Characterization and Assessment of a Novel Plate and Frame MD Module for Single Pass Wastewater Concentration–FEED Gap Air Gap Membrane Distillation
Abstract
1. Introduction
- (I)
- Implementing a plate and frame module design with replaceable inner parts;
- (II)
- Minimization of the number of components in contact with the highly corrosive feed solution;
- (III)
- Enabling of a single pass recovery rate high enough to operate the MD stage as a one- step process.
2. Materials and Methods
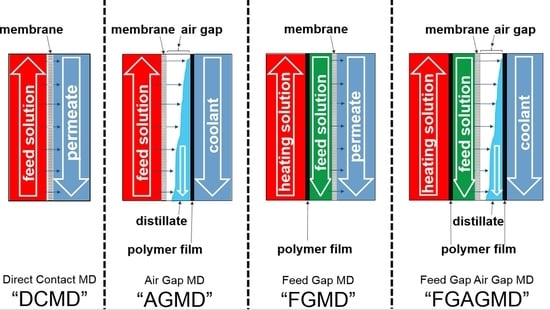
2.1. Membrane Distillation (MD) Process and the Evolution of Channel Configurations
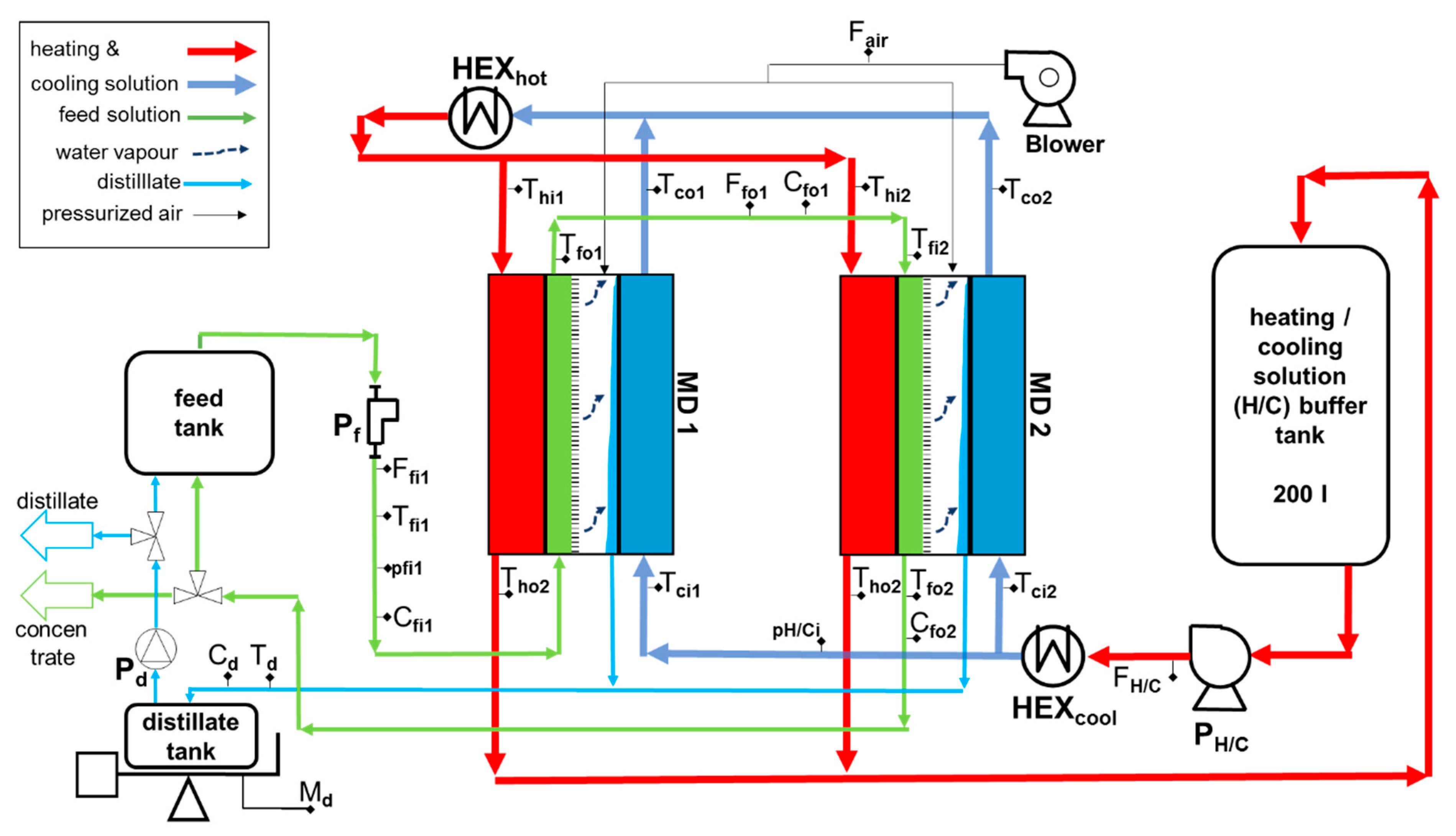
2.2. Module Design
2.3. Experimental Set-Up
2.3.1. Feed Loop
2.3.2. Heating and Cooling Loop
2.3.3. Distillate
