Post-Exertional Malaise May Be Related to Central Blood Pressure, Sympathetic Activity and Mental Fatigue in Chronic Fatigue Syndrome Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Enrolment
2.2. Measurement
Scales
2.3. Autonomic Symptom Assessment
2.4. Arterial Stiffness
2.5. Statistical Analysis
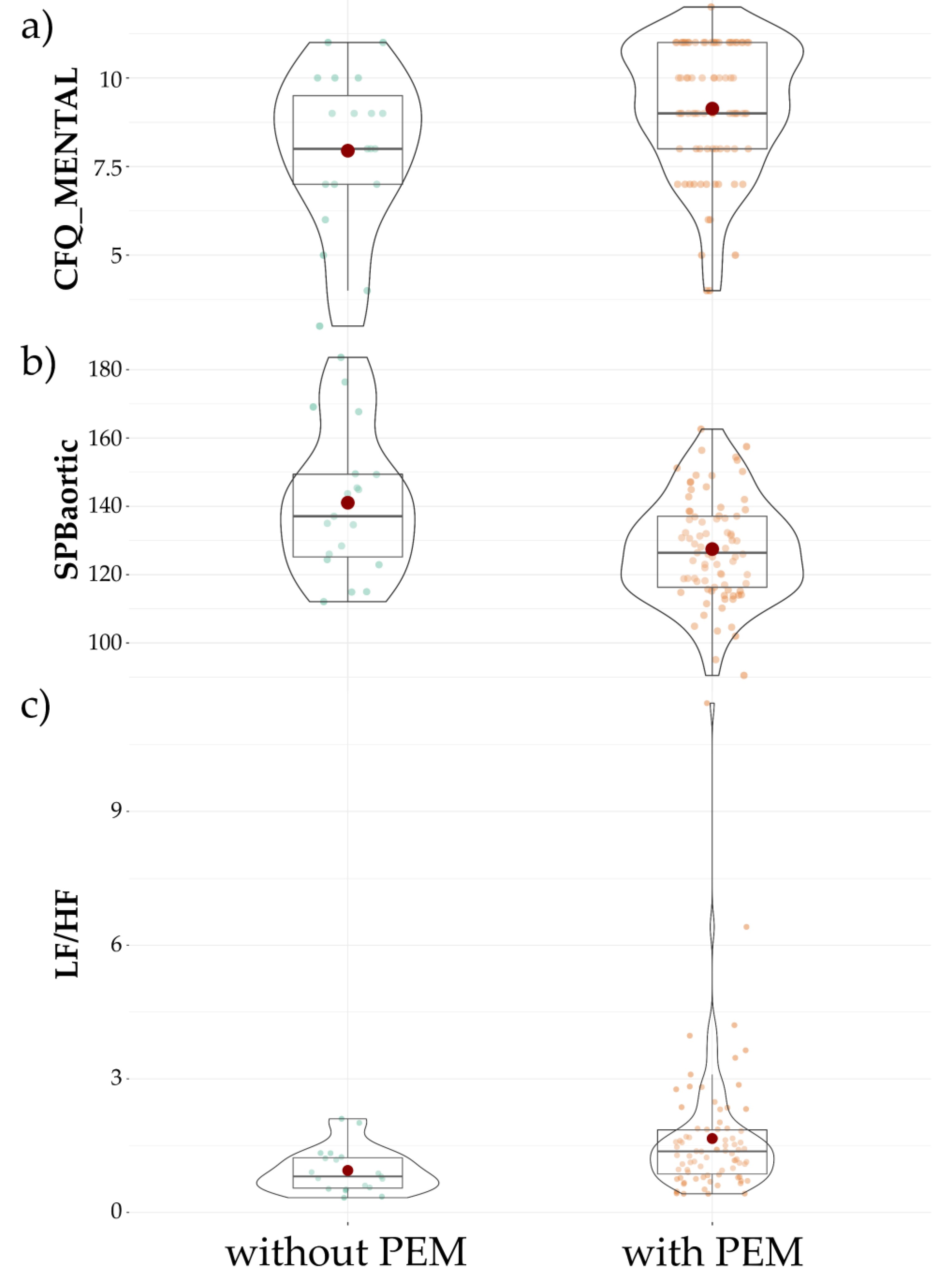
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments

Conflicts of Interest
Appendix A
| Term | Estimate | −95% CI | 95% CI | z Value | p-Value |
|---|---|---|---|---|---|
| CFQ_MENTAL_FATIGUE | 0.29 | 0.02 | 0.59 | 2.02 | 0.04 |
| SBPaortic | −0.05 | −0.09 | −0.02 | −2.66 | 0.01 |
| LF/HF | 1.46 | 0.43 | 2.78 | 2.45 | 0.01 |
References
- Oosterwijck, J.V.; Marusic, U.; De Wandele, I.; Paul, L.; Meeus, M.; Moorkens, G.; Lambrecht, L.; Danneels, L.; Nijs, J. The Role of Autonomic Function in Exercise-induced Endogenous Analgesia: A Case-control Study in Myalgic Encephalomyeli-tis/Chronic Fatigue Syndrome and Healthy People. Pain Physician 2017, 20, E389–E399. [Google Scholar]
- Committee on the Diagnostic Criteria for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome; Board on the Health of Select Populations; Institute of Medicine. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness. In The National Academies Collection: Reports Funded by National Institutes of Health; National Academies Press: Washington, DC, USA, 2015; ISBN 9780309316897. [Google Scholar]
- Carruthers, B.M.; Van De Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.C.; Dobbins, J.G.; Komaroff, A.; Schluederberg, A.; Jones, J.F.; Lloyd, A.R.; Wessely, S.; et al. The Chronic Fatigue Syndrome: A Comprehensive Approach to Its Definition and Study. Ann. Int. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Cotler, J.; Holtzman, C.; Dudun, C.; Jason, L.A. A Brief Questionnaire to Assess Post-Exertional Malaise. Diagnostics 2018, 8, 66. [Google Scholar] [CrossRef]
- Hawk, C.; Jason, L.A.; Torres-Harding, S. Differential diagnosis of chronic fatigue syndrome and major depressive disorder. Int. J. Behav. Med. 2006, 13, 244–251. [Google Scholar] [CrossRef]
- Cotler, J.; Katz, B.Z.; Reurts-Post, C.; Vermeulen, R.; Jason, L.A. A hierarchical logistic regression predicting rapid respiratory rates from post-exertional malaise. Fatigue Biomed. Health Behav. 2020, 17, 1–9. [Google Scholar] [CrossRef]
- Strahler, J.; Fischer, S.; Nater, U.M.; Ehlert, U.; Gaab, J. Norepinephrine and epinephrine responses to physiological and pharmacological stimulation in chronic fatigue syndrome. Biol. Psychol. 2013, 94, 160–166. [Google Scholar] [CrossRef]
