Wicking in Porous Polymeric Membranes: Determination of an Effective Capillary Radius to Predict the Flow Behavior in Lateral Flow Assays
Abstract
:1. Introduction
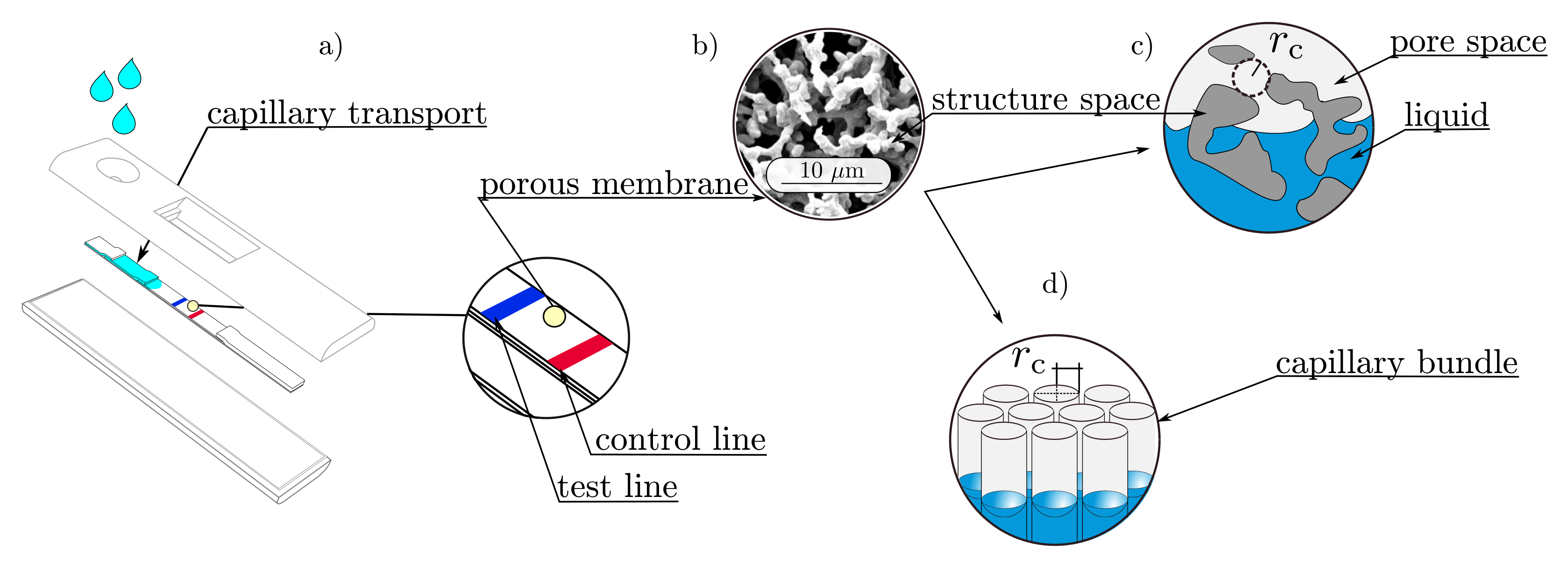
2. Materials and Methods
2.1. Mathematical Modeling
2.1.1. Two-Phase Phase-Field Approach
2.1.2. Macroscopic Flow Model for Wicking Processes
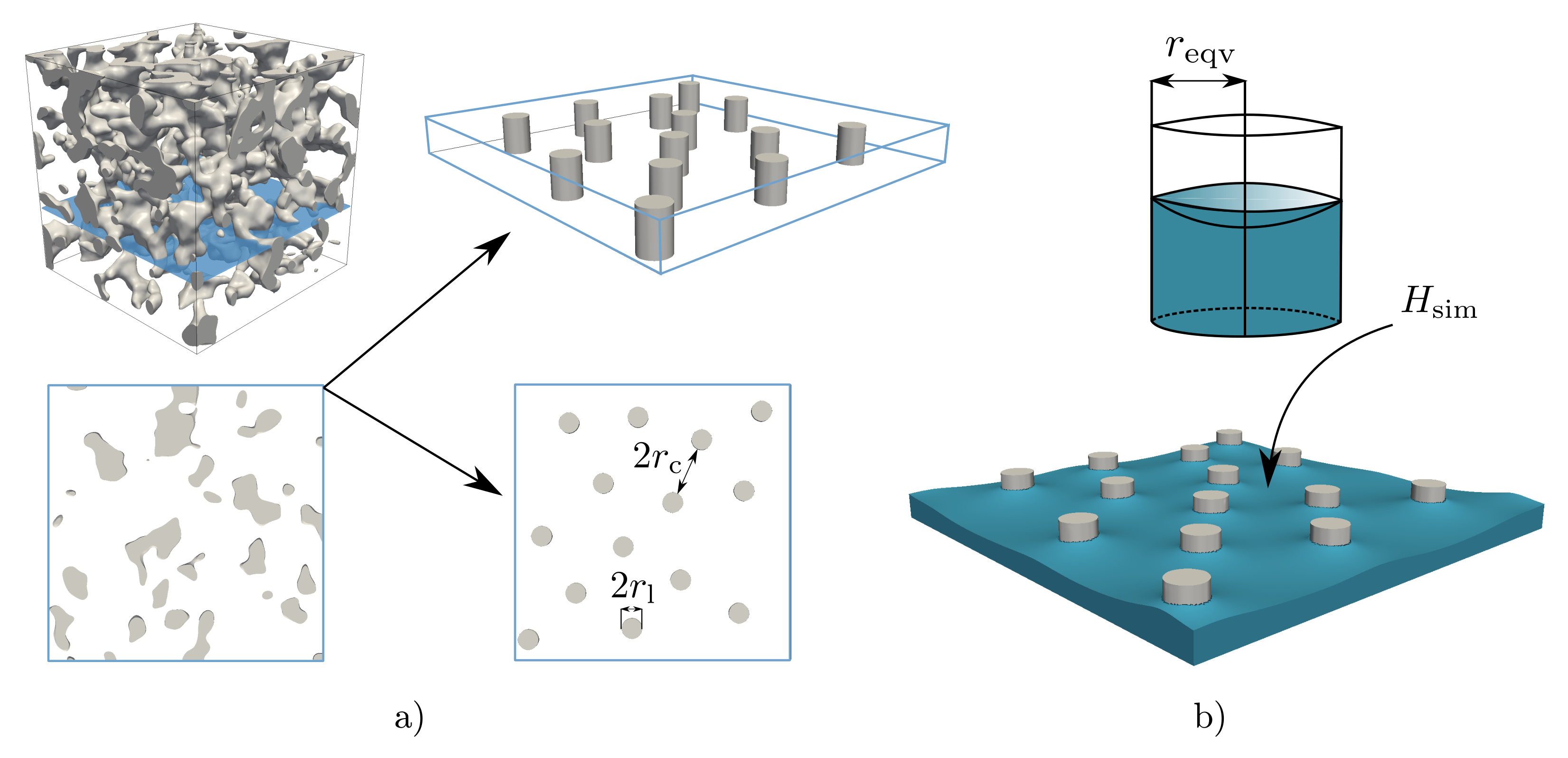
2.2. Digital Twins of Porous Polymeric Membranes
2.2.1. High-Resolution Computer Tomography (Nano CT)
2.2.2. Effective Properties
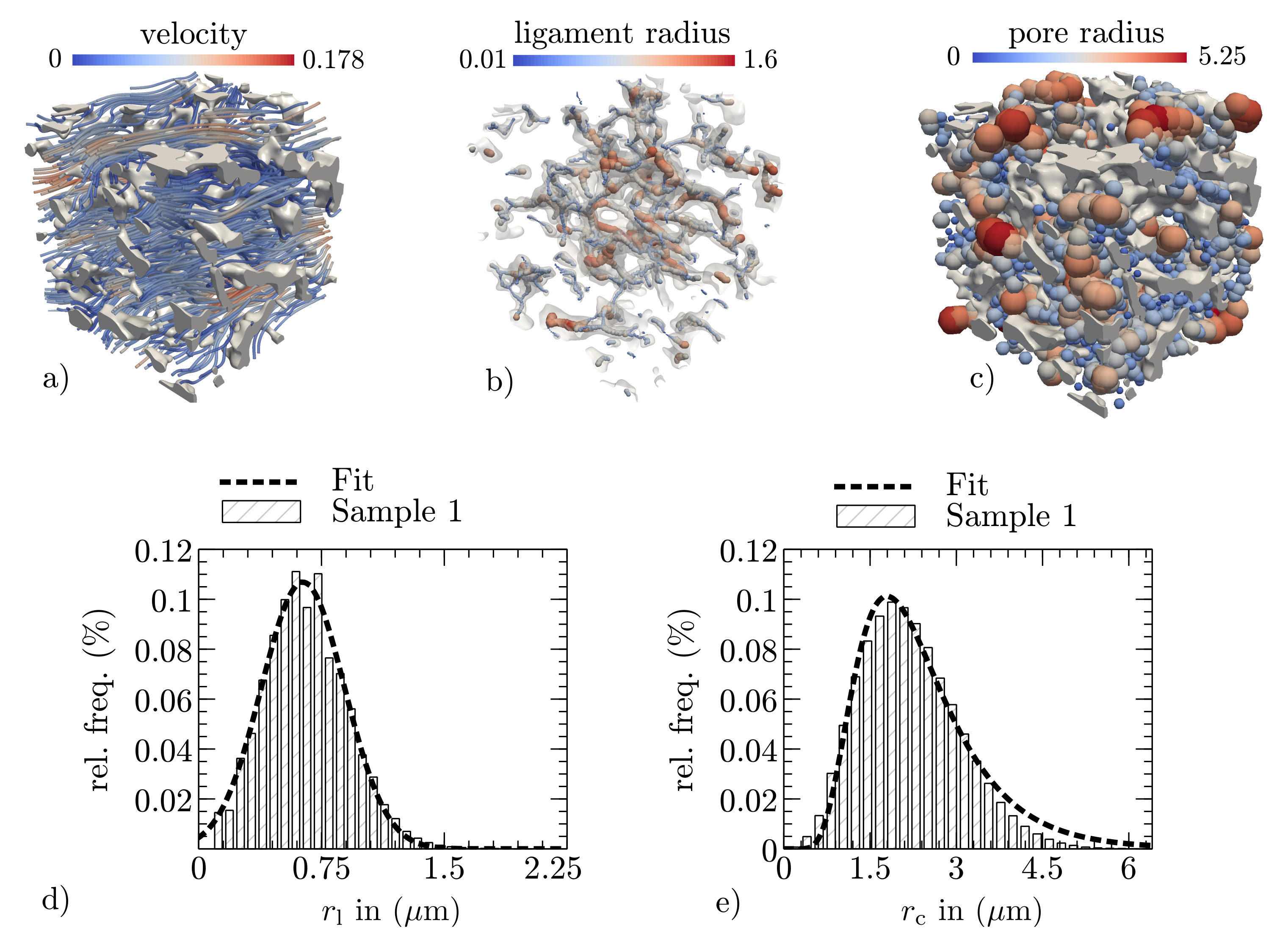
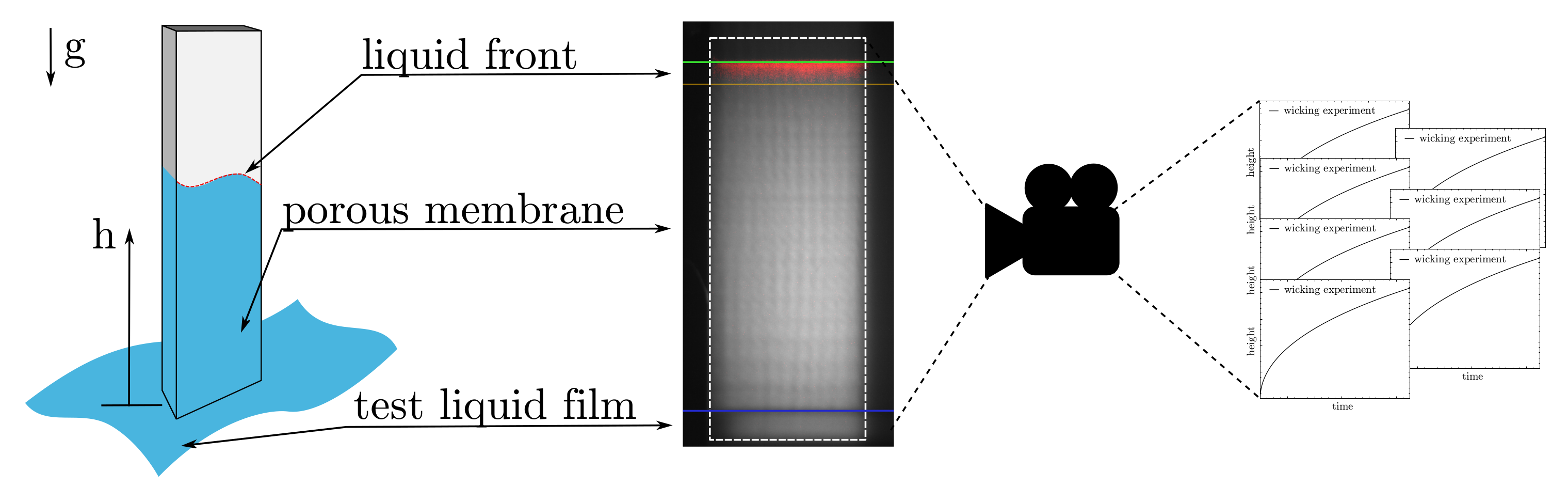
2.3. Wicking Experiment
3. Results
3.1. Simulation of Surface Curvature Formation in Two-Phase Equilibrium Conditions
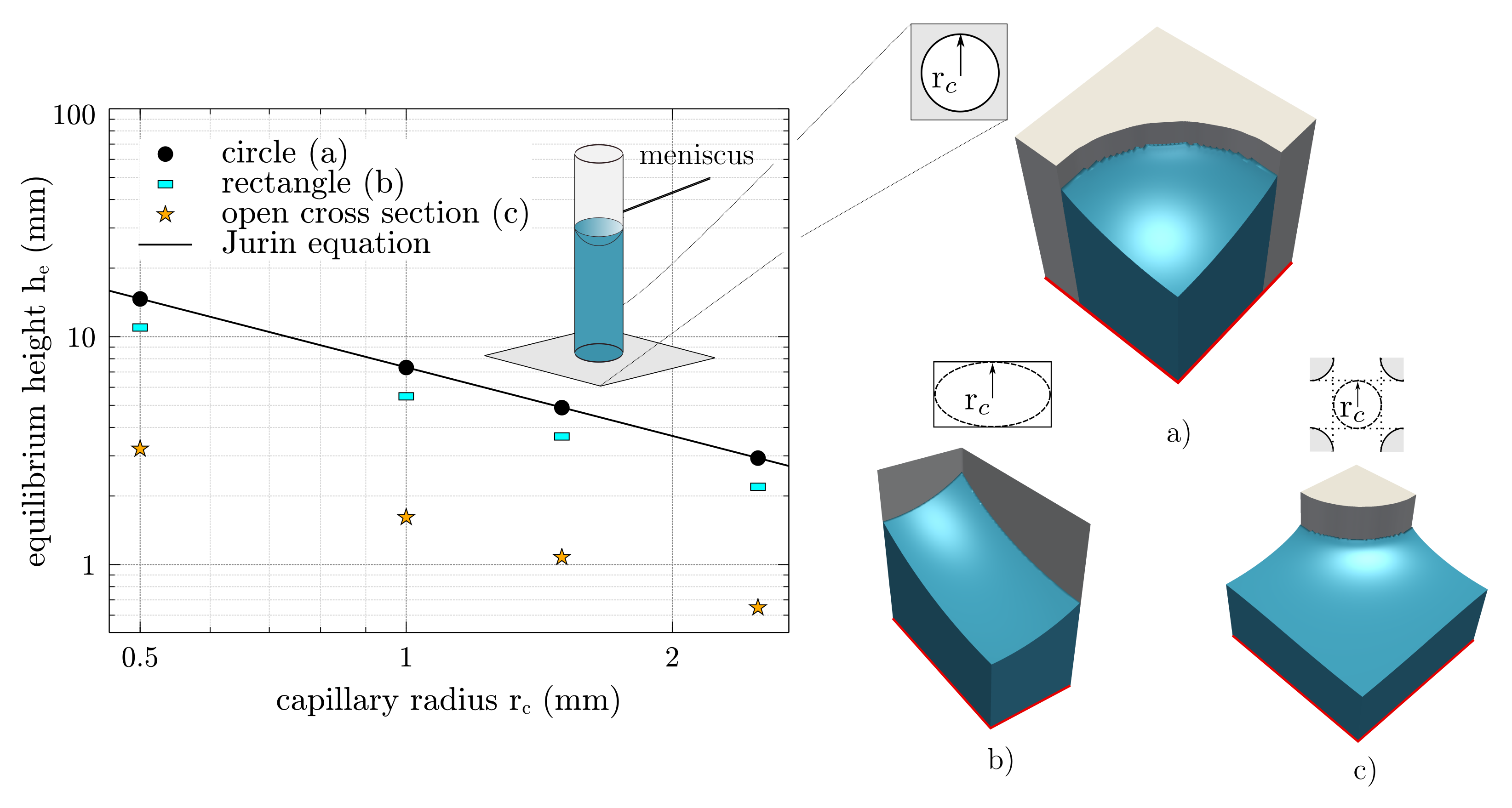
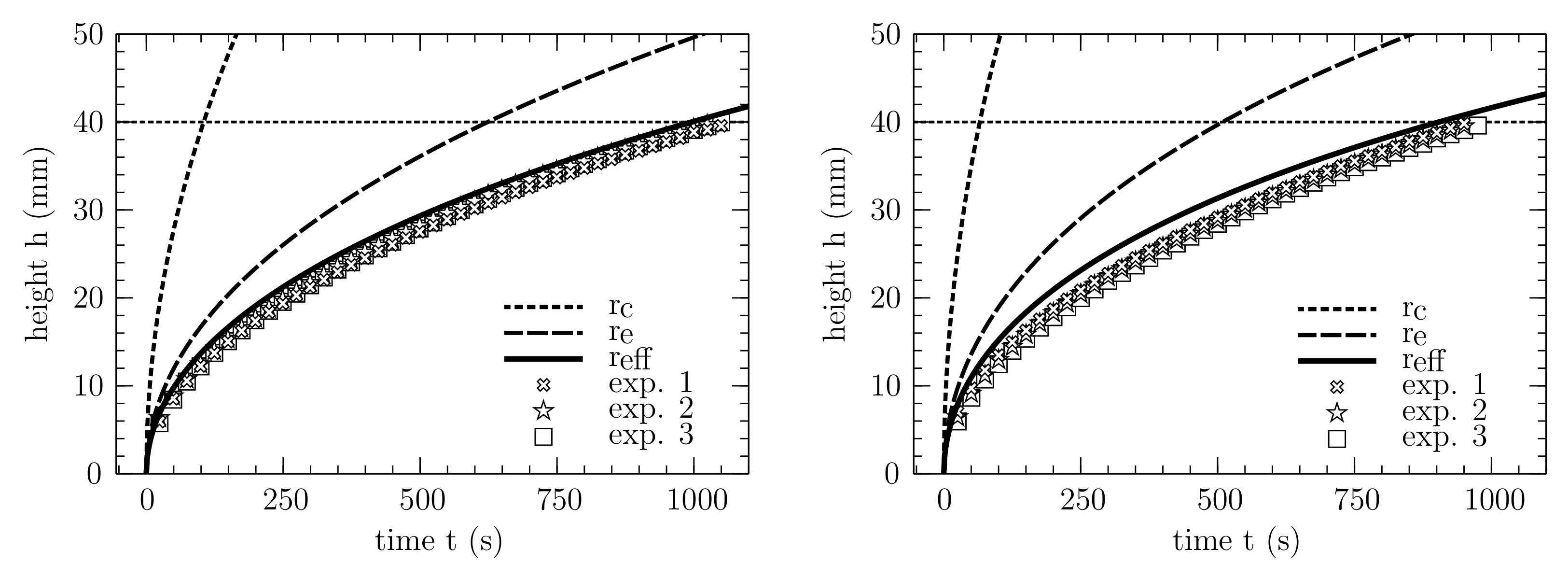
3.2. Methodological Determination of the Effective Pore Radius
3.3. Validation in an Ordered and Open Cross Section
3.4. Prediction of Wicking in the PPMs
| ( m) | ( m) | (-) | F (-) | ( m) | |
|---|---|---|---|---|---|
| Sample 1 | 2.16 | 13.85 | 0.296 | 9.6 | 20.7 |
| Sample 2 | 1.73 | 9.26 | 0.416 | 7.75 | 13.4 |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yetisen, A.K.; Akram, M.S.; Lowe, R.C. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 2013, 13, 2210–2251. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.M.; Sinton, D. Turning the page: Advancing paper-based microfluidics for broad diagnostic application. Chem. Rev. 2017, 117, 8447–8480. [Google Scholar] [CrossRef] [PubMed]
