Polymer-Induced Swelling of Solid-Supported Lipid Membranes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Dry State
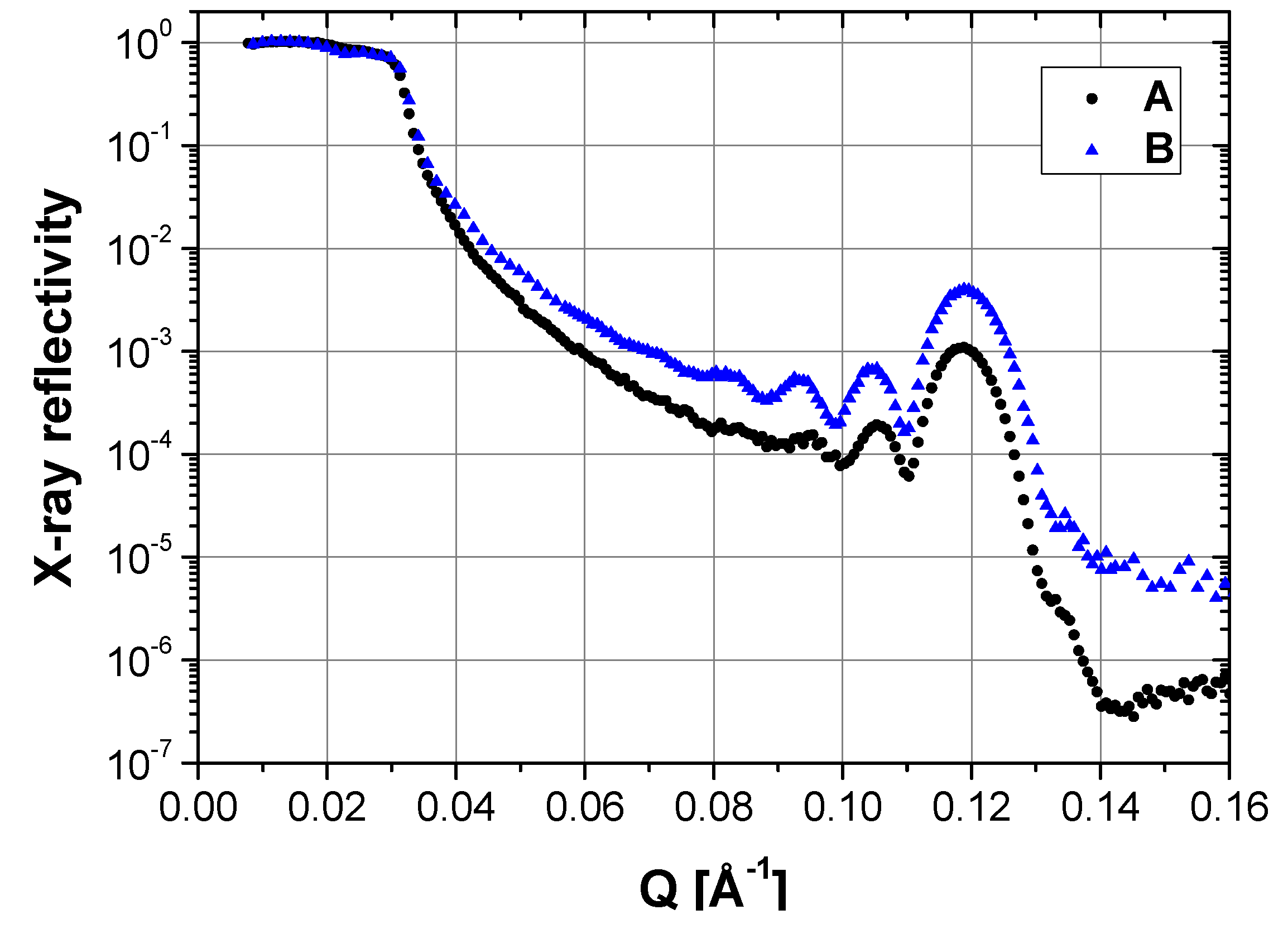
2.2. Wet State
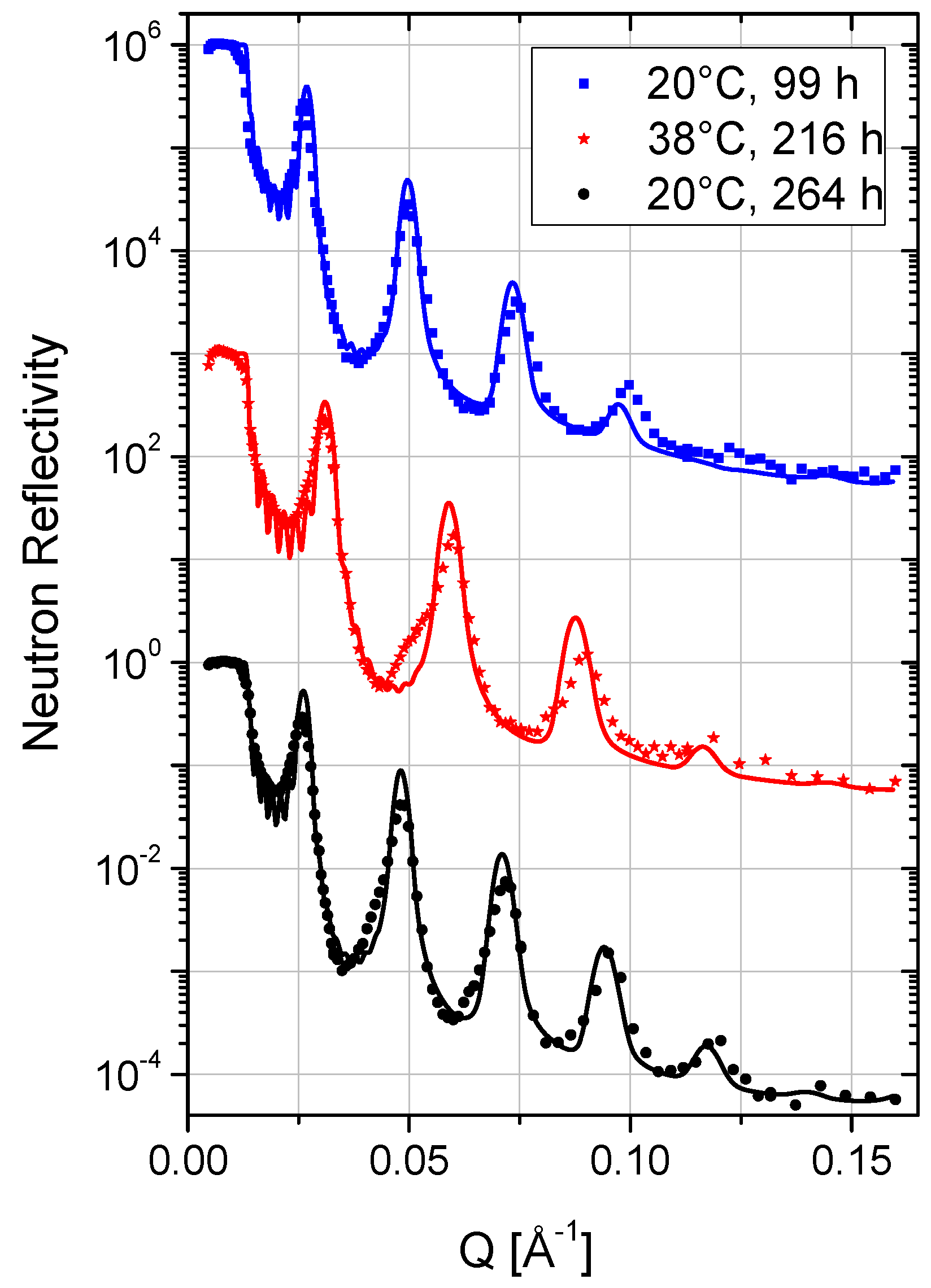
| Layer | 20 °C | 38 °C | 20 °C | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | ||
| Si | – | s-i | 2.07 | – | s-i | 2.07 | – | s-i | 2.07 | – | |
| SiO2 | 1 | 13.0 | 3.47 | 0 | 13.0 | 3.47 | 0 | 13.0 | 3.47 | 0 | |
| Headsa | 2 | 11.5 | 3.44 | 0 | 8.8 | 3.44 | 0 | 11.5 | 3.44 | 0 | |
| Tailsa | 3 | 30.3 | −0.2 | 0 ± 2 | 25.3 | −0.2 | 0 ± 6 | 30.3 | −0.2 | 0 ± 5 | |
| Headsa | 2 | 11.5 | 3.44 | 0 | 8.8 | 3.44 | 0 | 11.5 | 3.44 | 0 | |
| PAH | 4 | 9.5 | 0 | 60 | 12.4 | 0 | 60 | 7.5 | 0 | 60 | |
| 9 × | (Interlayer | 5 | 188.3 ± 2.9 | 5.7 | – | 149.7 ± 2.4 | 5.7 | – | 201.0 ± 2.6 | 5.7 | – |
| PAH § | 4 | 9.5 ± 2.8 | 0 | 60 ± 9 | 12.4 ± 2.4 | 0 | 60 ± 9 | 7.5 ± 2.5 | 0 | 60 ± 8 | |
| Heads $ | 6 | 11.5 | 3.44 | 45 | 8.8 | 3.44 | 29 | 11.5 | 3.44 | 22 | |
| Tails & | 7 | 30.3 | −0.2 | 45 ± 2 | 25.3 | −0.2 | 29±3 | 30.3 | −0.2 | 22 ± 2 | |
| Heads | 6 | 11.5 | 3.44 | 45 | 8.8 | 3.44 | 29 | 11.5 | 3.44 | 22 | |
| PAH) | 4 | 9.5 | 0 | 60 | 12.4 | 0 | 60 | 7.5 | 0 | 60 | |
| D2O £ | – | s-i | 5.7 | – | s-i | 5.7 | – | s-i | 5.7 | – | |
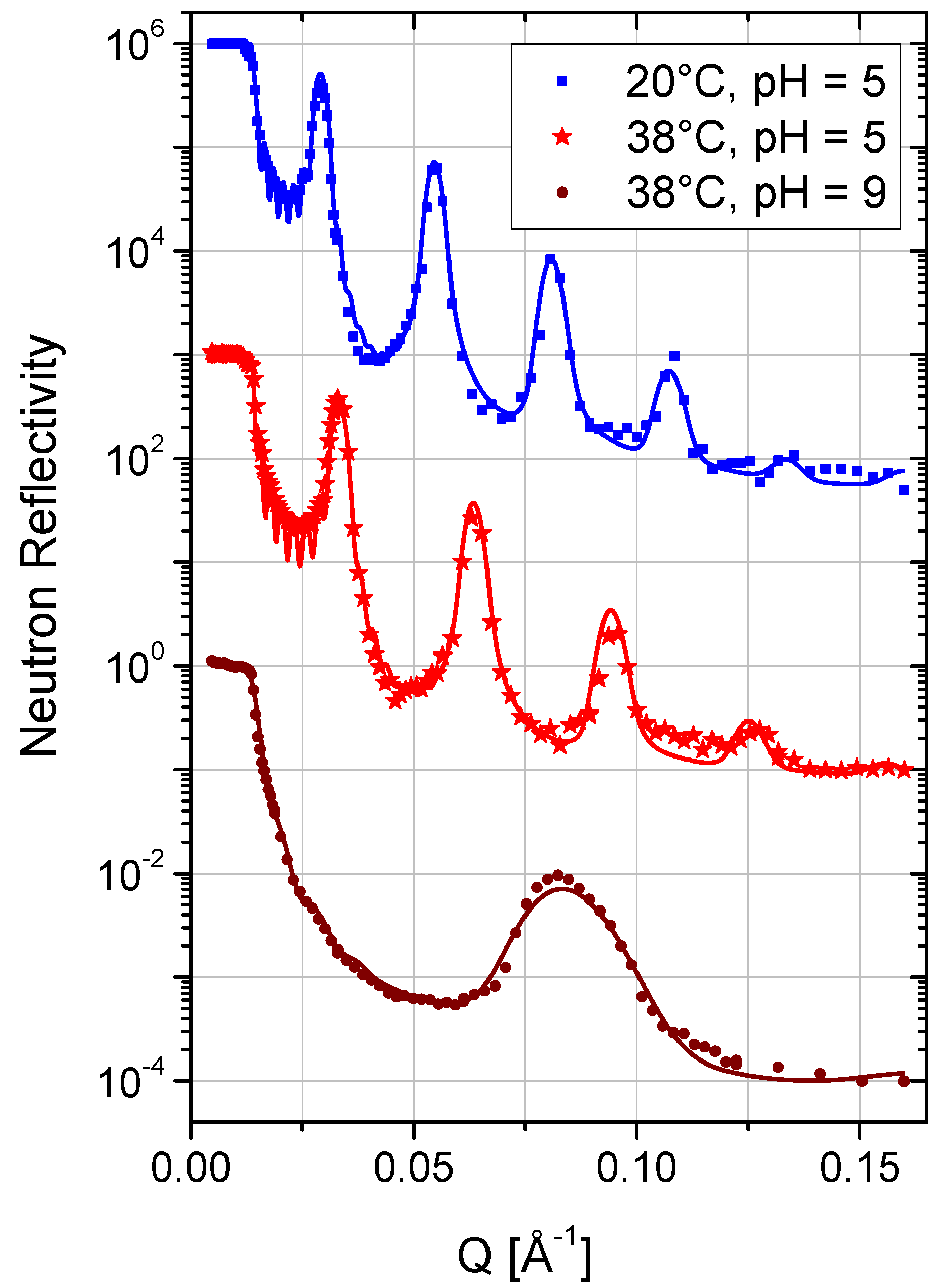
| Layer | 20 °C, pH = 5 | 38 °C, pH = 5 | 38 °C, pH = 9 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | d (Å) | SLD (10−6 Å−2) | ϕsol (%) | ||
| Si | – | s-i | 2.07 | – | s-i | 2.07 | – | s-i | 2.07 | – | |
| SiO2 | 1 | 13.0 | 3.47 | 0 | 13.0 | 3.47 | 0 | 13.0 | 3.47 | 0 | |
| Headsa | 2 | 11.5 | 3.56 | 0 | 8.8 | 3.56 | 0 | 8.8 | 3.56 | 0 | |
