outbreak size in Italy based on international case exportations. Lancet Infect. Dis. 2020, 20, 520. [Google Scholar] [CrossRef]
- Kumamoto Prefectural Government. Patients Associated with the New Coronavirus 2020. Available online: https://www.mhlw.go.jp/content/10906000/000599322.pdf (accessed on 5 March 2020). (In Japanese)
- Nagano Prefectural Government. Patients Associated with the New Coronavirus 2020. Available online: https://www.mhlw.go.jp/content/10906000/000599974.pdf (accessed on 5 March 2020). (In Japanese)
- Chiba Prefectural Government. Patients Associated with the New Coronavirus 2020. Available online: https://www.mhlw.go.jp/content/10906000/000599329.pdf (accessed on 5 March 2020). (In Japanese)
- Kyodo News. Patients Associated with the New Coronavirus 2020. Available online: https://english.kyodonews.net/news/2020/02/2622cef60938-malaysian-woman-tests-positive-for-coronavirus-after-japan-trip.html (accessed on 5 March 2020). (In Japanese).
- WHO Thailand. Coronavirus Disease 2019 (COVID-19) Situation Report—26 February 2020. Available online: https://www.who.int/docs/default-source/searo/thailand/20200226-tha-sitrep-09-covid-19-final.pdf (accessed on 5 March 2020). (In Japanese).
- Pan, X.; Chen, D.; Xia, Y. Asymptomatic cases in a family cluster with SARS-CoV-2 infection. Lancet Infect. Dis. 2020, 20, 410–411. [Google Scholar] [CrossRef]
- Hokkaido Prefecture, Japan. Basic Resident Register Information 2018. Available online: http://www.pref.hokkaido.lg.jp/ss/tuk/900brr/index2.htm (accessed on 2 March 2020).
- Sapporo City. National Census: Population and Employment Status by Employment Location and School Attendance and Population Moving 2015. Available online: https://www.city.sapporo.jp/toukei/tokusyu/kokuseityosa.html (accessed on 2 March 2020). (In Japanese).
- Kraemer, M.U.G.; Faria, N.R.; Reiner, R.C.; Golding, N.; Nikolay, B.; Stasse, S. Spread of yellow fever virus outbreak in Angola and the Democratic Republic of the Congo 2015–16: A modelling study. Lancet Infect. Dis. 2017, 17, 330–338. [Google Scholar] [CrossRef]
- Kraemer, M.U.G.; Golding, N.; Bisanzio, D. Utilizing general human movement models to predict the spread of emerging infectious diseases in resource poor settings. Sci. Rep. 2019, 9, 5151. [Google Scholar] [CrossRef]
- Golding, N.; Wuensch, K.; Schofield, A. SEEG-Oxford/movement: Modelling and Analysing Human Movement Data. R package version 0.4. Available online: https://github.com/SEEG-Oxford/movement/ (accessed on 5 March 2020).
- Spadon, G.; de Carvalho, A.C.P.L.F.; Rodrigues, J.F., Jr.; Alves, L.G.A. Reconstructing commuters network using machine learning and urban indicators. Sci. Rep. 2019, 9, 11801. [Google Scholar] [CrossRef]
- Simini, F.; Maritan, A.; Néda, Z. Human mobility in a continuum approach. PLoS ONE 2013, 8, e60069. [Google Scholar] [CrossRef]
- Simini, F.; González, M.C.; Maritan, A.; Barabási, A.-L. A universal model for mobility and migration patterns. Nature 2012, 484, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Dorigatti, I.; Hamlet, A.; Aguas, R. International risk of yellow fever spread from the ongoing outbreak in Brazil, December 2016 to May 2017. Eurosurveillance 2017, 22, 3057. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, S.; Lee, H.; Miura, F. Dynamics of the pneumonic plague epidemic in Madagascar, August to October 2017. Eurosurveillance 2017, 22, 17-00710. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Nishiura, H.; Imamura, A. Investigation of the proportion of diagnosed people living with HIV/AIDS among foreign residents in Japan. J. Clin. Med. 2019, 8, 804. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Ruan, F.; Huang, M. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020, 382, 1177–1179. [Google Scholar] [CrossRef]
- De Salazar, P.M.; Niehus, R.; Taylor, A.; Buckee, C.; Lipsitch, M. Identifying locations with possible undetected imported severe acute respiratory syndrome coronavirus 2 cases by using importation predictions. Emerg. Infect. Dis. 2020, 26, 1465–1469. [Google Scholar] [CrossRef]
- Reich, N.G.; Lessler, J.; Cummings, D.A.T.; Brookmeyer, R. Estimating incubation period distributions with coarse data. Stat. Med. 2009, 28, 2769–2784. [Google Scholar] [CrossRef]
- Carpenter, B.; Gelman, A.; Hoffman, M.D. Stan: A probabilistic programming language. J. Stat. Softw. 2017, 76, 1–32. [Google Scholar] [CrossRef]
- Salvatier, J.; Wiecki, T.V.; Fonnesbeck, C. Probabilistic programming in Python using PyMC3. PeerJ Comput. Sci. 2016, 2, e55. [Google Scholar] [CrossRef]
- Available online: http://github.com/aakhmetz/Covid19IncidenceHokkaidoFeb2020 (accessed on 27 May 2021).