- Gasperino, D.; Baughman, T.; Hsieh, H.V.; Bell, D.; Weigl, B.H. Improving lateral flow assay performance using computational modeling. Annu. Rev. Anal. Chem. 2018, 11, 219–244. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, M.A. Nitrocellulose membranes for lateral flow immunoassays: A technical treatise. In Lateral Flow Immunoassay; Humana Press: Totowa, NJ, USA, 2009; pp. 1–19. [Google Scholar] [CrossRef]
- Boţan, A.; Ulm, F.-J.; Pellenq, R.J.-M.; Coasne, B. Bottom-up model of adsorption and transport in multiscale porous media. Phys. Rev. E 2015, 91, 032133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altschuh, P. Skalenübergreifende Analyse Makroporöser Membranen im Kontext Digitaler Zwillinge. Ph.D. Thesis, Karlsruhe Institute of Technology, Karlsruhe, Germany, 2020. [Google Scholar] [CrossRef]
- Fries, N.; Dreyer, M. An analytic solution of capillary rise restrained by gravity. J. Colloid Interface Sci. 2008, 320, 259–263. [Google Scholar] [CrossRef]
- Fries, N.; Quéré, D. Capillary Transport Processes in Porous Materials-Experiment and Model; Cuvillier Verlag: Göttingen, Germany, 2010. [Google Scholar]
- Masoodi, R.; Pillai, K.M.; Varanasi, P.P. Darcy’s law-based models for liquid absorption in polymer wicks. AIChE J. 2007, 53, 2769–2782. [Google Scholar] [CrossRef]
- Masoodi, R.; Pillai, K.M. Darcy’s law-based model for wicking in paper-like swelling porous media. AIChE J. 2010, 56, 2257–2267. [Google Scholar] [CrossRef]
