Opioid-Free Anesthesia Benefit–Risk Balance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
:1. Introduction
2. Methods
2.1. Data Sources and Search Strategy
2.2. Eligibility Criteria
2.3. Data Extraction and Assessment of the Risk of Bias
2.4. Outcome Measures
2.5. Data Synthesis and Analysis
2.6. Strength of Evidence
3. Result
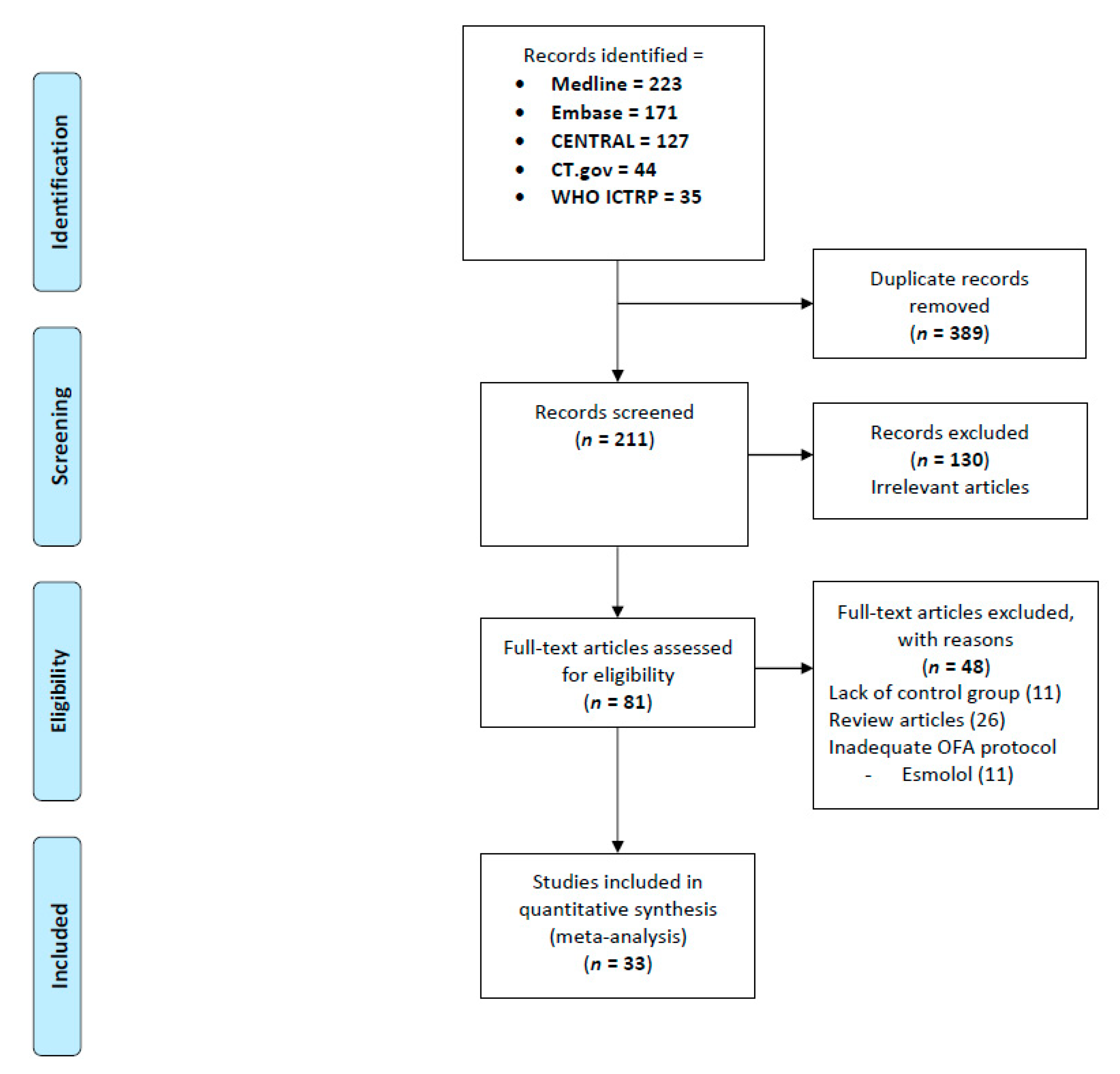
3.1. Search Results
3.2. Trial, Participants, and Intervention Characteristics
3.3. Risk of Bias Assessment of the Included Studies
3.4. Pain Intensity
3.5. Postoperative Morphine Use
3.6. Opioid-Related Adverse Events
3.7. Adverse Effects Linked to Substitution Drug
3.8. Severe Adverse Events
4. Discussion
4.1. No Clinical Benefit of Opioid-Free Anesthesia on Pain and Morphine Use after Surgery
4.2. OFA Is Associated with an Important Reduction in the Incidence of PONV and a Reduction in Sedation and Shivering
4.3. Other Evidences on the Potential Benefits Related to OFA
4.4. OFA May Be Responsible for Additional Side Effects
4.5. Strengths and Limitation of Our Meta-Analysis
4.6. Should We Use OFA?
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Egan, T.D. Are opioids indispensable for general anaesthesia? Br. J. Anaesth. 2019, 122, e127–e135. [Google Scholar] [CrossRef] [PubMed]
- Koepke, E.J.; Manning, E.L.; Miller, T.E.; Ganesh, A.; Williams, D.G.A.; Manning, M.W. The rising tide of opioid use and abuse: The role of the anesthesiologist. Perioper. Med. 2018, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Beloeil, H. Opioid-free anesthesia. Best Pract. Res. Clin. Anaesthesiol. 2019, 33, 353–360. [Google Scholar] [CrossRef]
- Frauenknecht, J.; Kirkham, K.R.; Jacot-Guillarmod, A.; Albrecht, E. Analgesic impact of intra-operative opioids vs. opioid-free anaesthesia: A systematic review and meta-analysis. Anaesthesia 2019, 74, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Grape, S.; Kirkham, K.R.; Frauenknecht, J.; Albrecht, E. Intra-operative analgesia with remifentanil