Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Spirometry
2.3. Survey on Respiratory Symptoms
2.4. Statistical Analysis
3. Results
3.1. Whole-Life Exposure to Air Pollution
3.2. Cohort Characteristics
3.3. Spirometry Variables and Respiratory or Allergic Symptoms
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization Air Pollution and Child Health: Prescribing Clean Air; World Health Organization: Geneva, Switzerland, 2019.
- Committee on Environmental Health. Ambient Air Pollution: Health Hazards to Children. Pediatrics 2004, 114, 1699–1707. [Google Scholar] [CrossRef]
- Hwang, B.-F.; Chen, Y.-H.; Lin, Y.-T.; Wu, X.-T.; Leo Lee, Y. Relationship between exposure to fine particulates and ozone and reduced lung function in children. Environ. Res. 2015, 137, 382–390. [Google Scholar] [CrossRef]
- Urman, R.; McConnell, R.; Islam, T.; Avol, E.L.; Lurmann, F.W.; Vora, H.; Linn, W.S.; Rappaport, E.B.; Gilliland, F.D.; Gauderman, W.J. Associations of children’s lung function with ambient air pollution: Joint effects of regional and near-roadway pollutants. Thorax 2014, 69, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chan, E.Y.Y.; Li, L.P.; He, Q.Q.; Wong, T.W. Chronic effects of ambient air pollution on lung function among Chinese children. Arch. Dis. Child. 2013, 98, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.B.; Rifas-Shiman, S.L.; Litonjua, A.A.; Oken, E.; Gillman, M.W.; Kloog, I.; Luttmann-Gibson, H.; Zanobetti, A.; Coull, B.A.; Schwartz, J.; et al. Lifetime Exposure to Ambient Pollution and Lung Function in Children. Am. J. Respir. Crit. Care Med. 2016, 193, 881–888. [Google Scholar] [CrossRef]
- Badyda, A.J.; Dąbrowiecki, P.; Czechowski, P.O.; Majewski, G. Risk of bronchi obstruction among non-smokers—Review of environmental factors affecting bronchoconstriction. Respir. Physiol. Neurobiol. 2015, 209, 39–46. [Google Scholar] [CrossRef]
- Dąbrowiecki, P.; Mucha, D.; Gayer, A.; Adamkiewicz, Ł.; Badyda, A.J. Assessment of Air Pollution Effects on the Respiratory System Based on Pulmonary Function Tests Performed During Spirometry Days. Adv. Exp. Med. Biol. 2015, 873, 43–52. [Google Scholar] [CrossRef]
- Bergstra, A.D.; Brunekreef, B.; Burdorf, A. The effect of industry-related air pollution on lung function and respiratory symptoms in school children. Environ. Health 2018, 17, 30. [Google Scholar] [CrossRef]
- Asgari, M.M.; Dubois, A.; Asgari, M.; Gent, J.; Beckett, W.S. Association of Ambient Air Quality with Children’s Lung Function in Urban and Rural Iran. Arch. Environ. Health Int. J. 1998, 53, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Priftis, K.N.; Anthracopoulos, M.B.; Paliatsos, A.G.; Tzavelas, G.; Nikolaou-Papanagiotou, A.; Douridas, P.; Nicolaidou, P.; Mantzouranis, E. Different effects of urban and rural environments in the respiratory status of Greek schoolchildren. Respir. Med. 2007, 101, 98–106. [Google Scholar] [CrossRef]