- Masoodi, R.; Tan, H.; Pillai, K.M. Numerical simulation of liquid absorption in paper-like swelling porous media. AIChE J. 2012, 58, 2536–2544. [Google Scholar] [CrossRef]
- Masoodi, R.; Pillai, K.M. Wicking in Porous Materials: Traditional and Modern Modeling Approaches; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Whitaker, S. Flow in porous media i: A theoretical derivation of darcy’s law. Transp. Porous Media 1986, 1, 3–25. [Google Scholar] [CrossRef]
- Alava, M.; Niskanen, K. The physics of paper. Rep. Prog. Phys. 2006, 69, 669. [Google Scholar] [CrossRef]
- Jiang, L.; Liu, Y.; Teng, Y.; Zhao, J.; Zhang, Y.; Yang, M.; Song, Y. Permeability estimation of porous media by using an improved capillary bundle model based on micro-ct derived pore geometries. Heat Mass Transf. 2017, 53, 49–58. [Google Scholar] [CrossRef]
- Yang, P.; Wen, Z.; Dou, R.; Liu, X. Permeability in multi-sized structures of random packed porous media using three-dimensional lattice boltzmann method. Int. J. Heat Mass Transf. 2017, 106, 1368–1375. [Google Scholar] [CrossRef]
- Zarandi, M.A.F.; Pillai, K.M.; Barari, B. Flow along and across glass-fiber wicks: Testing of permeability models through experiments and simulations. AIChE J. 2018, 64, 3491–3501. [Google Scholar] [CrossRef]
- Yabansu, Y.C.; Altschuh, P.; Hötzer, J.; Selzer, M.; Nestler, B.; Kalidindi, S.R. A digital workflow for learning the reduced-order structure-property linkages for permeability of porous membranes. Acta Mater. 2020, 195, 668–680. [Google Scholar] [CrossRef]