| Tailsa | 3 | 30.3 | −0.2 | 0 ± 3 | 25.3 | −0.2 | 0 ± 6 | 25.3 | −0.2 | 0 ± 4 | |
| Headsa | 2 | 11.5 | 3.56 | 0 | 8.8 | 3.56 | 0 | 8.8 | 3.56 | 0 | |
| PAH | 4 | 8.7 | 0 | 65 | 11.0 | 0 | 65 | 12.0 | 0 | 70 | |
| 9 × | (Interlayer | 5 | 165.5 ± 2.6 | 6.0 | – | 137.1 ± 2.3 | 6.0 | – | 9.2 ± 1.0 | 6.0 | – |
| PAH § | 4 | 8.7 ± 2.5 | 0 | 65 ± 4 | 11.0 ± 2.2 | 0 | 65 ± 4 | 12.0 ± 1.0 | 0 | 70 ± 4 | |
| Heads $ | 6 | 11.5 | 3.56 | 21 | 8.8 | 3.56 | 21 | 8.8 | 3.56 | 17 | |
| Tails & | 7 | 30.3 | −0.2 | 21 ± 2 | 25.3 | −0.2 | 21 ± 2 | 25.3 | −0.2 | 17 ± 2 | |
| Heads | 6 | 11.5 | 3.56 | 21 | 8.8 | 3.56 | 21 | 8.8 | 3.56 | 17 | |
| PAH) | 4 | 8.7 | 0 | 65 | 11.0 | 0 | 65 | 12.0 | 0 | 70 | |
| D2O ¥ | – | s-i | 6.0 | – | s-i | 6.0 | – | s-i | 6.0 | – | |
2.3. Data Modeling
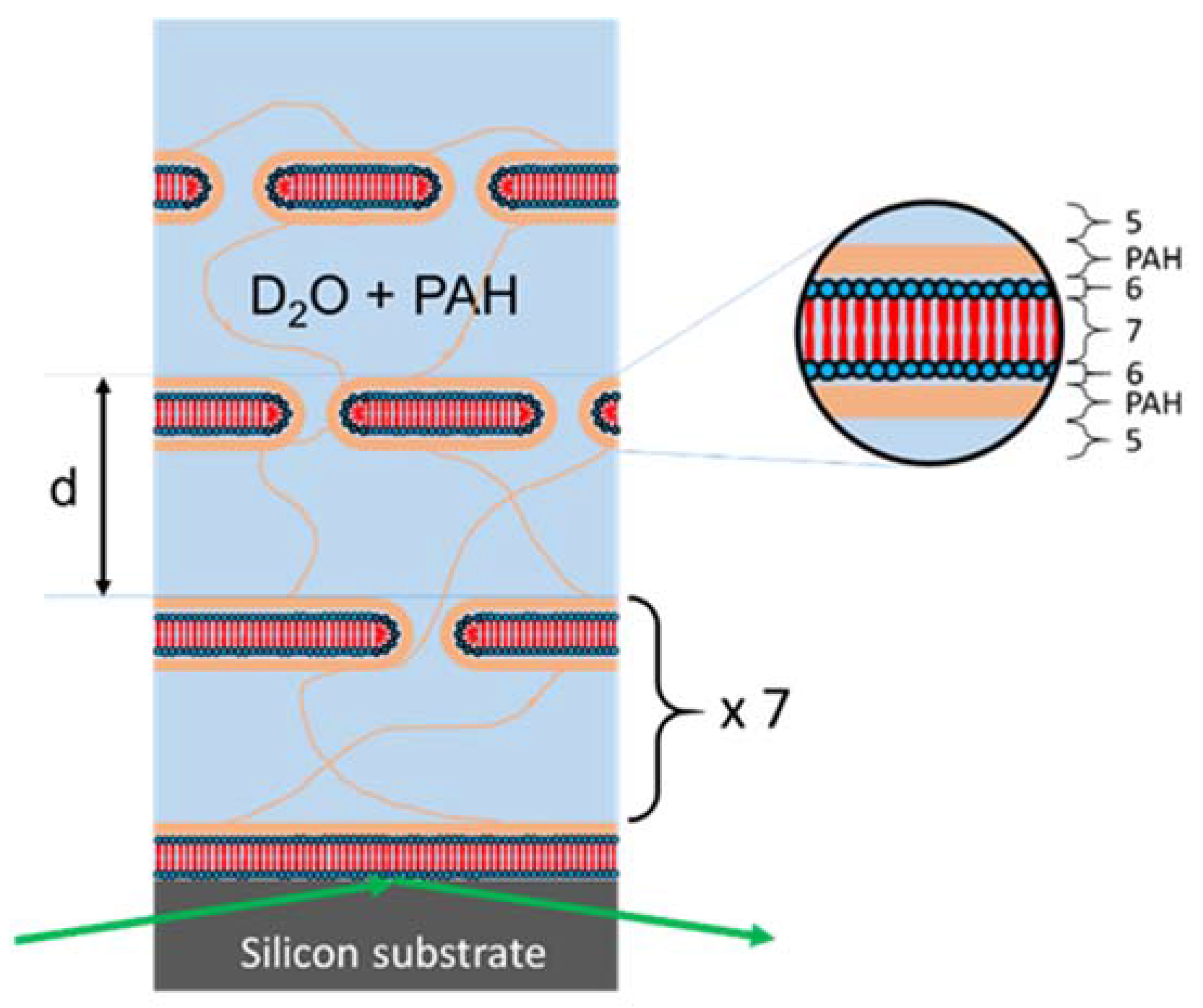
2.4. (Vertical) Structure
2.5. Temperature and Stability
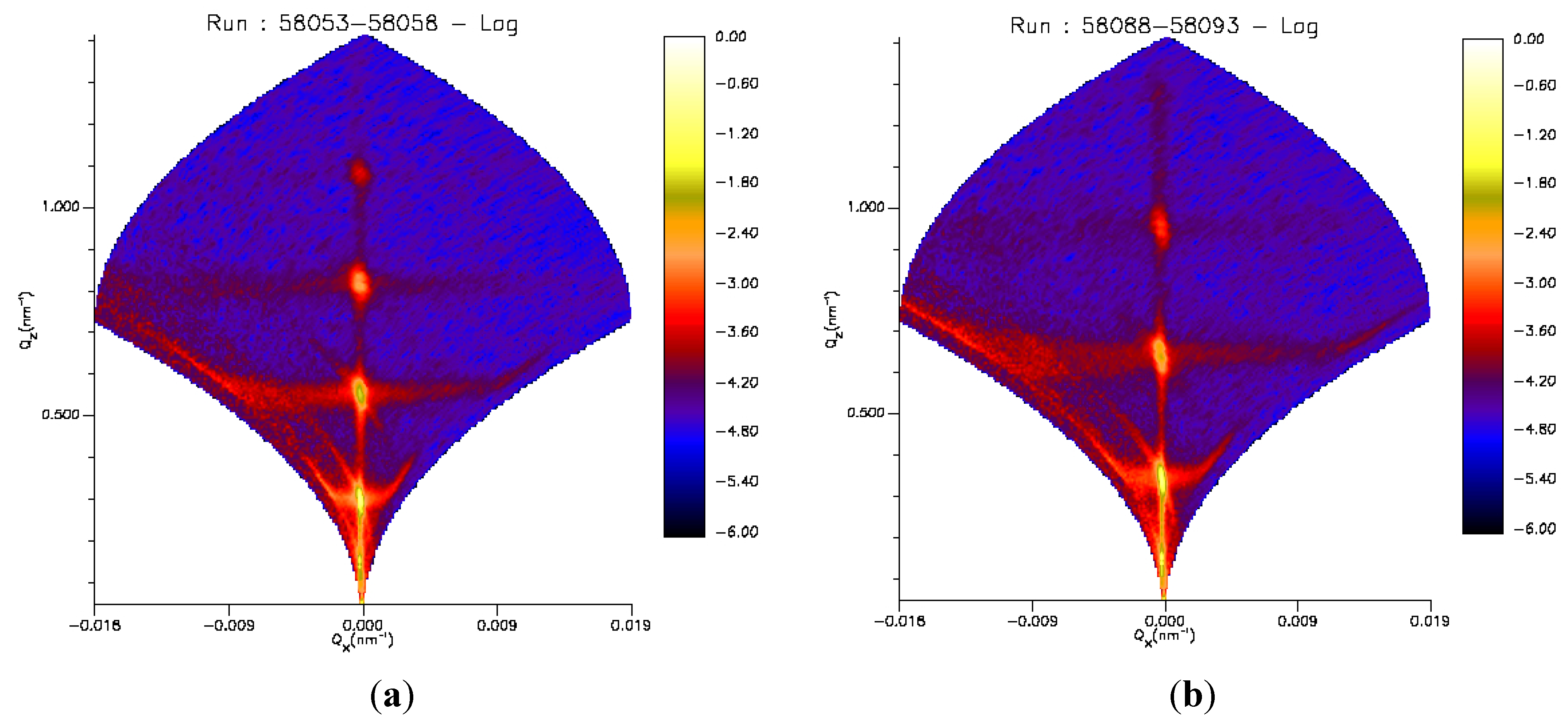
2.6. Time
2.7. Molecular Weight
| Sample | Solution D2O + Polymer (c = 3 mg/mL) | d @ 20 °C (Å) pH = 5 DIPAH = 95% | d @ 38 °C (Å) pH = 5 DIPAH = 95% | d @ 38 °C (Å) pH = 9 DIPAH = 45% | RE (Å) by Equation (3) |
|---|---|---|---|---|---|
| Lit1 [20] | – | 65 ± 1 | 63 ± 1 | n/a | – |
| Lit2 [13] | HA-769 | 234 ± 1 * | 247 ± 1 | n/a | 1637 |
| A | PAH-58 | 257 ± 2 a | 213 ± 2 | n/a | 388 |
| 270 ± 2 b | |||||
| B | PAH-15 | 239 ± 1 | 202 ± 1 | 76 ± 1 | 172 |
2.8. Charge
2.9. Balance of Forces

3. Experimental Section
3.1. Chemicals
3.2. Sample Preparation
3.3. X-Ray Reflectometry
3.4. Neutron Reflectometry
