Challenges and Solutions for Global Water Scarcity
Abstract
1. Introduction
2. Essential Measures to Address the Worldwide Water Shortage
2.1. Centralized Governance
2.2. Education
2.3. Water Catchment and Harvesting Technologies
2.4. Water Infrastructure
2.5. Irrigation and Agricultural Practices
2.6. Pollution Control
2.7. Novel Technologies
2.8. Transboundary Water Cooperation
3. Desalination
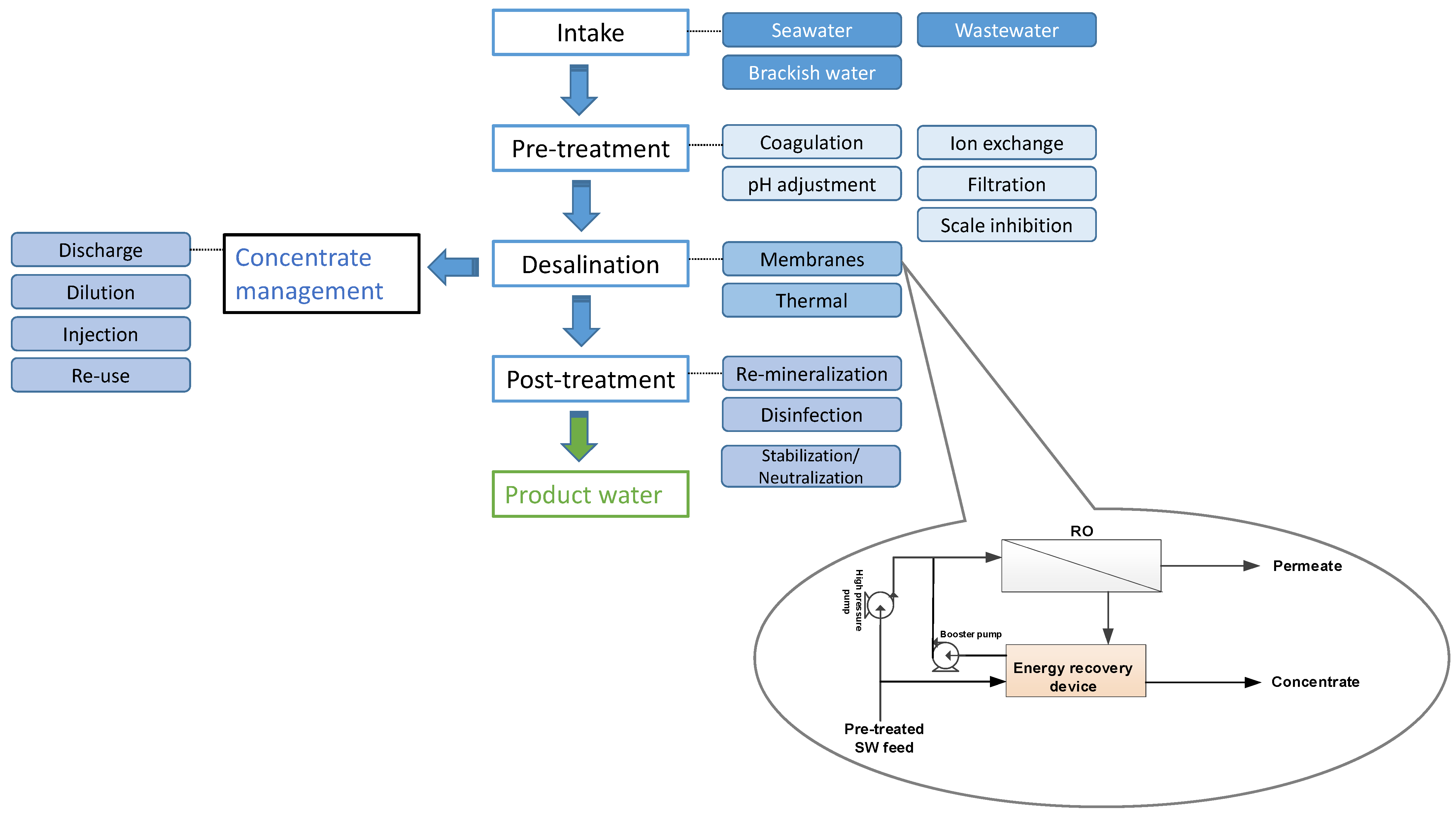
3.1. Overview
3.2. Seawater Reverse Osmosis
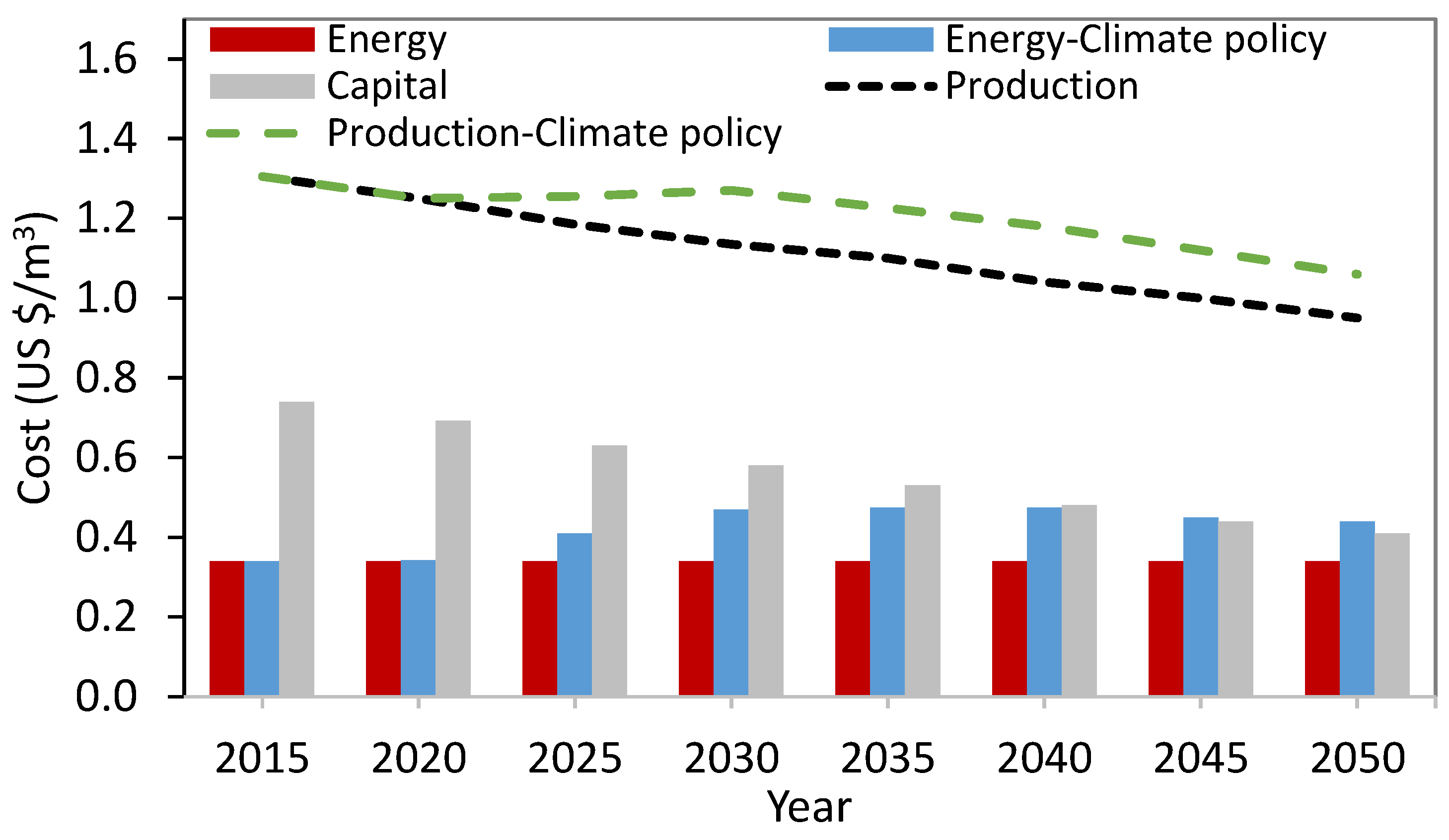
Energy Demand and Cost of SWRO Desalination