- Masoodi, R.; Pillai, K.M.; Varanasi, P.P. Role of hydraulic and capillary radii in improving the effectiveness of capillary model in wicking. In Proceedings of the Fluids Engineering Division Summer Meeting, Jacksonville, FL, USA, 10–14 August 2008; pp. 251–259. [Google Scholar] [CrossRef]
- Gennes, P.-G.D.; Brochard-Wyart, F.; Quéré, D. Capillarity and Wetting Phenomena: Drops, Bubbles, Pearls, Waves; Springer: Berlin, Germany, 2004; Volume 315. [Google Scholar]
- Berg, J.C. An Introduction to Interfaces & Colloids: The Bridge to Nanoscience; World Scientific: Singapore, 2010. [Google Scholar]
- Chwastiak, S. A wicking method for measuring wetting properties of carbon yarns. J. Colloid Interface Sci. 1973, 42, 298–309. [Google Scholar] [CrossRef]
- Cummins, B.M.; Chinthapatla, R.; Ligler, F.S.; Walker, G.M. Time-dependent model for fluid flow in porous materials with multiple pore sizes. Anal. Chem. 2017, 89, 4377–4381. [Google Scholar] [CrossRef]
- Ravi, S.; Dharmarajan, R.; Moghaddam, S. Measurement of capillary radius and contact angle within porous media. Langmuir 2015, 31, 12954–12959. [Google Scholar] [CrossRef]
- Sun, S.; Feng, S.; Ji, C.; Shi, M.; He, X.; Xu, F.; Lu, T.J. Microstructural effects on permeability of nitrocellulose membranes for biomedical applications. J. Membr. Sci. 2020, 595, 117502. [Google Scholar] [CrossRef]