vs. dexmedetomidine: A systematic review and meta-analysis with trial sequential analysis. Anaesthesia 2019, 74, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, E.; Grape, S.; Frauenknecht, J.; Kilchoer, L.; Kirkham, K.R. Low- versus high-dose intraoperative opioids: A systematic review with meta-analyses and trial sequential analyses. Acta Anaesthesiol. Scand. 2020, 64, 6–22. [Google Scholar] [CrossRef]
- Beloeil, H.; Garot, M.; Martinez, V. Comments on Albrecht et al. reviews. Anaesthesia 2019, 74, 1195–1196. [Google Scholar] [CrossRef] [Green Version]
- Demiri, M.; Antunes, T.; Fletcher, D.; Martinez, V. Perioperative adverse events attributed to alpha2-adrenoceptor agonists in patients not at risk of cardiovascular events: Systematic review and meta-analysis. Br. J. Anaesth. 2019, 123, 795–807. [Google Scholar] [CrossRef]
- Beloeil, H.; Laviolle, B.; Menard, C.; Paugam-Burtz, C.; Garot, M.; Asehnoune, K.; Minville, V.; Cuvillon, P.; Oger, S.; Nadaud, J.; et al. POFA trial study protocol: A multicentre, double-blind, randomised, controlled clinical trial comparing opioid-free versus opioid anaesthesia on postoperative opioid-related adverse events after major or intermediate non-cardiac surgery. BMJ Open 2018, 8, e020873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [Green Version]
- Visser, K.; Hassink, E.A.; Bonsel, G.J.; Moen, J.; Kalkman, C.J. Randomized controlled trial of total intravenous anesthesia with propofol versus inhalation anesthesia with isoflurane-nitrous oxide: Postoperative nausea with vomiting and economic analysis. Anesthesiology 2001, 95, 616–626. [Google Scholar] [CrossRef]
- Apfel, C.C.; Turan, A.; Souza, K.; Pergolizzi, J.; Hornuss, C. Intravenous acetaminophen reduces postoperative nausea and vomiting: A systematic review and meta-analysis. Pain 2013, 154, 677–689. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0 (Updated February 2011); Cochrane, 2011; Available online: www.cochrane-handbook.org (accessed on 12 May 2021).
