Chemical Enhancement for Retrofitting Moving Bed Biofilm and Integrated Fixed Film Activated Sludge Systems into Membrane Bioreactors
Abstract
1. Introduction
2. Materials and Methods
2.1. Mixed Liquors
2.2. Experimental Setup
2.3. Analytical Techniques
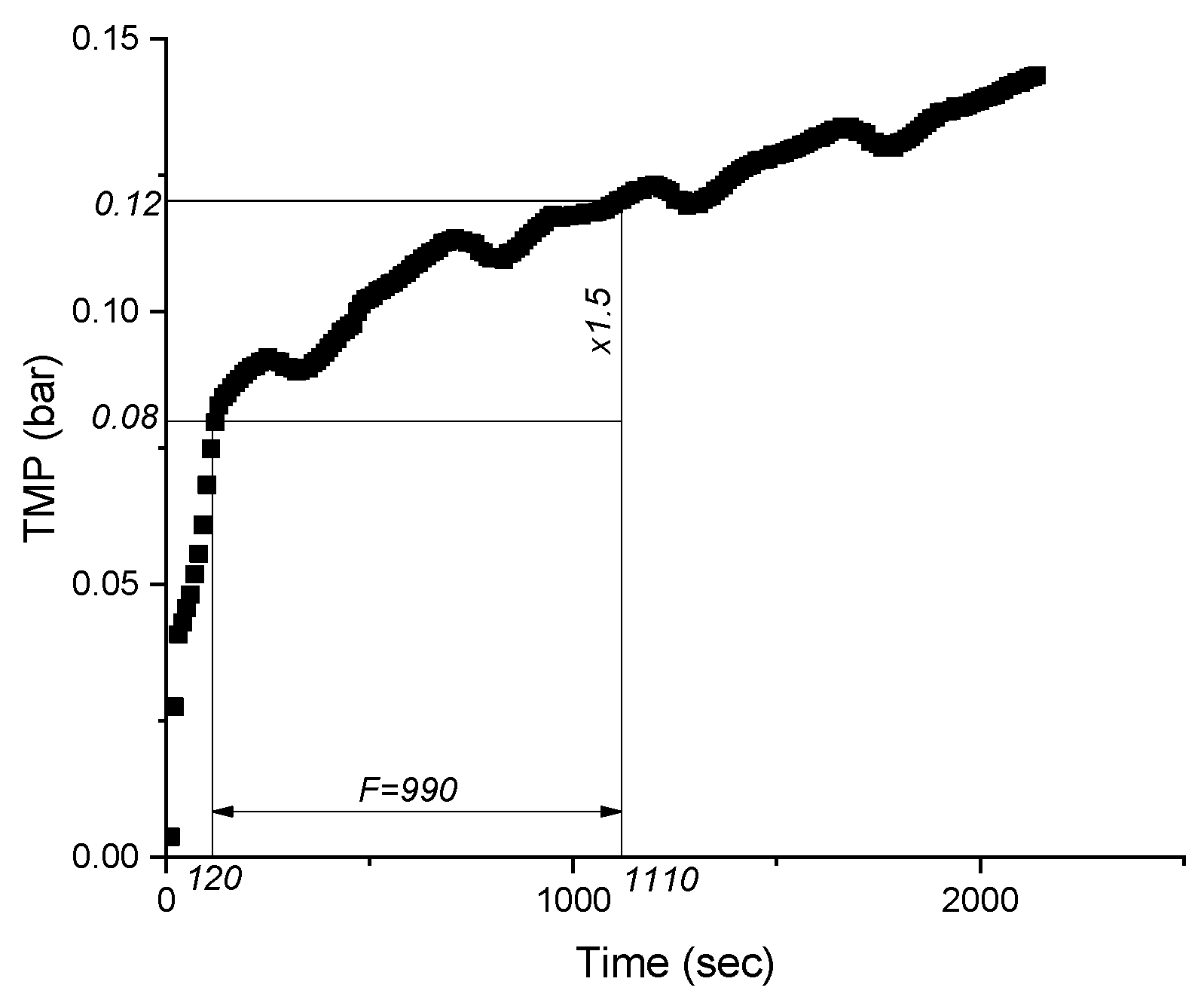
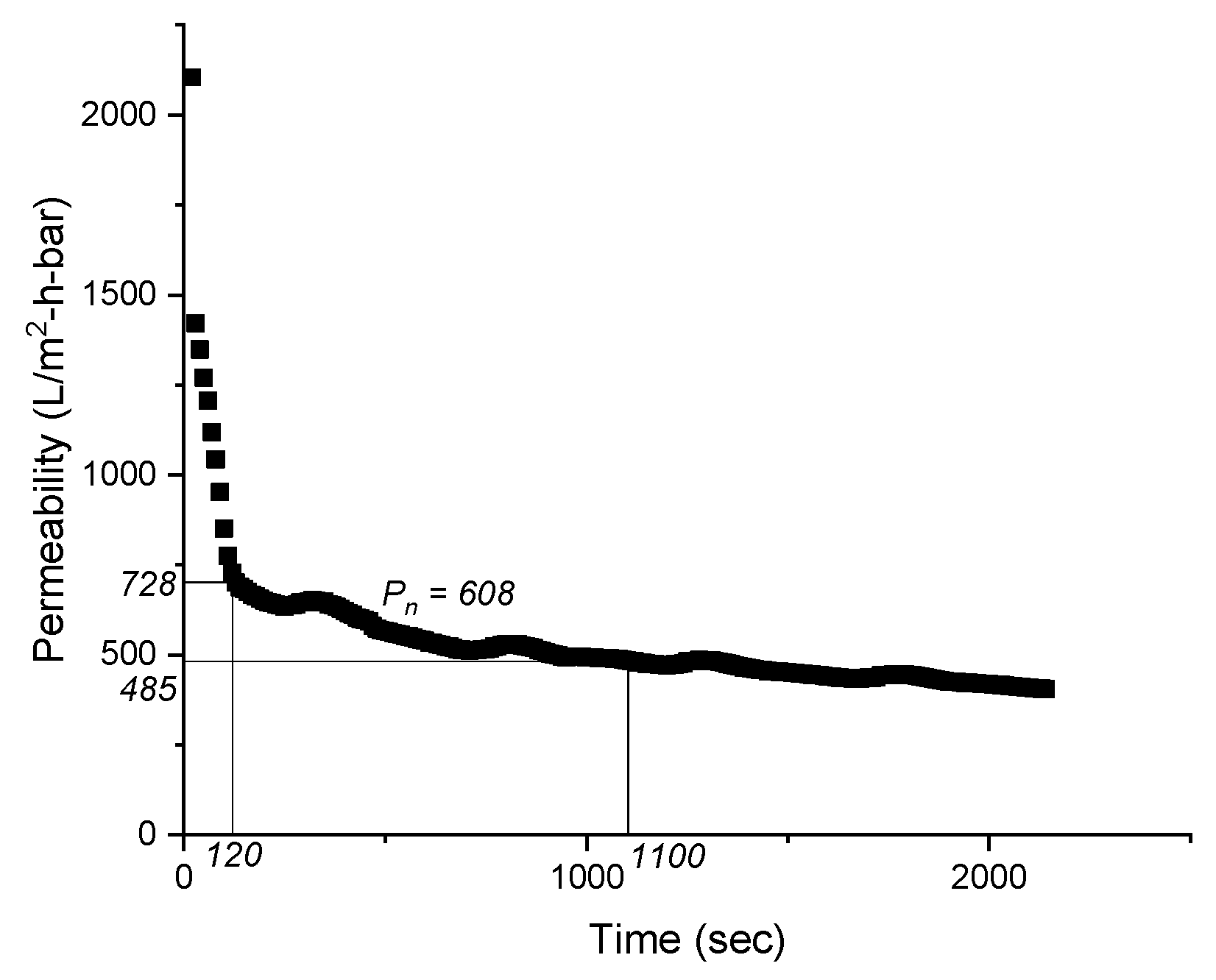
2.4. Data Processing
3. Results
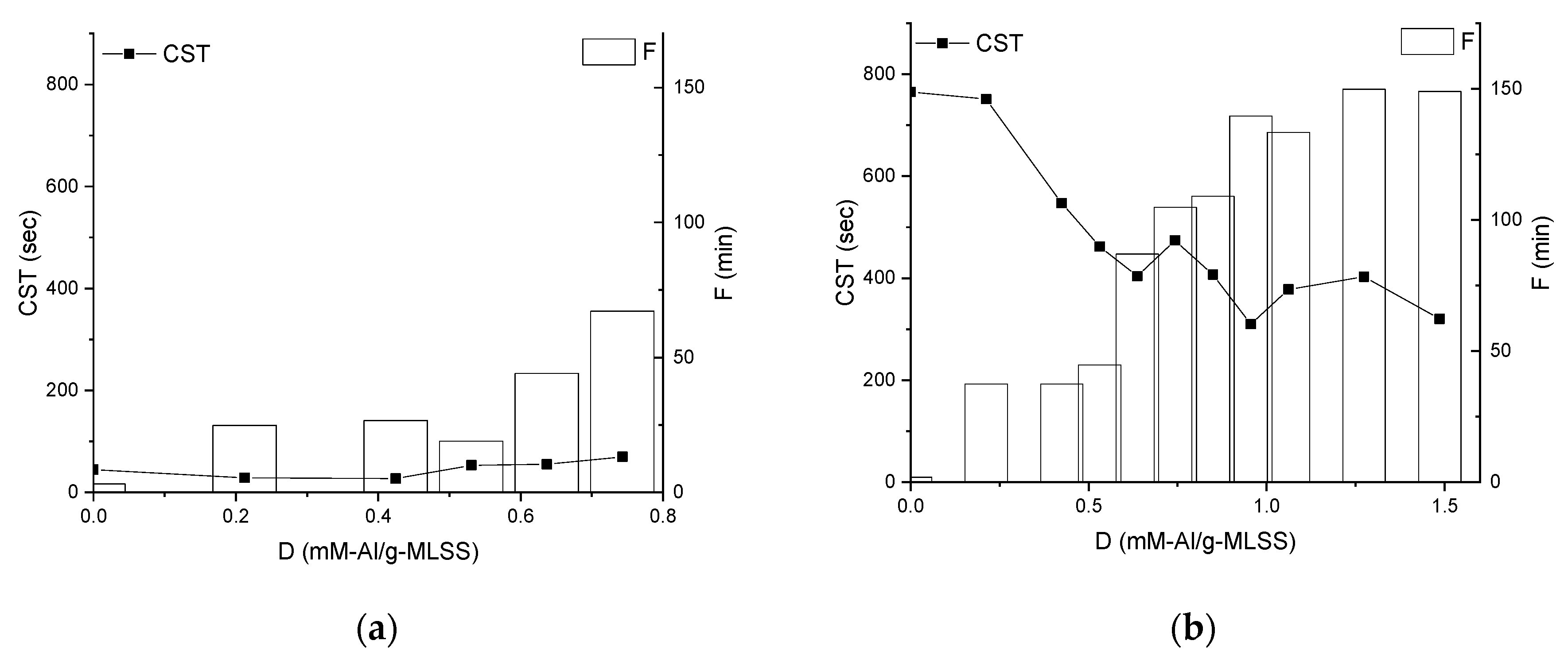
3.1. Membrane Filtration Performance
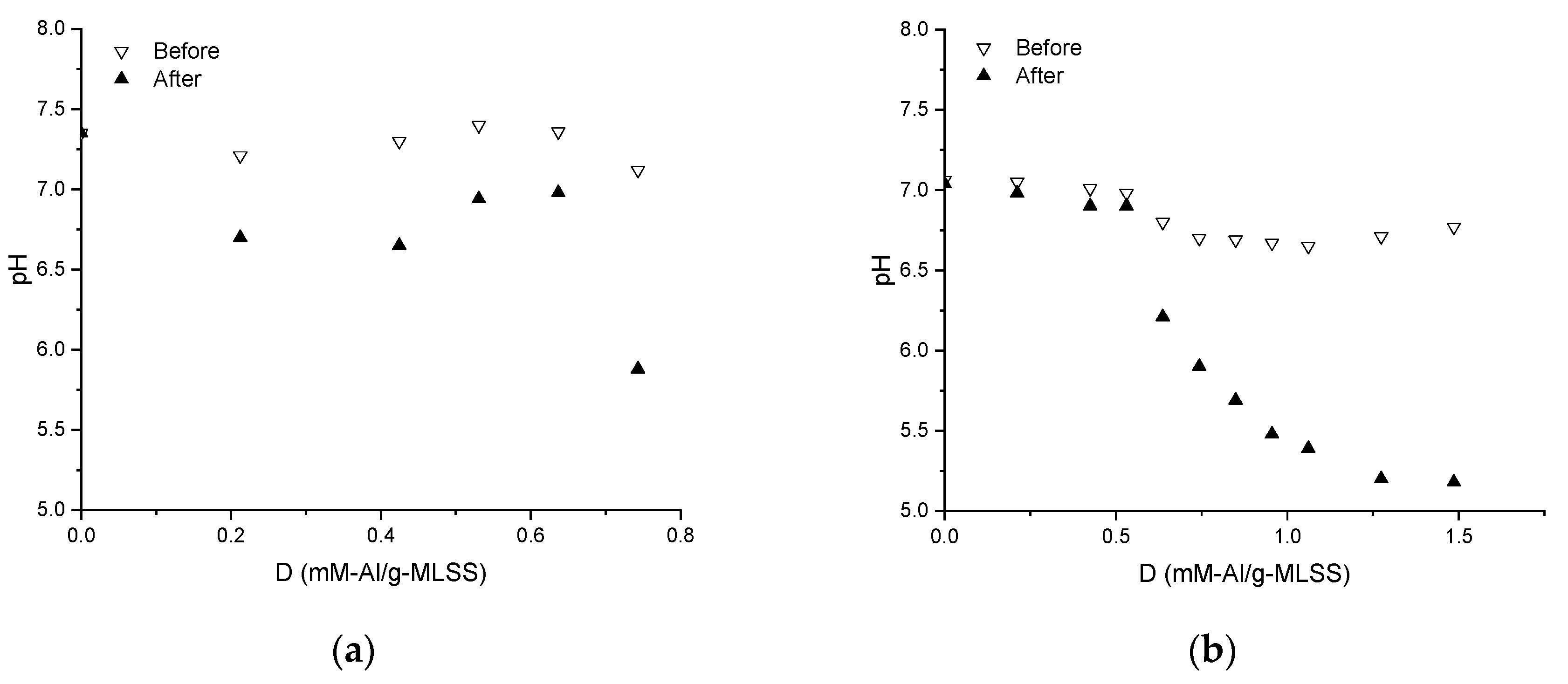
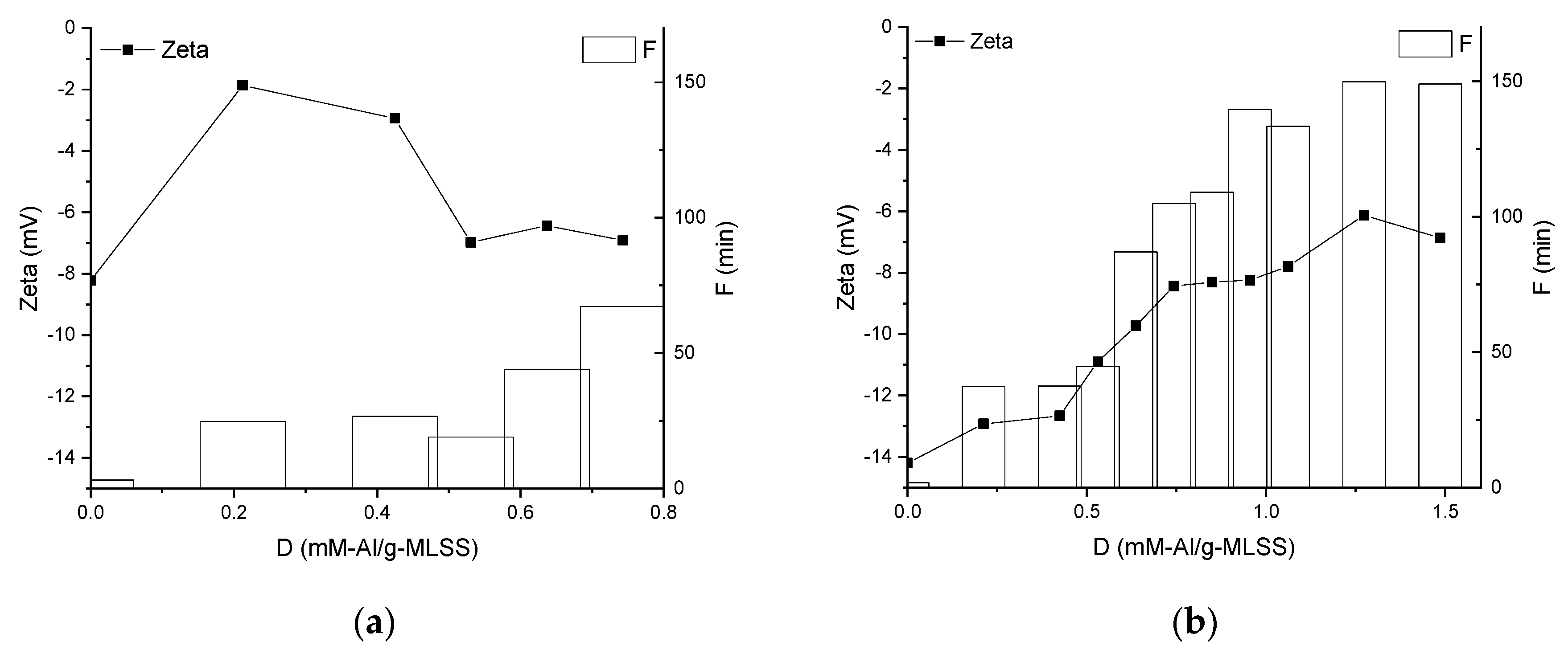
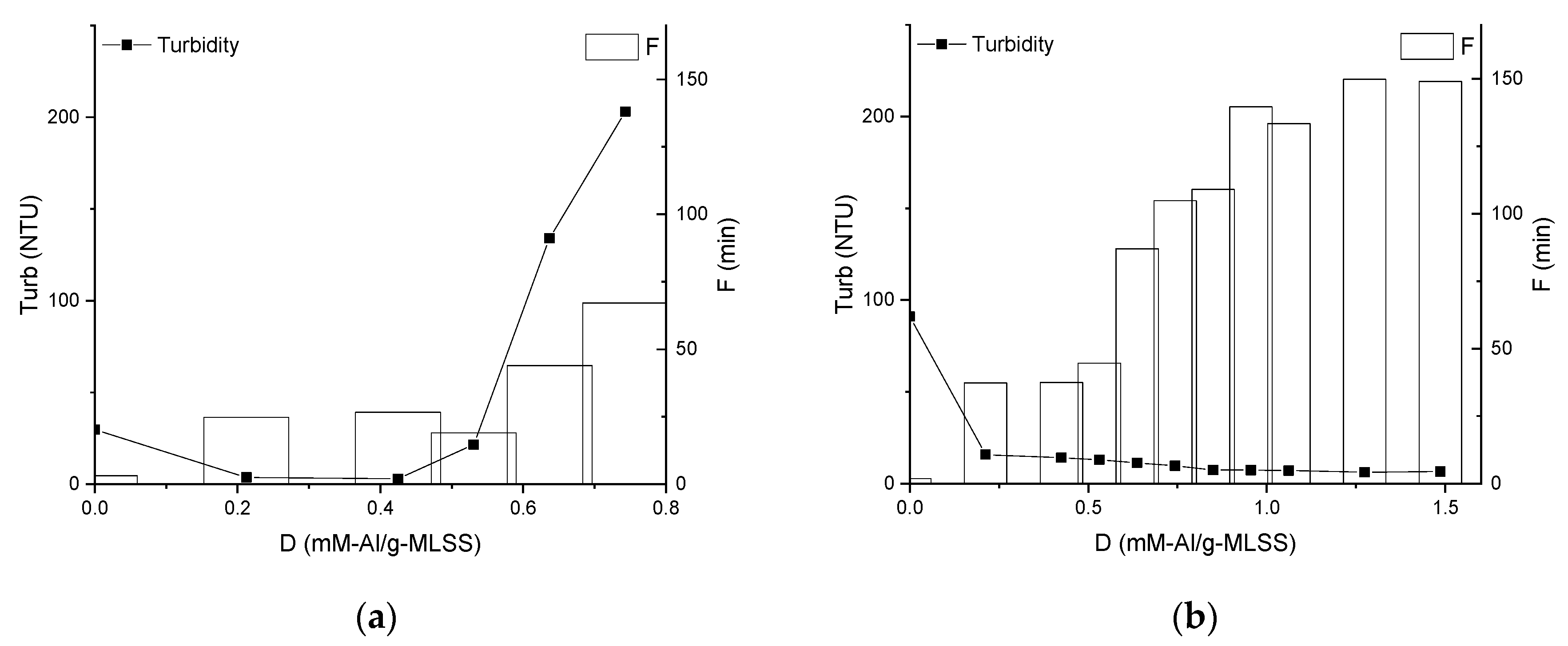
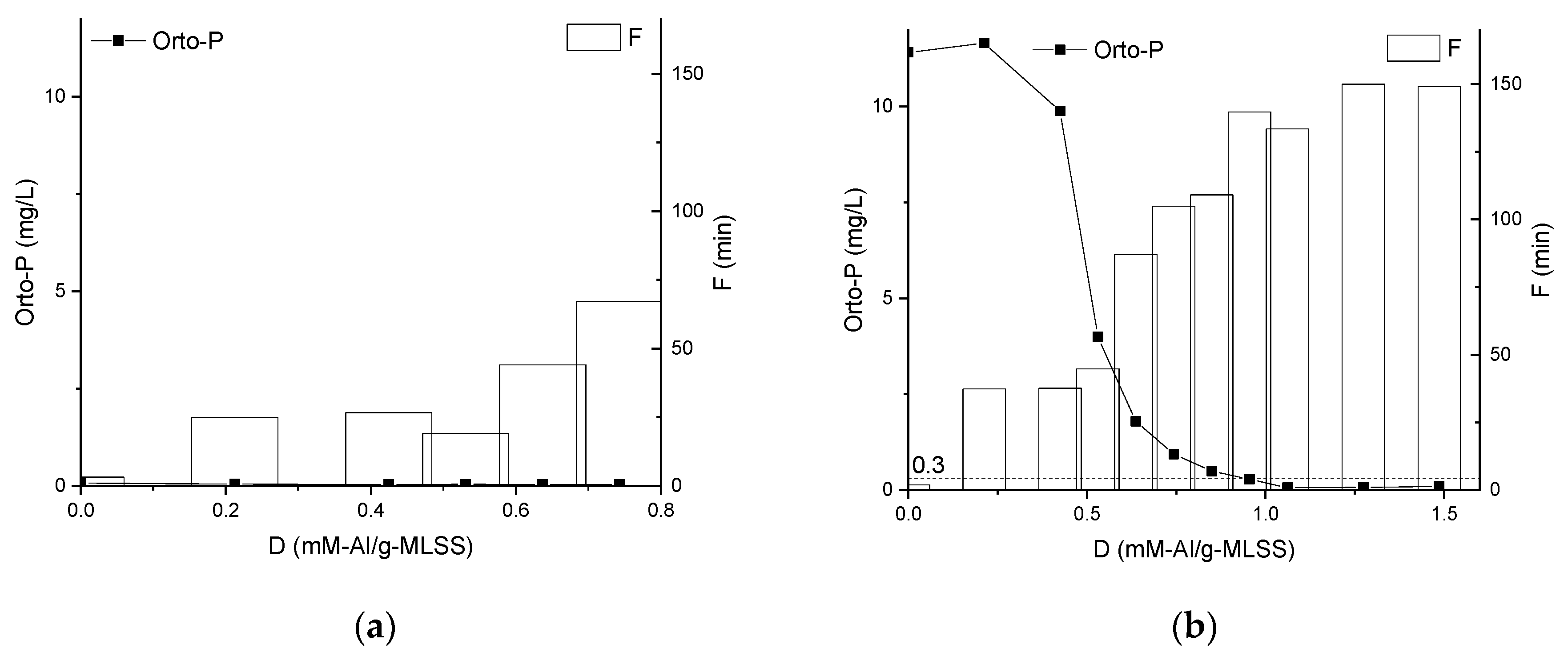
3.2. Properties of the Supernatants and Filtrates
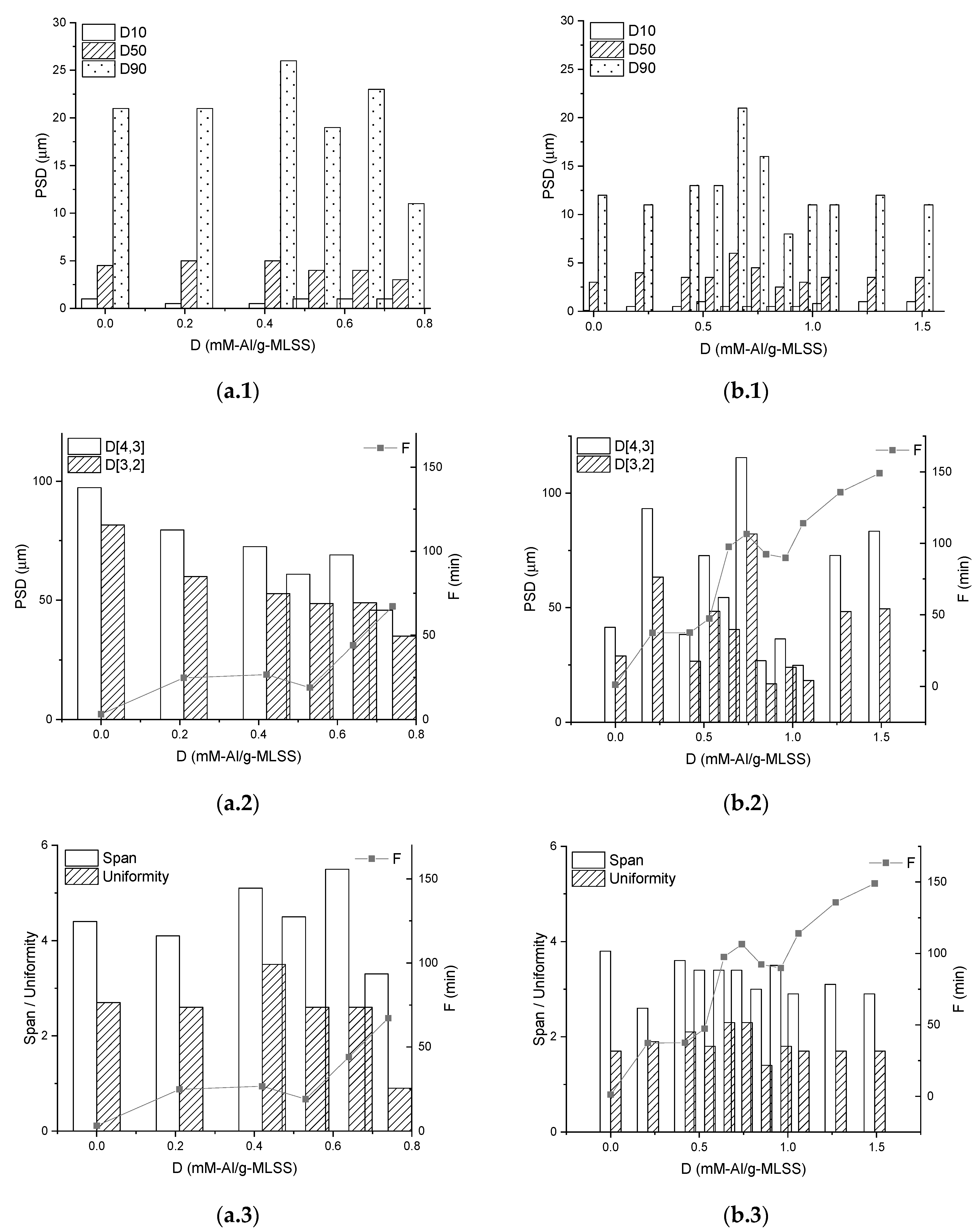
3.3. Properties of the Sediments
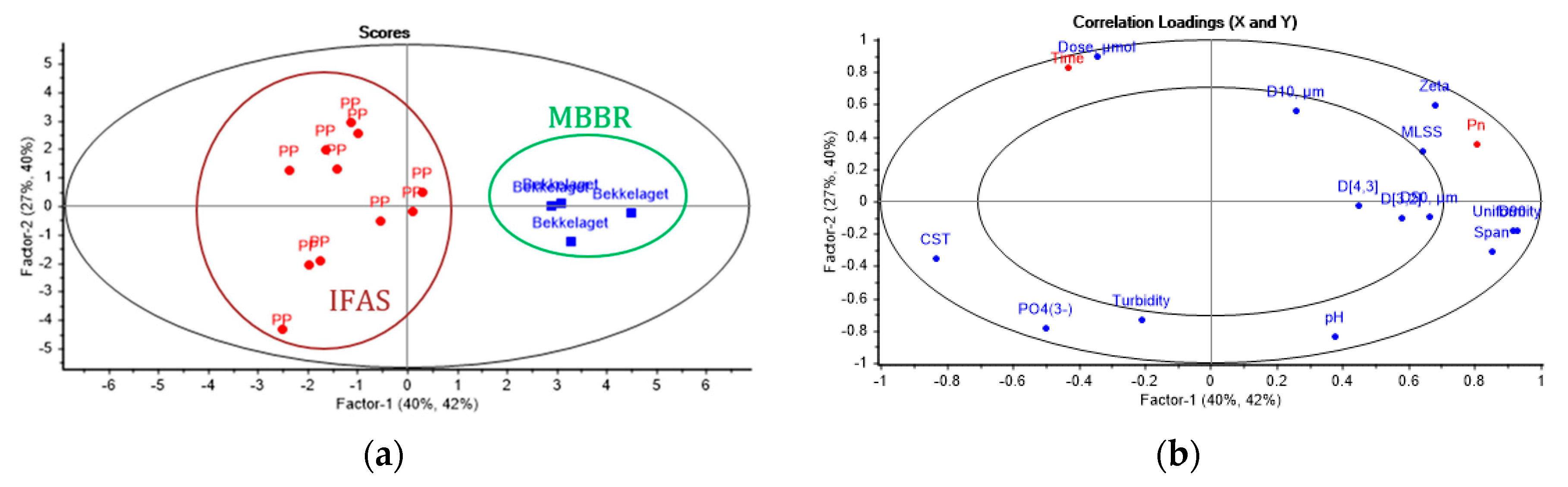
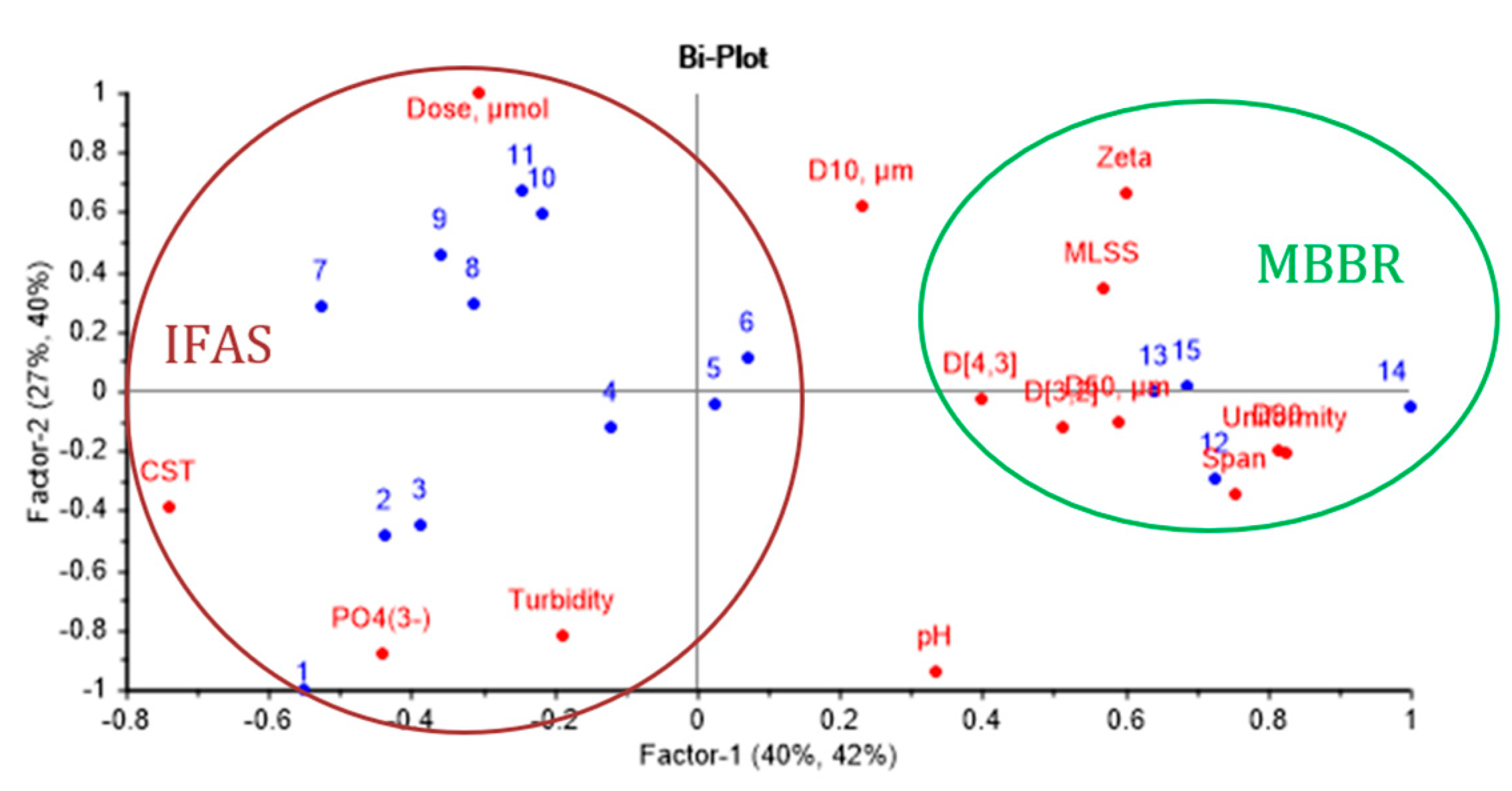
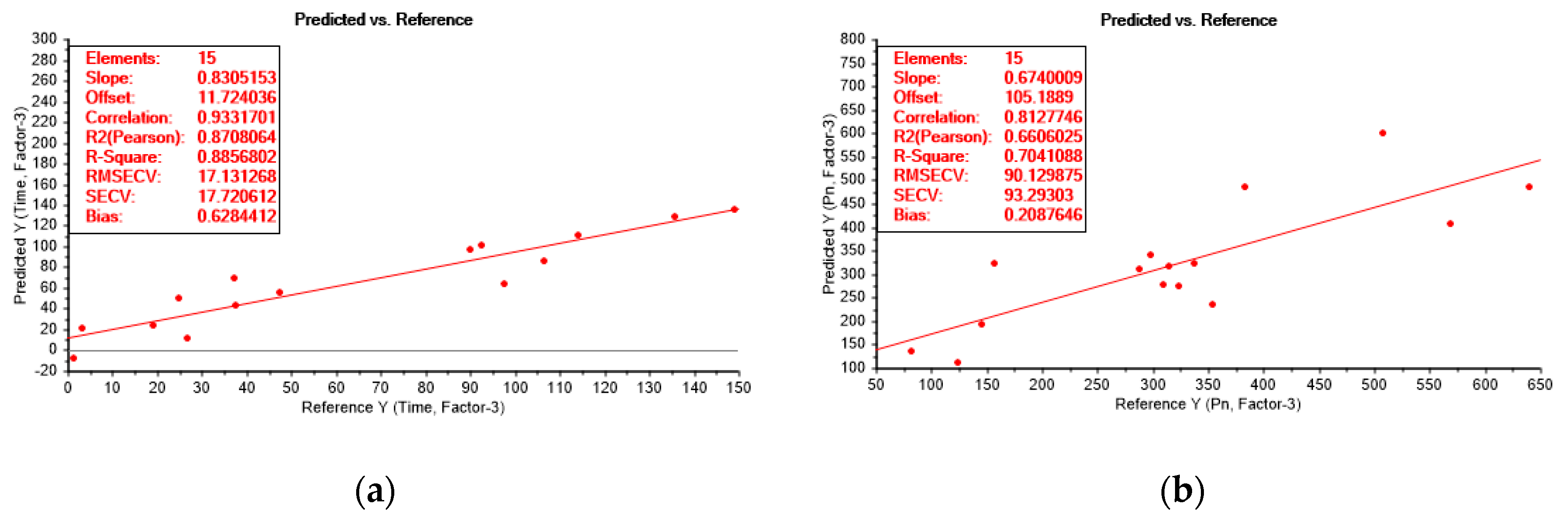
3.4. Partial Least Squares Regression Analysis
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- European Commission. Fitness Check of the EU Nature Legislation; no. November 2009; European Commission: Brussels, Belgium, 2016. [Google Scholar]
- European Commission. Implementation of the Circular Economy Action Plan; European Commission: Brussels, Belgium, 2019. [Google Scholar]