- Sonnappa, S.; Lum, S.; Kirkby, J.; Bonner, R.; Wade, A.; Subramanya, V.; Lakshman, P.T.; Rajan, B.; Nooyi, S.C.; Stocks, J. Disparities in Pulmonary Function in Healthy Children across the Indian Urban–Rural Continuum. Am. J. Respir. Crit. Care Med. 2015, 191, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Jakubiak-Lasocka, J.; Lasocki, J.; Badyda, A.J. The influence of particulate matter on respiratory morbidity and mortality in children and infants. Adv. Exp. Med. Biol. 2015, 849, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Badyda, A.J.; Feleszko, W.; Ratajczak, A.; Czechowski, P.O.; Czarnecki, A.; Dubrawski, M.; Dąbrowska, A. Influence of Particulate Matter on the Occurrence of Upper Respiratory Tract Symptoms in Children Aged 3–12 Years. Am. J. Respir. Crit. Care Med. 2020, 201, A6346. [Google Scholar] [CrossRef]
- Kurt, O.K.; Zhang, J.; Pinkerton, K.E. Pulmonary health effects of air pollution. Curr. Opin. Pulm. Med. 2016, 22, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Badyda, A.J.; Dąbrowiecki, P.; Czechowski, P.O.; Majewski, G.; Doboszyńska, A. Traffic-related air pollution and respiratory tract efficiency. Adv. Exp. Med. Biol. 2015, 834, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Provost, E.B.; Int Panis, L.; Saenen, N.D.; Kicinski, M.; Louwies, T.; Vrijens, K.; De Boever, P.; Nawrot, T.S. Recent versus chronic fine particulate air pollution exposure as determinant of the retinal microvasculature in school children. Environ. Res. 2017, 159, 103–110. [Google Scholar] [CrossRef]
- Pieters, N.; Koppen, G.; Van Poppel, M.; De Prins, S.; Cox, B.; Dons, E.; Nelen, V.; Panis, L.I.; Plusquin, M.; Schoeters, G.; et al. Blood Pressure and Same-Day Exposure to Air Pollution at School: Associations with Nano-Sized to Coarse PM in Children. Environ. Health Perspect. 2015, 123, 737–742. [Google Scholar] [CrossRef]
- Five Things We Learned from the World’s Biggest Air Pollution Database—Unearthed. Available online: https://unearthed.greenpeace.org/2018/05/02/air-pollution-cities-worst-global-data-world-health-organisation/ (accessed on 2 November 2020).
- Global Ambient Air Quality Database (Update 2018); World Health Organization: Geneva, Switzerland, 2019.
- Parascandola, M. Ambient air pollution and lung cancer in Poland: Research findings and gaps. J. Health Inequalities 2018, 4, 3–8. [Google Scholar] [CrossRef]
- Kupczyk, M.; Hofman, A.; Kołtowski, Ł.; Kuna, P.; Łukaszyk, M.; Buczyłko, K.; Bodzenta-Łukaszyk, A.; Nastałek, P.; Soliński, M.; Dąbrowiecki, P. Home self-monitoring in patients with asthma using a mobile spirometry system. J. Asthma 2021, 58, 505–511. [Google Scholar] [CrossRef]
- Graham, B.L.; Steenbruggen, I.; Miller, M.R.; Barjaktarevic, I.Z.; Cooper, B.G.; Hall, G.L.; Hallstrand, T.S.; Kaminsky, D.A.; McCarthy, K.; McCormack, M.C.; et al. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am. J. Respir. Crit. Care Med. 2019, 200, e70–e88. [Google Scholar] [CrossRef]