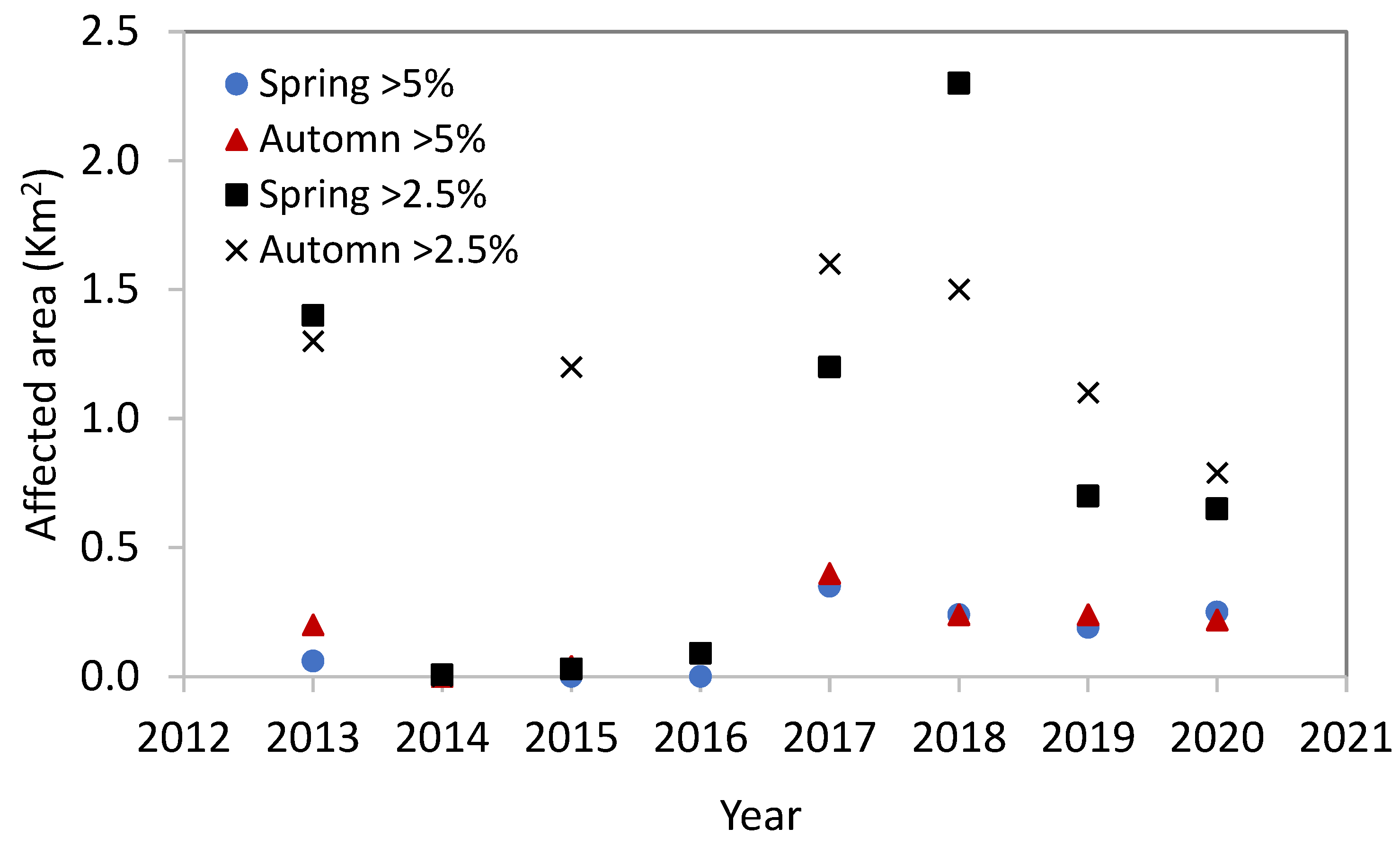
3.3. Environmental Aspects of SWRO Desalination
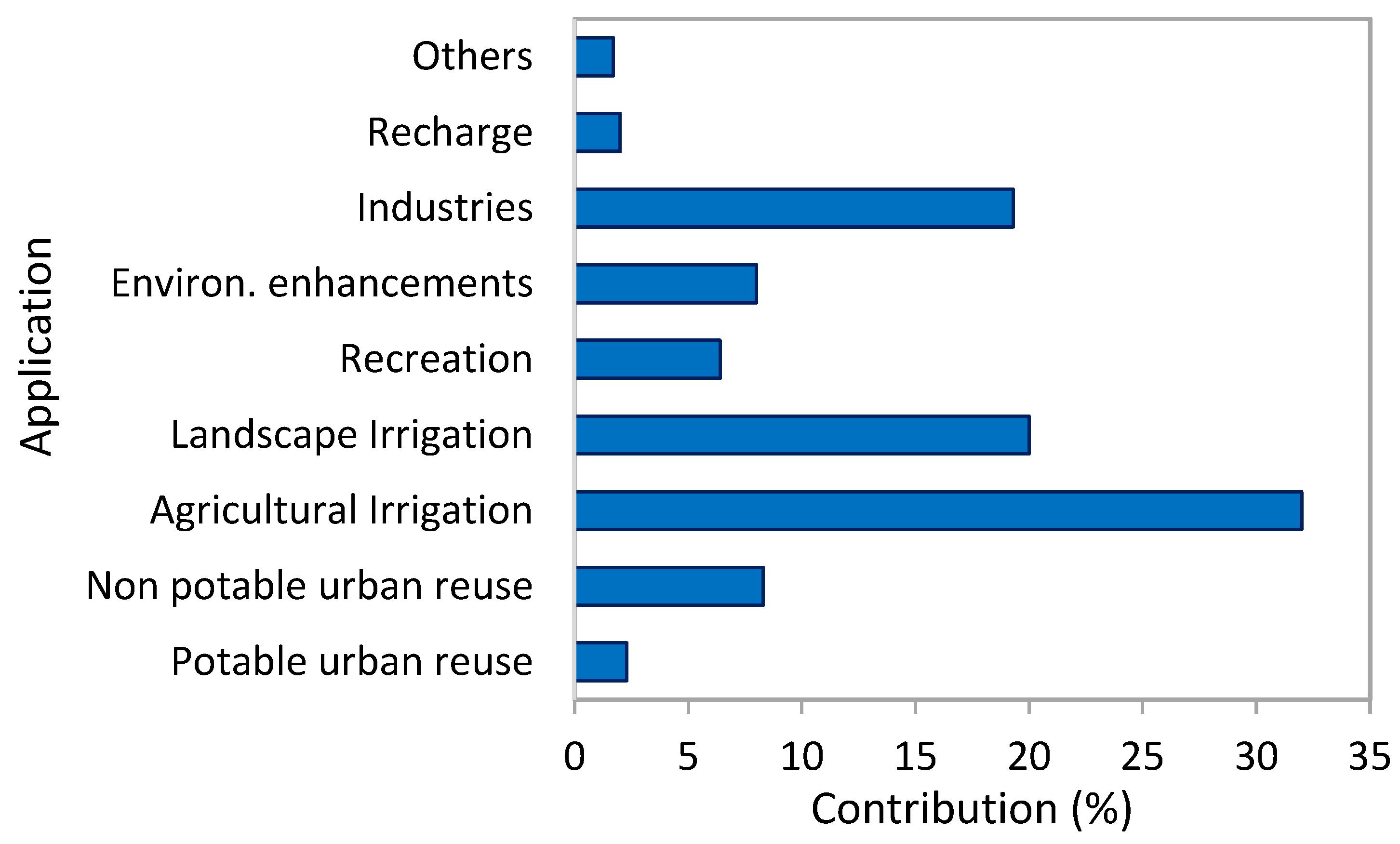
4. Water Reclamation
4.1. Overview
4.2. Microfiltration and Ultrafiltration
4.3. Reverse Osmosis and Nanofiltration
4.4. Membrane Bioreactors—MBR
4.5. Gravity-Driven Membrane-GDM
5. Summary
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations Sustainable Development Goals. Clean Water and Sanitation. Available online: https://unstats.un.org/sdgs/report/2022/The-Sustainable-Development-Goals-Report-2022.pdf/ (accessed on 20 June 2022).