- Brepols, C.; Dorgeloh, E.; Frechen, F.-B.; Fuchs, W.; Haider, S.; Joss, A.; de Korte, K.; Ruiken, C.; Schier, W.; van der Roest, H.; et al. Upgrading and retrofitting of municipal wastewater treatment plants by means of membrane bioreactor (MBR) technology. Desalination 2008, 231, 20–26. [Google Scholar] [CrossRef]
- Suzenet, G. Prospects in Water-related Technological Innovation, Tel Aviv. 2017. Available online: https://www.oecd.org/environment/resources/Session%201%20Prospects%20in%20water-related%20technological%20innovation.pdf (accessed on 17 August 2019).
- Diaz-Elsayed, N.; Xu, X.; Balaguer-Barbosa, M.; Zhang, Q. An evaluation of the sustainability of onsite wastewater treatment systems for nutrient management. Water Res. 2017, 121, 186–196. [Google Scholar] [CrossRef]
- Koottatep, T.; Cookey, P.E.; Polprasert, C. Regenerative Sanitation: A New Paradigm for Sanitation 4.0; IWA Publishing: London, UK, 2019. [Google Scholar]
- Morgan-Sagastume, F. Biofilm development, activity and the modification of carrier material surface properties in moving-bed biofilm reactors (MBBRs) for wastewater treatment. Crit. Rev. Environ. Sci. Technol. 2018, 48, 439–470. [Google Scholar] [CrossRef]