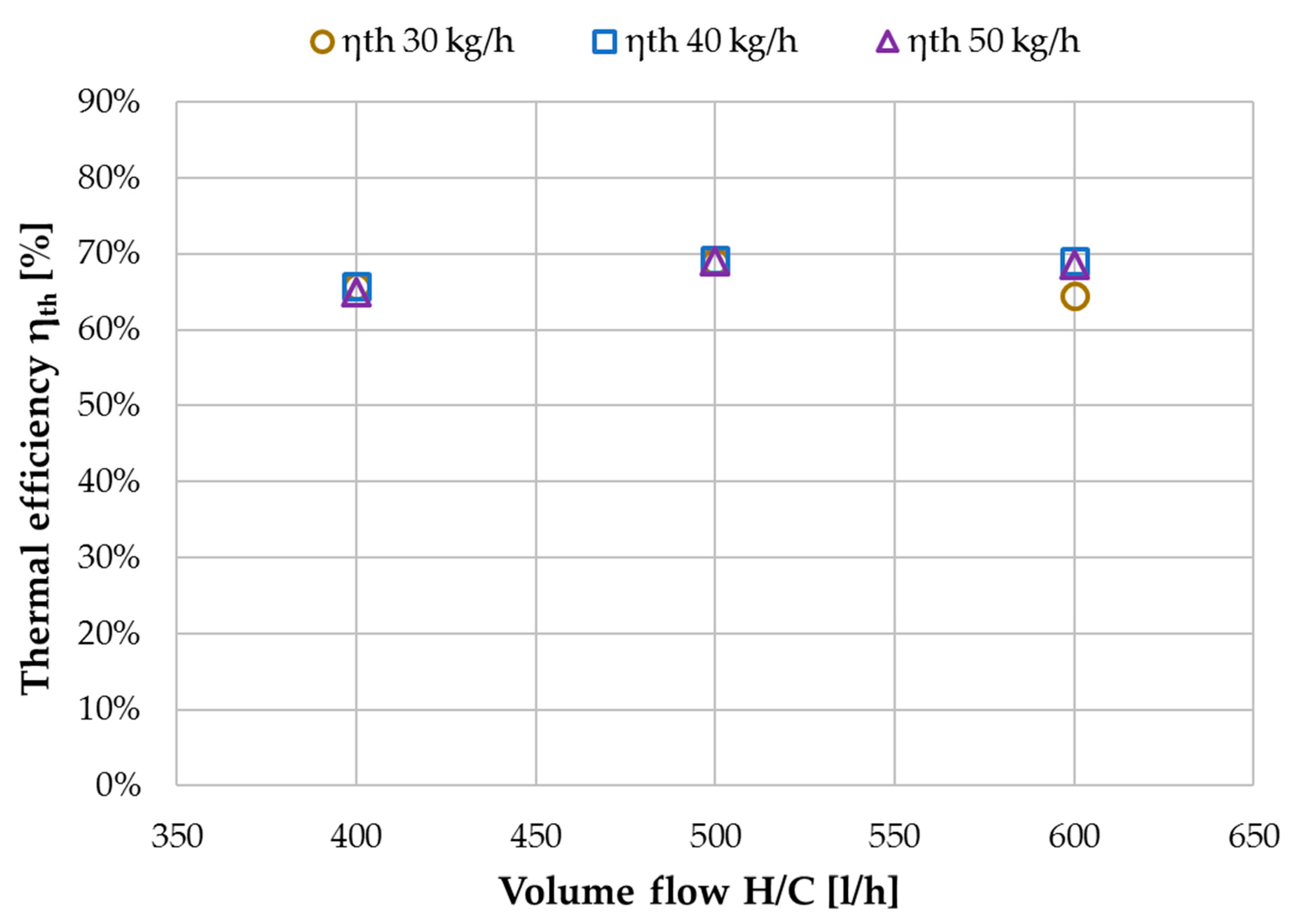
2.4. Key Performance Indicators
3. Results
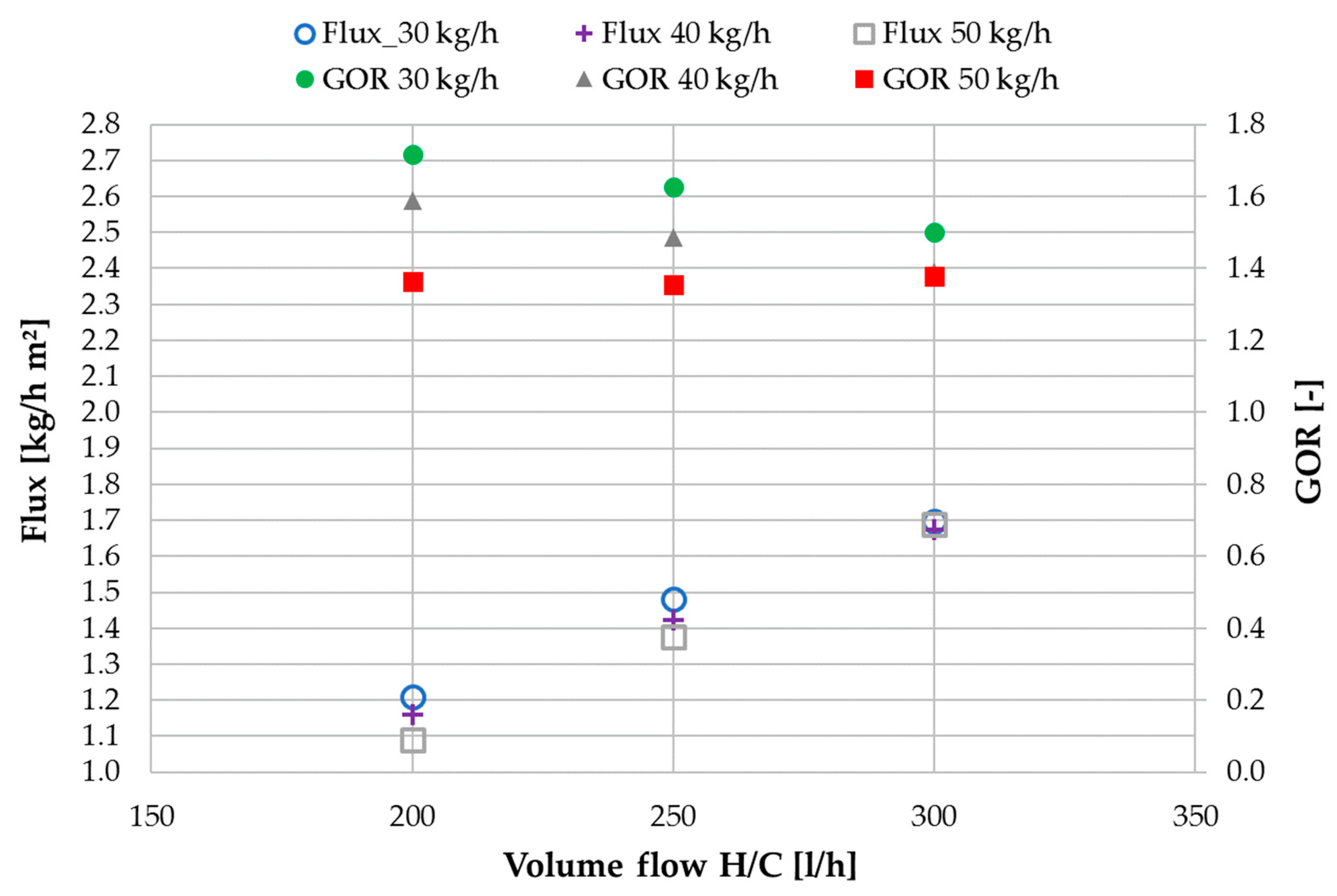
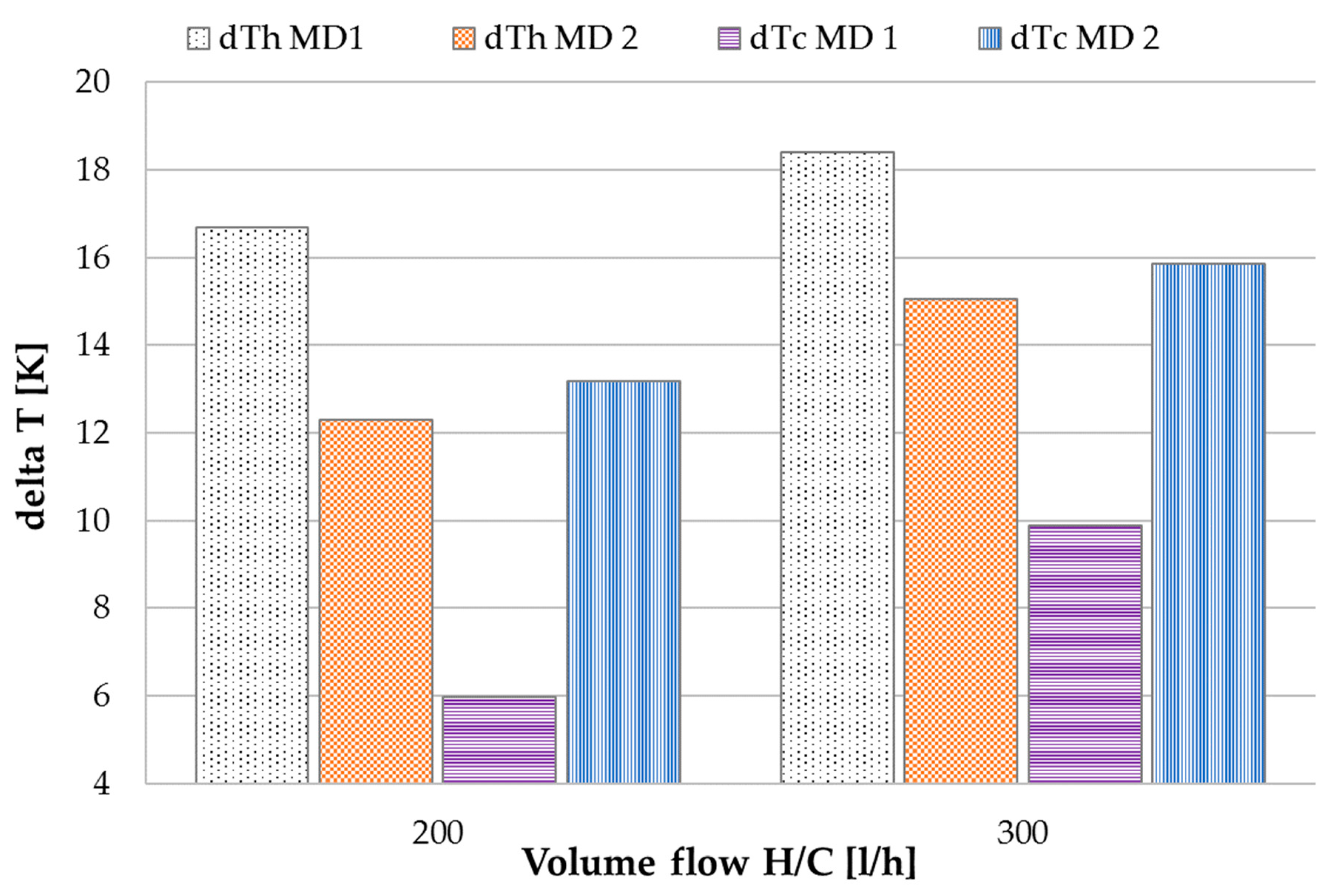
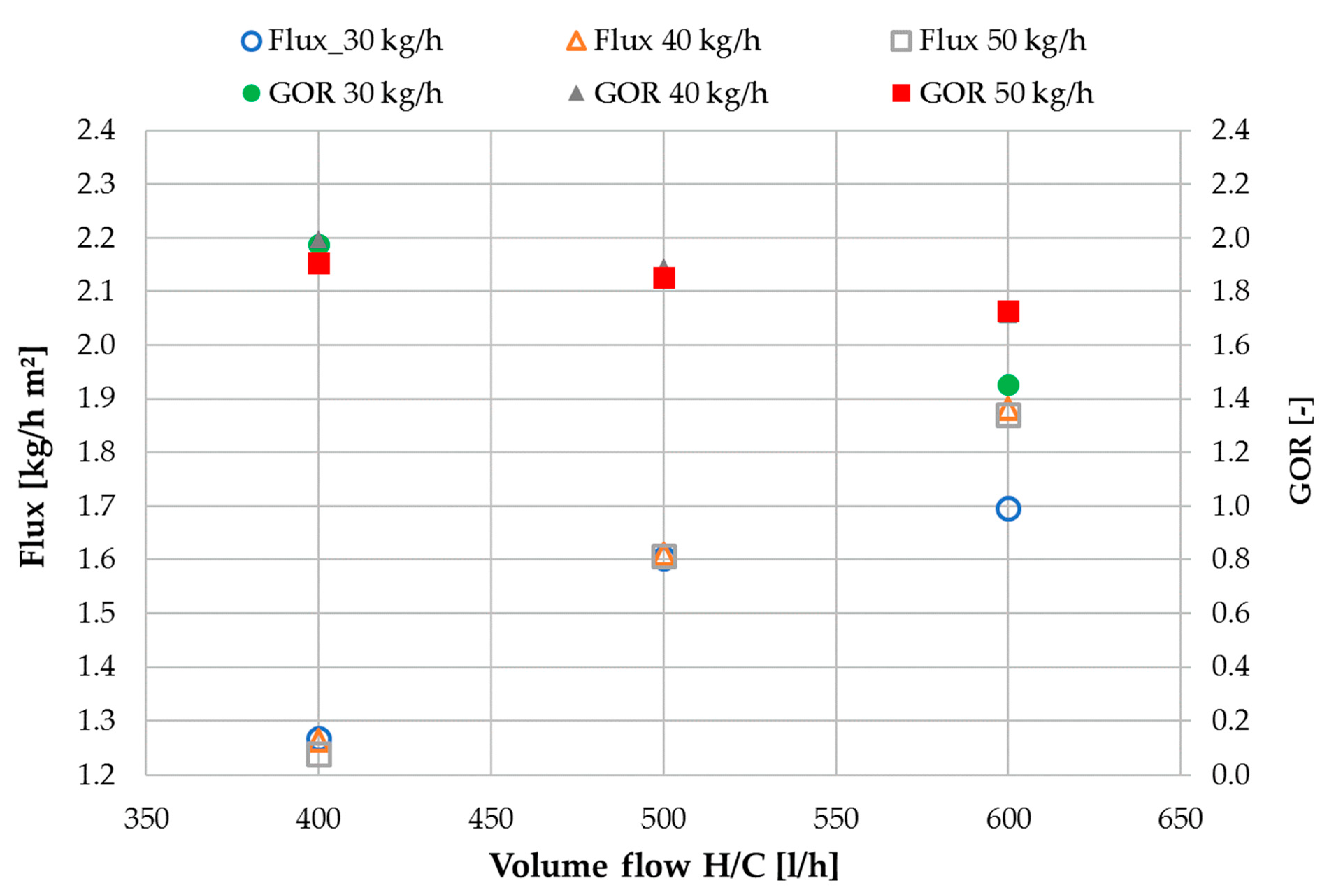
3.1. Performance Characterization with Tap Water
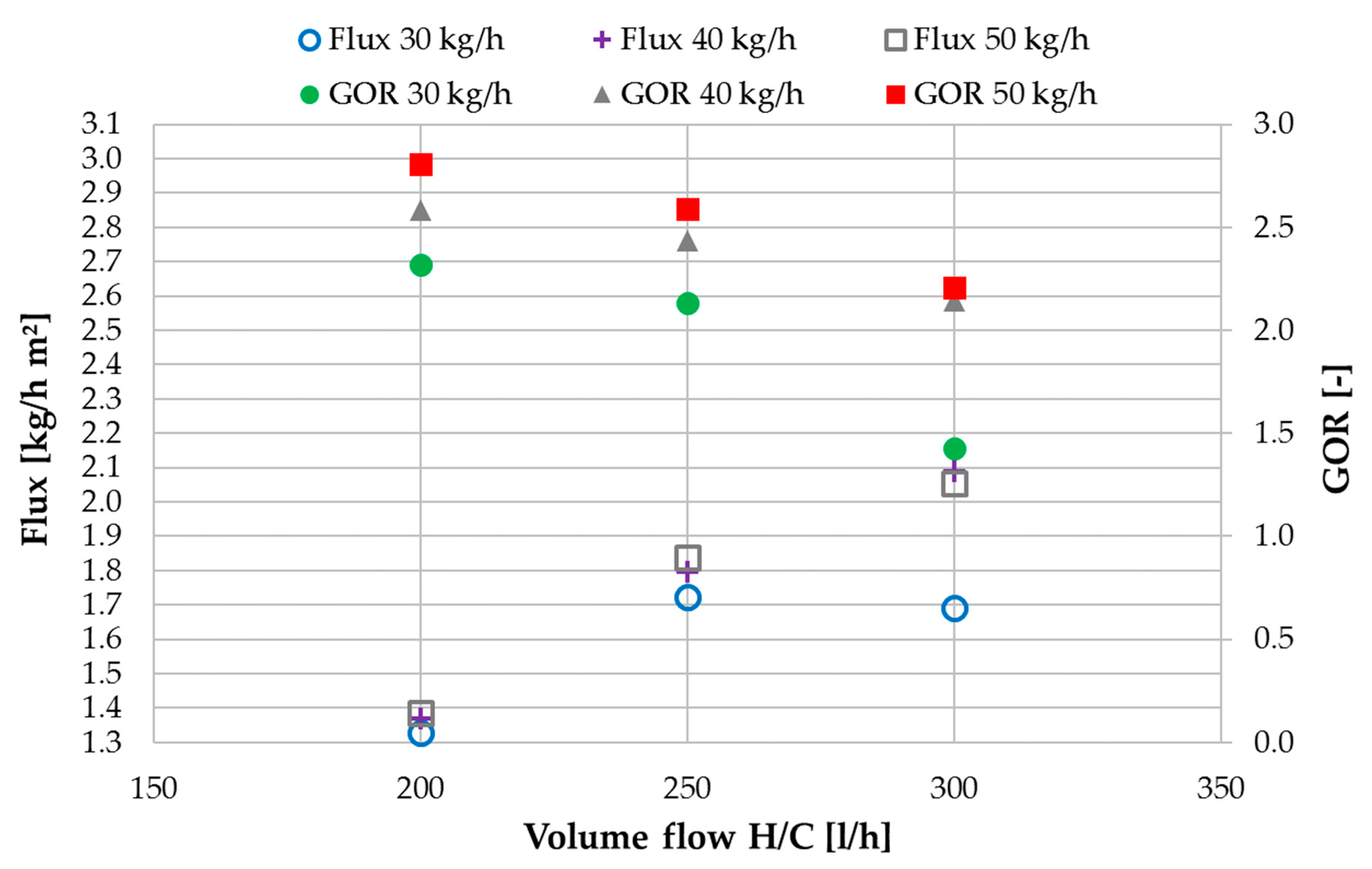
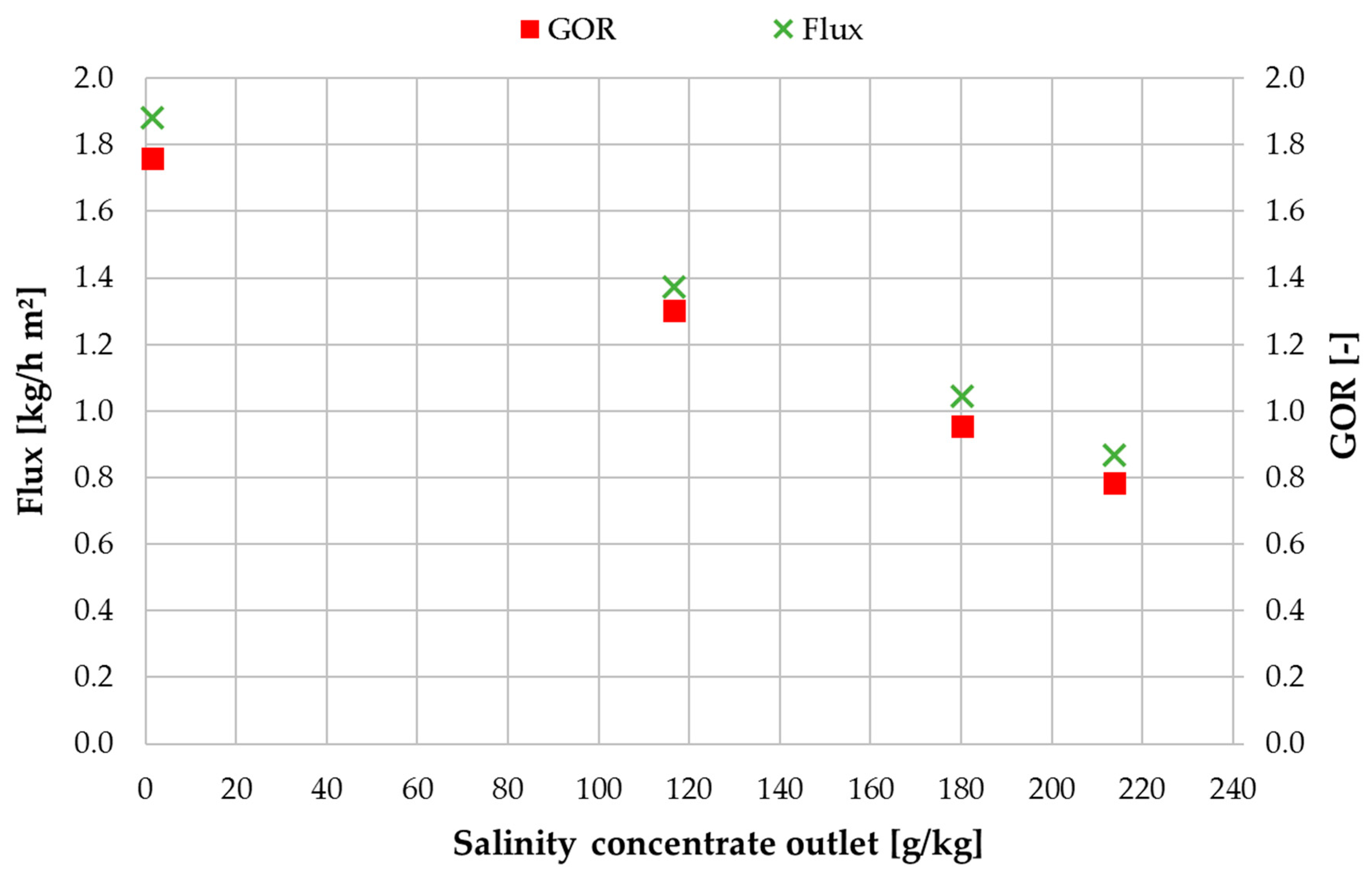
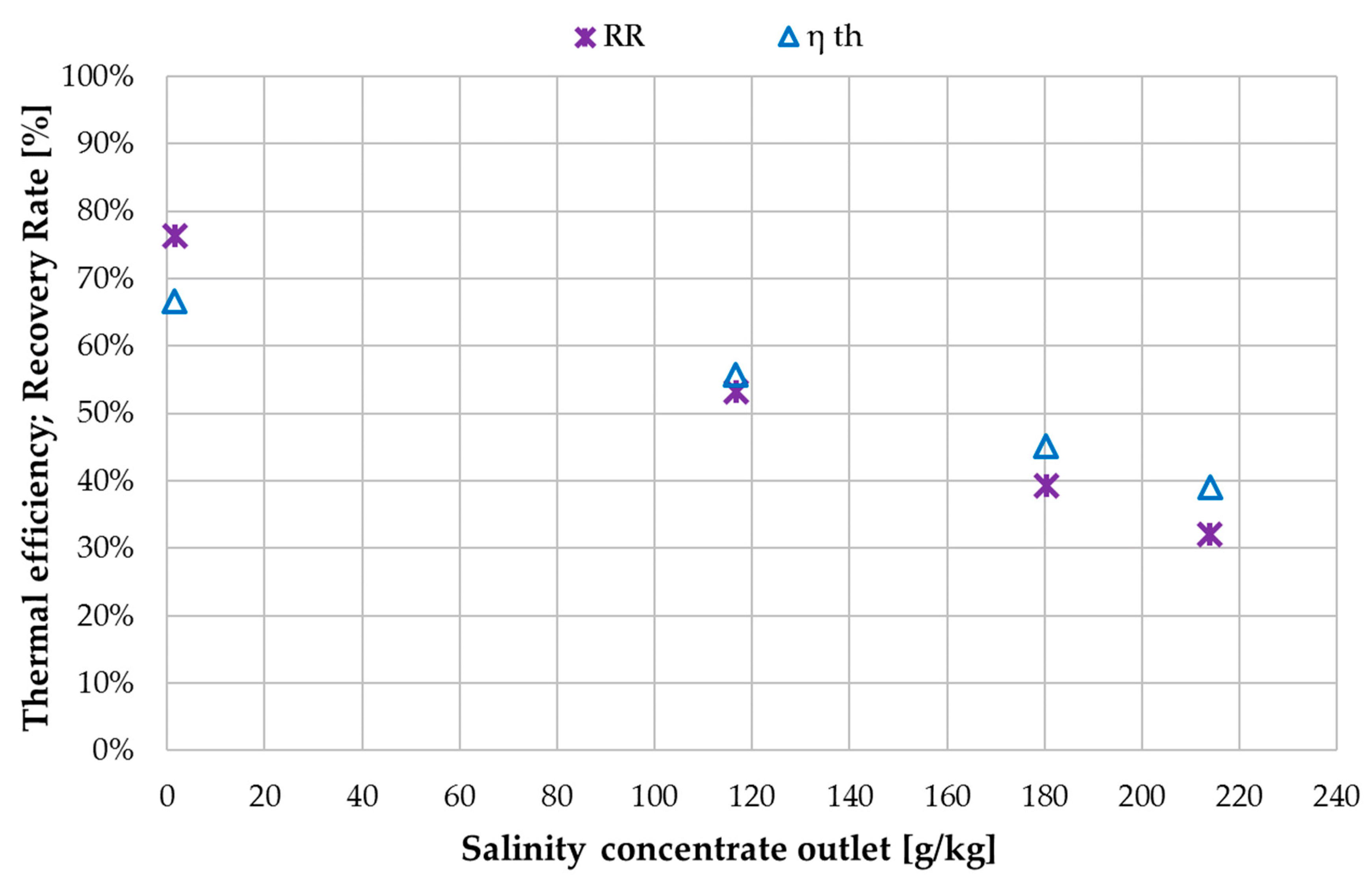