- Xing, Y.F.; Xu, Y.H.; Shi, M.H.; Lian, Y.X. The impact of PM2.5 on the human respiratory system. J. Thorac. Dis. 2016, 8, E69–E74. [Google Scholar]
- Wigenstam, E.; Elfsmark, L.; Bucht, A.; Jonasson, S. Inhaled sulfur dioxide causes pulmonary and systemic inflammation leading to fibrotic respiratory disease in a rat model of chemical-induced lung injury. Toxicology 2016, 368, 28–36. [Google Scholar] [CrossRef]
- Petit, P.C.; Fine, D.H.; Vásquez, G.B.; Gamero, L.; Slaughter, M.S.; Dasse, K.A. The Pathophysiology of Nitrogen Dioxide During Inhaled Nitric Oxide Therapy. ASAIO J. 2017, 63, 7–13. [Google Scholar] [CrossRef]
- Gauderman, W.J.; Avol, E.; Gilliland, F.; Vora, H.; Thomas, D.; Berhane, K.; McConnell, R.; Kuenzli, N.; Lurmann, F.; Rappaport, E.; et al. The Effect of Air Pollution on Lung Development from 10 to 18 Years of Age. N. Engl. J. Med. 2004, 351, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Martinez, R.; Perez-Padilla, R.; Olaiz-Fernandez, G.; Mendoza-Alvarado, L.; Moreno-Macias, H.; Fortoul, T.; McDonnell, W.; Loomis, D.; Romieu, I. Lung Function Growth in Children with Long-Term Exposure to Air Pollutants in Mexico City. Am. J. Respir. Crit. Care Med. 2007, 176, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.L.; Wang, W.-H.; Lu, C.-W.; Lin, Y.-H.; Hwang, B.-F. Effects of ambient air pollution on pulmonary function among schoolchildren. Int. J. Hyg. Environ. Health 2011, 214, 369–375. [Google Scholar] [CrossRef]
- Gehring, U.; Gruzieva, O.; Agius, R.M.; Beelen, R.; Custovic, A.; Cyrys, J.; Eeftens, M.; Flexeder, C.; Fuertes, E.; Heinrich, J.; et al. Air Pollution Exposure and Lung Function in Children: The ESCAPE Project. Environ. Health Perspect. 2013, 121, 1357–1364. [Google Scholar] [CrossRef]
- He, Q.-C.; Lioy, P.J.; Wilson, W.E.; Chapman, R.S. Effects of Air Pollution on Children’s Pulmonary Function in Urban and Suburban Areas of Wuhan, People’s Republic of China. Arch. Environ. Health Int. J. 1993, 48, 382–391. [Google Scholar] [CrossRef]
- Rusconi, F.; Catelan, D.; Accetta, G.; Peluso, M.; Pistelli, R.; Barbone, F.; Di Felice, E.; Munnia, A.; Murgia, P.; Paladini, L.; et al. Asthma Symptoms, Lung Function, and Markers of Oxidative Stress and Inflammation in Children Exposed to Oil Refinery Pollution. J. Asthma 2011, 48, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Wiwatanadate, P.; Trakultivakorn, M. Air pollution-related peak expiratory flow rates among asthmatic children in Chiang Mai, Thailand. Inhal. Toxicol. 2010, 22, 301–308. [Google Scholar] [CrossRef]
- Oftedal, B.; Brunekreef, B.; Nystad, W.; Madsen, C.; Walker, S.-E.; Nafstad, P. Residential Outdoor Air Pollution and Lung Function in Schoolchildren. Epidemiology 2008, 19, 129–137. [Google Scholar] [CrossRef]