- Suárez, A.; Guillamó, E.; Roig, T.; Blázquez, A.; Alegre, J.; Bermúdez, J.; Ventura, J.L.; García-Quintana, A.M.; Comella, A.; Segura, R.; et al. Nitric Oxide Metabolite Production During Exercise in Chronic Fatigue Syndrome: A Case-Control Study. J. Womens Health 2010, 19, 1073–1077. [Google Scholar] [CrossRef] [PubMed]
- Bond, J.; Nielsen, T.; Hodges, L. Effects of Post-Exertional Malaise on Markers of Arterial Stiffness in Individuals with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Int. J. Environ. Res. Public Health 2021, 18, 2366. [Google Scholar] [CrossRef]
- Sutton-Tyrrell, K.; Najjar, S.S.; Boudreau, R.M.; Venkitachalam, L.; Kupelian, V.; Simonsick, E.M.; Havlik, R.; Lakatta, E.G.; Spurgeon, H.; Kritchevsky, S.; et al. Elevated Aortic Pulse Wave Velocity, a Marker of Arterial Stiffness, Predicts Cardiovascular Events in Well-Functioning Older Adults. Circulation 2005, 111, 3384–3390. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.; Murray, A.; Di Maria, C.; Newton, J.L. Chronic fatigue syndrome and impaired peripheral pulse characteristics on orthostasis–A new potential diagnostic biomarker. Physiol. Meas. 2012, 33, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Słomko, J.; Estévez-López, F.; Kujawski, S.; Zawadka-Kunikowska, M.; Tafil-Klawe, M.; Klawe, J.J.; Morten, K.J.; Szrajda, J.; Murovska, M.; Newton, J.L.; et al. Autonomic Phenotypes in Chronic Fatigue Syndrome (CFS) are Associated with Illness Severity: A Cluster Analysis. J. Clin. Med. 2020, 9, 2531. [Google Scholar] [CrossRef] [PubMed]
- Hunter, G.R.; Neumeier, W.H.; Bickel, C.S.; McCarthy, J.P.; Fisher, G.; Chandler-Laney, P.C.; Glasser, S.P. Arterial Elasticity, Strength, Fatigue, and Endurance in Older Women. BioMed. Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Twisk, F.N.M.; Johnson, C. Myalgic Encephalomyelitis (ME), Chronic Fatigue Syndrome (CFS), and Chronic Fatigue (CF) are distinguished accurately: Results of supervised learning techniques applied on clinical and inflammatory data. Psychiatry Res. 2012, 200, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, B.M.; Jain, A.K.; De Meirleir, K.L.; Peterson, D.L.; Klimas, N.G.; Lerner, A.M.; van de Sande, M.I. Myalgic encephalomyelitis/chronic fatigue syndrome: Clinical working case definition, diagnostic and treatment protocols. J. Chronic Fatigue Syndr. 2003, 11, 7–115. [Google Scholar] [CrossRef]
- May, M.; Milrad, S.F.; Perdomo, D.M.; Czaja, S.J.; Fletcher, M.A.; Jutagir, D.R.; Hall, D.L.; Klimas, N.; Antoni, M.H. Post-exertional malaise is associated with greater symptom burden and psychological distress in patients diagnosed with Chronic Fatigue Syndrome. J. Psychosom. Res. 2020, 129, 109893. [Google Scholar] [CrossRef]