3.5. Reflectometry—Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tanaka, M.; Sackmann, E. Polymer-supported membranes as models of the cell surface. Nature 2005, 437, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Chan, J.M.; Gu, F.X.; Rhee, J.-W.; Wang, A.Z.; Radovic-Moreno, A.F.; Alexis, F.; Langer, R.; Farokhzad, O.C. Self-assembled lipid-polymer hybrid nanoparticles: A robust drug delivery platform. ACS Nano 2008, 2, 1696–1702. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.C.; Langer, R. Impact of nanotechnology on drug delivery. ACS Nano 2009, 3, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Lankalapalli, S.; Kolapalli, V.R.M. Polyelectrolyte complexes: A review of their applicability in drug delivery technology. Indian J. Pharm. Sci. 2009, 71, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Froehlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, Y.; Mao, Z.; Gao, C. Cellular uptake of covalent poly(allylamine hydrochloride) microcapsules and its influences on cell functions. Macromol. Biosci. 2012, 12, 1534–1545. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.V.; Granick, S.; Gewirth, A.A. Modification of a supported lipid bilayer by polyelectrolyte adsorption. Langmuir 2004, 20, 8796–8804. [Google Scholar] [CrossRef] [PubMed]
- Quemeneur, F.; Rinaudo, M.; Pepin-Donat, B. Influence of polyelectrolyte chemical structure on their interaction with lipid membrane of zwitterionic liposomes. Biomacromolecules 2008, 9, 2237–2243. [Google Scholar] [CrossRef] [PubMed]
- Quemeneur, F.; Rinaudo, M.; Maret, G.; Pepin-Donat, B. Decoration of lipid vesicles by polyelectrolytes: Mechanism and structure. Soft Matter 2010, 6, 4471–4481. [Google Scholar] [CrossRef]
- Kunze, K.K.; Netz, R.R. Salt-induced DNA-histone complexation. Phys. Rev. Lett. 2000, 85, 4389–4392. [Google Scholar] [CrossRef] [PubMed]