- Islam, M.A.; Ulbricht, M. Microfiltration membrane characterization by gas-liquid displacement porometry: Matching experimental pore number distribution with liquid permeability and bulk porosity. J. Membr. Sci. 2019, 569, 104–116. [Google Scholar] [CrossRef]
- Ley, A.; Altschuh, P.; Thom, V.; Selzer, M.; Nestler, B.; Vana, P. Characterization of a macro porous polymer membrane at micron-scale by confocal-laser-scanning microscopy and 3d image analysis. J. Membr. Sci. 2018, 564, 543–551. [Google Scholar] [CrossRef]
- Altschuh, P.; Yabansu, Y.C.; Hötzer, J.; Selzer, M.; Nestler, B.; Kalidindi, S.R. Data science approaches for microstructure quantification and feature identification in porous membranes. J. Membr. Sci. 2017, 540, 88–97. [Google Scholar] [CrossRef]
- Tang, Y.; Min, J.; Zhang, X.; Liu, G. Meniscus behaviors and capillary pressures in capillary channels having various cross-sectional geometries. Chin. J. Chem. Eng. 2018, 26, 2014–2022. [Google Scholar] [CrossRef]
- Benltoufa, S.; Fayala, F.; BenNasrallah, S. Capillary rise in macro and micro pores of jersey knitting structure. J. Eng. Fabr. Fibers 2008, 3, 155892500800300305. [Google Scholar] [CrossRef] [Green Version]
- Walji, N.; MacDonald, B.D. Influence of geometry and surrounding conditions on fluid flow in paper-based devices. Micromachines 2016, 7, 73. [Google Scholar] [CrossRef] [Green Version]
- Masoodi, R.; Pillai, K. A general formula for capillary suction-pressure in porous media. J. Porous Media 2012, 15, 775–783. [Google Scholar] [CrossRef]