- Jason, L.A.; Evans, M.; So, S.; Scott, J.; Brown, A. Problems in Defining Post-Exertional Malaise. J. Prev. Interv. Community 2015, 43, 20–31. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Steer, R.A.; Ball, R.; Ranieri, W.F. Comparison of Beck Depression Inventories-IA and-II in Psychiatric Outpatients. J. Pers. Assess. 1996, 67, 588–597. [Google Scholar] [CrossRef]
- Morriss, R.; Wearden, A.; Mullis, R. Exploring the validity of the chalder fatigue scale in chronic fatigue syndrome. J. Psychosom. Res. 1998, 45, 411–417. [Google Scholar] [CrossRef]
- Valko, P.O.; Bassetti, C.L.; Bloch, K.E.; Held, U.; Baumann, C.R. Validation of the Fatigue Severity Scale in a Swiss Cohort. Sleep 2008, 31, 1601–1607. [Google Scholar] [CrossRef] [PubMed]
- Frith, J.; Newton, J. Fatigue Impact Scale. Occup. Med. 2010, 60, 159. [Google Scholar] [CrossRef] [PubMed]
- Johns, M.W. A New Method for Measuring Daytime Sleepiness: The Epworth Sleepiness Scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Suarez, G.A.; Opfer-Gehrking, T.L.; Offord, K.P.; Atkinson, E.J.; O’Brien, P.C.; Low, P.A. The Autonomic Symptom Profile: A new instrument to assess autonomic symptoms. Neurology 1999, 52, 523. [Google Scholar] [CrossRef] [PubMed]
- Sletten, D.M.; Suarez, G.A.; Low, P.A.; Mandrekar, J.; Singer, W. COMPASS 31: A Refined and Abbreviated Composite Autonomic Symptom Score. Mayo Clin. Proc. 2012, 87, 1196–1201. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-A.; Lee, H.; Park, K.-J.; Lim, J.-G. Autonomic dysfunction in patients with orthostatic dizziness: Validation of orthostatic grading scale and comparison of Valsalva maneuver and head-up tilt testing results. J. Neurol. Sci. 2013, 325, 61–66. [Google Scholar] [CrossRef]
- Bianchi, A.M.; Mainardi, L.T.; Meloni, C.; Chierchiu, S.; Cerutti, S. Continuous monitoring of the sympatho-vagal balance through spectral analysis. IEEE Eng. Med. Biol. Mag. 1997, 16, 64–73. [Google Scholar] [CrossRef]
- Németh, Z.; Móczár, K.; Deák, G. Evaluation of the Tensioday ambulatory blood pressure monitor according to the protocols of the British Hypertension Society and the Association for the Advancement of Medical Instrumentation. Blood Press. Monit. 2002, 7, 191–197. [Google Scholar] [CrossRef]
- Ring, M.; Eriksson, M.J.; Zierath, J.R.; Caidahl, K. Arterial stiffness estimation in healthy subjects: A validation of oscillometric (Arteriograph) and tonometric (SphygmoCor) techniques. Hypertens. Res. 2014, 37, 999–1007. [Google Scholar] [CrossRef]
- Horvath, I.G.; Nemeth, A.; Lenkey, Z.; Alessandri, N.; Tufano, F.; Kis, P.; Gaszner, B.; Cziraki, A. Invasive validation of a new oscillometric device (arteriogrph) for measuring augmentation index, central blood pressure and aortic pulse wave velocity. J Hypertens. 2010, 28, 2068–2075. [Google Scholar] [CrossRef]