- Shreve, M.J.; Brennan, R.A. Trace organic contaminant removal in six full-scale integrated fixed-film activated sludge (IFAS) systems treating municipal wastewater. Water Res. 2019, 151, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Li, S.; Han, L.; Song, Y.; Zhou, B.; Zhang, J. Comparison between moving bed-membrane bioreactor and conventional membrane bioreactor systems. Part I: Membrane fouling. Environ. Earth Sci. 2015, 73, 4881–4890. [Google Scholar] [CrossRef]
- Ivanovic, I.; Leiknes, T.O. The biofilm membrane bioreactor (BF-MBR)—A review. Desalin. Water Treat. 2012, 37, 288–295. [Google Scholar] [CrossRef]
- Zhang, W.; Tang, B.; Bin, L. Research Progress in Biofilm-Membrane Bioreactor: A Critical Review. Ind. Eng. Chem. Res. 2017, 56, 6900–6909. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, A.; Leyva-Diaz, J.C.; Gonzalez-Lopez, J.; Poyatos, J.M. Membrane bioreactor and hybrid moving bed biofilm reactor-membrane bioreactor for the treatment of variable salinity wastewater: Influence of biomass concentration and hydraulic retention time. Chem. Eng. J. 2018, 336, 102–111. [Google Scholar] [CrossRef]
