Electrospun Hydrophobic Interaction Chromatography (HIC) Membranes for Protein Purification
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of Electrospun Membrane Substrate
2.3. Ligand Grafting
2.4. Characterization of the Functionalized HIC Membranes
2.5. The Static and Dynamic Binding Capacities of HIC Membranes
3. Results and Discussion
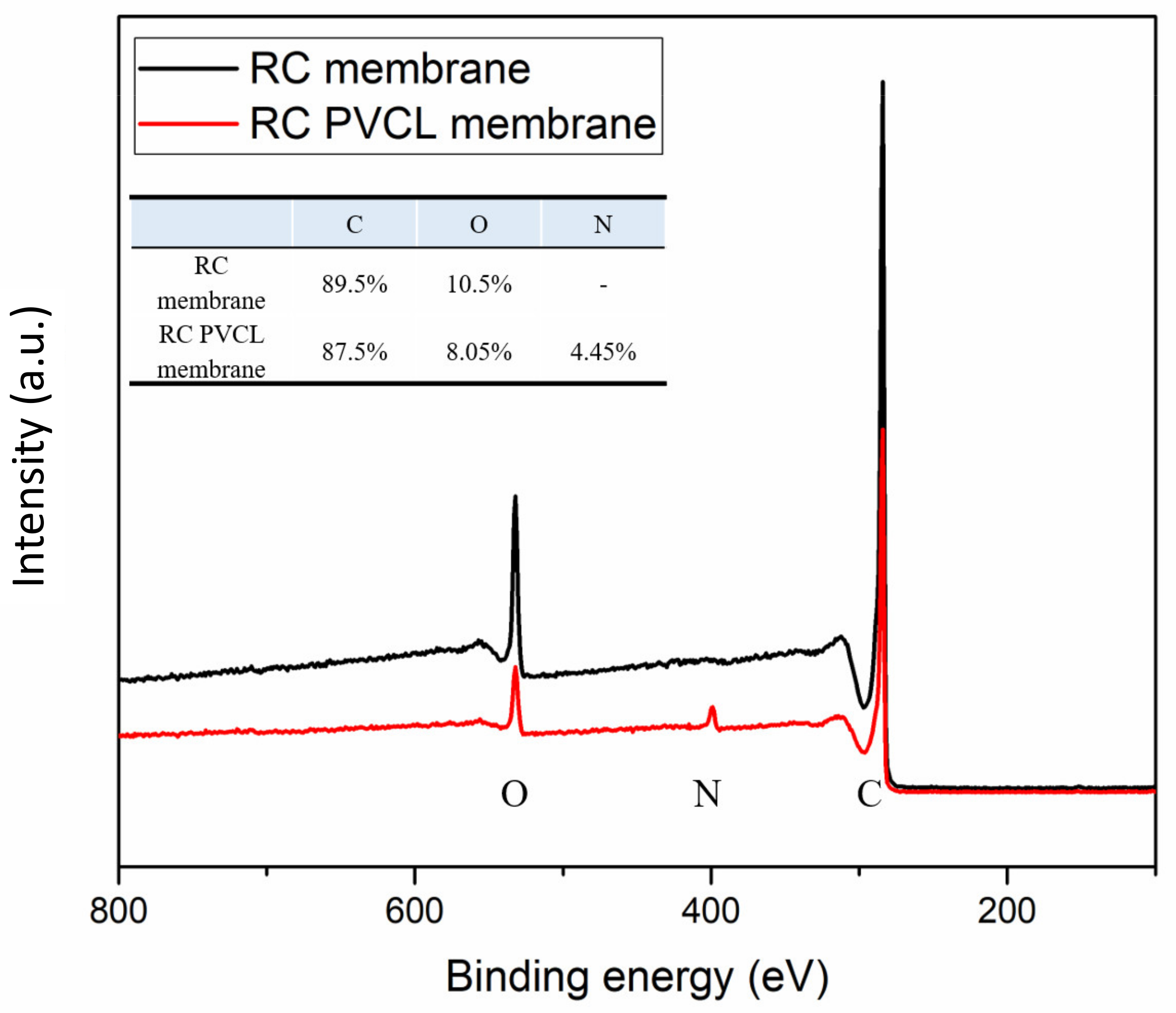
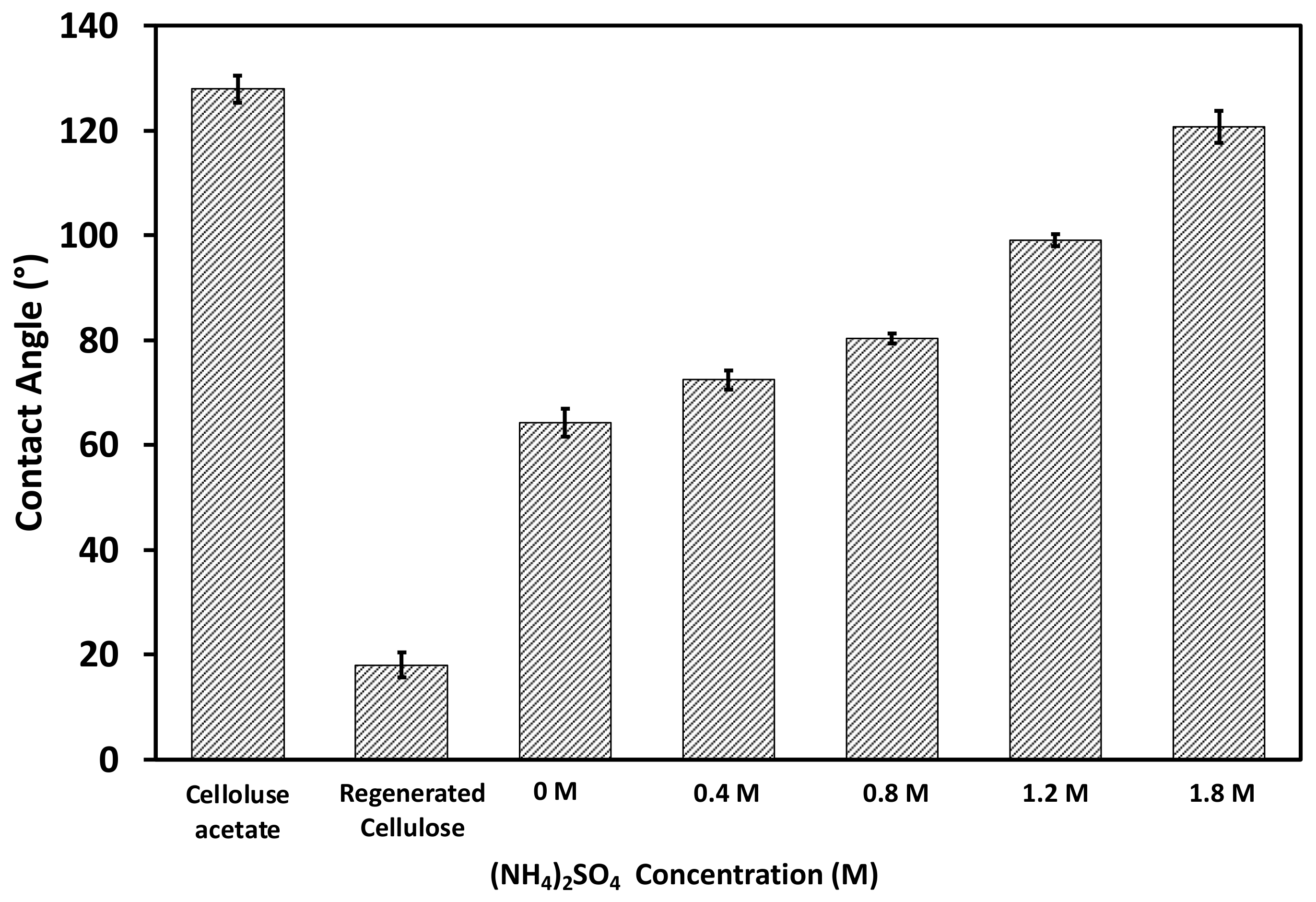
3.1. Physicochemical Properties of Fabricated HIC Membranes
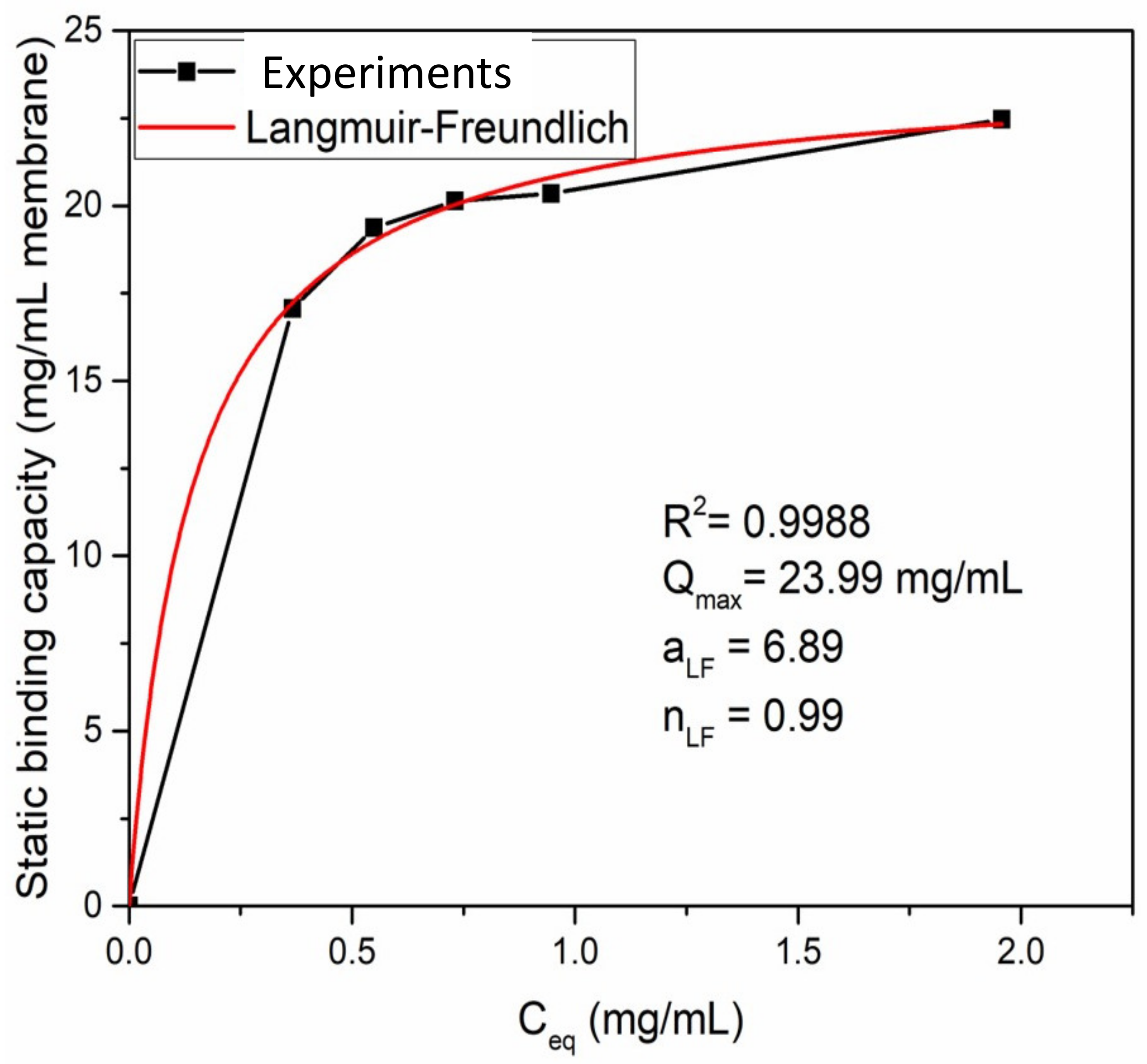
3.2. Static Binding Capacity
3.3. Protein Dynamic Binding Capacity and Recovery
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cramer, S.M.; Holstein, M.A. Downstream bioprocessing: Recent advances and future promise. Curr. Opin. Chem. Eng. 2011, 1, 27–37. [Google Scholar] [CrossRef]
- Liu, Z.; Wickramasinghe, S.R.; Qian, X. Membrane chromatography for protein purifications from ligand design to functionalization. Sep. Sci. Technol. 2017, 52, 299–319. [Google Scholar] [CrossRef]
- Devi, N.; Patel, S.K.S.; Kumar, P.; Singh, A.; Thakur, N.; Lata, J.; Pandey, D.; Thakur, V.; Chand, D. Bioprocess Scale-up for Acetohydroxamic Acid Production by Hyperactive Acyltransferase of Immobilized Rhodococcus Pyridinivorans. Catal. Lett. 2022, 152, 944–953. [Google Scholar] [CrossRef]
- Eriksson, K.O. Hydrophobic Interaction Chromatography, Biopharmaceutical Processing; Elsevier: Amsterdam, The Netherlands, 2018; pp. 401–408. [Google Scholar]
- Ghosh, R. Separation of proteins using hydrophobic interaction membrane chromatography. J. Chromatogr. A 2001, 923, 59–64. [Google Scholar] [CrossRef]
- Liu, Z.; Wickramasinghe, S.R.; Qian, X. Ion-specificity in protein binding and recovery for the responsive hydrophobic poly (vinylcaprolactam) ligand. RSC Adv. 2017, 7, 36351–36360. [Google Scholar] [CrossRef] [Green Version]