- Ayboga, E. Policy, and Impacts of Dams in the Euphrates and Tigris Basin. Mesopotamia Water Forum 6–8 April 2019, Sulaimani, Kurdistan Region of Iraq. Available online: https://www.savethetigris.org/wp-content/uploads/2019/01/Paper-Challenge-B-Dams-FINAL-to-be-published.pdf (accessed on 12 December 2022).
- Dhakal, N.; Salinas-Rodriguez, S.G.; Hamdani, J.; Abushaban, A.; Sawalha, H.; Schippers, J.C.; Kennedy, M.D. Is Desalination a Solution to Freshwater Scarcity in Developing Countries? Membranes 2022, 12, 381. [Google Scholar] [CrossRef]
- Domènech, L. Rethinking water management: From centralised to decentralised water supplyand sanitation models. Doc. D’anàlisi Geogràfica 2011, 57, 293. [Google Scholar] [CrossRef]
- Kislev, Y. The Water Economy of Israel. Taub Center for Social Policy Studies in Israel Jerusalem. Policy Paper No. 2011.15, Nov. 2011. Available online: https://openscholar.huji.ac.il/sites/default/files/agri_economics/files/38-2011_water_economy_taub_center.pdf (accessed on 10 January 2023).
- Bar-Nahum, Z.; Reznik, A.; Finkelshtain, I.; Kan, I. Centralized water management under lobbying: Economic analysis of desalination in Israel. Ecol. Econ. 2021, 193, 107320. [Google Scholar] [CrossRef]
- Pugel, K.; Javernick-Will, A.; Peabody, S.; Nyaga, C.; Mussa, M.; Mekonta, L.; Dimtse, D.; Watsisi, M.; Buhungiro, E.; Mulatu, T.; et al. Pathways for collaboratively strengthening water and sanitation systems. Sci. Total Environ. 2021, 802, 149854. [Google Scholar] [CrossRef]
- Galvez, V.; Rojas, R.; Bennison, G.; Prats, C.; Claro, E. Collaborate or perish: Water resources management under contentious water use in a semiarid basin. Int. J. River Basin Manag. 2020, 18, 421. [Google Scholar] [CrossRef]
- Sachidananda, M.; Webb, D.P.; Rahimifard, S. A Concept of Water Usage Efficiency to Support Water Reduction in Manufacturing Industry. Sustainability 2016, 8, 1222. [Google Scholar] [CrossRef]
- Bauer, S.; Wagner, M. Possibilities and Challenges of Wastewater Reuse—Planning Aspects and Realized Examples. Water 2022, 14, 1619. [Google Scholar] [CrossRef]
- Baba, A.; Tsatsanifos, C.; El Gohary, F.; Palerm, J.; Khan, S.; Mahmoudian, S.A.; Ahmed, A.T.; Tayfur, G.; Dialynas, Y.G.; Angelakis, A.N. Developments in water dams and water harvesting systems throughout history in different civilizations. Int. J. Hydrol. 2018, 2, 62–65. [Google Scholar] [CrossRef]
- Alim, M.A.; Rahman, A.; Tao, Z.; Samali, B.; Khan, M.M.; Shirin, S. Suitability of roof harvested rainwater for potential potable water production: A scoping review. J. Clean. Prod. 2020, 248, 119226. [Google Scholar] [CrossRef]
- Rojas, E.M.; Ortiz, E.A.D.; Tafur, C.A.M.; García, L.; Oliva, M.; Briceño, N.B.R. A Rainwater Harvesting and Treatment System for Domestic Use and Human Consumption in Native Communities in Amazonas (NW Peru): Technical and Economic Validation. Scientifica 2021, 2021, 4136379. [Google Scholar] [CrossRef]
- Yildirim, G.; Alim, M.A.; Rahman, A. Review of Rainwater Harvesting Research by a Bibliometric Analysis. Water 2022, 14, 3200. [Google Scholar] [CrossRef]
- Xu, J.; Dai, J.; Wu, X.; Wu, S.; Zhang, Y.; Wang, F.; Gao, A.; Tan, Y. Urban rainwater utilization: A review of management modes and harvesting systems. Front. Environ. Sci. 2023, 11, 118. [Google Scholar] [CrossRef]
- Silva, A.C.R.d.S.; Bimbato, A.M.; Balestieri, J.A.P.; Vilanova, M.R.N. Exploring environmental, economic and social aspects of rainwater harvesting systems: A review. Sustain. Cities Soc. 2022, 76, 103475. [Google Scholar] [CrossRef]
- Singh, S.; Yadav, R.; Kathi, S.; Singh, A.N. Chapter 14-Treatment of harvested rainwater and reuse: Practices, prospects, and challenges. In Cost Effective Technologies for Solid Waste and Wastewater Treatment; Kathi, S., Devipriya, S., Thamaraiselvi, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 161–178. [Google Scholar] [CrossRef]
- Pala, G.K.; Pathivada, A.P.; Velugoti, S.J.H.; Yerramsetti, C.; Veeranki, S. Rainwater harvesting—A review on conservation, creation & cost-effectiveness. Mater. Today Proc. 2021, 45, 6567–6571. [Google Scholar] [CrossRef]
- Semaan, M.; Day, S.D.; Garvin, M.; Ramakrishnan, N.; Pearce, A. Optimal sizing of rainwater harvesting systems for domestic water usages: A systematic literature review. Resour. Conserv. Recycl. X 2020, 6, 100033. [Google Scholar] [CrossRef]
- Słyś, D.; Stec, A. Centralized or Decentralized Rainwater Harvesting Systems: A Case Study. Resources 2020, 9, 5. [Google Scholar] [CrossRef]
- Velasco-Muñoz, J.F.; Aznar-Sánchez, J.A.; Batlles-Delafuente, A.; Fidelibus, M.D. Rainwater Harvesting for Agricultural Irrigation: An Analysis of Global Research. Water 2019, 11, 1320. [Google Scholar] [CrossRef]
- Yannopoulos, S.; Giannopoulou, I.; Kaiafa-Saropoulou, M. Investigation of the Current Situation and Prospects for the Development of Rainwater Harvesting as a Tool to Confront Water Scarcity Worldwide. Water 2019, 11, 2168. [Google Scholar] [CrossRef]
- Malik, R.; Giordano, M.; Sharma, V. Examining farm-level perceptions, costs, and benefits of small water harvesting structures in Dewas, Madhya Pradesh. Agric. Water Manag. 2014, 131, 204–211. [Google Scholar] [CrossRef]
- Basel, B.; Quiroz, N.H.; Herrera, R.V.; Alonso, C.S.; Hoogesteger, J. Bee mietii rak rkabni nis (The people know how to seed water): A Zapotec experience in adapting to water scarcity and drought. Clim. Dev. 2020, 13, 792–806. [Google Scholar] [CrossRef]
- Basel, B.; Hoogesteger, J.; Hellegers, P. Promise and paradox: A critical sociohydrological perspective on small-scale managed aquifer recharge. Front. Water 2022, 4, 1–14. [Google Scholar] [CrossRef]
- Seddon, N.; Chausson, A.; Berry, P.; Girardin, C.A.J.; Smith, A.; Turner, B. Understanding the value and limits of nature-based solutions to climate change and other global challenges. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190120. [Google Scholar] [CrossRef]
- Pamidimukkala, A.; Kermanshachi, S.; Adepu, N.; Safapour, E. Resilience in Water Infrastructures: A Review of Challenges and Adoption Strategies. Sustainability 2021, 13, 12986. [Google Scholar] [CrossRef]