- Delajon, C.; Gutberlet, T.; Steitz, R.; Mohwald, H.; Krastev, R. Formation of polyelectrolyte multilayer architectures with embedded DMPC studied in situ by neutron reflectometry. Langmuir 2005, 21, 8509–8514. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Junghans, A.; Tian, J.; Dubey, M.; Gnanakaran, S.; Chlistunoff, J.; Majewski, J. Polyelectrolyte multilayers as a platform for pH-responsive lipid bilayers. Soft Matter 2013, 9, 8938–8948. [Google Scholar] [CrossRef]
- Kreuzer, M.; Strobl, M.; Reinhardt, M.; Hemmer, M.C.; Hauß, T.; Dahint, R.; Steitz, R. Impact of a model synovial fluid on supported lipid membranes. Biochim. Biophys. Acta (BBA) Biomembr. 2012, 1818, 2648–2659. [Google Scholar] [CrossRef] [PubMed]
- Kreuzer, M.; Kaltofen, T.; Steitz, R.; Zehnder, B.H.; Dahint, R. Pressure cell for investigations of solid-liquid interfaces by neutron reflectivity. Rev. Sci. Instrum. 2011, 82. [Google Scholar] [CrossRef] [PubMed]
- Chu, N.; Kucerka, N.; Liu, Y.F.; Tristram-Nagle, S.; Nagle, J.F. Anomalous swelling of lipid bilayer stacks is caused by softening of the bending modulus. Phys. Rev. E 2005, 71. [Google Scholar] [CrossRef] [PubMed]
- Mennicke, U.; Salditt, T. Preparation of solid-supported lipid bilayers by spin-coating. Langmuir 2002, 18, 8172–8177. [Google Scholar] [CrossRef]
- Losche, M.; Schmitt, J.; Decher, G.; Bouwman, W.G.; Kjaer, K. Detailed structure of molecularly thin polyelectrolyte multilayer films on solid substrates as revealed by neutron reflectometry. Macromolecules 1998, 31, 8893–8906. [Google Scholar] [CrossRef]
- Sears, V.F. Neutron scattering lengths and cross sections. Neutron News 1992, 3, 26–37. [Google Scholar] [CrossRef]