- Lienqueo, M.E.; Mahn, A.; Salgado, J.C.; Asenjo, J.A. Current insights on protein behaviour in hydrophobic interaction chromatography. J. Chromatogr. B 2007, 849, 53–68. [Google Scholar] [CrossRef]
- Liu, Z.; Wickramasinghe, S.R.; Qian, X. The architecture of responsive polymeric ligands on protein binding and recovery. RSC Adv. 2017, 7, 27823–27832. [Google Scholar] [CrossRef] [Green Version]
- Baumann, P.; Baumgartner, K.; Hubbuch, J. Influence of binding pH and protein solubility on the dynamic binding capacity in hydrophobic interaction chromatography. J. Chromatogr. A 2015, 1396, 77–85. [Google Scholar] [CrossRef]
- Vu, A.; Qian, X.; Wickramasinghe, S.R. Membrane-based hydrophobic interaction chromatography. Sep. Sci. Technol. 2017, 52, 287–298. [Google Scholar] [CrossRef]
- Liu, Z.; Du, H.; Wickramasinghe, S.R.; Qian, X. Membrane surface engineering for protein separations: Experiments and simulations. Langmuir 2014, 30, 10651–10660. [Google Scholar] [CrossRef]
- Boi, C.; Malavasi, A.; Carbonell, R.G.; Gilleskie, G. A direct comparison between membrane adsorber and packed column chromatography performance. J. Chromatogr. A 2020, 1612, 460629. [Google Scholar] [CrossRef] [PubMed]
- Chenette, H.C.; Robinson, J.R.; Hobley, E.; Husson, S.M. Development of high-productivity, strong cation-exchange adsorbers for protein capture by graft polymerization from membranes with different pore sizes. J. Membr. Sci. 2012, 423, 43–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.-T.; Wickramasinghe, S.R.; Qian, X. Electrospun weak anion-exchange fibrous membranes for protein purification. Membranes 2020, 10, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, T.; Kelly, G.M.; Emery, W.R. Use of mobile phase additives for the elution of bispecific and monoclonal antibodies from phenyl based hydrophobic interaction chromatography resins. J. Chromatogr. B 2018, 1096, 20–30. [Google Scholar] [CrossRef]