- Higgins, J.P.; White, I.R.; Wood, A.M. Imputation methods for missing outcome data in meta-analysis of clinical trials. Clin. Trials 2008, 5, 225–239. [Google Scholar] [CrossRef]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef] [Green Version]
- Verret, M.; Lauzier, F.; Zarychanski, R.; Perron, C.; Savard, X.; Pinard, A.M.; Leblanc, G.; Cossi, M.J.; Neveu, X.; Turgeon, A.F.; et al. Perioperative Use of Gabapentinoids for the Management of Postoperative Acute Pain: A Systematic Review and Meta-analysis. Anesthesiology 2020, 133, 265–279. [Google Scholar] [CrossRef]
- Nielsen, S.; Degenhardt, L.; Hoban, B.; Gisev, N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharm. Drug Saf. 2016, 25, 733–737. [Google Scholar] [CrossRef]
- Laigaard, J.; Pedersen, C.; Ronsbo, T.N.; Mathiesen, O.; Karlsen, A.P.H. Minimal clinically important differences in randomised clinical trials on pain management after total hip and knee arthroplasty: A systematic review. Br. J. Anaesth. 2021. [Google Scholar] [CrossRef]
- Tsertsvadze, A.; Maglione, M.; Chou, R.; Garritty, C.; Coleman, C.; Lux, L.; Bass, E.; Balshem, H.; Moher, D. Updating comparative effectiveness reviews: Current efforts in AHRQ’s Effective Health Care Program. J. Clin. Epidemiol. 2011, 64, 1208–1215. [Google Scholar] [CrossRef] [PubMed]
- Bakan, M.; Umutoglu, T.; Topuz, U.; Uysal, H.; Bayram, M.; Kadioglu, H.; Salihoglu, Z. Opioid-free total intravenous anesthesia with propofol, dexmedetomidine and lidocaine infusions for laparoscopic cholecystectomy: A prospective, randomized, double-blinded study. Braz. J. Anesthesiol. 2015, 65, 191–199. [Google Scholar] [CrossRef] [Green Version]
- Choi, J.W.; Joo, J.D.; Kim, D.W.; In, J.H.; Kwon, S.Y.; Seo, K.; Han, D.; Cheon, G.Y.; Jung, H.S. Comparison of an Intraoperative Infusion of Dexmedetomidine, Fentanyl, and Remifentanil on Perioperative Hemodynamics, Sedation Quality, and Postoperative Pain Control. J. Korean Med. Sci. 2016, 31, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.K.; Seo, Y.; Lim, D.G.; Park, S. Postoperative nausea and vomiting after thyroidectomy: A comparison between dexmedetomidine and remifentanil as part of balanced anesthesia. Korean J. Anesthesiol. 2017, 70, 299–304. [Google Scholar] [CrossRef] [Green Version]
- Curry, C.S.; Darby, J.R.; Janssen, B.R. Evaluation of pain following electrocautery tubal ligation and effect of intraoperative fentanyl. J. Clin. Anesth. 1996, 8, 216–219. [Google Scholar] [CrossRef]
- Feld, J.M.; Hoffman, W.E.; Stechert, M.M.; Hoffman, I.W.; Ananda, R.C. Fentanyl or dexmedetomidine combined with desflurane for bariatric surgery. J. Clin. Anesth. 2006, 18, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Goyal, S.; Gupta, K.K.; Mahajan, V. A Comparative Evaluation of Intravenous Dexmedetomidine and Fentanyl in Breast Cancer Surgery: A Prospective, Randomized, and Controlled Trial. Anesth. Essays Res. 2017, 11, 611–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, W.; Lee, J.; Park, J.; Joo, J. Dexmedetomidine versus remifentanil in postoperative pain control after spinal surgery: A randomized controlled study. BMC Anesthesiol. 2015, 15, 21. [Google Scholar] [CrossRef] [Green Version]