- Leyva-Díaz, J.C.; Martín-Pascual, J.; Poyatos, J.M. Moving bed biofilm reactor to treat wastewater. Int. J. Environ. Sci. Technol. 2017, 14, 881–910. [Google Scholar] [CrossRef]
- Leiknes, T.; Ødegaard, H. The development of a biofilm membrane bioreactor. Desalination 2007, 202, 135–143. [Google Scholar] [CrossRef]
- Wang, X.C.; Liu, Q.; Liu, Y.J. Membrane Fouling Control of Hybrid Membrane Bioreactor: Effect of Extracellular Polymeric Substances. Sep. Sci. Technol. 2010, 45, 928–934. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhang, W.; Tang, B.; Ding, J.; Zheng, Y.; Zhang, Z. Membrane fouling mechanism of biofilm-membrane bioreactor (BF-MBR): Pore blocking model and membrane cleaning. Bioresour. Technol. 2018, 250, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Chae, S.R.; Drews, A.; Kraume, M.; Shin, H.S.; Yang, F. Recent advances in membrane bioreactors (MBRs): Membrane fouling and membrane material. Water Res. 2009, 43, 1489–1512. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Yang, F.; Fu, Z.; Lei, R. Comparison between a moving bed membrane bioreactor and a conventional membrane bioreactor on organic carbon and nitrogen removal. Bioresour. Technol. 2009, 100, 2369–2374. [Google Scholar] [CrossRef]