- Almoughni, H.; Gong, H. Capillary flow of liquid water through yarns: A theoretical model. Text. Res. J. 2015, 85, 722–732. [Google Scholar] [CrossRef]
- Fei, Y.R.; Batty, C.; Grinspun, E.; Zheng, C. A multi-scale model for simulating liquid-fabric interactions. ACM Trans. Graph. 2018, 37, 51. [Google Scholar] [CrossRef]
- Armstrong, R.T.; Porter, M.L.; Wildenschild, D. Linking pore-scale interfacial curvature to column-scale capillary pressure. Adv. Water Resour. 2012, 46, 55–62. [Google Scholar] [CrossRef]
- Nam, Y.; Sharratt, S.; Byon, C.; Kim, S.J.; Ju, Y.S. Fabrication and characterization of the capillary performance of superhydrophilic cu micropost arrays. J. Microelectromech. Syst. 2010, 19, 581–588. [Google Scholar] [CrossRef]
- Hale, R.; Ranjan, R.; Hidrovo, C. Capillary flow through rectangular micropillar arrays. Int. J. Heat Mass Transf. 2014, 75, 710–717. [Google Scholar] [CrossRef]
- Nestler, B.; Wendler, F.; Selzer, M.; Stinner, B.; Garcke, H. Phase-field model for multiphase systems with preserved volume fractions. Phys. Rev. E 2008, 78, 011604. [Google Scholar] [CrossRef] [Green Version]
- Said, M.B.; Selzer, M.; Nestler, B.; Braun, D.; Greiner, C.; Garcke, H. A phase-field approach for wetting phenomena of multiphase droplets on solid surfaces. Langmuir 2014, 30, 4033–4039. [Google Scholar] [CrossRef]
- Garcke, H.; Nestler, B.; Stinner, B.; Wendler, F. Allen-cahn systems with volume constraints. Math. Model. Methods Appl. Sci. 2008, 18, 1347–1381. [Google Scholar] [CrossRef]