- Senol Karatas, S.; Eti, Z.; Saracoglu, K.T.; Gogus, F.Y. Does perioperative opioid infusion increase postoperative opioid requirement? AGRI 2015, 27, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.; Clairoux, M.; Redahan, C.; Kavanagh, B.P.; Carroll, S.; Nierenberg, H.; Jackson, M.; Beattie, J.; Taddio, A.; Sandler, A.N. High dose alfentanil pre-empts pain after abdominal hysterectomy. Pain 1996, 68, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Kim, Y.D.; Kim, J.N. Antihyperalgesic effects of dexmedetomidine on high-dose remifentanil-induced hyperalgesia. Korean J. Anesthesiol. 2013, 64, 301–307. [Google Scholar] [CrossRef] [Green Version]
- Hontoir, S.; Saxena, S.; Gatto, P.; Khalife, M.; Ben Aziz, A.M.; Paesmans, M.; Sosnowski, M. Opioid-free anesthesia: What about patient comfort? A prospective, randomized, controlled trial. Acta Anaesthesiol. Belg. 2016, 67, 183–190. [Google Scholar]
- Beloeil, H.; Garot, M.; Lebuffe, G.; Gerbaud, A.; Bila, J.; Cuvillon, P.; Dubout, E.; Oger, S.; Nadaud, J.; Becret, A.; et al. Balanced Opioid-free Anesthesia with Dexmedetomidine versus Balanced Anesthesia with Remifentanil for Major or Intermediate Noncardiac Surgery. Anesthesiology 2021, 134, 541–551. [Google Scholar] [CrossRef]
- Salman, N.; Uzun, S.; Coskun, F.; Salman, M.A.; Salman, A.E.; Aypar, U. Dexmedetomidine as a substitute for remifentanil in ambulatory gynecologic laparoscopic surgery. Saudi Med. J. 2009, 30, 77–81. [Google Scholar]
- Toleska, M.; Dimitrovski, A. Is Opioid-Free General Anesthesia More Superior for Postoperative Pain Versus Opioid General Anesthesia in Laparoscopic Cholecystectomy? Prilozi 2019, 40, 81–87. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.; Song, Y.K.; Lee, J.H.; Ha, S.M. The effects of intraoperative adenosine infusion on acute opioid tolerance and opioid induced hyperalgesia induced by remifentanil in adult patients undergoing tonsillectomy. Korean J. Pain 2011, 24, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, S.; Garg, K.; Devgan, S. Comparison of opioid-based and opioid-free TIVA for laparoscopic urological procedures in obese patients. J. Anaesthesiol. Clin. Pharmacol. 2019, 35, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Gazi, M.; Abitagaoglu, S.; Turan, G.; Koksal, C.; Akgun, F.N.; Ari, D.E. Evaluation of the effects of dexmedetomidine and remifentanil on pain with the analgesia nociception index in the perioperative period in hysteroscopies under general anesthesia. A randomized prospective study. Saudi Med. J. 2018, 39, 1017–1022. [Google Scholar] [CrossRef]
- Hakim, K.Y.K.; Wahba, W.Z.B. Opioid-Free Total Intravenous Anesthesia Improves Postoperative Quality of Recovery after Ambulatory Gynecologic Laparoscopy. Anesth. Essays Res. 2019, 13, 199–203. [Google Scholar] [CrossRef]
- Kataria, A.P.; Attri, J.P.; Kashyap, R.; Mahajan, L. Efficacy of dexmedetomidine and fentanyl on pressor response and pneumoperitoneum in laparoscopic cholecystectomy. Anesth. Essays Res. 2016, 10, 446–450. [Google Scholar] [CrossRef] [Green Version]