- Andri, S.; Ken, A.; Andreas, A.; Nanina, A.; Tomas, A.; Chandima, A.; Antti, A.; Adrian, B.; Kamil, B.; Ben, B.; et al. DescTools: Tools for Descriptive Statistics. R package version 0.99.36. 2020. Available online: https://cran.r-project.org/package=DescTools (accessed on 12 March 2021).
- Solt, F.; Hu, Y. Dotwhisker: Dot-and-Whisker Plots of Regression Results. The Comprehensive R ArchiveNetwork (CRAN). 2015. Available online: https://cran.r-project.org/web/packages/dotwhisker/vignettes/dotwhisker-vignette.html (accessed on 18 March 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org/ (accessed on 12 March 2021).
- Patil, I.; Powell, C. Ggstatsplot: ‘ggplot2’ Based Plots with Statistical Details. 2018. Available online: https://CRAN.R-project.org/package=ggstatsplot (accessed on 12 March 2021).
- Frith, J.; Zalewski, P.; Klawe, J.J.; Pairman, J.; Bitner, A.; Tafil-Klawe, M.; Newton, J.L. Impaired blood pressure variability in chronic fatigue syndrome—a potential biomarker. QJM Int. J. Med. 2012, 105, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.L.; Sheth, A.; Shin, J.; Pairman, J.; Wilton, K.; Burt, J.A.; Jones, D.E.J. Lower Ambulatory Blood Pressure in Chronic Fatigue Syndrome. Psychosom. Med. 2009, 71, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, I.B.; Maccallum, H.; Flint, L.; Cockcroft, J.R.; Newby, D.E.; Webb, D.J. The influence of heart rate on augmentation index and central arterial pressure in humans. J. Physiol. 2000, 525, 263–270. [Google Scholar] [CrossRef]
- Sunbul, M.; Kanar, B.G.; Durmus, E.; Kivrak, T.; Sari, I. Acute sleep deprivation is associated with increased arterial stiffness in healthy young adults. Sleep Breath. 2013, 18, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.; Scheibenbogen, C. A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Recognitions from the finding of autoantibodies against ß2-adrenergic receptors. Autoimmun. Rev. 2020, 19, 102527. [Google Scholar] [CrossRef]
- Yoshiuchi, K.; Farkas, J.; Natelson, B.H. Patients with chronic fatigue syndrome have reduced absolute cortical blood flow. Clin. Physiol. Funct. Imaging 2006, 26, 83–86. [Google Scholar] [CrossRef]
- Boissoneault, J.; Letzen, J.; Robinson, M.; Staud, R. Cerebral blood flow and heart rate variability predict fatigue severity in patients with chronic fatigue syndrome. Brain Imaging Behav. 2018, 13, 789–797. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Hollingsworth, K.G.; Newton, J.L.; Blamire, A.M. Cerebral vascular control is associated with skeletal muscle pH in chronic fatigue syndrome patients both at rest and during dynamic stimulation. NeuroImage Clin. 2013, 2, 168–173. [Google Scholar] [CrossRef]