- Hötzer, J.; Reiter, A.; Hierl, H.; Steinmetz, P.; Selzer, M.; Nestler, B. The parallel multi-physics phase-field framework pace3d. J. Comput. Sci. 2018, 26, 1–12. [Google Scholar] [CrossRef]
- Sophocleous, M. Understanding and explaining surface tension and capillarity: An introduction to fundamental physics for water professionals. Hydrogeol. J. 2010, 18, 811–821. [Google Scholar] [CrossRef]
- Løvoll, G.; Méheust, Y.; Måløy, K.J.; Aker, E.; Schmittbuhl, J. Competition of gravity, capillary and viscous forces during drainage in a two-dimensional porous medium, a pore scale study. Energy 2005, 30, 861–872. [Google Scholar] [CrossRef]
- Byon, C.; Kim, S.J. The effect of meniscus on the permeability of micro-post arrays. J. Micromech. Microeng. 2011, 21, 115011. [Google Scholar] [CrossRef]
- Martínez-Criado, G.; Villanova, J.; Tucoulou, R.; Salomon, D.; Suuronen, J.-P.; Labouré, S.; Guilloud, C.; Valls, V.; Barrett, R.; Gagliardini, E.; et al. Id16b: A hard x-ray nanoprobe beamline at the esrf for nano-analysis. J. Synchrotron Radiat. 2016, 23, 344–352. [Google Scholar] [CrossRef] [Green Version]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. Nih image to imagej: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Liakopoulos, A.C. Darcy’s coefficient of permeability as symmetric tensor of second rank. Hydrol. Sci. J. 1965, 10, 41–48. [Google Scholar] [CrossRef] [Green Version]
- POROMETER. Wetting Liquids. Available online: http://www.porometer.com/porometers/wetting-liquids/ (accessed on 14 February 2022).