- Mansour, M.A.; Mahmoud, A.A.; Geddawy, M. Nonopioid versus opioid based general anesthesia technique for bariatric surgery: A randomized double-blind study. Saudi J. Anaesth. 2013, 7, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.H.; Sohn, I.S.; Do, S.H. Controlled hypotension for middle ear surgery: A comparison between remifentanil and magnesium sulphate. Br. J. Anaesth. 2009, 103, 490–495. [Google Scholar] [CrossRef] [Green Version]
- Techanivate, A.; Dusitkasem, S.; Anuwattanavit, C. Dexmedetomidine compare with fentanyl for postoperative analgesia in outpatient gynecologic laparoscopy: A randomized controlled trial. J. Med. Assoc. Thai 2012, 95, 383–390. [Google Scholar] [PubMed]
- Tverskoy, M.; Oz, Y.; Isakson, A.; Finger, J.; Bradley, E.L., Jr.; Kissin, I. Preemptive effect of fentanyl and ketamine on postoperative pain and wound hyperalgesia. Anesth. Analg. 1994, 78, 205–209. [Google Scholar] [CrossRef]
- Bulow, N.M.; Barbosa, N.V.; Rocha, J.B. Opioid consumption in total intravenous anesthesia is reduced with dexmedetomidine: A comparative study with remifentanil in gynecologic videolaparoscopic surgery. J. Clin. Anesth. 2007, 19, 280–285. [Google Scholar] [CrossRef]
- Inoue, Y.; Koga, K.; Sata, T.; Shigematsu, A. Effects of fentanyl on emergence characteristics from anesthesia in adult cervical spine surgery: A comparison of fentanyl-based and sevoflurane-based anesthesia. J. Anesth. 2005, 19, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Javaherforooshzadeh, F.; Monajemzadeh, S.A.; Soltanzadeh, M.; Janatmakan, F.; Salari, A.; Saeed, H. A Comparative Study of the Amount of Bleeding and Hemodynamic Changes between Dexmedetomidine Infusion and Remifentanil Infusion for Controlled Hypotensive Anesthesia in Lumbar Discopathy Surgery: A Double-Blind, Randomized, Clinical Trial. Anesth. Pain Med. 2018, 8, e66959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortinez, L.I.; Brandes, V.; Munoz, H.R.; Guerrero, M.E.; Mur, M. No clinical evidence of acute opioid tolerance after remifentanil-based anaesthesia. Br. J. Anaesth. 2001, 87, 866–869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, H.S.; Joo, J.D.; Jeon, Y.S.; Lee, J.A.; Kim, D.W.; In, J.H.; Rhee, H.Y.; Choi, J.W. Comparison of an intraoperative infusion of dexmedetomidine or remifentanil on perioperative haemodynamics, hypnosis and sedation, and postoperative pain control. J. Int. Med. Res. 2011, 39, 1890–1899. [Google Scholar] [CrossRef] [Green Version]
- Mogahed, M.; Anwar, A. The Effects of Dexmedetomidine or Remifentanil Continuous Infusion on End- Tidal Sevoflurane Concentration in Patients Undergoing Laparoscopic Cholecestectomies, Monitored by Bispectral Analysis. J. Anesth. Clin. Res. 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Mulier, J.; Wouters, R.; Dillemans, B.; Dekock, M. A Randomized Controlled, Double-Blind Trial Evaluating the Effect of Opioid-Free Versus Opioid General Anaesthesia on Postoperative Pain and Discomfort Measured by the QoR-40. J. Clin. Anesth. Pain Med. 2018, 2, 6. [Google Scholar]