- Butler, D.; Ward, S.; Sweetapple, C.; Astaraie-Imani, M.; Diao, K.; Farmani, R.; Fu, G. Reliable, resilient and sustainable water management: The Safe & SuRe approach. Glob. Chall. 2016, 1, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Kang, H. Challenges for water infrastructure asset management in South Korea. Water Policy 2019, 21, 934–944. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, L.; Yang, M. Digitalisation for Water Sustainability: Barriers to Implementing Circular Economy in Smart Water Management. Sustainability 2021, 13, 11868. [Google Scholar] [CrossRef]
- Taormina, R.; Galelli, S.; Tippenhauer, N.O.; Salomons, E.; Ostfeld, A. Characterizing Cyber-Physical Attacks on Water Distribution Systems. J. Water Resour. Plan. Manag. 2017, 143, 04017009. [Google Scholar] [CrossRef]
- FAO. The State of Food and Agriculture 2020, Overcoming Water Challenges in Agriculture; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Bwambale, E.; Abagale, F.K.; Anornu, G.K. Smart irrigation monitoring and control strategies for improving water use efficiency in precision agriculture: A review. Agric. Water Manag. 2021, 260, 107324. [Google Scholar] [CrossRef]
- Levidow, L.; Zaccaria, D.; Maia, R.; Vivas, E.; Todorovic, M.; Scardigno, A. Improving water-efficient irrigation: Prospects and difficulties of innovative practices. Agric. Water Manag. 2014, 146, 84–94. [Google Scholar] [CrossRef]
- Bertule, M.; Appelquist, L.R.; Spensley, J.; Trærup, S.L.M.; Naswa, P. Climate Change Adaptation Technologies for Water: A Practitioner’s Guide to Adaptation Technologies for Increased Water Sector Resilience; CTCN Publications: Copenhagen, Denmark, 2018. [Google Scholar]
- Tal, A. Rethinking the sustainability of Israel’s irrigation practices in the Drylands. Water Res. 2016, 90, 387–394. [Google Scholar] [CrossRef]
- Sauvé, S.; Desrosiers, M. A review of what is an emerging contaminant. Chem. Cent. J. 2014, 8, 15. [Google Scholar] [CrossRef]
- Zhao, L.; Deng, J.; Sun, P.; Liu, J.; Ji, Y.; Nakada, N.; Qiao, Z.; Tanaka, H.; Yang, Y. Nanomaterials for treating emerging contaminants in water by adsorption and photocatalysis: Systematic review and bibliometric analysis. Sci. Total Environ. 2018, 627, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- Rathi, B.S.; Kumar, P.S.; Show, P.-L. A review on effective removal of emerging contaminants from aquatic systems: Current trends and scope for further research. J. Hazard. Mater. 2020, 409, 124413. [Google Scholar] [CrossRef] [PubMed]
- Sivaranjanee, R.; Kumar, P.S. A review on remedial measures for effective separation of emerging contaminants from wastewater. Environ. Technol. Innov. 2021, 23, 101741. [Google Scholar] [CrossRef]
- Shahid, M.K.; Kashif, A.; Fuwad, A.; Choi, Y. Current advances in treatment technologies for removal of emerging contaminants from water—A critical review. Coord. Chem. Rev. 2021, 442, 213993. [Google Scholar] [CrossRef]
- Kumar, R.; Qureshi, M.; Vishwakarma, D.K.; Al-Ansari, N.; Kuriqi, A.; Elbeltagi, A.; Saraswat, A. A review on emerging water contaminants and the application of sustainable removal technologies. Case Stud. Chem. Environ. Eng. 2022, 6, 100219. [Google Scholar] [CrossRef]
- Varsha, M.; Kumar, P.S.; Rathi, B.S. A review on recent trends in the removal of emerging contaminants from aquatic environment using low-cost adsorbents. Chemosphere 2022, 287, 132270. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Lichtfouse, E.; Fourmentin, M.; Ribeiro, A.R.L.; Noutsopoulos, C.; Mapelli, F.; Fenyvesi, É.; Vieira, M.G.A.; Picos-Corrales, L.A.; Moreno-Piraján, J.C.; et al. Removal of emerging contaminants from wastewater using advanced treatments. A review. Environ. Chem. Lett. 2022, 20, 1333–1375. [Google Scholar] [CrossRef]
- Sengupta, A.; Jebur, M.; Kamaz, M.; Wickramasinghe, S.R. Removal of Emerging Contaminants from Wastewater Streams Using Membrane Bioreactors: A Review. Membranes 2022, 12, 60. [Google Scholar] [CrossRef]
- Priyadarshini, M.; Das, I.; Ghangrekar, M.M.; Blaney, L. Advanced oxidation processes: Performance, advantages, and scale-up of emerging technologies. J. Environ. Manag. 2022, 316, 115295. [Google Scholar] [CrossRef]
- Kumar, V.; Bilal, M.; Ferreira, L.F.R. Editorial: Recent Trends in Integrated Wastewater Treatment for Sustainable Development. Front. Microbiol. 2022, 13, 1101. [Google Scholar] [CrossRef]
- Söderholm, P. The green economy transition: The challenges of technological change for sustainability. Sustain. Earth 2020, 3, 6. [Google Scholar] [CrossRef]
- Ahmed, F.; Johnson, D.; Hashaikeh, R.; Hilal, N. Barriers to Innovation in Water Treatment. Water 2023, 15, 773. [Google Scholar] [CrossRef]
- Süsser, D. Accelerating Cleantech Commercialization in Israel: Green Innovation as a Catalyst for Sustainable Development; Policy Paper Series “Decarbonization Strategies in Germany and Israel”; Institute for Advanced Sustainability Studies (IASS): Potsdam, Germany; Israel Public Policy Institute (IPPI): Tel Aviv, Israel; Heinrich Böll-Stiftung: Tel Aviv, Israel, 2020. [Google Scholar] [CrossRef]
- Hao, J. A Study of Cleantech Innovations in the Israeli Entrepreneurial Ecosystem. MIT Reap. 2018. Available online: https://reap.mit.edu/assets/Junli-Final.pdf (accessed on 5 May 2023).
- Petersen-Perlman, J.D.; Veilleux, J.C.; Wolf, A.T. International water conflict and cooperation: Challenges and opportunities. Water Int. 2017, 42, 105–120. [Google Scholar] [CrossRef]
- UNECE. Policy Guidance Note on the Benefits of Transboundary Water Cooperation: Identification, Assessment, and Communication. Convention on the Protection and Use of Transboundary Watercourses and International Lakes. United Nations Economic Commission for Europe (UNECE). 2015. Available online: https://unece.org/environment-policy/publications/policy-guidance-note-benefits-transboundary-water-cooperation (accessed on 7 May 2023).
- CADRI Partnership. Good Practices on Transboundary Water Resources Management and Cooperation. CADRI Partnership under the Leadership of FAO with Inputs from UN Environment and the United Nations Economic Commission for Europe. 2020. Available online: https://www.cadri.net/system/files/2021-09 (accessed on 5 May 2023).
- Mohammed, I.N.; Bolten, J.D.; Souter, N.J.; Shaad, K.; Vollmer, D. Diagnosing challenges and setting priorities for sustainable water resource management under climate change. Sci. Rep. 2022, 12, 796. [Google Scholar] [CrossRef]
- UN World Development Report 2022. Groundwater, Making the Invisible Visible. Available online: https://www.unesco.org/reports/wwdr/2022/en (accessed on 12 May 2023).