- Sun, X.; Qian, X. Atomistic Molecular Dynamics Simulations of the Lower Critical Solution Temperature Transition of Poly (N-vinylcaprolactam) in Aqueous Solutions. J. Phys. Chem. B 2019, 123, 4986–4995. [Google Scholar] [CrossRef]
- Zhang, Y.; Furyk, S.; Bergbreiter, D.E.; Cremer, P.S. Specific ion effects on the water solubility of macromolecules: PNIPAM and the Hofmeister series. J. Am. Chem. Soc. 2005, 127, 14505–14510. [Google Scholar] [CrossRef]
- Du, H.; Wickramasinghe, R.; Qian, X. Effects of salt on the lower critical solution temperature of poly (N-isopropylacrylamide). J. Phys. Chem. B 2010, 114, 16594–16604. [Google Scholar] [CrossRef]
- Du, H.; Qian, X. The Interactions between Salt Ions and Thermo-Responsive Poly (N-Isopropylacrylamide) from Molecular Dynamics Simulations. Responsive Membr. Mater. 2012, 10, 229–242. [Google Scholar] [CrossRef]
- Du, H.; Wickramasinghe, S.R.; Qian, X. Specificity in cationic interaction with poly (N-isopropylacrylamide). J. Phys. Chem. B 2013, 117, 5090–5101. [Google Scholar] [CrossRef]
- Zhang, Y.; Furyk, S.; Sagle, L.B.; Cho, Y.; Bergbreiter, D.E.; Cremer, P.S. Effects of Hofmeister anions on the LCST of PNIPAM as a function of molecular weight. J. Phys. Chem. C 2007, 111, 8916–8924. [Google Scholar] [CrossRef] [Green Version]