3.2. Performance Characterization with NaCl Solution
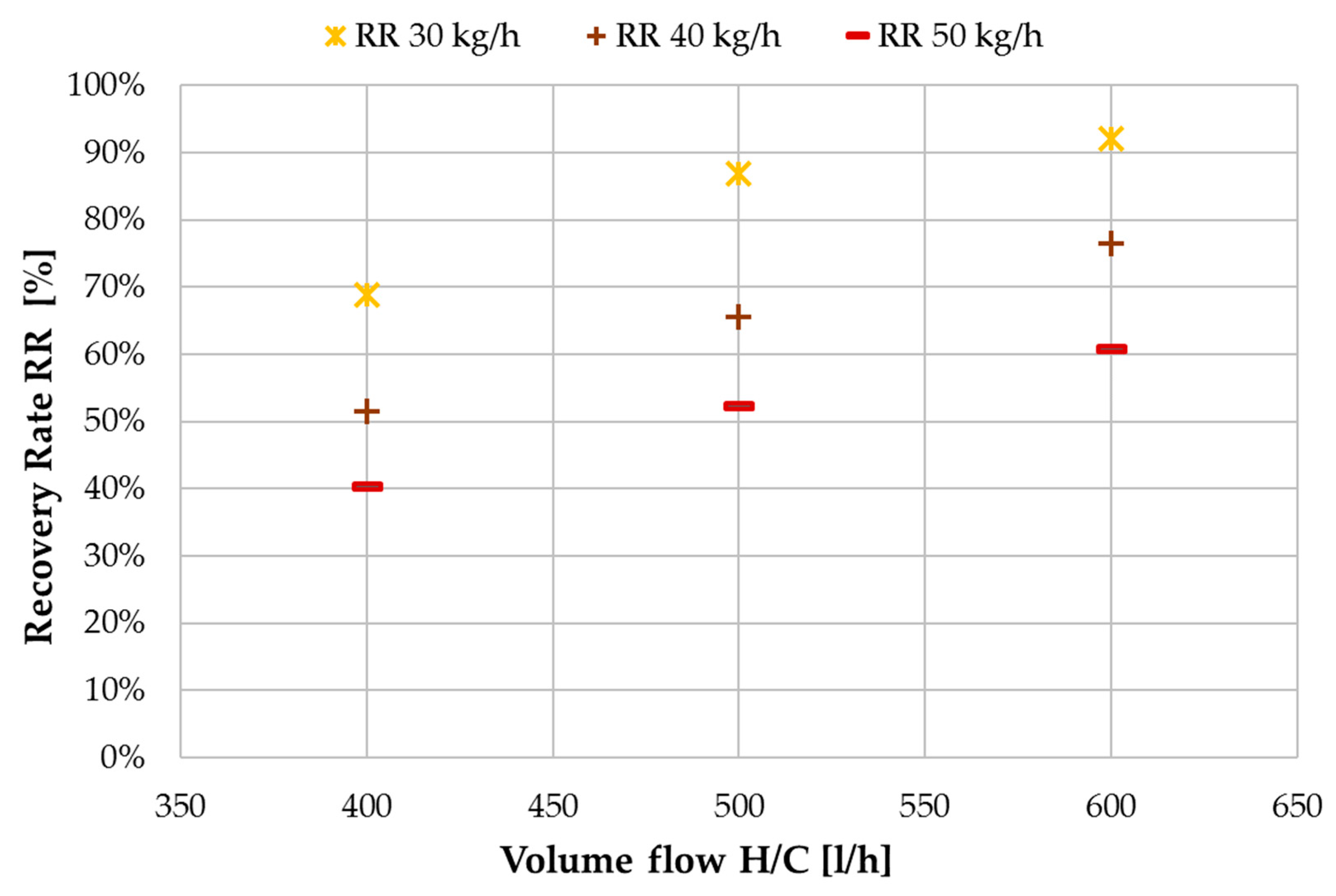
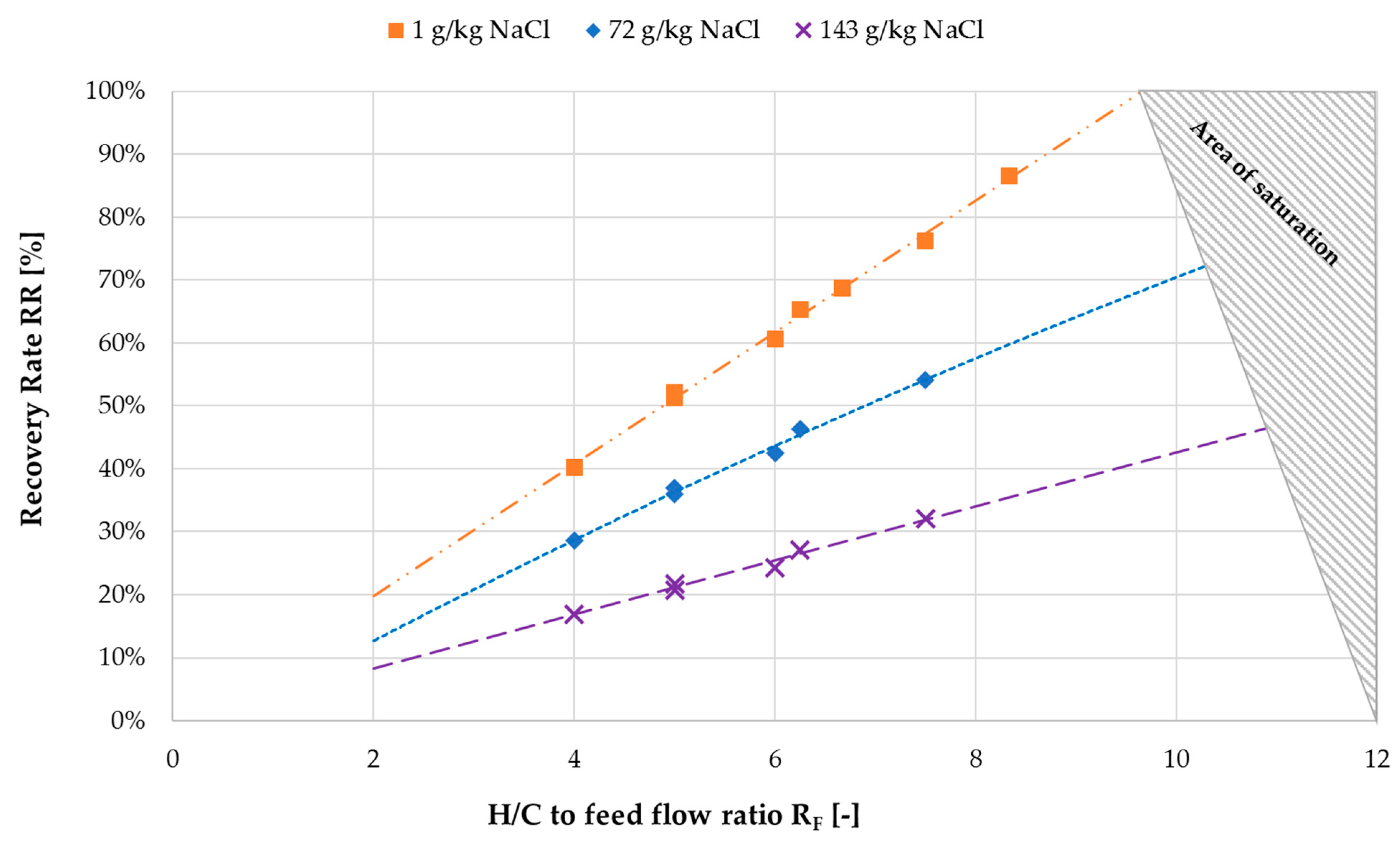
3.3. Ratio of H/C Solution Flow to Feed Flow
- Determination of feed concentration;
- Selection of desired outlet concentration;
- Selection of required RF;
- Determination of minimum feed flow;
- Calculation of H/C flow rate according to RF.