- Petrache, H.I.; Tristram-Nagle, S.; Nagle, J.F. Fluid phase structure of EPC and DMPC bilayers. Chem. Phys. Lipids 1998, 95, 83–94. [Google Scholar] [CrossRef]
- Kučerka, N.; Nieh, M.-P.; Katsaras, J. Fluid phase lipid areas and bilayer thicknesses of commonly used phosphatidylcholines as a function of temperature. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 2761–2771. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A. Co-refinement of multiple-contrast neutron/X-ray reflectivity data using motofit. J. Appl. Crystallogr. 2006, 39, 273–276. [Google Scholar] [CrossRef]
- Sun, W.J.; Tristram-Nagle, S.; Suter, R.M.; Nagle, J.F. Structure of the ripple phase in lecithin bilayers. Proc. Natl. Acad. Sci. USA 1996, 93, 7008–7012. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, K.; Raghunathan, V.A.; Katsaras, J. Novel structural features of the ripple phase of phospholipids. EPL Europhys. Lett. 2000, 49. [Google Scholar] [CrossRef]
- Harroun, T.A.; Nieh, M.P.; Watson, M.J.; Raghunathan, V.A.; Pabst, G.; Morrow, M.R.; Katsaras, J. Relationship between the unbinding and main transition temperatures of phospholipid bilayers under pressure. Phys. Rev. E 2004, 69. [Google Scholar] [CrossRef] [PubMed]
- Nagle, J.F.; Petrache, H.I.; Gouliaev, N.; Tristram-Nagle, S.; Liu, Y.; Suter, R.M.; Gawrisch, K. Multiple mechanisms for critical behavior in the biologically relevant phase of lecithin bilayers. Phys. Rev. E 1998, 58. [Google Scholar] [CrossRef]
- Tristram-Nagle, S.; Liu, Y.F.; Legleiter, J.; Nagle, J.F. Structure of gel phase DMPC determined by X-ray diffraction. Biophys. J. 2002, 83, 3324–3335. [Google Scholar] [CrossRef]