- Subasi, H.; Kol, I.; Kaygusuz, K.I.; Isbir, A.C.; Gursoy, S. Dexmedetomidine and remifentanil as adjuncts to total intravenous anesthesia with propofol. Pain Intensive Care 2019, 20, 323–329. [Google Scholar]
- Sahoo, J.; Sujata, P. Comparative study betweeen dexmedetomidine and remifentanyl for efficient pain and ponv management in propofol based total intravenous anesthesia after laparoscopic gynaecological surgeries. Int. J. Pharm. Sci. Rev. Res. 2016, 36, 212–216. [Google Scholar]
- Hossam, M. Intravenous Dexmedetomidine Infusion Compared with that of Fentanyl in Patients UndergoingArthroscopic Shoulder Surgery under General Anesthesia. Anesth. Essays Res. 2017, 11, 1070–1074. [Google Scholar]
- Marret, E.; Kurdi, O.; Zufferey, P.; Bonnet, F. Effects of nonsteroidal antiinflammatory drugs on patient-controlled analgesia morphine side effects: Meta-analysis of randomized controlled trials. Anesthesiology 2005, 102, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- Martinez, V.; Beloeil, H.; Marret, E.; Fletcher, D.; Ravaud, P.; Trinquart, L. Non-opioid analgesics in adults after major surgery: Systematic review with network meta-analysis of randomized trials. Br. J. Anaesth. 2017, 118, 22–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fletcher, D.; Martinez, V. Opioid-induced hyperalgesia in patients after surgery: A systematic review and a meta-analysis. Br. J. Anaesth. 2014, 112, 991–1004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apfel, C.C.; Korttila, K.; Abdalla, M.; Kerger, H.; Turan, A.; Vedder, I.; Zernak, C.; Danner, K.; Jokela, R.; Pocock, S.J.; et al. A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. N. Engl. J. Med. 2004, 350, 2441–2451. [Google Scholar] [CrossRef] [Green Version]
- Tramer, M.R. A rational approach to the control of postoperative nausea and vomiting: Evidence from systematic reviews. Part II. Recommendations for prevention and treatment, and research agenda. Acta Anaesthesiol. Scand. 2001, 45, 14–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harkouk, H.; Fletcher, D.; Beloeil, H. Opioid free anaesthesia: Myth or reality? Anaesth. Crit. Care Pain Med. 2019, 38, 111–112. [Google Scholar] [CrossRef] [PubMed]
- Kharasch, E.D.; Clark, J.D. Opioid-free Anesthesia: Time to Regain Our Balance. Anesthesiology 2021, 134, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Martinez, V.; Fletcher, D. Prevention of opioid-induced hyperalgesia in surgical patients: Does it really matter? Br. J. Anaesth. 2012, 109, 302–304. [Google Scholar] [CrossRef] [Green Version]


| Publication year, median (range). | 2015 (1994–2021) |
| Single-center trials, n (%) | 32 (96) |
| Trial size, median (range) | 60 (27–316) |
| Continent of origin | |
| Europe, n (%) | 9 (27) |
| Asia, n (%) | 15 (45) |
| North America, n (%) | 3 (9) |
| South America, n (%) | 2 (6) |
| Africa, n (%) | 4 (12) |
| Type of surgery | |
| Abdominal, n (%) | 9 (27) |
| Uro-gynecological, n (%) | 13 (39) |
| Orthopedic, n (%) | 1 (3) |
| Neurological, n (%) | 3 (9) |
| ENT, n (%) | 4 (12) |
| Thoracic, n (%) | 2 (6) |
| Mixed, n (%) | 1 (3) |
| Anesthesia maintenance | |
| Propofol, n (%) | 9 (27%) |