- Shehata, N.; Egirani, D.; Olabi, A.; Inayat, A.; Abdelkareem, M.A.; Chae, K.-J.; Sayed, E.T. Membrane-based water and wastewater treatment technologies: Issues, current trends, challenges, and role in achieving sustainable development goals, and circular economy. Chemosphere 2023, 320, 137993. [Google Scholar] [CrossRef] [PubMed]
- Curto, D.; Franzitta, V.; Guercio, A. A Review of the Water Desalination Technologies. Appl. Sci. 2021, 11, 670. [Google Scholar] [CrossRef]
- Najim, A. A review of advances in freeze desalination and future prospects. Npj Clean Water 2022, 5, 15. [Google Scholar] [CrossRef]
- Bo, Z.; Huang, Z.; Xu, C.; Chen, Y.; Wu, E.; Yan, J.; Cen, K.; Yang, H.; Ostrikov, K. Anion-kinetics-selective graphene anode and cation-energy-selective MXene cathode for high-performance capacitive deionization. Energy Storage Mater. 2022, 50, 395–406. [Google Scholar] [CrossRef]
- Zhao, X.; Wei, H.; Zhao, H.; Wang, Y.; Tang, N. Electrode materials for capacitive deionization: A review. J. Electroanal. Chem. 2020, 873, 114416. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality: First Addendum, 4th ed.; WHO: Geneva, Switzerland, 2017; pp. 323–324. [Google Scholar]
- Vera, A.; Bastida, F.; Patiño-García, M.; Moreno, J.L. The effects of boron-enriched water irrigation on soil microbial community are dependent on crop species. Appl. Soil Ecol. 2023, 181, 104677. [Google Scholar] [CrossRef]
- Jacob, C. Seawater desalination: Boron removal by ion exchange technology. Desalination 2007, 205, 47–52. [Google Scholar] [CrossRef]
- Landsman, M.R.; Lawler, D.F.; Katz, L.E. Application of electrodialysis pretreatment to enhance boron removal and reduce fouling during desalination by nanofiltration/reverse osmosis. Desalination 2020, 491, 114563. [Google Scholar] [CrossRef]
- Milne, N.A.; O’Reilly, T.; Sanciolo, P.; Ostarcevic, E.; Beighton, M.; Taylor, K.; Mullett, M.; Tarquin, A.J.; Gray, S.R. Chemistry of silica scale mitigation for RO desalination with particular reference to remote operations. Water Res. 2014, 65, 107–133. [Google Scholar] [CrossRef] [PubMed]
- Haidari, A.; Witkamp, G.; Heijman, S. High silica concentration in RO concentrate. Water Resour. Ind. 2022, 27, 100171. [Google Scholar] [CrossRef]
- Matin, A.; Rahman, F.; Shafi, H.Z.; Zubair, S.M. Scaling of reverse osmosis membranes used in water desalination: Phenomena, impact, and control; future directions. Desalination 2019, 455, 135–157. [Google Scholar] [CrossRef]
- Valavala, R.; Sohn, J.-S.; Han, J.-H.; Her, N.-G.; Yoon, Y.-M. Pretreatment in Reverse Osmosis Seawater Desalination: A Short Review. Environ. Eng. Res. 2011, 16, 205–212. [Google Scholar] [CrossRef]
- Charcosset, C. Classical and Recent Developments of Membrane Processes for Desalination and Natural Water Treatment. Membranes 2022, 12, 267. [Google Scholar] [CrossRef]
- Hasson, D.; Semiat, R. Scale Control in Saline and Wastewater Desalination. Isr. J. Chem. 2006, 46, 97–104. [Google Scholar] [CrossRef]
- Cohen, Y.; Semiat, R.; Rahardianto, A. A perspective on reverse osmosis water desalination: Quest for sustainability. AIChE J. 2017, 63, 1771–1784. [Google Scholar] [CrossRef]
- Lesimple, A.; Ahmed, F.E.; Hilal, N. Remineralization of desalinated water: Methods and environmental impact. Desalination 2020, 496, 114692. [Google Scholar] [CrossRef]
- Shemer, H.; Hasson, D.; Semiat, R. State-of-the-art review on post-treatment technologies. Desalination 2015, 356, 285–293. [Google Scholar] [CrossRef]
- Missimer, T.M.; Maliva, R.G. Environmental issues in seawater reverse osmosis desalination: Intakes and outfalls. Desalination 2018, 434, 198–215. [Google Scholar] [CrossRef]
- Backer, S.N.; Bouaziz, I.; Kallayi, N.; Thomas, R.T.; Preethikumar, G.; Takriff, M.S.; Laoui, T.; Atieh, M.A. Review: Brine Solution: Current Status, Future Management and Technology Development. Sustainability 2022, 14, 6752. [Google Scholar] [CrossRef]
- Bello, A.S.; Zouari, N.; Da’Ana, D.A.; Hahladakis, J.N.; Al-Ghouti, M.A. An overview of brine management: Emerging desalination technologies, life cycle assessment, and metal recovery methodologies. J. Environ. Manag. 2021, 288, 112358. [Google Scholar] [CrossRef]
- Al-Absi, R.S.; Abu-Dieyeh, M.; Al-Ghouti, M.A. Brine management strategies, technologies, and recovery using adsorption processes. Environ. Technol. Innov. 2021, 22, 101541. [Google Scholar] [CrossRef]
- Soliman, M.N.; Guen, F.Z.; Ahmed, S.A.; Saleem, H.; Khalil, M.J.; Zaidi, S.J. Energy consumption and environmental impact assessment of desalination plants and brine disposal strategies. Process. Saf. Environ. Prot. 2021, 147, 589–608. [Google Scholar] [CrossRef]
- Voutchkov, N.; Semiat, R. Seawater Desalination, in Advanced Membrane Technology and Applications; Li, N.N., Ho, W.S., Fane, A.G., Matsuura, T., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar]
- Wang, X.-N.; Ma, M.-Y.; Pan, X.-H.; Hao, J.; Zhang, C.-N. Quality of product water by three full-scale seawater reverse osmosis desalination in China. Desalin. Water Treat 2020, 174, 46–52. [Google Scholar] [CrossRef]
- Semiat, R. Water Purification: Materials and Technologies. In Encyclopedia of Materials: Science and Technology, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2010; pp. 1–4. [Google Scholar] [CrossRef]
- Shokri, A.; Fard, M.S. Techno-economic assessment of water desalination: Future outlooks and challenges. Process. Saf. Environ. Prot. 2023, 169, 564–578. [Google Scholar] [CrossRef]
- Gao, L.; Yoshikawa, S.; Iseri, Y.; Fujimori, S.; Kanae, S. An Economic Assessment of the Global Potential for Seawater Desalination to 2050. Water 2017, 9, 763. [Google Scholar] [CrossRef]
- Miller, S.; Shemer, H.; Semiat, R. Energy and environmental issues in desalination. Desalination 2015, 366, 2–8. [Google Scholar] [CrossRef]
- Shemer, H.; Semiat, R. Sustainable RO desalination—Energy demand and environmental impact. Desalination 2017, 424, 10–16. [Google Scholar] [CrossRef]
- Park, J.; Lee, S. Desalination Technology in South Korea: A Comprehensive Review of Technology Trends and Future Outlook. Membranes 2022, 12, 204. [Google Scholar] [CrossRef]
- Chu, S.; Zhang, S.; Ma, X.; Li, Y.; Qiu, D.; Ge, W.; Kou, L. Experimental Study on the Influence of Flexible Control on Key Parameters in Reverse Osmosis Desalination. IEEE Access 2022, 10, 4844–4860. [Google Scholar] [CrossRef]
- Himeur, Y.; Elnour, M.; Fadli, F.; Meskin, N.; Petri, I.; Rezgui, Y.; Bensaali, F.; Amira, A. AI-big data analytics for building automation and management systems: A survey, actual challenges and future perspectives. Artif. Intell. Rev. 2023, 56, 4929–5021. [Google Scholar] [CrossRef]
- Kurihara, M. Current Status and Future Trend of Dominant Commercial Reverse Osmosis Membranes. Membranes 2021, 11, 906. [Google Scholar] [CrossRef] [PubMed]
- Kettani, M.; Bandelier, P. Techno-economic assessment of solar energy coupling with large-scale desalination plant: The case of Morocco. Desalination 2020, 494, 114627. [Google Scholar] [CrossRef]
- Wilf, M. Fundamentals of RO-NF technology. In Proceedings of the International Conference on Desalination Costing, Limassol, Cyprus, 6–8 December 2004. [Google Scholar]
- Glueckstern, P. History of desalination cost estimations. In Proceedings of the International Conference on Desalination Costing, Limassol, Cyprus, 6–8 December 2004. [Google Scholar]