- Schwörer, F.; Trapp, M.; Ballauff, M.; Dahint, R.; Steitz, R. Surface-active lipid linings under shear load—A combined in-situ neutron reflectivity and ATR-FTIR study. Langmuir 2015, 31, 11539–11548. [Google Scholar] [CrossRef] [PubMed]
- Tatur, S.; Maccarini, M.; Barker, R.; Nelson, A.; Fragneto, G. Effect of functionalized gold nanoparticles on floating lipid bilayers. Langmuir 2013, 29, 6606–6614. [Google Scholar] [CrossRef] [PubMed]
- Watkins, E.B.; El-Khouri, R.J.; Miller, C.E.; Seaby, B.G.; Majewski, J.; Marques, C.M.; Kuhl, T.L. Structure and thermodynamics of lipid bilayers on polyethylene glycol cushions: Fact and fiction of PEG cushioned membranes. Langmuir 2011, 27, 13618–13628. [Google Scholar] [CrossRef] [PubMed]
- McGillivray, D.J.; Valincius, G.; Vanderah, D.J.; Febo-Ayala, W.; Woodward, J.T.; Heinrich, F.; Kasianowicz, J.J.; Losche, M. Molecular-scale structural and functional characterization of sparsely tethered bilayer lipid membranes. Biointerphases 2007, 2, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Teichert, A.; Krist, T.; Steitz, R. Substrate-stress-induced magnetic and nonmagnetic structural correlations in Fe/Si multilayers. J. Appl. Crystallogr. 2015, 48, 1023–1033. [Google Scholar] [CrossRef]
- Salditt, T.; Münster, C.; Lu, J.; Vogel, M.; Fenzl, W.; Souvorov, A. Specular and diffuse scattering of highly aligned phospholipid membranes. Phys. Rev. E 1999, 60. [Google Scholar] [CrossRef]
- Nagle, J.F. Theory of the main lipid bilayer phase transition. Annu. Rev. Phys. Chem. 1980, 31, 157–196. [Google Scholar] [CrossRef]