- Hasibuan, I.E.; Supriatmo, M.N.; Faisal, A.; Panggabean, G.; Daulay, R.M.; Siregar, Z.; Lubis, H.M. Peak expiratory flow rate of primary school children in high and low air pollution level areas. Paediatr. Indones. 2016, 43, 10. [Google Scholar] [CrossRef][Green Version]
- Croft, D.P.; Zhang, W.; Lin, S.; Thurston, S.W.; Hopke, P.K.; Masiol, M.; Squizzato, S.; van Wijngaarden, E.; Utell, M.J.; Rich, D.Q. The Association between Respiratory Infection and Air Pollution in the Setting of Air Quality Policy and Economic Change. Ann. Am. Thorac. Soc. 2018, 16, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.-F.; Jaakkola, J.J.; Lee, Y.-L.; Lin, Y.-C.; Leon Guo, Y. Relation between air pollution and allergic rhinitis in Taiwanese schoolchildren. Respir. Res. 2006, 7, 23. [Google Scholar] [CrossRef]
- Calderón-Garcidueñas, L.; Mora-Tiscareño, A.; Fordham, L.A.; Chung, C.J.; Valencia-Salazar, G.; Flores-Gómez, S.; Solt, A.C.; Campo, A.G.; Jardón-Torres, R.; Henríquez-Roldán, C.; et al. Lung Radiology and Pulmonary Function of Children Chronically Exposed to Air Pollution. Environ. Health Perspect. 2006, 114, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Araviiskaia, E.; Berardesca, E.; Bieber, T.; Gontijo, G.; Sanchez Viera, M.; Marrot, L.; Chuberre, B.; Dreno, B. The impact of airborne pollution on skin. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1496–1505. [Google Scholar] [CrossRef]
- Gauderman, W.J.; Urman, R.; Avol, E.; Berhane, K.; McConnell, R.; Rappaport, E.; Chang, R.; Lurmann, F.; Gilliland, F. Association of Improved Air Quality with Lung Development in Children. N. Engl. J. Med. 2015, 372, 905–913. [Google Scholar] [CrossRef] [PubMed]
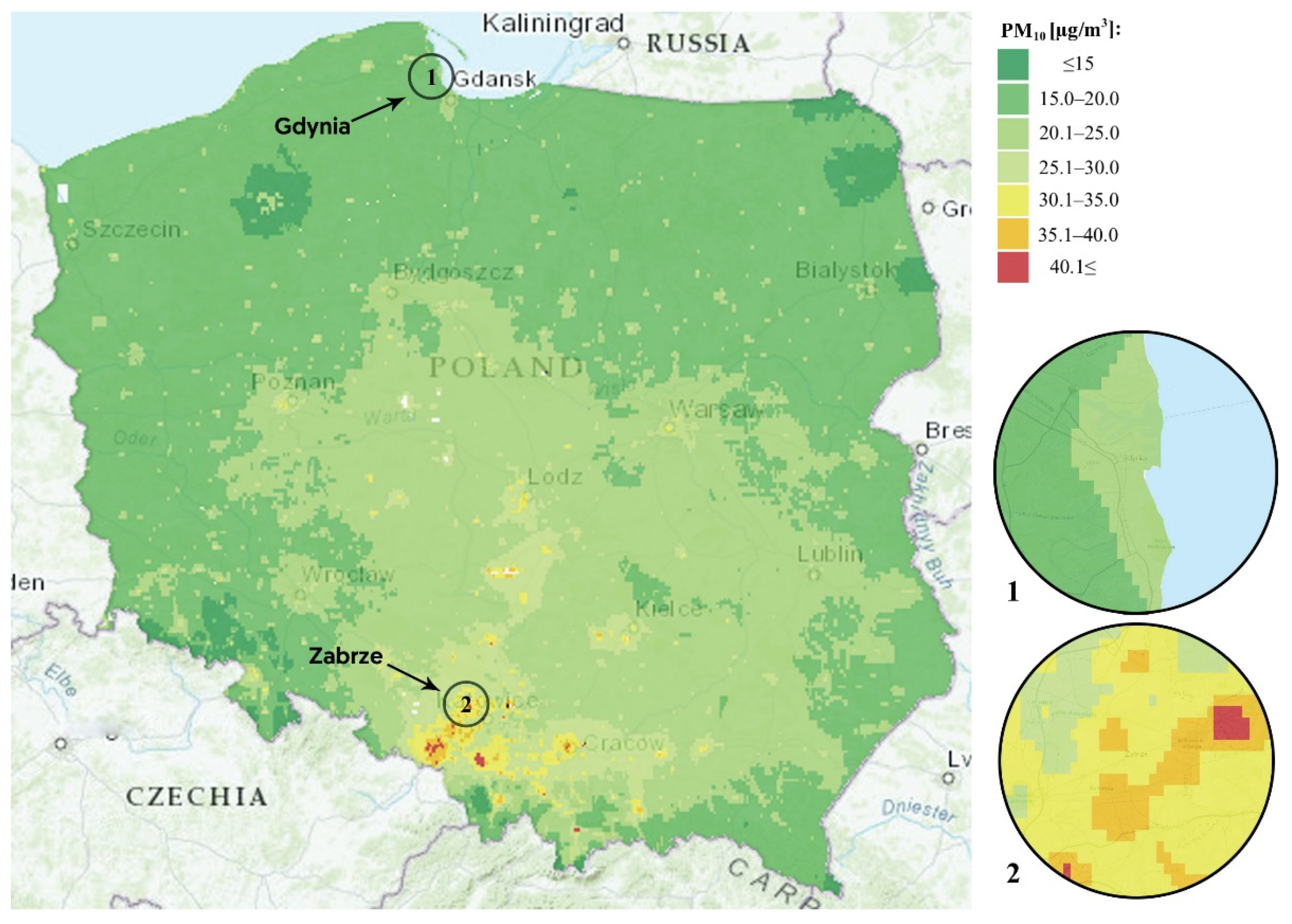
| Age Group | Pollutant | |||||||
|---|---|---|---|---|---|---|---|---|
| PM10, µg/m3 | SO2, µg/m3 | NO2, µg/m3 | O3, µg/m3 | |||||
| Gdynia | Zabrze | Gdynia | Zabrze | Gdynia | Zabrze | Gdynia | Zabrze | |
| 9-year-olds | 21.96 | 50.06 | 3.81 | 18.18 | 16.34 | 25.16 | 52.40 | 42.11 |
| 10-year-olds | 21.73 | 50.05 | 3.71 | 17.68 | 16.37 | 24.86 | 52.44 | 41.24 |
| 11-year-olds | 18.83 | 49.30 | 3.55 | 16.85 | 16.51 | 24.42 | 51.79 | 40.97 |
| 12-year-olds | 18.41 | 49.74 | 3.49 | 16.97 | 16.79 | 24.60 | 51.28 | 41.80 |
| 13-year-olds | 18.06 | 50.31 | 3.42 | 17.08 | 16.87 | 24.65 | 50.78 | 42.80 |
| 14-year-olds | 17.59 | 50.68 | 3.41 | 16.91 | 16.99 | 24.64 | 50.98 | 42.81 |
| 15-year-olds | 17.16 | 50.34 | 3.30 | 16.07 | 16.44 | 24.28 | 50.99 | 43.37 |
| Variable | Gdynia (n = 258) | Zabrze (n = 512) | p-Value |
|---|---|---|---|
| Age (years), mean ± SD | 11.11 ± 1.32 | 11.23 ± 1.44 | 0.254 |
| Girls, n (%) | 123 (47.67) | 264 (51.56) | 0.308 |
| Height (cm), mean ± SD | 152.23 ± 10.41 | 151.54 ± 11.08 | 0.406 |
| Weight (kg), mean ± SD | 45.45 ± 11.60 | 45.87 ± 13.43 | 0.673 |