- Hiruta, Y.; Nagumo, Y.; Suzuki, Y.; Funatsu, T.; Ishikawa, Y.; Kanazawa, H. The effects of anionic electrolytes and human serum albumin on the LCST of poly (N-isopropylacrylamide)-based temperature-responsive copolymers. Colloids Surf. B Biointerfaces 2015, 132, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Himstedt, H.H.; Qian, X.; Weaver, J.R.; Wickramasinghe, S.R. Responsive membranes for hydrophobic interaction chromatography. J. Membr. Sci. 2013, 447, 335–344. [Google Scholar] [CrossRef]
- Darvishmanesh, S.; Qian, X.; Wickramasinghe, S.R. Responsive membranes for advanced separations. Curr. Opin. Chem. Eng. 2015, 8, 98–104. [Google Scholar] [CrossRef]
- Okubo, K.; Ikeda, K.; Oaku, A.; Hiruta, Y.; Nagase, K.; Kanazawa, H. Protein purification using solid-phase extraction on temperature-responsive hydrogel-modified silica beads. J. Chromatogr. A 2018, 1568, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Nagase, K.; Ishii, S.; Ikeda, K.; Yamada, S.; Ichikawa, D.; Akimoto, A.M.; Hattori, Y.; Kanazawa, H. Antibody drug separation using thermoresponsive anionic polymer brush modified beads with optimised electrostatic and hydrophobic interactions. Sci. Rep. 2020, 10, 11896. [Google Scholar] [CrossRef]
- Nomoto, D.; Nagase, K.; Nakamura, Y.; Kanazawa, H.; Citterio, D.; Hiruta, Y. Anion species-triggered antibody separation system utilizing a thermo-responsive polymer column under optimized constant temperature. Colloids Surf. B Biointerfaces 2021, 205, 111890. [Google Scholar] [CrossRef]
- Huang, L.; Arena, J.T.; McCutcheon, J.R. Surface modified PVDF nanofiber supported thin film composite membranes for forward osmosis. J. Membr. Sci. 2016, 499, 352–360. [Google Scholar] [CrossRef] [Green Version]
- Chitpong, N.; Husson, S.M. Polyacid functionalized cellulose nanofiber membranes for removal of heavy metals from impaired waters. J. Membr. Sci. 2017, 523, 418–429. [Google Scholar] [CrossRef]