- Sombatsompop, K.; Visvanathan, C.; Aim, R.B. Evaluation of biofouling phenomenon in suspended and attached growth membrane bioreactor systems. Desalination 2006, 201, 138–149. [Google Scholar] [CrossRef]
- Zhang, W.; Luo, J.; Ding, L.; Jaffrin, M.Y.A. Review on Flux Decline Control Strategies in Pressure-Driven Membrane Processes. Ind. Eng. Chem. Res. 2015, 54, 2843–2861. [Google Scholar] [CrossRef]
- Rahimi, Y.; Torabian, A.; Mehrdadi, N.; Habibi-Rezaie, M.; Pezeshk, H.; Nabi-Bidhendi, G.R. Optimizing aeration rates for minimizing membrane fouling and its effect on sludge characteristics in a moving bed membrane bioreactor. J. Hazard. Mater. 2014, 186, 1097–1102. [Google Scholar] [CrossRef]
- Aslam, M.; McCarty, P.L.; Bae, J.; Kim, J. The effect of fluidized media characteristics on membrane fouling and energy consumption in anaerobic fluidized membrane bioreactors. Sep. Purif. Technol. 2014, 132, 10–15. [Google Scholar] [CrossRef]
- Munz, G.; Gori, R.; Mori, G.; Lubello, C. Powdered activated carbon and membrane bioreactors (MBRPAC) for tannery wastewater treatment: Long term effect on biological and filtration process performances. Desalination 2007, 207, 349–360. [Google Scholar] [CrossRef]