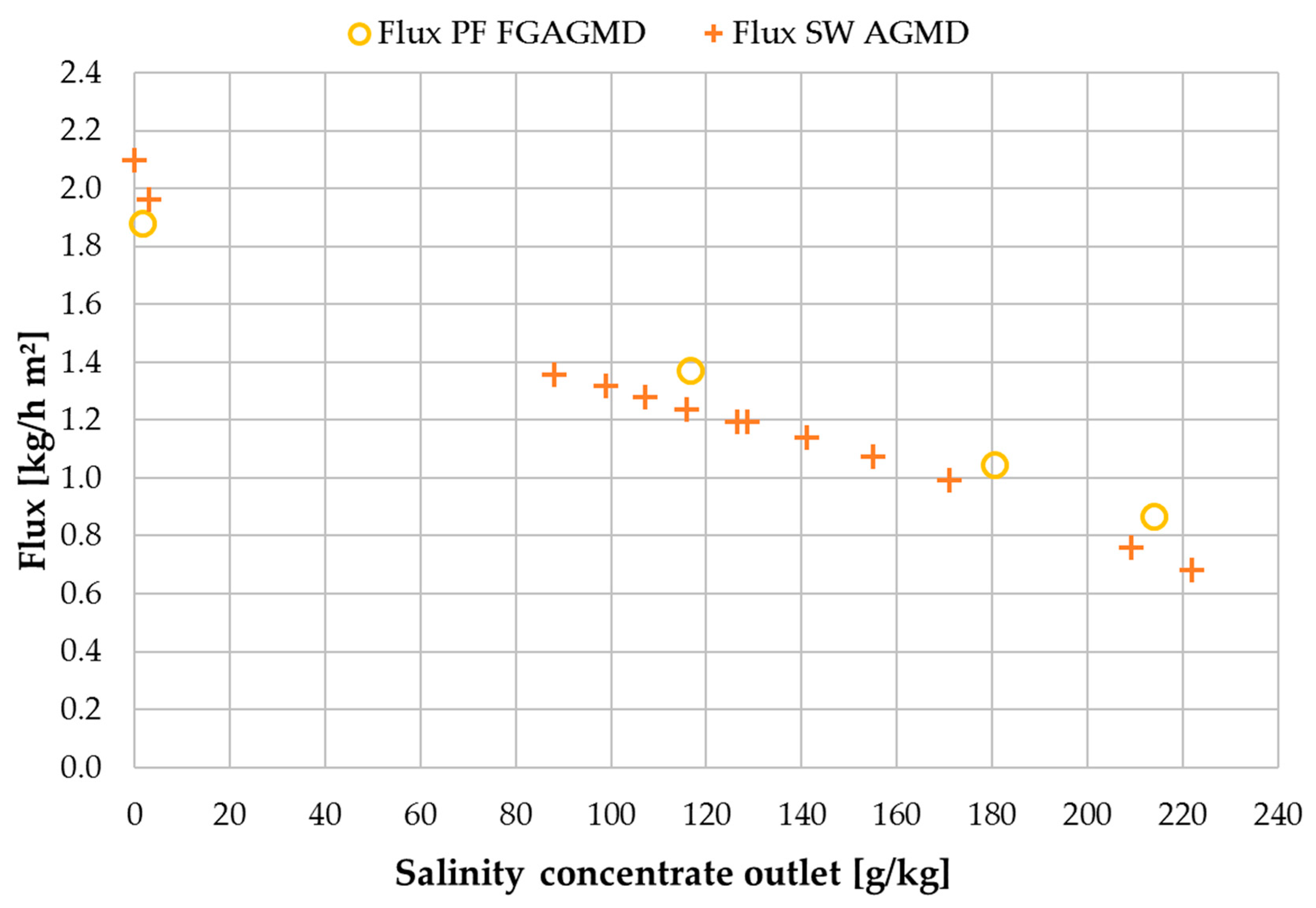
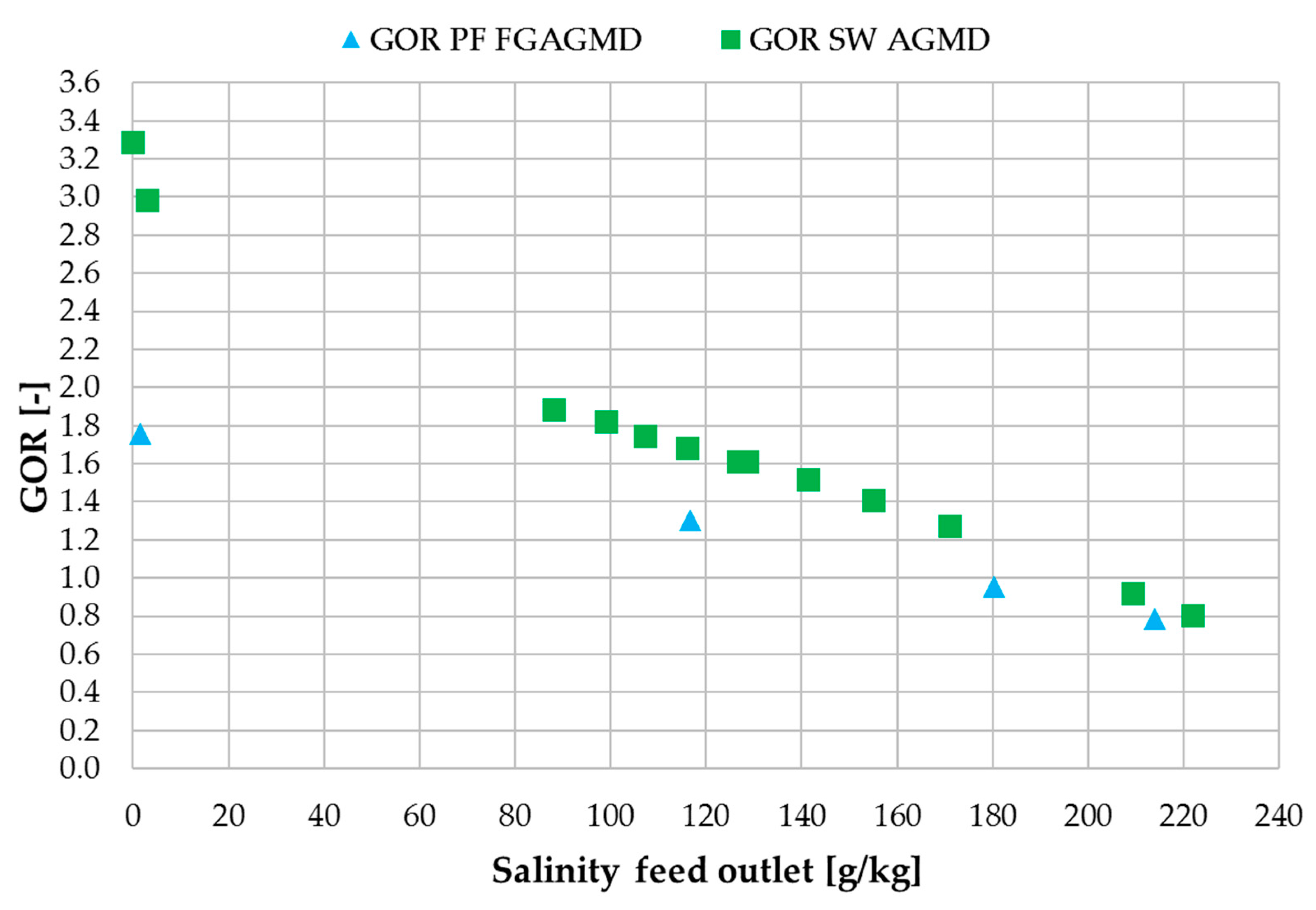
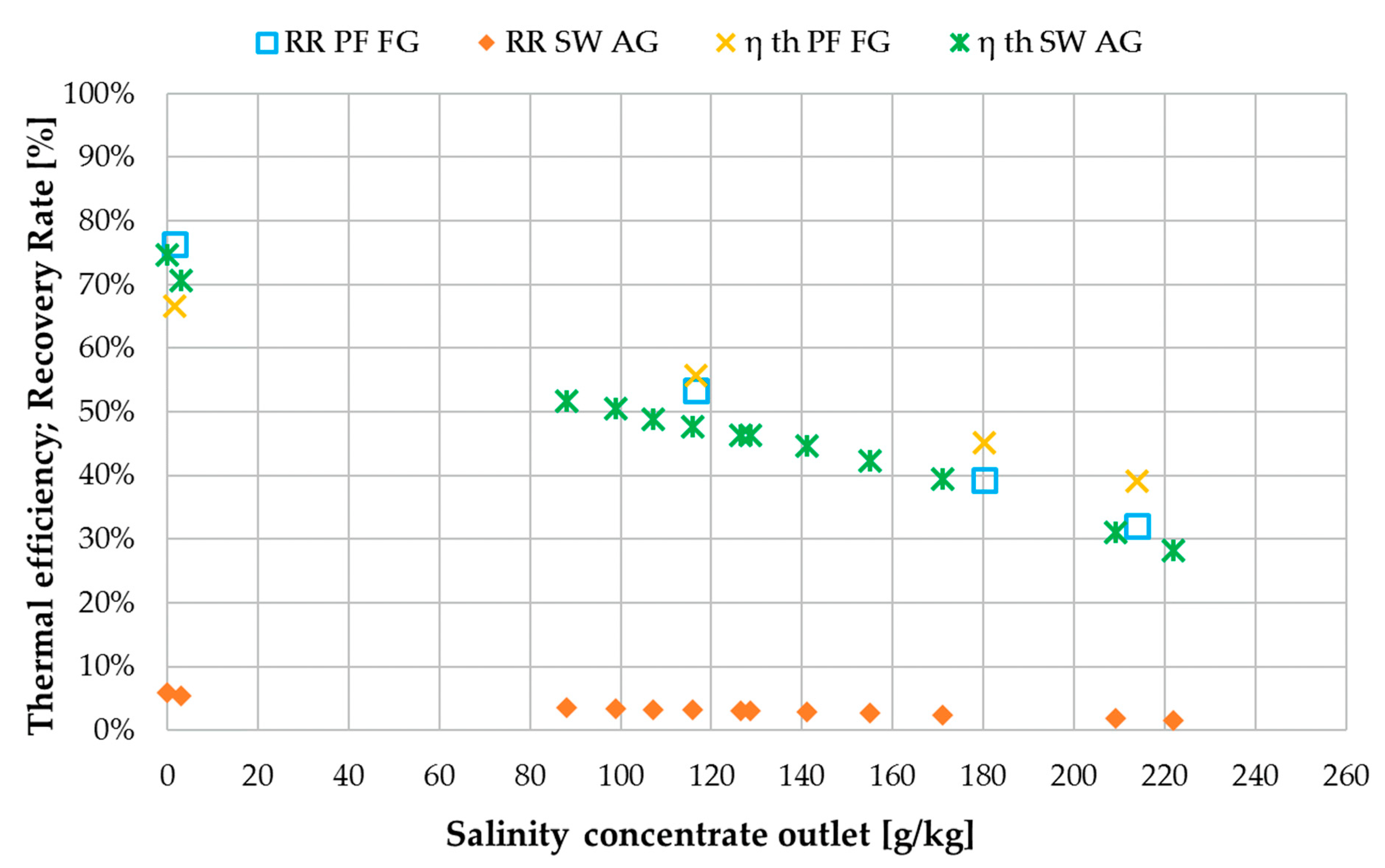
3.4. Comparison with Spiral Wound Air Gap Membrane Distillation (AGMD) Module
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| A | Membrane area |
| AGMD | Air gap membrane distillation |
| C | Conductivity (mS/cm; µS/cm) |
| cp | Specific heat capacity (kJ/kg K) |
| DCMD | Direct contact membrane distillation |
| delta T | Driving force temperature difference (K) |
| dTh | Temperature difference on the hot side of the module |
| dTc | Temperature difference on the cold side of the module |
| F | Flow (L/h) |
| FGMD | Feed Gap Membrane Distillation |
| FGAGMD | Feed Gap Air Gap Membrane Distillation |
| GOR | Gained output ratio ( - ) |
| h | Height (m) |
| HEX | Heat exchanger |
| KPI | Key performance indicator |
| m | Mass flow (kg/h) |
| MD | Membrane Distillation |
| MED | Multi Effect Distillation |
| MSF | Multi Stage Flash |
| j | Transmembrane flux (kg/m² h) |
| L | Channel length (m) |
| p | Pressure (bar) |
| P | Pump |
| PP | Polypropylene |
| PTFE | Polytetrafluorethylene |
| PGMD | Permeate gap membrane distillation |
| R | Ratio |
| RR | Recovery ratio |
| RO | Reverse Osmosis |
| S | Salinity (g/kg) |
| SWRO | Seawater Reverse Osmosis |
| T | Temperature (°C) |
| ρ | Density (kg/m³] |
| η | Efficiency (%) |
| v-AGMD | Vacuum air gap membrane distillation |
| v | Volume flow (L/h) |
| ∆hv | Evaporation enthalpy (kJ/kg) |
Indices
| ci | cooling inlet |
| co | cooling outlet |
| d | distillate |
| hi | heating inlet |
| ho | heating outlet |
| f | feed |
| F | feed ratio |
| H/C | heating and cooling |
| hx | heat exchanger |
| m | mean |
| th | thermal |
References
- Criscuoli, A.; Drioli, E.; Capuano, A.; Memoli, B.; Andreucci, V. Human plasma ultrafiltrate purification by membrane distillation: process optimisation and evaluation of its possible application on-line. Desalination 2002, 147, 147–148. [Google Scholar] [CrossRef]
- Ding, Z.; Liu, L.; Li, Z.; Ma, R.; Yang, Z. Experimental study of ammonia removal from water by membrane distillation (MD): The comparison of three configurations. J. Membr. Sci. 2006, 286, 93–103. [Google Scholar] [CrossRef]
- Grzechulska-Damszel, J.; Tomaszewska, M.; Morawski, A. Integration of photocatalysis with membrane processes for purification of water contaminated with organic dyes. Desalination 2009, 241, 118–126. [Google Scholar] [CrossRef]
- Khayet, M. Treatment of radioactive wastewater solutions by direct contact membrane distillation using surface modified membranes. Desalination 2013, 321, 60–66. [Google Scholar] [CrossRef]
- Ma, R.; El-Bourawi, M.; Khayet, M.; Ding, Z.; Li, Z.; Zhang, X. Application of vacuum membrane distillation for ammonia removal. J. Membr. Sci. 2007, 301, 200–209. [Google Scholar]
- Sarti, B. Concentration of must through vacuum membrane distillation. Desalination 2002, 149, 253–259. [Google Scholar]
- Lagana, F.; Barbieri, G.; Drioli, E. Direct contact membrane distillation: modelling and concentration experiments. J. Membr. Sci. 2000, 166, 1–11. [Google Scholar] [CrossRef]
- Hitsov, I.; Maere, T.; de Sitter, K.; Dotremont, C.; Nopens, I. Modelling approaches in membrane distillation: A critical review. Sep. Purif. Technol. 2015, 142, 48–64. [Google Scholar] [CrossRef]
- Winter, D. Membrane Distillation—A Thermodynamic, Technological and Economic Analysis. Ph.D. Thesis, University of Kaiserslautern, Rhineland-Palatinate, Germany, 2015. Available online: http://www.reiner-lemoine-stiftung.de/pdf/dissertationen/Dissertation-Winter.pdf (accessed on 6 September 2019).