- Nishiura, H.; Linton, N.M.; Akhmetzhanov, A.R. Serial interval of novel coronavirus (COVID-19) infections. Int. J. Infect. Dis. 2020, 93, 284–286. [Google Scholar] [CrossRef]
- Imai, N.; Dorigatti, I.; Cori, A. Report 2: Estimating the Potential Total Number of Novel Coronavirus Cases in Wuhan City, China; Imperial College London: London, UK, 2020. [Google Scholar]
- Anzai, A.; Kobayashi, T.; Linton, N.M. Assessing the impact of reduced travel on exportation dynamics of novel coronavirus infection (COVID-19). J. Clin. Med. 2020, 9, 601. [Google Scholar] [CrossRef]
- Quilty, B.J.; Clifford, S.; CMMID nCoV Working Group. Effectiveness of airport screening at detecting travellers infected with novel coronavirus (2019-nCoV). Eurosurveillance 2020, 25. [Google Scholar] [CrossRef]
- Niehus, R.; De Salazar, P.M.; Taylor, A.R.; Lipsitch, M. Using observational data to quantify bias of traveller-derived COVID-19 prevalence estimates in Wuhan, China. Lancet Infect. Dis. 2020, 20, 803–808. [Google Scholar] [CrossRef]
- Nishiura, H.; Oshitani, H.; Kobayashi, T. Closed environments facilitate secondary transmission of coronavirus disease 2019 (COVID-19). medRxiv 2020. [Google Scholar] [CrossRef]
- Wang, C.J.; Ng, C.Y.; Brook, R.H. Response to COVID-19 in Taiwan: Big data analytics, new technology, and proactive testing. JAMA 2020, 323, 1341. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Litvinova, M.; Wang, W. Evolving epidemiology and transmission dynamics of coronavirus disease 2019 outside Hubei province, China: A descriptive and modelling study. Lancet Infect. Dis. 2020, 20, 793–802. [Google Scholar] [CrossRef]
- Ferretti, L.; Wymant, C.; Kendall, M.; Zhao, L.; Nurtay, A.; Abeler-Dorner, L.; Parker, M.; Bonsall, D.; Fraser, C. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science 2020, 368, eabb6936. [Google Scholar] [CrossRef] [PubMed]
- Jian, S.-H.; Cheng, H.-Y.; Huang, X.-T.; Liu, D.-P. Contact tracing with digital assistance in Taiwan’s COVID-19 outbreak response. Intern. J. Infect. Dis. 2020, 101, 348–352. [Google Scholar] [CrossRef] [PubMed]
| Case Number | Diagnosis Location | Date of Illness Onset | Date of Report | Cumulative Count | Estimated Incidence in Hokkaido (95% CI) |
|---|---|---|---|---|---|
| 1 | Kumamoto | 15 February 2020 | 22 February 2020 | 2 | 24 (4–74) |
| 2 | Chiba | 16 February 2020 | |||
| 3 | Nagano | 20 February 2020 | 25 February 2020 | 3 | 36 (9–93) |
| 4–5 | Thailand | 20 February 2020 | 26 February 2020 | 6 | 3446 (857–8931) # 2298 (382–7091) ## |
| NA | |||||
| 6 | Malaysia | 25 February 2020 | 27 February 2020 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akhmetzhanov, A.R.; Mizumoto, K.; Jung, S.-M.; Linton, N.M.; Omori, R.; Nishiura, H. Estimation of the Actual Incidence of Coronavirus Disease (COVID-19) in Emergent Hotspots: The Example of Hokkaido, Japan during February–March 2020. J. Clin. Med. 2021, 10, 2392. https://doi.org/10.3390/jcm10112392
Akhmetzhanov AR, Mizumoto K, Jung S-M, Linton NM, Omori R, Nishiura H. Estimation of the Actual Incidence of Coronavirus Disease (COVID-19) in Emergent Hotspots: The Example of Hokkaido, Japan during February–March 2020. Journal of Clinical Medicine. 2021; 10(11):2392. https://doi.org/10.3390/jcm10112392
Chicago/Turabian StyleAkhmetzhanov, Andrei R., Kenji Mizumoto, Sung-Mok Jung, Natalie M. Linton, Ryosuke Omori, and Hiroshi Nishiura. 2021. "Estimation of the Actual Incidence of Coronavirus Disease (COVID-19) in Emergent Hotspots: The Example of Hokkaido, Japan during February–March 2020" Journal of Clinical Medicine 10, no. 11: 2392. https://doi.org/10.3390/jcm10112392
APA StyleAkhmetzhanov, A. R., Mizumoto, K., Jung, S.-M., Linton, N. M., Omori, R., & Nishiura, H. (2021). Estimation of the Actual Incidence of Coronavirus Disease (COVID-19) in Emergent Hotspots: The Example of Hokkaido, Japan during February–March 2020. Journal of Clinical Medicine, 10(11), 2392. https://doi.org/10.3390/jcm10112392