- Fu, Q.; Wang, X.; Si, Y.; Liu, L.; Yu, J.; Ding, B. Scalable fabrication of electrospun nanofibrous membranes functionalized with citric acid for high-performance protein adsorption. ACS Appl. Mater. Interfaces 2016, 8, 11819–11829. [Google Scholar] [CrossRef]
- Dods, S.R.; Hardick, O.; Stevens, B.; Bracewell, D.G. Fabricating electrospun cellulose nanofibre adsorbents for ion-exchange chromatography. J. Chromatogr. A 2015, 1376, 74–83. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, C.; Zhang, Y.; White, C.J.B.; Xue, Y.; Nie, H.; Zhu, L. Elaboration, characterization and study of a novel affinity membrane made from electrospun hybrid chitosan/nylon-6 nanofibers for papain purification. J. Mater. Sci. 2010, 45, 2296–2304. [Google Scholar] [CrossRef]
- Ma, H.; Hsiao, B.S.; Chu, B. Electrospun nanofibrous membrane for heavy metal ion adsorption. Curr. Org. Chem. 2013, 17, 1361–1370. [Google Scholar] [CrossRef]
- Ma, Z.; Lan, Z.; Matsuura, T.; Ramakrishna, S. Electrospun polyethersulfone affinity membrane: Membrane preparation and performance evaluation. J. Chromatogr. B 2009, 877, 3686–3694. [Google Scholar] [CrossRef] [PubMed]
- Schneiderman, S.; Zhang, L.; Fong, H.; Menkhaus, T.J. Surface-functionalized electrospun carbon nanofiber mats as an innovative type of protein adsorption/purification medium with high capacity and high throughput. J. Chromatogr. A 2011, 1218, 8989–8995. [Google Scholar] [CrossRef]
- Chen, S.-T.; Wickramasinghe, S.R.; Qian, X. High Performance Mixed-Matrix Electrospun Membranes for Ammonium Removal from Wastewaters. Membranes 2021, 11, 440. [Google Scholar] [CrossRef]
- Li, G.; Xiao, J.; Zhang, W. Knoevenagel condensation catalyzed by a tertiary-amine functionalized polyacrylonitrile fiber. Green Chem. 2011, 13, 1828–1836. [Google Scholar] [CrossRef]