- Kulesha, O.; Maletskyi, Z.; Ratnaweera, H. State-of-the-art of membrane flux enhancement in membrane bioreactor. Cogent Eng. 2018, 5, 1489700. [Google Scholar] [CrossRef]
- Wu, J.; Chen, F.; Huang, X.; Geng, W.; Wen, X. Using inorganic coagulants to control membrane fouling in a submerged membrane bioreactor. Desalination 2006, 197, 124–136. [Google Scholar] [CrossRef]
- Bezirgiannidis, A.; Plesia-Efstathopoulou, A.; Ntougias, S.; Melidis, P. Combined chemically enhanced primary sedimentation and biofiltration process for low cost municipal wastewater treatment. J. Environ. Sci. Health Part A 2019. [Google Scholar] [CrossRef] [PubMed]
- Ivanovic, I.; Leiknes, T.O. Improved Performance through Particle Surface Modifications by Coagulation with Inorganic Coagulants in a Biofilm Membrane Bioreactor (BF-MBR). Sep. Sci. Technol. 2012, 48, 288–294. [Google Scholar] [CrossRef]
- Kulesha, O.; Maletskyi, Z.; Kvaal, K.; Ratnaweera, H. Strategy for Flux Enhancement in Biofilm Ceramic Membrane Bioreactor Applying Prepolymerized and Non-Prepolymerized Inorganic Coagulants. Water 2019, 11, 446. [Google Scholar] [CrossRef]