- Cvejic, E.; Sandler, C.X.; Keech, A.; Barry, B.K.; Lloyd, A.R.; Vollmer-Conna, U. Autonomic nervous system function, activity patterns, and sleep after physical or cognitive challenge in people with chronic fatigue syndrome. J. Psychosom. Res. 2017, 103, 91–94. [Google Scholar] [CrossRef]
- Thomas, G.D. Neural control of the circulation. Adv. Physiol. Educ. 2011, 35, 28–32. [Google Scholar] [CrossRef]
- Estévez-López, F.; Mudie, K.; Wang-Steverding, X.; Bakken, I.J.; Ivanovs, A.; Castro-Marrero, J.; Nacul, L.; Alegre, J.; Zalewski, P.; Słomko, J.; et al. Systematic Review of the Epidemiological Burden of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Across Europe: Current Evidence and EUROMENE Research Recommendations for Epidemiology. J. Clin. Med. 2020, 9, 1557. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Jason, L.A. Meta-analysis investigating post-exertional malaise between patients and controls. J. Health Psychol. 2020, 25, 2053–2071. [Google Scholar] [CrossRef] [PubMed]

| Variable | Total Sample | PEM Mean ± SD | Without PEM Mean ± SD | p-Value | FDR p-Value |
|---|---|---|---|---|---|
| Age [years] | 38.15 ± 8.0 | 38.23 ± 8.1 | 37.79 ± 8.0 | 0.83 | 0.88 |
| Height [cm] | 171.55 ± 8.4 | 172.07 ± 8.7 | 169.32 ± 6.6 | 0.20 | 0.15 |
| Weight [kg] | 72.22 ± 12.6 | 72.77 ± 12.6 | 69.84 ± 12.5 | 0.36 | 0.61 |
| BMI | 24.47 ± 3.6 | 24.51 ± 3.5 | 24.31 ± 3.8 | 0.83 | 0.85 |
| Symptoms duration [years] | 4.54 ± 4.1 | 4.75 ± 4.2 | 3.64 ± 3.7 | 0.20 | 0.44 |
| CFQ [points] | 23.68 ± 4.6 | 24.07 ± 4.6 | 22.00 ± 4.4 | 0.08 | 0.21 |
| CFQ_BINARY [points] | 14.06 ± 4.8 | 14.49 ± 4.6 | 12.21 ± 5.1 | 0.07 | 0.22 |
| CFQ_PHYSICAL [points] | 10.47 ± 3.9 | 10.39 ± 4.0 | 10.79 ± 3.7 | 0.50 | 0.69 |
| CFQ_MENTAL [points] | 8.91 ± 2.0 | 9.13 ± 1.8 | 7.95 ± 2.2 | 0.03 | 0.11 |
| FSS [points] | 47.03 ± 10.1 | 47.57 ± 9.4 | 44.68 ± 12.8 | 0.46 | 0.68 |
| FIS [points] | 78.12 ± 31.6 | 81.61 ± 30.3 | 63.05 ± 33.9 | 0.02 | 0.09 |
| HADS_A [points] | 9.27 ± 3.5 | 9.22 ± 3.4 | 9.47 ± 4.2 | 0.78 | 0.87 |
| HADS_D [points] | 7.85 ± 3.4 | 8.00 ± 3.5 | 7.21 ± 2.9 | 0.36 | 0.63 |
| BDI [points] | 15.83 ± 7.9 | 16.15 ± 8.1 | 14.39 ± 6.9 | 0.57 | 0.75 |
| ESS [points] | 10.86 ± 5.6 | 11.01 ± 5.5 | 10.21 ± 6.3 | 0.65 | 0.78 |
| OGS [points] | 3.64 ± 3.2 | 3.71 ± 3.4 | 3.37 ± 2.5 | 0.98 | 0.98 |
| Orthostatic intolerance [points] | 11.96 ± 11.0 | 12.24 ± 11.3 | 10.74 ± 10.0 | 0.67 | 0.77 |
| Vasomotor [points] | 0.83 ± 1.4 | 0.86 ± 1.4 | 0.70 ± 1.4 | 0.57 | 0.73 |
| Secretomotor [points] | 5.52 ± 3.9 | 5.64 ± 3.9 | 4.96 ± 3.9 | 0.49 | 0.70 |
| Gastrointestinal [points] | 5.09 ± 4.2 | 4.89 ± 4.1 | 5.97 ± 4.7 | 0.37 | 0.60 |