- Eykens, L.; de Sitter, K.; Dotremont, C.; Pinoy, L.; van der Bruggen, B. Characterization and performance evaluation of commercially available hydrophobic membranes for direct contact membrane distillation. Desalination 2016, 392, 63–73. [Google Scholar] [CrossRef]
- Li, X.; Shan, H.; Cao, M.; Li, B. Facile fabrication of omniphobic PVDF composite membrane via a waterborne coating for anti-wetting and anti-fouling membrane distillation. J. Membr. Sci. 2019, 589, 117262. [Google Scholar] [CrossRef]
- Fane, A.; Tun, C.; Matheickal, J.; Sheikholeslami, R. Membrane distillation crystallisation of concentrated salts—Flux and crystal formation. J. Membr. Sci. 2005, 257, 144–155. [Google Scholar]
- Arafat, A.; Saffarini, R.; Mansoor, B.; Thomas, R. Effect of temperature-dependent microstructure evolution on pore wetting in PTFE membranes under membrane distillation conditions. J. Membr. Sci. 2013, 429, 282–294. [Google Scholar]
- Guillen-Burieza, E.; Mavukkandy, M.; Bilad, M.R.; Arafat, H. Understanding wetting phenomena in membrane distillation and how operational parameters can affect it. J. Membr. Sci. 2016, 515, 163–174. [Google Scholar] [CrossRef]
- Guillen-Burrieza, E. Membrane structure and surface morphology impact on the wetting of MD membranes. J. Membr. Sci. 2015, 483, 94–103. [Google Scholar] [CrossRef]
- Lienhard, J.; Warsinger, D.; Servi, A.; Connors, G.; Mavukkandy, M.; Arafat, H.; Gleason, K. Reversing membrane wetting in membrane distillation: comparing dryout to backwashing with pressurized air. Environ. Sci. Water Res. Technol. 2017, 3, 930–939. [Google Scholar]
- Peng, Y.; Ge, J.; Li, Z.; Chen, P.; Wang, S. Membrane fouling and wetting in a DCMD process for RO brine concentration. Desalination 2014, 344, 97–107. [Google Scholar]
- Rezaei, M.; Warsinger, D.M.; Lienhard, J.H.; Samhaber, W.M. Wetting prevention in membrane distillation through superhydrophobicity and recharging an air layer on the membrane surface. J. Membr. Sci. 2017, 530, 42–52. [Google Scholar] [CrossRef]
- Rezaei, M.; Warsinger, D.; Lienhard, J.; Duke, M.; Matsuura, T.; Samhaber, W. Wetting phenomena in membrane distillation: Mechanisms, reversal, and prevention. Water Res. 2018, 329–352. [Google Scholar] [CrossRef] [PubMed]
- Nashed, A. High recovery rate solar driven reverse osmosis and membrane distillation plants for brackish groundwater desalination in Egypt. Ph.D. Thesis, University of New South Wales, Sydney, Australia, 2014. [Google Scholar]
- Volpin, F.; Chekli, L.; Ghaffour, N.; Vrouwenvelder, J.; Shon, H.K. Optimisation of a forward osmosis and membrane distillation hybrid system for the treatment of source-separated urine. Sep. Purif. Technol. 2019, 212, 368–375. [Google Scholar] [CrossRef]
- Schwantes, R.; Chavan, K.; Winter, D.; Felsmann, C.; Pfafferott, J. Techno-economic comparison of membrane distillation and MVC in a zero liquid discharge application. Desalination 2018, 428, 50–68. [Google Scholar] [CrossRef]
- Tavakkoli, S.; Lokare, O.; Vidic, R.; Khanna, V. A techno-economic assessment of membrane distillation for treatment of Marcellus shale produced water. Desalination 2017, 416, 24–34. [Google Scholar] [CrossRef]
- Al-Asheh, S.; Banat, F.; Qtaishat, M.; Al-Khateeb, M. Concentration of sucrose solutions via vacuum membrane distillation. Desalination 2006, 195, 60–68. [Google Scholar] [CrossRef]
- Hilal, N.; Gunko, S.; Verbych, S.; Bryk, M. Concentration of apple juice using direct contact membrane distillation. Desalination 2006, 190, 117–124. [Google Scholar]
- Macedonio, F.; Quist-Jensen, C.; Coindi, C.; Cassano, A.; Aljlil, S.; Alharbi, O.; Drioli, E. Direct contact membrane distillation for the concentration of clarified orange juice. J. Food Eng. 2016, 187, 37–43. [Google Scholar]
- Alkhudhiri, A. Membrane Distillation: A comprehensive review. Desalination 2012, 287, 1–7. [Google Scholar] [CrossRef]
- Drioli, E. Membrane distillation: Recent developments and perspectives. Desalination 2015, 356, 56–84. [Google Scholar] [CrossRef]
- Warsinger, D.M. Scaling and fouling in membrane distillation for desalination applications: A review. Desalination 2015, 365, 294–313. [Google Scholar] [CrossRef]
- Choudhury, M.; Anwar, N.; Jassby, D.; Rahaman, M. Fouling and wetting in the membrane distillation driven wastewater reclamation process—A review. Adv. Colloid Interface Sci. 2019, 269, 370–399. [Google Scholar] [CrossRef]
- Singh, D.; Li, L.; Obuscovic, G.; Chau, J.; Sirkar, K. Novel Cylindrical Cross-Flow Hollow Fiber Membrane Module for Direct Contact Membrane Distillation-based Desalination. J. Membr. Sci. 2018, 545, 312–322. [Google Scholar] [CrossRef]
- Song, L.; Ma, Z.; Liao, X.; Kosaraju, P.; Irish, J.; Sirkar, K. Pilot plant studies of novel membranes and devices for direct contact membrane distillation-based desalination. J. Membr. Sci. 2008, 232, 257–270. [Google Scholar] [CrossRef]
- He, F.; Gilron, J.; Sirkar, K. High water recovery in direct contact membrane distillation using a series of cascades. Desalination 2013, 323, 48–54. [Google Scholar] [CrossRef]
- Singh, D.; Sirkar, K. Desalination by air gap membrane distillation using a two hollow-fiber-set membrane module. J. Membr. Sci. 2012, 421–422, 172–179. [Google Scholar] [CrossRef]
- Hitsov, I.; de Sitter, K.; Dotremont, C.; Cauwenberg, P.; Nopens, I. Full-scale validated Air Gap Membrane Distillation (AGMD) model without calibration parameters. J. Membr. Sci. 2017, 533, 309–320. [Google Scholar] [CrossRef]
- Lee, H.; He, F.; Song, L.; Gilron, J.; Sirkar, K. Desalination with a Cascade of Cross-Flow Hollow Fiber Membrane Distillation Devices Integrated with a Heat Exchanger. AIChE 2011, 57, 1780–1795. [Google Scholar] [CrossRef]
- Gilron, J.; Song, L.; Sirkar, K. Design for Cascade of Crossflow Direct Contact Membrane Distillation. Ind. Eng. Chem. Res. 2007, 46, 2324–2334. [Google Scholar] [CrossRef]
- Winter, D.; Koschikowski, J.; Gross, F.; Maucher, D.; Düver, D.; Jositz, M.; Mann, T.; Hagedorn, A. Comparative analysis of full-scale membrane distillation contactors—Methods and modules. J. Membr. Sci. 2017, 524, 758–771. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Hilal, N. Air gap membrane distillation: A detailed study of high saline solution. Desalination 2017, 403, 179–186. [Google Scholar] [CrossRef]
- Schofield, R.; Fane, A.; Fell, C. Heat and mass transfer in membrane distillation. J. Membr. Sci. 1987, 33, 299–313. [Google Scholar] [CrossRef]
- Bodell, B. Distillation of saline water using silicone rubber membrane. U.S. Patent Patent 3361645A, 3 June 1963. [Google Scholar]
- Wang, P.; Chung, T.-S. Recent advances in membrane distillation processes: Membrane development, configuration design and application exploring. J. Membr. Sci. 2015, 474, 39–56. [Google Scholar] [CrossRef]
- Yang, X.; Wang, R.; Fane, A. Novel designs for improving the performance of hollow fiber membranedistillation modules. J. Membr. Sci. 2011, 384, 52–62. [Google Scholar] [CrossRef]
- Eykens, L.; Reyns, T.; de Sitter, K.; Dotremont, C.; Pinoy, L.; van der Bruggen, B. How to select a membrane distillation configuration? Process conditions and membrane influence unraveled. Desalination 2016, 399, 105–115. [Google Scholar] [CrossRef]
- Ruiz-Aguirre, A.; Andres-Manas, J.; Fernandes-Sevilla, J.; Zaragoza, G. Experimental characterization and optimization of multi-channel spiral wound air gap membrane distillation modules for seawater desalination. Sep. Purif. Technol. 2018, 205, 212–222. [Google Scholar] [CrossRef]
- Memsys GmbH. Available online: Available: http://www.memsys.eu/ (accessed on 10 March 2016).