- Chiao, Y.-H.; Sengupta, A.; Chen, S.-T.; Huang, S.-H.; Hu, C.-C.; Hung, W.-S.; Chang, Y.; Qian, X.; Wickramasinghe, S.R.; Lee, K.-R. Zwitterion augmented polyamide membrane for improved forward osmosis performance with significant antifouling characteristics. Sep. Purif. Technol. 2019, 212, 316–325. [Google Scholar] [CrossRef]
- Du, Y.; Li, Y.; Wu, T. A superhydrophilic and underwater superoleophobic chitosan–TiO2 composite membrane for fast oil-in-water emulsion separation. RSC Adv. 2017, 7, 41838–41846. [Google Scholar] [CrossRef] [Green Version]
- Umpleby, R.J.; Baxter, S.C.; Chen, Y.; Shah, R.N.; Shimizu, K.D. Characterization of molecularly imprinted polymers with the Langmuir–Freundlich isotherm. Anal. Chem. 2001, 73, 4584–4591. [Google Scholar] [CrossRef]
- Jeppu, G.P.; Clement, T.P. A modified Langmuir-Freundlich isotherm model for simulating pH-dependent adsorption effects. J. Contam. Hydrol. 2012, 129, 46–53. [Google Scholar] [CrossRef]





| Grafting Degree (DG) (%) | BSA Loading Concentration (g/L) | Protein Binding Capacity (mg/mL) | Recovery (%) |
|---|---|---|---|
| 6 | 0.10 | 7.95 ± 0.75 | 98.17 ± 0.02 |
| 6 | 1.0 | 30.69 ± 0.34 | 51.54 ± 1.96 |
| 9 | 0.10 | 14.29 | 80.64 |
| 9 | 1.0 | 43.43 | 21.09 |
| Initiator (mM) | BSA Loading Concentration (g/L) | Protein Binding Capacity (mg/mL) | Recovery (%) |
|---|---|---|---|
| 40 | 0.1 | 4.79 ± 0.11 | 97.54 ± 2.27 |
| 40 | 1.0 | 24.95 ± 0.56 | 57.24 ± 2.33 |
| 80 | 0.1 | 7.95 ± 0.75 | 98.17 ± 0.02 |
| 80 | 1.0 | 30.69 ± 0.34 | 51.54 ± 1.96 |
| 200 | 0.1 | 6.22 ± 0.11 | 83.39 ± 0.22 |
| 200 | 1.0 | 28.38 ± 1.75 | 41.44 ± 0.01 |
| BSA Loading Concentration (g/L) | Protein Binding Capacity (mg/mL) | Recovery (%) |
|---|---|---|
| 0.1 | 7.95 ± 0.75 | 98.17 ± 0.02 |
| 0.3 | 9.93 | 91.56 |