| Bladder [points] | 0.54 ± 0.9 | 0.57 ± 1.0 | 0.41 ± 0.8 | 0.60 | 0.74 |
| Pupillomotor [points] | 1.15 ± 1.2 | 1.22 ± 1.2 | 0.86 ± 0.9 | 0.38 | 0.59 |
| Compass-31 Total [points] | 25.09 ± 14.7 | 25.43 ± 15.0 | 23.64 ± 13.4 | 0.81 | 0.88 |
| Variable | PEM Mean ± SD | Without PEM Mean ± SD | p-Value | FDR p-Value |
|---|---|---|---|---|
| PWVaortic [m/s] | 8.33 ± 1.7 | 8.65 ± 1.8 | 0.21 | 0.41 |
| Aixaortic [%] | 28.11 ± 14.4 | 32.95 ± 15.5 | 0.20 | 0.41 |
| sBPaortic [mmHg] | 127.49 ± 15 | 141.05 ± 21.1 | 0.02 | 0.12 |
| central-peripheral sBP [mmHg] | 9.56 ± 21.7 | 23.41 ± 25.7 | 0.06 | 0.20 |
| Variable | PEM Mean ± SD | Without PEM Mean ± SD | p-Value | FDR p-Value |
|---|---|---|---|---|
| Spectral analysis of HR variability | ||||
| LFnu-RRI | 56.61 ± 16.7 | 46.64 ± 13.9 | 0.01 | 0.12 |
| HFnu-RRI | 43.39 ± 16.7 | 53.36 ± 13.9 | 0.01 | 0.19 |
| LF/HF-RRI | 1.96 ± 2.2 | 1.13 ± 0.9 | 0.02 | 0.08 |
| LF/HF | 1.66 ± 1.5 | 0.94 ± 0.5 | 0.003 | 0.11 |
| Spectral analysis of BP variability | ||||
| LFnu-dBP | 53.88 ± 14.4 | 43.45 ± 13.8 | 0.01 | 0.09 |
| HFnu-dBP | 11.67 ± 7.3 | 18.20 ± 16.0 | 0.07 | 0.20 |
| LF/HF-dBP | 7.27 ± 6.0 | 4.32 ± 3.6 | 0.02 | 0.11 |
| LFnu-sBP | 42.86 ± 14.0 | 38.14 ± 13.7 | 0.19 | 0.44 |
| HFnu-sBP | 14.86 ± 9.3 | 18.74 ± 14.0 | 0.31 | 0.57 |
| LF/HF-sBP | 4.10 ± 2.8 | 3.02 ± 1.8 | 0.17 | 0.42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujawski, S.; Słomko, J.; Hodges, L.; Pheby, D.F.H.; Murovska, M.; Newton, J.L.; Zalewski, P. Post-Exertional Malaise May Be Related to Central Blood Pressure, Sympathetic Activity and Mental Fatigue in Chronic Fatigue Syndrome Patients. J. Clin. Med. 2021, 10, 2327. https://doi.org/10.3390/jcm10112327
Kujawski S, Słomko J, Hodges L, Pheby DFH, Murovska M, Newton JL, Zalewski P. Post-Exertional Malaise May Be Related to Central Blood Pressure, Sympathetic Activity and Mental Fatigue in Chronic Fatigue Syndrome Patients. Journal of Clinical Medicine. 2021; 10(11):2327. https://doi.org/10.3390/jcm10112327
Chicago/Turabian StyleKujawski, Sławomir, Joanna Słomko, Lynette Hodges, Derek F. H. Pheby, Modra Murovska, Julia L. Newton, and Paweł Zalewski. 2021. "Post-Exertional Malaise May Be Related to Central Blood Pressure, Sympathetic Activity and Mental Fatigue in Chronic Fatigue Syndrome Patients" Journal of Clinical Medicine 10, no. 11: 2327. https://doi.org/10.3390/jcm10112327
APA StyleKujawski, S., Słomko, J., Hodges, L., Pheby, D. F. H., Murovska, M., Newton, J. L., & Zalewski, P. (2021). Post-Exertional Malaise May Be Related to Central Blood Pressure, Sympathetic Activity and Mental Fatigue in Chronic Fatigue Syndrome Patients. Journal of Clinical Medicine, 10(11), 2327. https://doi.org/10.3390/jcm10112327