- Wittholz, M.K.; O’Neill, B.K.; Colby, C.B.; Lewis, D. Estimating the cost of desalination plants using a cost database. Desalination 2008, 229, 10–20. [Google Scholar] [CrossRef]
- Reddy, K.; Ghaffour, N. Overview of the cost of desalinated water and costing methodologies. Desalination 2007, 205, 340–353. [Google Scholar] [CrossRef]
- Reddy, K.V. Review and evaluation of desalination cost and costing methodologies. Int. J. Nucl. Desalin. 2008, 3, 79. [Google Scholar] [CrossRef]
- Jones, E.; Qadir, M.; van Vliet, M.T.; Smakhtin, V.; Kang, S.-M. The state of desalination and brine production: A global outlook. Sci. Total Environ. 2019, 657, 1343–1356. [Google Scholar] [CrossRef]
- Lokiec, F. Sustainable desalination: Environmental approaches. In Proceedings of the International Desalination Association World Congress on Desalination and Water Reuse, Tianjin, China, 20–25 October 2013. [Google Scholar]
- Thi, H.T.D.; Pasztor, T.; Fozer, D.; Manenti, F.; Toth, A.J. Comparison of Desalination Technologies Using Renewable Energy Sources with Life Cycle, PESTLE, and Multi-Criteria Decision Analyses. Water 2021, 13, 3023. [Google Scholar] [CrossRef]
- El-Ghonemy, A. RETRACTED: Water desalination systems Powered by Renewable energy sources, Review. Renew. Sustain. Energy Rev. 2012, 16, 1537–1556. [Google Scholar] [CrossRef]
- Mohammad, N.; Ishak, W.W.M.; Mustapa, S.I.; Ayodele, B.V. Natural Gas as a Key Alternative Energy Source in Sustainable Renewable Energy Transition: A Mini Review. Front. Energy Res. 2021, 9, 625023. [Google Scholar] [CrossRef]
- Safari, A.; Das, N.; Langhelle, O.; Roy, J.; Assadi, M. Natural gas: A transition fuel for sustainable energy system transformation? Energy Sci. Eng. 2019, 7, 1075–1094. [Google Scholar] [CrossRef]
- Pankratz, T. Overview of Intake Systems for Seawater Reverse Osmosis Facilities. In Intakes and Outfalls for Seawater Reverse-Osmosis Desalination Facilities: Innovations and Environmental Impacts; Missimer, T.M., Jones, B., Maliva, R.G., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 3–17. [Google Scholar] [CrossRef]
- Musfique, A.; Rifat, A. An Assessment of the environmental impact of brine disposal in the marine environment. Int. J. Mod. Eng. Res. 2012, 2, 2756–2761. [Google Scholar]
- Panagopoulos, A.; Haralambous, K.-J.; Loizidou, M. Desalination brine disposal methods and treatment technologies—A review. Sci. Total Environ. 2019, 693, 133545. [Google Scholar] [CrossRef]
- Einav, R.; Harussi, K.; Perry, D. The footprint of the desalination processes on the environment. Desalination 2003, 152, 141–154. [Google Scholar] [CrossRef]
- Ketsetzi, A.; Stathoulopoulou, A.; Demadis, K.D. Being “green” in chemical water treatment technologies: Issues, challenges and developments. Desalination 2008, 223, 487–493. [Google Scholar] [CrossRef]
- Roberts, D.A.; Johnston, E.L.; Knott, N.A. Impacts of desalination plant discharges on the marine environment: A critical review of published studies. Water Res. 2010, 44, 5117–5128. [Google Scholar] [CrossRef]
- Palomar, P.; Losada, I.J. Impacts of Brine Discharge on the Marine Environment. Modeling as a Predictive Tool. Chapter 13. In Desalination Trends and Technologies; Schorr, M., Ed.; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Alameddine, I.; El-Fadel, M. Brine discharge from desalination plants: A modeling approach to an optimized outfall design. Desalination 2007, 214, 241–260. [Google Scholar] [CrossRef]
- Voutchkov, N. Overview of seawater concentrate disposal alternatives. Desalination 2011, 273, 205–219. [Google Scholar] [CrossRef]
- Abessi, O.; Roberts, P.J.W. Multiport diffusers for dense discharges. J. Hydraul. Eng. 2014, 140, 04014032. [Google Scholar] [CrossRef]
- Omerspahic, M.; Al-Jabri, H.; Siddiqui, S.A.; Saadaoui, I. Characteristics of Desalination Brine and Its Impacts on Marine Chemistry and Health, With Emphasis on the Persian/Arabian Gulf: A Review. Front. Mar. Sci. 2022, 9, 525. [Google Scholar] [CrossRef]
- Frank, H.; Fussmann, K.E.; Rahav, E.; Bar Zeev, E. Chronic effects of brine discharge from large-scale seawater reverse osmosis desalination facilities on benthic bacteria. Water Res. 2019, 151, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Monitoring of the Marine and Coastal Environment of the Rutenberg Power Station. The Ashkelon Desalination Plant, the Brackish Water Desalination Plants, and the Power Station of Dorad Energy, Intel Kiryat Gat; RELP-2-2021; Israel Electric Co.: Haifa, Israel, 2011. (In Hebrew) [Google Scholar]
- Kress, N.; Shoham-Frider, E.; Lubinevsky, H. Monitoring the Effect of Brine Discharge on the Marine Environment of the Palmachim and Sorek Desalination Plants; IOLR Report H16/2020; Israel Oceanographic and Limnological Research: Haifa, Israel, 2020. (In Hebrew) [Google Scholar]
- Sanchez, J.L.; Zarzo, D. Environmental challenges on design, construction, and operation of desalination plants worldwide. TexasDesal 2017. In Proceedings of the Developing a Drought-Proof Water Supply Conference, Austin, TX, USA, 21–22 September 2017. [Google Scholar]
- Arconada, B.; Delgado, P.; García, Á. Minimizing environmental risks on constructing marine pipelines: Aguilas desalination plant. Desalin. Water Treat. 2013, 51, 246–261. [Google Scholar] [CrossRef]
- Voutchkov, N.; Kaiser, G.; Stover, R.; Lienhart, J.; Awerbuch, L. Sustainable management of desalination plant concentrate-desalination industry position. In Proceedings of the International Desalination Association World Congress on Desalination and Water Reuse, IDAWC19, Dubai, United Arab Emirates, 20–24 October 2019. [Google Scholar]
- Morote, Á.-F.; Rico, A.-M.; Moltó, E. Critical review of desalination in Spain: A resource for the future? Geogr. Res. 2017, 55, 412–423. [Google Scholar] [CrossRef]
- Pulido-Bosch, A.; Vallejos, A.; Sola, F. Methods to supply seawater to desalination plants along the Spanish mediterranean coast and their associated issues. Environ. Earth Sci. 2019, 78, 322. [Google Scholar] [CrossRef]
- Ibrahim, H.D.; Eltahir, E.A.B. Impact of Brine Discharge from Seawater Desalination Plants on Persian/Arabian Gulf Salinity. J. Environ. Eng. 2019, 145, 04019084. [Google Scholar] [CrossRef]
- Petersen, K.L.; Heck, N.; Reguero, B.G.; Potts, D.; Hovagimian, A.; Paytan, A. Biological and Physical Effects of Brine Discharge from the Carlsbad Desalination Plant and Implications for Future Desalination Plant Constructions. Water 2019, 11, 208. [Google Scholar] [CrossRef]
- Clark, G.F.; Knott, N.A.; Miller, B.M.; Kelaher, B.P.; Coleman, M.A.; Ushiama, S.; Johnston, E.L. First large-scale ecological impact study of desalination outfall reveals trade-offs in effects of hypersalinity and hydrodynamics. Water Res. 2018, 145, 757–768. [Google Scholar] [CrossRef]
- Kelaher, B.P.; Clark, G.F.; Johnston, E.L.; Coleman, M.A. Effect of Desalination Discharge on the Abundance and Diversity of Reef Fishes. Environ. Sci. Technol. 2020, 54, 735–744. [Google Scholar] [CrossRef]
- Marine & Estuarine Monitoring Program-Detailed Design (Version 3) Preferred Project Report for Sydney’s Desalination Project (Sydney Water). Available online: https://www.sydneydesal.com.au/media/1092/marine-and-estuarine-monitoring-program.pdf (accessed on 17 May 2022).