| Halogenated agent, n (%) | 25 (76%) |
| OFA group | |
| Active drug, n (%) | 24 (72) |
| Dexmedetomidine and Lidocaine combined | 22 (66) 4 |
| Ketamine, n (%) | 2 |
| Magnesium, n (%) | 1 |
| Combination, n (%) | 3 * |
| Clonidine, n (%) | 1 |
| Saline, n (%) | 7 (21) |
| Outcomes | Studies | Patients | Effect Size (95% CI) or | Heterogeneity, I2 (%) | NNTH (95% CI) | GRADE |
|---|---|---|---|---|---|---|
| Morphine consumption | ||||||
| PACU | 13 | 551 | WMD −1.61 (−2.69, −0.53) | 86% | ⊗⊗⊗ Z | |
| 24 h | 10 | 427 | WMD −1.73 (−2.82, −0.65) | 73% | ⊗⊗⊗ Z | |
| 48 h | 5 | 318 | WMD −3.14 (−10.34, 4.05) | 88% | ⊗⊗⊗ Z | |
| Pain score | ||||||
| PACU | 26 | 1568 | −0.75 (−1.18, −0.32) | 92% | ⊗⊗⊗ Z | |
| 24 h | 11 | 487 | 0.11 (−0.38, 0.60) | 91% | ⊗⊗⊗ Z | |
| 48 h | 5 | 158 | 0.16 (−0.09, 0.41) | 53% | ⊗⊗ | |
| Intraoperative Incidence | ||||||
| Tachycardia | 2 | 125 | RR 0.89 (0.51; 1.54) | 0% | ---- | ⊗ *Y |
| Bradycardia | 8 | 745 | RR 0.86 (0.39; 1.92) | 55% | ---- | ⊗⊗ Z |
| Hypertension | 4 | 505 | RR 1.07 (0.59; 1.93) | 56% | ---- | ⊗⊗ YZ |
| Hypotension | 10 | 845 | RR 0.92 (0.79; 1.08) | 46% | ---- | ⊗⊗⊗ |
| Postoperative Incidence | ||||||
| Tachycardia | 0 | 0 | Not estimable | ---- | ---- | |
| Bradycardia | 5 | 310 | RR 1.65 (0.67; 4.08) | 0% | ---- | ⊗⊗ * |
| Hypertension | 0 | 0 | Not estimable | ---- | ---- | |
| Hypotension | 3 | 200 | RR 1.11 (0.21; 5.76) | 45% | ---- | ⊗⊗⊗ *Y |
| Nausea, PACU | 20 | 1521 | RR 0.46 (0.38; 0.56) | 9% | 3 (3; 4) | ⊗⊗⊗⊗ |
| Nausea, H24 | 6 | 450 | RR 0.55 (0.32; 0.95) | 71% | 2 (2; 4) | ⊗⊗⊗ Z |
| Vomiting, PACU | 12 | 788 | RR 0.34 (0.21; 0.56) | 16% | 9 (8; 13) | ⊗⊗⊗⊗ |
| Shivering | 9 | 581 | RR 0.48 (0.33; 0.70) | 12% | 6 (5; 10) | ⊗⊗⊗ |
| Respiratory depression | 5 | 557 | RR 0.51 (0.07; 3.63) | 87% | ---- | ⊗⊗ Z* |
| Cognitive dysfunction | 1 | 308 | RR 5.06 (0.25; 105) | --- | ---- | ---- |
| Sedation | 6 | 336 | SMD −0.81 (−1.05, −0.58) | 8% | ||
| Serious adverse events | ||||||
| Requiring drug administration | 8 | 691 | RR 1.23 (0.71; 2.15) | 49% | ---- | ⊗⊗⊗ |
| Reported as SAE | 3 | 417 | RR 1.47 (0.87; 2.48) | 84% | ---- | ⊗⊗⊗ *YZ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salomé, A.; Harkouk, H.; Fletcher, D.; Martinez, V. Opioid-Free Anesthesia Benefit–Risk Balance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2021, 10, 2069. https://doi.org/10.3390/jcm10102069
Salomé A, Harkouk H, Fletcher D, Martinez V. Opioid-Free Anesthesia Benefit–Risk Balance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Journal of Clinical Medicine. 2021; 10(10):2069. https://doi.org/10.3390/jcm10102069
Chicago/Turabian StyleSalomé, Arthur, Hakim Harkouk, Dominique Fletcher, and Valeria Martinez. 2021. "Opioid-Free Anesthesia Benefit–Risk Balance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Journal of Clinical Medicine 10, no. 10: 2069. https://doi.org/10.3390/jcm10102069
APA StyleSalomé, A., Harkouk, H., Fletcher, D., & Martinez, V. (2021). Opioid-Free Anesthesia Benefit–Risk Balance: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Journal of Clinical Medicine, 10(10), 2069. https://doi.org/10.3390/jcm10102069