- Koseoglu, H.; Yigit, N.; Ivetrsen, V.; Drews, A.; Kitis, M.; Lesjean, B.; Kraume, M. Effects of several different flux enhancing chemicals on filterability and fouling reduction of membrane bioreactor (MBR) mixed liquors. J. Membr. Sci. 2008, 320, 57–64. [Google Scholar] [CrossRef]
- Yoon, S.-H.; Collins, J.H. A novel flux enhancing method for membrane bioreactor (MBR) process using polymer. Desalination 2007, 191, 52–61. [Google Scholar] [CrossRef]
- Lee, W.-N.; Chang, I.-S.; Hwang, B.-K.; Park, P.-K.; Lee, C.-H.; Huang, X. Changes in biofilm architecture with addition of membrane fouling reducer in a membrane bioreactor. Process Biochem. 2007, 42, 655–661. [Google Scholar] [CrossRef]
- Fu, C.; Yue, X.; Shi, X.; Ng, K.K.; Ng, H.Y. Membrane fouling between a membrane bioreactor and a moving bed membrane bioreactor: Effects of solids retention time. Chem. Eng. J. 2017, 309, 397–408. [Google Scholar] [CrossRef]
- Oslo Kommune. Utbygging Av Bekkelaget Renseanlegg (UBRA)—Prosjekter—Oslo commune. Oslo.Kommune.no. 2015. Available online: https://www.oslo.kommune.no/politikk-og-administrasjon/slik-bygger-vi-oslo/utbygging-av-bekkelaget-renseanlegg-ubra/#gref (accessed on 12 August 2019).