- Gil-Meseguer, E.; Espin, J.M.G.; Bernabe-Crespo, M.B. Desalination and water security in Southeastern Spain. J. Politi-Ecol. 2019, 26, 486–499. [Google Scholar] [CrossRef]
- Iso, S.; Suizu, S.; Maejima, A. The lethal effect of hypertonic solutions and avoidance of marine organisms in relation to discharged brine from a destination plant. Desalination 1994, 97, 389–399. [Google Scholar] [CrossRef]
- Seibel, F.B.; Giubel, G.O.M.; Brião, V.B.; Shabani, M.; Pontié, M. End-of-life reverse osmosis membranes: Recycle procedure and its applications for the treatment of brackish and surface water. J. Appl. Res. Water Wastewater 2021, 8, 77–87. [Google Scholar]
- Pontié, M.; Awad, S.; Tazerout, M.; Chaouachi, O.; Chaouachi, B. Recycling and energy recovery solutions of end-of-life reverse osmosis (RO) membrane materials: A sustainable approach. Desalination 2017, 423, 30–40. [Google Scholar] [CrossRef]
- De Paula, E.C.; Gomes, J.C.L.; Amaral, M. Recycling of end-of-life reverse osmosis membranes by oxidative treatment: A technical evaluation. Water Sci. Technol. 2017, 76, 605–622. [Google Scholar] [CrossRef]
- Contreras-Martínez, J.; Sanmartino, J.; Khayet, M.; García-Payo, M. Chapter 11-Reuse and recycling of end-of-life reverse osmosis membranes. In Advancement in Polymer-Based Membranes for Water Remediation; Nayak, S.K., Dutta, K., Gohil, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 381–417. [Google Scholar] [CrossRef]
- Veza, J.M.; Rodriguez-Gonzalez, J.J. Second use for old reverse osmosis membranes: Wastewater treatment. Desalination 2003, 157, 65–72. [Google Scholar] [CrossRef]
- Lahlou, F.-Z.; Mackey, H.R.; Al-Ansari, T. Role of wastewater in achieving carbon and water neutral agricultural production. J. Clean. Prod. 2022, 339, 130706. [Google Scholar] [CrossRef]
- Jones, E.R.; van Vliet, M.T.H.; Qadir, M.; Bierkens, M.F.P. Country-level and gridded estimates of wastewater production, collection, treatment and reuse. Earth Syst. Sci. Data 2021, 13, 237–254. [Google Scholar] [CrossRef]
- EU Policy on the Environment, Water Reuse. 2018. Available online: http://ec.europa.eu/environment/water/reuse.htm (accessed on 9 March 2020).
- Zaidi, M.K. (Ed.) Wastewater Reuse–Risk Assessment, Decision-Making and Environmental Security; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar] [CrossRef]
- Yang, J.; Monnot, M.; Ercolei, L.; Moulin, P. Membrane-Based Processes Used in Municipal Wastewater Treatment for Water Reuse: State-of-the-Art and Performance Analysis. Membranes 2020, 10, 131. [Google Scholar] [CrossRef]
- Shtull-Trauring, E.; Cohen, A.; Ben-Hur, M.; Tanny, J.; Bernstein, N. Reducing salinity of treated waste water with large scale desalination. Water Res. 2020, 186, 116322. [Google Scholar] [CrossRef]
- Portman, M.E.; Vdov, O.; Schuetze, M.; Gilboa, Y.; Friedler, E. Public perceptions and perspectives on alternative sources of water for reuse generated at the household level. J. Water Reuse Desalin. 2022, 12, 157–174. [Google Scholar] [CrossRef]
- Furlong, C.; Jegatheesan, J.; Currell, M.; Iyer-Raniga, U.; Khan, T.; Ball, A.S. Is the global public willing to drink recycled water? A review for researchers and practitioners. Util. Policy 2019, 56, 53–61. [Google Scholar] [CrossRef]
- Ormerod, K.J. Illuminating Elimination: Public Perception and the Production of Potable Water Reuse. Wiley Interdiscip. Rev. Water 2016, 3, 537–547. [Google Scholar] [CrossRef]
- Tortajada, C.; Nambiar, S. Communications on Technological Innovations: Potable Water Reuse. Water 2019, 11, 251. [Google Scholar] [CrossRef]
- Tang, C.Y.; Yang, Z.; Guo, H.; Wen, J.J.; Nghiem, L.D.; Cornelissen, E. Potable Water Reuse through Advanced Membrane Technology. Environ. Sci. Technol. 2018, 52, 10215–10223. [Google Scholar] [CrossRef] [PubMed]
- Partyka, M.L.; Bond, R.F. Wastewater reuse for irrigation of produce: A review of research, regulations, and risks. Sci. Total Environ. 2022, 828, 154385. [Google Scholar] [CrossRef]
- Helmecke, M.; Fries, E.; Schulte, C. Regulating water reuse for agricultural irrigation: Risks related to organic micro-contaminants. Environ. Sci. Eur. 2020, 32, 4. [Google Scholar] [CrossRef]
- Chojnacka, K.; Witek-Krowiak, A.; Moustakas, K.; Skrzypczak, D.; Mikula, K.; Loizidou, M. A transition from conventional irrigation to fertigation with reclaimed wastewater: Prospects and challenges. Renew. Sustain. Energy Rev. 2020, 130, 109959. [Google Scholar] [CrossRef]
- Owusu-Ansah, E.D.-G.J.; Sampson, A.; Amponsah, S.K.; Abaidoo, R.C.; Hald, T. Performance, Compliance and Reliability of Waste Stabilization Pond: Effluent Discharge Quality and Environmental Protection Agency Standards in Ghana. Res. J. Appl. Sci. Eng. Technol. 2015, 10, 1293–1302. [Google Scholar] [CrossRef]
- Lu, X.; Zhou, B.; Vogt, R.D.; Seip, H.M.; Xin, Z.; Ekengren, Ö. Rethinking China’s water policy: The worst water quality despite the most stringent standards. Water Int. 2016, 41, 1044–1048. [Google Scholar] [CrossRef]
- Regulation (EU) 2020/741 of the European Parliament and of the Council of 25 May 2020. Available online: https://climate-adapt.eea.europa.eu/en/metadata/guidances/regulation-on-minimum-requirements-for-water-reuse (accessed on 7 April 2023).
- Israeli Wastewater Quality Standards for Wastewater Treatment 2010. Available online: https://www.nevo.co.il/law_html/law01/500_306.htm (accessed on 17 May 2023). (In Hebrew).
- USEPA Basic Information about Water Reuse. Available online: https://www.epa.gov/waterreuse/basic-information-about-water-reuse (accessed on 25 March 2023).