- Heinzl, W.; Zhao, K.; Wenzel, M.; Büttner, S.; Bollen, F.; Lange, G.; Heinzl, S.; Sarda, N. Experimental study of the memsys vacuum-multi-effect-membrane-distillation (V-MEMD) module. Desalination 2013, 323, 150–160. [Google Scholar]
- Eykens, L.; Hitsov, I.; de Sitter, K.; Dotremont, C.; Pinoy, L.; van der Bruggen, B. Direct contact and air gap membrane distillation: Differences and similarities between lab and pilot scale. Desalination 2017, 422, 91–100. [Google Scholar] [CrossRef]
- Schwantes, R.; Bauer, L.; Chavan, K.; Dücker, D.; Felsmann, C.; Pfafferott, J. Air gap membrane distillation for hypersaline brine concentration: Operational analysis of a full-scale module–New strategies for wetting mitigation. Desalination 2018, 444, 13–25. [Google Scholar] [CrossRef]
- Schwantes, R.; Cipollina, A.; Gross, F.; Koschikowski, J.; Pfeifle, D.; Subiela, V.; Rolletschek, M. Membrane distillation: Solar and waste heat driven demonstration plants for desalination. Desalination 2013, 323, 93–106. [Google Scholar] [CrossRef]
- Raluy, G.; Schwantes, R.; Ortin, V.S.; Penate, B.; Melian, G.; Betancort, J. Operational experience of a solar membrane distillation demonstration plant in Pozo Izquierdo-Gran Canaria Island (Spain). Desalination 2012, 290, 1–13. [Google Scholar] [CrossRef]
- Winter, D.; Koschikowski, J.; Wieghaus, M. Desalination using membrane distillation: Experimental studies on full scale membrane distillation modules. J. Membr. Sci. 2011, 375, 104–112. [Google Scholar] [CrossRef]
- Koschikowski, J.; Wieghaus, M.; Rommel, M.; Ortin, V.S.; Penate, B.; Betancort, J. Experimental investigations on solar driven stand-alone membrane distillation systems for remote areas. Desalination 2009, 248, 125–131. [Google Scholar] [CrossRef]
- Banat, F.; Jwaied, N.; Rommel, M.; Koschikowski, J.; Wieghaus, M. Desalination by a “compact SMADES” autonomous solarpowered membrane distillation unit. Desalination 2007, 217, 29–37. [Google Scholar] [CrossRef]
- Swaminathan, J.; Chung, H.; Warsinger, D.; Lienhard, J. Energy efficiency of membrane distillation up to high salinity: Evaluating critical system size and optimal membrane thickness. Appl. Energy 2018, 211, 715–734. [Google Scholar] [CrossRef]
- Hausmann, A.; Sanciolo, P.; Vasiljevic, T.; Weeks, M.; Duke, M. Membrane distillation in the dairy industry: process integration and membrane performance. In Proceedings of the International Workshop on Membrane Distillation and Related Technologies, Ravello, Italy, 9–12 October 2011; pp. 93–96. [Google Scholar]
- Hausmann, A.; Sanciolo, P.; Vasiljevic, T.; Weeks, M.; Duke, M. Integration of Membrane Distillation into Heat Paths of Industrial Processes. Chem. Eng. J. 2012, 211–212, 378–387. [Google Scholar] [CrossRef]
- El-Bourawi, M.; Ding, Z.; Ma, R.; Khayet, M. A framework for better understanding membrane distillation separation process. J. Membr. Sci. 2006, 285, 4–29. [Google Scholar] [CrossRef]
- Khayet, M. Membranes and theoretical modeling of membrane distillation: A review. Adv. Colloid Interface Sci. 2011, 164, 56–88. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, A.; Arumugam, S. Experimental Study on Specific Heat of Hot Brine for Salt Gradient Solar Pond Application. Int. J. Chemtech Res. 2012, 4, 56–961. [Google Scholar]
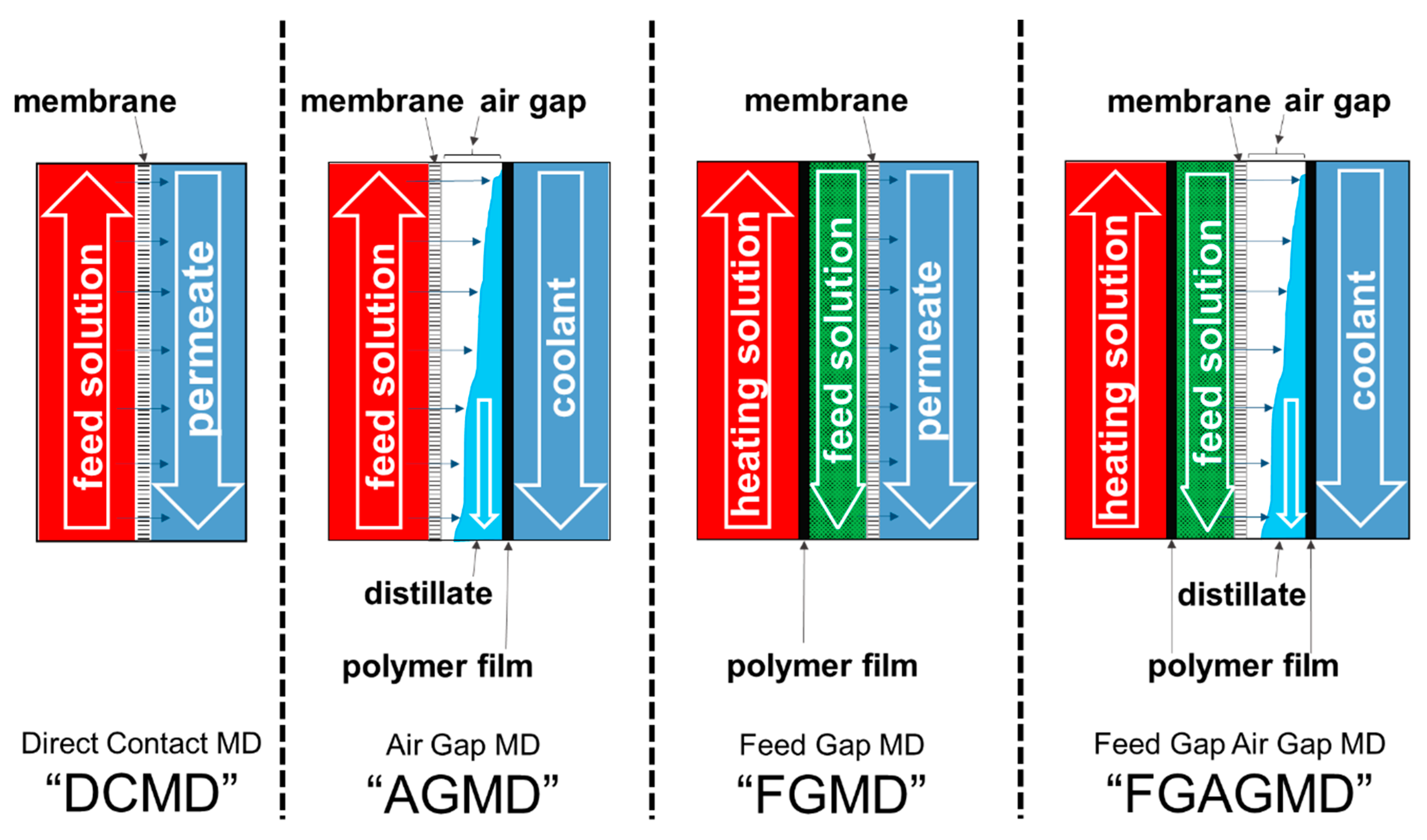
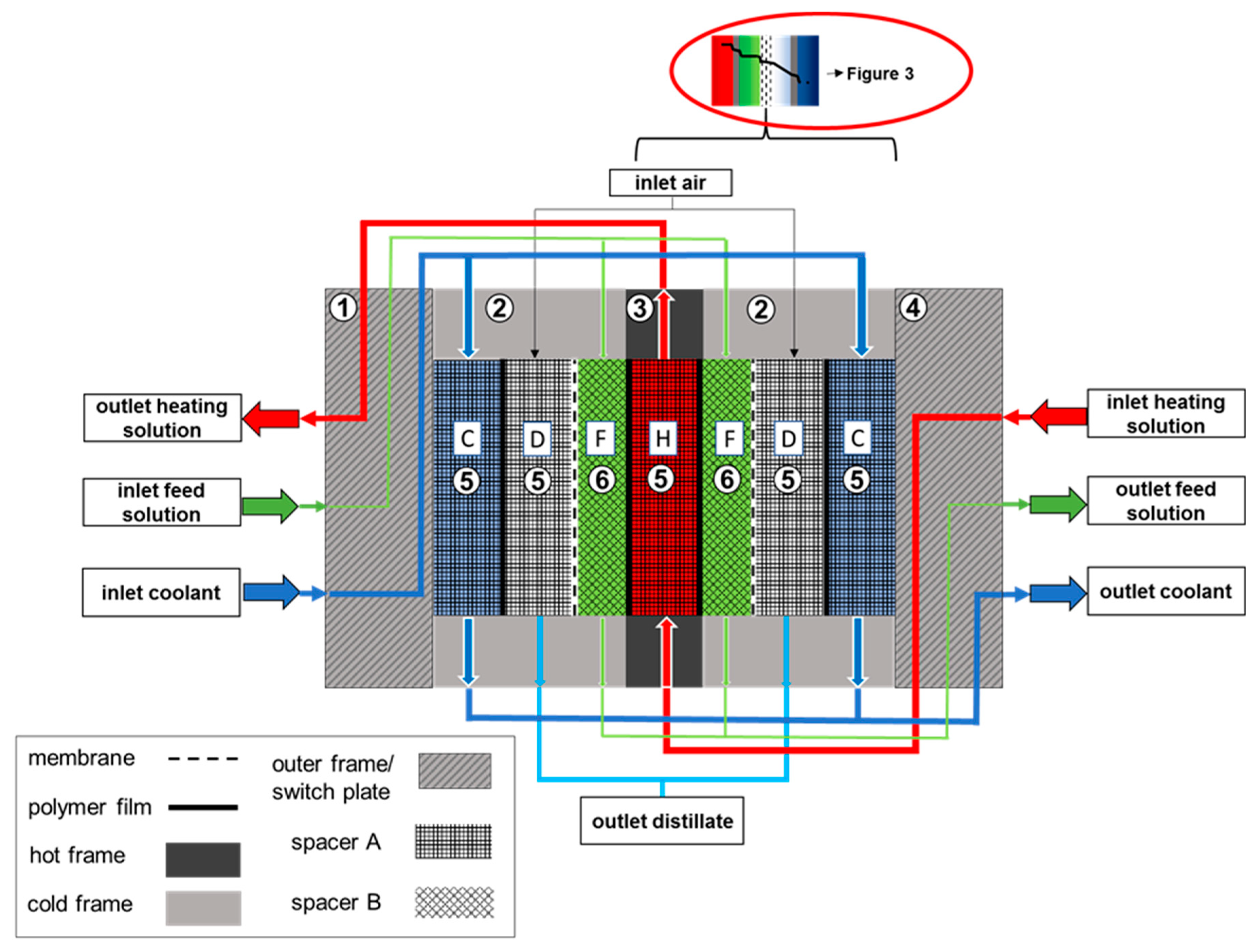
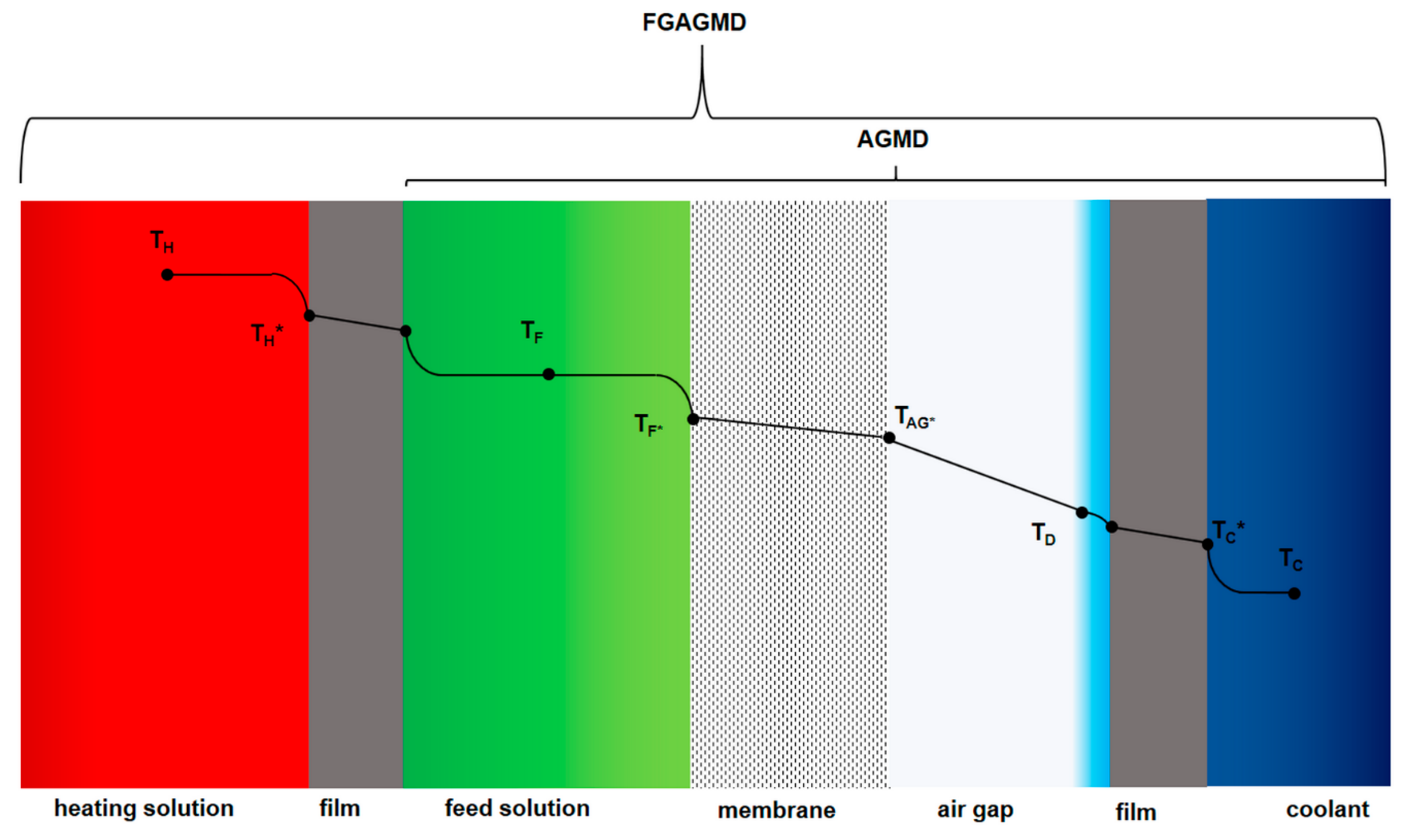


| No | Material | Thickness | Polymer | Approx. Porosity (%) | Nominal Pore Diameter (µm) |
|---|---|---|---|---|---|
| 1 + 4 | Outer- and switch plate | 30 mm | Polypropylene (PP) | - | - |