- Ramli, M.R.M.; Ahmad, A.L.; Leo, C.P. Surface modification of polytetrafluoroethylene hollow fiber membrane for direct contact membrane distillation through low-density polyethylene solution coating. ACS Omega 2021, 6, 4609–4618. [Google Scholar] [CrossRef]
- Lee, K.; Ivanova, N.; Starov, V.; Hilal, N.; Dutschk, V. Kinetics of wetting and spreading by aqueous surfactant solutions. Adv. Colloid Interface Sci. 2008, 144, 54–65. [Google Scholar] [CrossRef]
- Williams, T.; Kelley, C. Gnuplot 5.2: An Interactive Plotting Program. Available online: http://gnuplot.sourceforge.net/ (accessed on 14 February 2022).
- Wang, F.; Ratke, L.; Zhang, H.; Altschuh, P.; Nestler, B. A phase-field study on polymerization-induced phase separation occasioned by diffusion and capillary flow—A mechanism for the formation of porous microstructures in membranes. J. Sol-Gel Sci. Technol. 2020, 94, 356–374. [Google Scholar] [CrossRef]
- Brandt, N.; Griem, L.; Herrmann, C.; Schoof, E.; Tosato, G.; Zhao, Y.; Zschumme, P.; Selzer, M. Kadi4Mat: A Research Data Infrastructure for Materials Science. Data Sci. J. 2021, 20, 8. [Google Scholar] [CrossRef]


| Effective Properties | Porefil® Properties [48] | ||||||
|---|---|---|---|---|---|---|---|
| Sample | (-) | K (10 ) | ( m) | ( m) | (mN m−1) | () | (mPa s) |
| 1 | 0.89 | 16.83 | 2.16 | 0.64 | >16.0 | >0.0 | >2.2 |
| 2 | 0.82 | 7.78 | 1.73 | 0.72 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altschuh, P.; Kunz, W.; Bremerich, M.; Reiter, A.; Selzer, M.; Nestler, B. Wicking in Porous Polymeric Membranes: Determination of an Effective Capillary Radius to Predict the Flow Behavior in Lateral Flow Assays. Membranes 2022, 12, 638. https://doi.org/10.3390/membranes12070638
Altschuh P, Kunz W, Bremerich M, Reiter A, Selzer M, Nestler B. Wicking in Porous Polymeric Membranes: Determination of an Effective Capillary Radius to Predict the Flow Behavior in Lateral Flow Assays. Membranes. 2022; 12(7):638. https://doi.org/10.3390/membranes12070638
Chicago/Turabian StyleAltschuh, Patrick, Willfried Kunz, Marcel Bremerich, Andreas Reiter, Michael Selzer, and Britta Nestler. 2022. "Wicking in Porous Polymeric Membranes: Determination of an Effective Capillary Radius to Predict the Flow Behavior in Lateral Flow Assays" Membranes 12, no. 7: 638. https://doi.org/10.3390/membranes12070638
APA StyleAltschuh, P., Kunz, W., Bremerich, M., Reiter, A., Selzer, M., & Nestler, B. (2022). Wicking in Porous Polymeric Membranes: Determination of an Effective Capillary Radius to Predict the Flow Behavior in Lateral Flow Assays. Membranes, 12(7), 638. https://doi.org/10.3390/membranes12070638