- Vogel, M.; Munster, C.; Fenzl, W.; Salditt, T. Thermal unbinding of highly oriented phospholipid membranes. Phys. Rev. Lett. 2000, 84, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Rubner, M.F. Influence of the degree of ionization on weak polyelectrolyte multilayer assembly. Macromolecules 2005, 38, 116–124. [Google Scholar] [CrossRef]
- Ohshima, H.; Mitsui, T. Theory of effects of calcium-ions on lamellar phase of dipalmitoyl lecithin. J. Colloid Interface Sci. 1978, 63, 525–537. [Google Scholar] [CrossRef]
- Lis, L.J.; McAlister, M.; Fuller, N.; Rand, R.P.; Parsegian, V.A. Interactions between neutral phospholipid-bilayer membranes. Biophys. J. 1982, 37, 657–665. [Google Scholar] [PubMed]
- Rand, R.P.; Parsegian, V.A. Hydration forces between phospholipid-bilayers. Biochim. Biophys. Acta 1989, 988, 351–376. [Google Scholar] [CrossRef]
- Paunov, V.N.; Binks, B.P. Analytical expression for the electrostatic disjoining pressure taking into account the excluded volume of the hydrated ions between charged interfaces in electrolyte. Langmuir 1999, 15, 2015–2021. [Google Scholar] [CrossRef]
- Cowley, A.C.; Fuller, N.L.; Rand, R.P.; Parsegian, V.A. Measurement of repulsive forces between charged phospholipid bilayers. Biochemistry 1978, 17, 3163–3168. [Google Scholar] [CrossRef] [PubMed]
- Butt, H.-J.; Graf, K.; Kappl, M. Physics and Chemistry of Interfaces; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Netz, R.R.; Andelman, D. Polyelectrolytes in solution and at surfaces. In Encyclopedia of Electrochemistry; Urbakh, M., Giladi, E., Eds.; Wiley-VCH: Weinheim, Germany, 2002; Volume 1, pp. 282–322. [Google Scholar]