- Capodaglio, A.G. Fit-for-purpose urban wastewater reuse: Analysis of issues and available technologies for sustainable multiple barrier approaches. Crit. Rev. Environ. Sci. Technol. 2020, 51, 1619–1666. [Google Scholar] [CrossRef]
- Bai, Y.; Shan, F.; Zhu, Y.-Y.; Xu, J.-Y.; Wu, Y.-S.; Luo, X.-G.; Wu, Y.-H.; Hu, H.-Y.; Zhang, B.-L. Long-term performance and economic evaluation of full-scale MF and RO process—A case study of the changi NEWater Project Phase 2 in Singapore. Water Cycle 2020, 1, 128–135. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, Y.; Wang, J.; Zheng, X.; Chen, Y. Municipal wastewater reclamation and reuse using membrane-based technologies: A review. Desalin. Water Treat. 2021, 224, 65–82. [Google Scholar] [CrossRef]
- Gurreri, L.; Tamburini, A.; Cipollina, A.; Micale, G. Electrodialysis Applications in Wastewater Treatment for Environmental Protection and Resources Recovery: A Systematic Review on Progress and Perspectives. Membranes 2020, 10, 146. [Google Scholar] [CrossRef]
- Tibi, F.; Charfi, A.; Cho, J.; Kim, J. Fabrication of polymeric membranes for membrane distillation process and application for wastewater treatment: Critical review. Process. Saf. Environ. Prot. 2020, 141, 190–201. [Google Scholar] [CrossRef]
- Wu, B.; Kim, J. Anaerobic Membrane Bioreactors for Nonpotable Water Reuse and Energy Recovery. J. Environ. Eng. 2020, 146, 03119002. [Google Scholar] [CrossRef]
- Kharraz, J.A.; Khanzada, N.K.; Farid, M.U.; Kim, J.; Jeong, S.; An, A.K. Membrane distillation bioreactor (MDBR) for wastewater treatment, water reuse, and resource recovery: A review. J. Water Process. Eng. 2022, 47, 102687. [Google Scholar] [CrossRef]
- Aslam, A.; Khan, S.J.; Shahzad, H.M.A. Anaerobic membrane bioreactors (AnMBRs) for municipal wastewater treatment- potential benefits, constraints, and future perspectives: An updated review. Sci. Total Environ. 2021, 802, 149612. [Google Scholar] [CrossRef]
- Ang, W.L.; Mohammad, A.W.; Hilal, N.; Leo, C.P. A review on the applicability of integrated/hybrid membrane processes in water treatment and desalination plants. Desalination 2015, 363, 2–18. [Google Scholar] [CrossRef]
- Kammakakam, I.; Lai, Z. Next-generation ultrafiltration membranes: A review of material design, properties, recent progress, and challenges. Chemosphere 2023, 316, 137669. [Google Scholar] [CrossRef] [PubMed]
- Anis, S.F.; Hashaikeh, R.; Hilal, N. Microfiltration membrane processes: A review of research trends over the past decade. J. Water Process. Eng. 2019, 32, 100941. [Google Scholar] [CrossRef]
- Qu, F.; Wang, H.; He, J.; Fan, G.; Pan, Z.; Tian, J.; Rong, H.; Li, G.; Yu, H. Tertiary treatment of secondary effluent using ultrafiltration for wastewater reuse: Correlating membrane fouling with rejection of effluent organic matter and hydrophobic pharmaceuticals. Environ. Sci. Water Res. Technol. 2019, 5, 672–683. [Google Scholar] [CrossRef]
- Bartels, C.R. Reverse osmosis membranes for wastewater reclamation. WaterWorld 2006, 21, 37–38. [Google Scholar]
- Snyder, S.; Adham, S.; Redding, A.M.; Cannon, F.S.; Decarolis, J.; Oppenheimer, J.; Wert, E.C.; Yoon, Y. Role of membranes and activated carbon in the removal of endocrine disruptors and pharmaceuticals. Desalination 2007, 202, 156–181. [Google Scholar] [CrossRef]
- Sreejon, D.; Nillohit, M.R.; Wan, J.; Khan, A.; Chakraborty, T.; Madhumita, B.R. Micropollutants in Wastewater: Fate and Removal Processes. In Physico-Chemical Wastewater Treatment and Resource Recovery; Farooq, R., Ahmad, Z., Eds.; InTech: Yokohama, Japan, 2017. [Google Scholar]
- Comerton, A.M.; Andrews, R.C.; Bagley, D.M.; Hao, C. The rejection of endocrine disrupting and pharmaceutically active compounds by NF and RO membranes as a function of compound and water matrix properties. J. Membr. Sci. 2008, 313, 323–335. [Google Scholar] [CrossRef]
- Judd, S. The status of membrane bioreactor technology. Trends Biotechnol. 2008, 26, 109–116. [Google Scholar] [CrossRef]
- Banti, D.C.; Tsangas, M.; Samaras, P.; Zorpas, A. LCA of a membrane bioreactor compared to activated sludge system for municipal wastewater treatment. Membranes 2020, 10, 421. [Google Scholar] [CrossRef]
- Al-Asheh, S.; Bagheri, M.; Aidan, A. Membrane bioreactor for wastewater treatment: A review. Case Stud. Chem. Environ. Eng. 2021, 4, 100109. [Google Scholar] [CrossRef]
- Stoffel, D.; Rigo, E.; Derlon, N.; Staaks, C.; Heijnen, M.; Morgenroth, E.; Jacquin, C. Low maintenance gravity-driven membrane filtration using hollow fibers: Effect of reducing space for biofilm growth and control strategies on permeate flux. Sci. Total Environ. 2022, 811, 152307. [Google Scholar] [CrossRef] [PubMed]
- Pronk, W.; Ding, A.; Morgenroth, E.; Derlon, N.; Desmond, P.; Burkhardt, M.; Wu, B.; Fane, A.G. Gravity-driven membrane filtration for water and wastewater treatment: A review. Water Res. 2018, 149, 553–565. [Google Scholar] [CrossRef] [PubMed]
| Economic | Societal | Environmental |
|---|---|---|
| Expand activity and productivity in agriculture, mining, energy, and nature | Reduced risk of water-related disasters | Preservation of resources |
| The reduced economic impact of water- related hazards such as floods and droughts | Employment and reduced poverty | Increase ecological integrity |
| Increased value of property | Improved access to services such as electricity and water supply | Reduced habitat degradation and biodiversity loss |
| Development of regional markets for goods, services, and labor | Strengthened scientific knowledge of water status | |
| Increase in cross-border investments | Strengthening of international law | |
| Development of transnational infrastructure networks | Increased geopolitical stability and strengthened diplomatic relations | |
| Joint initiatives and investments | Creation of a shared basin identity | |
| Avoided cost of conflicts |
| Parameter | US EPA [148] | EU [149,150] | China [149] | Israel [151] | Japan [149] |
|---|---|---|---|---|---|
| pH | 6–9 | 6–9 | 6.5–8.5 | 5–9 | |
| COD (mg/L) | 250 | 125 | 50 | 100 | 120 |
| BOD5 (mg/L) | 50 | ≤10 | 10 | 10 | 120 |
| TSS (mg/L) | 50 | ≤10 | 10 | 10 | 150 |
| TN (mg/L) | 50 | 10 | 15 | 25 | 60 |
| NH4-N (mg/L) | 1.0 | 5 | 10 | ||
| TP (mg/L) | 2.0 | 1.0 | 0.5 | 5.0 | 8.0 |
| E-coli (MPN/100 mL) | 10 | ≤10 | 10 |
| Advantages | Disadvantages |
|---|---|
| Smaller footprint | Membrane fouling |
| There are no limitations on the concentration of the mixed liquor suspended solids (MLSS) in the MBR, so the generation of waste AS is reduced. The maximum concentration of MLSS in CAS is around 5000 mg/L while the optimum level in MBR is around 8000–12,000 mg/L. | Higher capital and operational costs due to the cost of the membranes and antifouling strategies. |
| Fine control of the solid retention time (SRT) can be achieved in MBR due to the elimination of secondary sedimentation tanks. | Higher aeration requirement contributes to the increased foaming propensity. |
| Applicability of longer SRT in MBR (more than 20 days) in comparison to CAS (generally 5–15 days) provides higher effluent quality. | Higher power consumption during operation. In some cases, it is double the electricity consumption of CAS. |
| The generation of high-quality effluent due to a membrane separation eliminates the need for tertiary treatment. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shemer, H.; Wald, S.; Semiat, R. Challenges and Solutions for Global Water Scarcity. Membranes 2023, 13, 612. https://doi.org/10.3390/membranes13060612
Shemer H, Wald S, Semiat R. Challenges and Solutions for Global Water Scarcity. Membranes. 2023; 13(6):612. https://doi.org/10.3390/membranes13060612
Chicago/Turabian StyleShemer, Hilla, Shlomo Wald, and Raphael Semiat. 2023. "Challenges and Solutions for Global Water Scarcity" Membranes 13, no. 6: 612. https://doi.org/10.3390/membranes13060612
APA StyleShemer, H., Wald, S., & Semiat, R. (2023). Challenges and Solutions for Global Water Scarcity. Membranes, 13(6), 612. https://doi.org/10.3390/membranes13060612