- Henze, M.; van Loosdrecht, M.C.M.; Ekama, G.A.; Brdjanovic, D. Biological Wastewater Treatment: Principles, Modelling and Design. Water Intell. Online 2015, 7. [Google Scholar] [CrossRef]
- Skambraks, A.-K.; Kjerstadius, H.; Meier, M.; Davidsson, Å.; Wuttke, M.; Giese, T. Source separation sewage systems as a trend in urban wastewater management: Drivers for the implementation of pilot areas in Northern Europe. Sustain. Cities Soc. 2017, 28, 287–296. [Google Scholar] [CrossRef]
- van Loosdrecht, M.C.M.; Nielsen, P.H.; Lopez-Vazquez, C.M.; Brdjanovic, D. Experimental Methods in Wastewater Treatment. Water Intell. Online 2016, 15. [Google Scholar] [CrossRef]
- Baird, R.B.; Eaton, A.D.; Clesceri, L.S. Standard Methods for the Examination of Water and Wastewater; Rice, E.W., Ed.; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- Wills, B.A.; Finch, J.A.; Wills, B.A.; Finch, J.A. Particle Size Analysis. Wills’ Miner. Process Technol. 2016, 8, 91–107. [Google Scholar]
- ISO. Representation of Results of Particle Size Analysis—Part 2: Calculation of Average Particle Sizes/Diameters and Moments from Particle Size Distributions; ISO 8276-2; ISO: Geneva, Switzerland, 2002. [Google Scholar]
- Van den Broeck, R.V.D.; Krzeminski, P.; Van Dierdonck, J.; Gins, G.; Lousada-Ferreira, M.; Van Impe, J.; Van Der Graaf, J.; Smets, I.; Van Lier, J. Activated sludge characteristics affecting sludge filterability in municipal and industrial MBRs: Unraveling correlations using multi-component regression analysis. J. Membr. Sci. 2011, 378, 330–338. [Google Scholar] [CrossRef]
- Drews, A. Membrane fouling in membrane Bioreactors-Characterisation, contradictions, cause and cures. J. Memb. Sci. 2010, 363, 1–28. [Google Scholar] [CrossRef]
- Bratby, J. Coagulation and Flocculation in Water and Wastewater Treatment; IWA Publishing: London, UK, 2016. [Google Scholar]
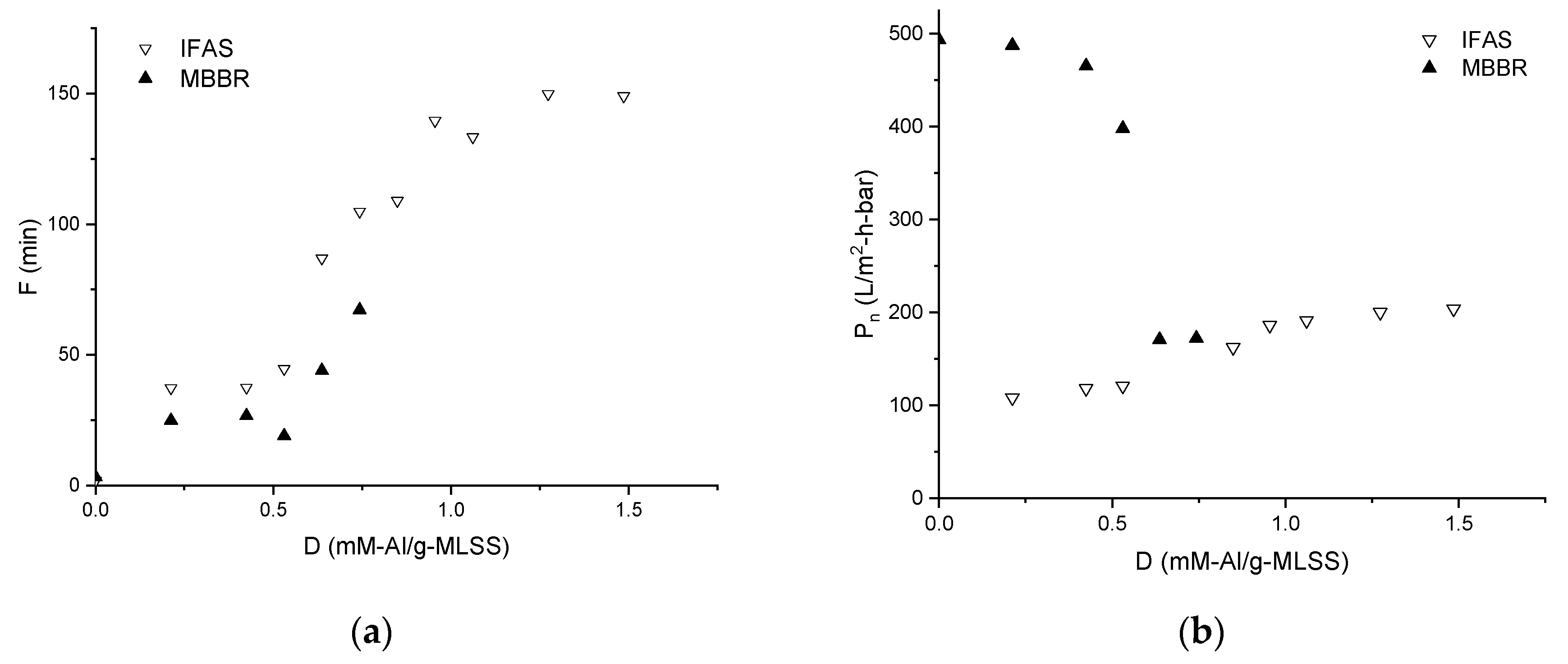


| Parameters | ML Type | |
|---|---|---|
| MBBR | IFAS | |
| MLSS, g/L | 4.72 | 3.53 |
| Turbidity, NTU | 29.7 | 91.1 |
| pH | 7.3–7.5 | 6.6–7.0 |
| Zeta, mV | −8.2 | −14.2 |
| CST, sec. | 45 | 765 |
| PO43−, mg/L | 0.07 | 11.4 |
| Viscosity, mPa/s | 1.11 | 1.05 |
| Predictors (X) | Designation |
|---|---|
| Coagulant dosage, mM-Al/g-MLSS | D |
| Capillary Suction Time of sediments, sec | CST |
| Supernatant turbidity, NTU | Turb |
| Supernatant zeta potential, mV | Zeta |
| Mixed Liquor Suspended Solids, g/L | MLSS |
| Orthophosphates in permeate, mg/L | PO43− |
| Supernatant pH | pH |
| Particle Size Analysis | |
| 10%, 50% 90% distribution points | D10, D50, D90 |
| Surface Area Moment Mean | D[3,2] |
| Volume Moment Mean | D[4,3] |
| Span | Span |
| Uniformity | Uniformity |
| Responses (Y) | |
| Average permeability, L/m2⋅h⋅bar | Pn |
| Filtration duration | F |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maletskyi, Z.; Zigta, D.K.; Kulesha, O.; Ratnaweera, H. Chemical Enhancement for Retrofitting Moving Bed Biofilm and Integrated Fixed Film Activated Sludge Systems into Membrane Bioreactors. Membranes 2019, 9, 135. https://doi.org/10.3390/membranes9100135
Maletskyi Z, Zigta DK, Kulesha O, Ratnaweera H. Chemical Enhancement for Retrofitting Moving Bed Biofilm and Integrated Fixed Film Activated Sludge Systems into Membrane Bioreactors. Membranes. 2019; 9(10):135. https://doi.org/10.3390/membranes9100135
Chicago/Turabian StyleMaletskyi, Zakhar, Dawit K. Zigta, Olga Kulesha, and Harsha Ratnaweera. 2019. "Chemical Enhancement for Retrofitting Moving Bed Biofilm and Integrated Fixed Film Activated Sludge Systems into Membrane Bioreactors" Membranes 9, no. 10: 135. https://doi.org/10.3390/membranes9100135
APA StyleMaletskyi, Z., Zigta, D. K., Kulesha, O., & Ratnaweera, H. (2019). Chemical Enhancement for Retrofitting Moving Bed Biofilm and Integrated Fixed Film Activated Sludge Systems into Membrane Bioreactors. Membranes, 9(10), 135. https://doi.org/10.3390/membranes9100135