- Schneck, E.; Sedlmeier, F.; Netz, R.R. Hydration repulsion between biomembranes results from an interplay of dehydration and depolarization. Proc. Natl. Acad. Sci. USA 2012, 109, 14405–14409. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, Y.G.; Aeffner, S.; Risselada, H.J.; Salditt, T.; Marrink, S.J.; Mueller, M.; Knecht, V. Interbilayer repulsion forces between tension-free lipid bilayers from simulation. Soft Matter 2013, 9, 10705–10718. [Google Scholar] [CrossRef]
- Howse, J.R.; Steitz, R.; Pannek, M.; Simon, P.; Schubert, D.W.; Findenegg, G.H. Adsorbed surfactant layers at polymer/liquid interfaces. A neutron reflectivity study. Phys. Chem. Chem. Phys. 2001, 3, 4044–4051. [Google Scholar] [CrossRef]
- Howse, J.R.; Manzanares-Papayanopoulos, E.; McLure, I.A.; Bowers, J.; Steitz, R.; Findenegg, G.H. Critical adsorption and boundary layer structure of 2-butoxyethanol + D2O mixtures at a hydrophilic silica surface. J. Chem. Phys. 2002, 116, 7177–7188. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kreuzer, M.; Trapp, M.; Dahint, R.; Steitz, R. Polymer-Induced Swelling of Solid-Supported Lipid Membranes. Membranes 2016, 6, 2. https://doi.org/10.3390/membranes6010002
Kreuzer M, Trapp M, Dahint R, Steitz R. Polymer-Induced Swelling of Solid-Supported Lipid Membranes. Membranes. 2016; 6(1):2. https://doi.org/10.3390/membranes6010002
Chicago/Turabian StyleKreuzer, Martin, Marcus Trapp, Reiner Dahint, and Roland Steitz. 2016. "Polymer-Induced Swelling of Solid-Supported Lipid Membranes" Membranes 6, no. 1: 2. https://doi.org/10.3390/membranes6010002
APA StyleKreuzer, M., Trapp, M., Dahint, R., & Steitz, R. (2016). Polymer-Induced Swelling of Solid-Supported Lipid Membranes. Membranes, 6(1), 2. https://doi.org/10.3390/membranes6010002