| 2 + 3 | Hot and cold channels | 4 mm/2 mm | PP | - | - |
| 5 | Spacer A | 2 mm | High density Polyethylene (HDPE) | 80 | - |
| 6 | Spacer B | 1 mm | PP | 80 | - |
| - | Membrane (+ backing) | 76 (+280) µm | Polytetrafluorethylene (PTFE), (PP) | 80 (50) | 0.2 |
| - | Polymer film | 100 µm | PP | - | - |
| Component | Producer | Type | Accuracy | Name in Schematic |
|---|---|---|---|---|
| Conductivity meters | Jumo, Fulda, Germany | CTI500 24VDC | ≤0.5% of measuring range (0–500 mS/cm) | Cfi1, Cfo2 |
| Conductivity meter distillate | Jumo, Fulda, Germany | BlackLine CR-EC | ≤2% of measuring range (0–5000 µS/cm) | Cd |
| Volume flow H/C | Krohne, Duisburg, Germany | Optiflux 4300C | 0.5% of measuring value | FH/C |
| Volume flow Feed | MIB GmbH, Breisach am Rhein, Germany | Flowmax 42i | ±2% of measured value ± 3mm/s | |
| Temperature sensors | TC direct, Mönchengladbach, Germany | Pt100 Klasse A | ±(0.15 + 0.002 × t) | all Temperatures |
| Feed pump | KNF Neuberger GmbH, Freiburg, Germany | PML14169-NF 300 | - | Pf |
| Heating and Cooling pump | Dunkermotoren, 79848 Bonndorf im Schwarzwald, Germany | BG 65X50 SI | - | |
| Pressure sensors | Jumo, Fulda, Germany | Midas, C18 SW -1,6 | 1.6% of measuring value | pH/Ci, pfi1 |
| PLC | Advantech Europe BV, Hilden, Germany | 2271G-E1-C20170517 | - | - |
| Pressurized air pump | KNF, Neuberger GmbH, Freiburg, Germany | KNF N828 KNE | - | Blower |
| Balance distillate | Soehnle Industrial Solutions GmbH, Backnang, Germany | Table balance | 1 g for 0–32 kg | Md |
| Volume flow air | First Sensor AG, Berlin, Germany | WTA | ±2% of reading +0.25% of measuring value | Fair |
| Heating and cooling pump | Harton Anlagentechnik GmbH, Alsdorf, Germany | DC 40/10BL | - | PH/Ci |
| H/C | FG in M1 | FG in M2 | FG out M2 |
|---|---|---|---|
| (L/h) | (kg/h) | (kg/h) | (kg/h) |
| 400 | 30 | 20.2 | 9.5 |
| 400 | 40 | 30.7 | 19.6 |
| 400 | 50 | 41.2 | 30.0 |
| 500 | 30 | 18.1 | 4.2 |
| 500 | 40 | 28.5 | 14.0 |
| 500 | 50 | 38.9 | 24.1 |
| 600 | 30 | 16.3 | 2.7 |
| 600 | 40 | 26.5 | 9.7 |
| 600 | 50 | 36.4 | 19.9 |
| Flux | GOR | RR | ηth | |
|---|---|---|---|---|
| (kg/m² h) | (-) | (%) | (%) | |
| PF FGAGMD | 1.2 | 1.1 | 45 | 50 |
| SW AGMD | 1.1 | 1.4 | 3 | 46 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwantes, R.; Seger, J.; Bauer, L.; Winter, D.; Hogen, T.; Koschikowski, J.; Geißen, S.-U. Characterization and Assessment of a Novel Plate and Frame MD Module for Single Pass Wastewater Concentration–FEED Gap Air Gap Membrane Distillation. Membranes 2019, 9, 118. https://doi.org/10.3390/membranes9090118
Schwantes R, Seger J, Bauer L, Winter D, Hogen T, Koschikowski J, Geißen S-U. Characterization and Assessment of a Novel Plate and Frame MD Module for Single Pass Wastewater Concentration–FEED Gap Air Gap Membrane Distillation. Membranes. 2019; 9(9):118. https://doi.org/10.3390/membranes9090118
Chicago/Turabian StyleSchwantes, Rebecca, Jakob Seger, Lorenz Bauer, Daniel Winter, Tobias Hogen, Joachim Koschikowski, and Sven-Uwe Geißen. 2019. "Characterization and Assessment of a Novel Plate and Frame MD Module for Single Pass Wastewater Concentration–FEED Gap Air Gap Membrane Distillation" Membranes 9, no. 9: 118. https://doi.org/10.3390/membranes9090118
APA StyleSchwantes, R., Seger, J., Bauer, L., Winter, D., Hogen, T., Koschikowski, J., & Geißen, S.-U. (2019). Characterization and Assessment of a Novel Plate and Frame MD Module for Single Pass Wastewater Concentration–FEED Gap Air Gap Membrane Distillation. Membranes, 9(9), 118. https://doi.org/10.3390/membranes9090118