| 0.5 | 16.55 ± 2.09 | 75.67 ± 3.32 |
| 1.0 | 30.69 ± 0.34 | 51.54 ± 1.96 |
| BSA Loading Concentration (g/L) | Flowrate (mL/min) | Protein Binding Capacity (mg/mL) | Recovery (%) |
|---|---|---|---|
| 1 | 0.5 | 28.87 ± 0.79 | 57.51 ± 1.23 |
| 1 | 1 | 30.69 ± 0.34 | 51.54 ± 1.96 |
| 1 | 2 | 27.36 ± 0.93 | 41.72 ± 1.80 |
| Voltage (kV) | Fiber Diameter (nm) | Pore Size (μm) | BSA (g/L) | DBC (mg/mL) | Recovery (%) |
|---|---|---|---|---|---|
| 12.5 | 330–440 | 3.24 ± 0.27 | 0.1 | 7.95 ± 0.75 | 98.17 ± 0.02 |
| 1.0 | 30.69 ± 0.34 | 51.54 ± 1.96 | |||
| 20 | 220–350 | 2.88 ± 0.39 | 0.1 | 9.84 | 79.08 |
| 1.0 | 33.58 | 37.86 |
| IgG4 Loading Concentration (g/L) | Protein Binding Capacity (mg/mL) | Recovery (%) |
|---|---|---|
| 0.1 | 16.31 ± 0.23 | 85.32 ± 1.38 |
| 0.5 | 28.78 ± 0.18 | 23.95 ± 1.17 |
| 1 | 47.68 ± 1.29 | 14.22 ± 1.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.-T.; Wickramasinghe, S.R.; Qian, X. Electrospun Hydrophobic Interaction Chromatography (HIC) Membranes for Protein Purification. Membranes 2022, 12, 714. https://doi.org/10.3390/membranes12070714
Chen S-T, Wickramasinghe SR, Qian X. Electrospun Hydrophobic Interaction Chromatography (HIC) Membranes for Protein Purification. Membranes. 2022; 12(7):714. https://doi.org/10.3390/membranes12070714
Chicago/Turabian StyleChen, Shu-Ting, Sumith Ranil Wickramasinghe, and Xianghong Qian. 2022. "Electrospun Hydrophobic Interaction Chromatography (HIC) Membranes for Protein Purification" Membranes 12, no. 7: 714. https://doi.org/10.3390/membranes12070714
APA StyleChen, S.-T., Wickramasinghe, S. R., & Qian, X. (2022). Electrospun Hydrophobic Interaction Chromatography (HIC) Membranes for Protein Purification. Membranes, 12(7), 714. https://doi.org/10.3390/membranes12070714