| BMI (kg/m2), mean ± SD | 19.38 ± 3.32 | 19.70 ± 4.17 | 0.289 |
| Distance to a major road, n (%) | <0.001 | ||
| <100 m | 56 (24.24) | 173 (37.94) | |
| 100–200 m | 64 (27.70) | 123 (26.97) | |
| 200–500 m | 59 (25.54) | 101 (22.15) | |
| >500 m | 52 (22.51) | 59 (12.94) | |
| Coal- or wood-burning stove, n (%) | 0 | 11 (2.21) | 0.040 |
| Heating furnace in a living area, n (%) | 17 (6.80) | 63 (12.70) | 0.014 |
| Smoking parent, n (%) | 59 (23.51) | 144 (28.97) | 0.112 |
| Physical activity, n (%) | 0.011 | ||
| Not active | 2 (0.78) | 5 (1.01) | |
| Irregular | 150 (59.76) | 238 (48.18) | |
| Regular | 99 (39.44) | 251 (50.81) |
| Variable | Gdynia | Zabrze | p-Value * |
|---|---|---|---|
| FVC, mean ± SD (L) | 2.42 ± 0.77 | 2.21 ± 0.73 | <0.001 |
| FEV1, mean ± SD (L) | 2.17 ± 0.60 | 2.07 ± 0.67 | 0.032 |
| FEV1/FVC, mean ± SD | 0.91 ± 0.08 | 0.94 ± 0.07 | <0.001 |
| PEF, mean ± SD, (L/min) | 259.34 ± 83.90 | 244.64 ± 81.64 | 0.018 |
| Variable | Gdynia | Zabrze | p-Value |
|---|---|---|---|
| All-year rhinorrhea, n (%) | 15 (6.10) | 27 (5.92) | 0.925 |
| Seasonal rhinorrhea, n (%) | 99 (39.60) | 241 (48.98) | 0.015 |
| Acute allergic reactions, n (%) | 66 (26.29) | 1107 (21.49) | 0.140 |
| Dyspnea episodes, n (%) | 26 (10.53) | 58 (11.88) | 0.584 |
| Coughing episodes, n (%) | 56 (22.40) | 148 (30.39) | 0.022 |
| Wheezing episodes, n (%) | 23 (9.27) | 52 (10.68) | 0.552 |
| Skin changes, n (%) | 108 (43.20) | 194 (39.59) | 0.345 |
| Allergies (any), n (%) | 81 (32.66) | 134 (27.52) | 0.147 |
| Allergic skin reactions, n (%) | 110 (44.18) | 185 (37.23) | 0.067 |
| Recurring bronchitis, n (%) | 46 (18.33) | 99 (19.88) | 0.612 |
| Wheezing (ever), n (%) | 32 (12.80) | 69 (14.02) | 0.646 |
| Asthma diagnosis, n (%) | 16 (6.56) | 37 (7.65) | 0.594 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dąbrowiecki, P.; Adamkiewicz, Ł.; Mucha, D.; Czechowski, P.O.; Soliński, M.; Chciałowski, A.; Badyda, A. Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland. J. Clin. Med. 2021, 10, 2375. https://doi.org/10.3390/jcm10112375
Dąbrowiecki P, Adamkiewicz Ł, Mucha D, Czechowski PO, Soliński M, Chciałowski A, Badyda A. Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland. Journal of Clinical Medicine. 2021; 10(11):2375. https://doi.org/10.3390/jcm10112375
Chicago/Turabian StyleDąbrowiecki, Piotr, Łukasz Adamkiewicz, Dominika Mucha, Piotr Oskar Czechowski, Mateusz Soliński, Andrzej Chciałowski, and Artur Badyda. 2021. "Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland" Journal of Clinical Medicine 10, no. 11: 2375. https://doi.org/10.3390/jcm10112375
APA StyleDąbrowiecki, P., Adamkiewicz, Ł., Mucha, D., Czechowski, P. O., Soliński, M., Chciałowski, A., & Badyda, A. (2021). Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland. Journal of Clinical Medicine, 10(11), 2375. https://doi.org/10.3390/jcm10112375








